
A NEW METHOD FOR DETECTION
OF TOTAL HIP REPLACEMENT LOOSENING
Development and First Results of a Novel Mechano-acoustical Sensor
Cathérine Ruther, Daniel Kluess, Andreas Fritsche, Wolfram Mittelmeier, Rainer Bader
Department of Orthopaedics, University of Rostock
Doberaner Str. 142, 18057 Rostock, Germany
Hartmut Ewald
Department of General Electronics, University of Rostock, Justus-von-Liebig-Weg 2, 18059 Rostock, Germany
Keywords: Total hip replacement, Implant loosening, Sensor, Diagnosis tool.
Abstract: Currently applied diagnostic methods for loosening of total hip replacements often result in imprecise
identification of implant fixation and in the worst case unnecessary revision surgery. Developed sensors
integrated in implants require adequate energy supply, which in most cases is achieved by inductive
coupling and complex data telemetry. In order to avoid a telemetric apparatus, we developed a passive
concept of a novel in vivo method to improve diagnostic investigations of total hip replacement loosening.
A mechano-acoustical sensor, attached on small membranes inside the femoral hip stem, is proposed and
enables osseous anchorage detection. The sensor is excited and detected by extracorporeal coils. First
functional models show significant differences between different material layers located at the membrane.
The novel in vivo sensor system has a promising potential to detect implant loosening.
1 INTRODUCTION
Prediction of total hip replacement (THR) loosening
using ex vivo techniques such as imaging often fails
to deliver reliable and localized results
(Temmerman, 2005). Moreover, monitoring of
implant anchorage is an important factor in
determining the longterm success rate of implants
(Pastrav, 2009). Currently diagnostic methods used
to identify THR loosening do not provide satisfying
results. Hence, researchers strive for supporting
techniques or even attempt to replace ex vivo
techniques by novel in vivo sensors such as
accelerometers in vibrometry (Li, 1995), which
showed that implant instability can be identified by
several harmonics in the output signal of the sensors.
However, the low signal-to-noise ratio of presently
available accelerometers needs to be addressed
(Marschner, 2007).
Besides miniaturized design and
biocompatibility, a reliable power supply is a
challenge in development of in vivo sensors. The
technology of choice for vibrometry is wireless
powering and data telemetry (Puers, 1999). With this
system, energy can be provided externally and
transcutaneous. This is achieved by a primary coil,
used outside the human body, and a secondary coil
integrated in the implant in vivo.
Inductive powering and passive data telemetry is
effective for implants where both coils are
positioned with small separation distance and fixed
orientation. In many cases this cannot be achieved
because of possible changes in the patients´ weight.
The influence of variable distances between both
coils leads to lower coupling factors and therefore
poor signal transmission. Additionally, interferences
of the inductive telemetry during patients´
movements could occur. Another problem is the use
of only low frequencies, which affect the damping of
the signals caused by eddy currents, especially in
metals. This is an evident drawback for orthopaedic
implants, especially for THR where biocompatible
metal alloys are used.
Due to the aforementioned reasons we propose a
novel and passive sensor concept without inductive
70
Ruther C., Ewald H., Mittelmeier W., Kluess D., Fritsche A. and Bader R..
A NEW METHOD FOR DETECTION OF TOTAL HIP REPLACEMENT LOOSENING - Development and First Results of a Novel Mechano-acoustical
Sensor.
DOI: 10.5220/0003165900700073
In Proceedings of the International Conference on Biomedical Electronics and Devices (BIODEVICES-2011), pages 70-73
ISBN: 978-989-8425-37-9
Copyright
c
2011 SCITEPRESS (Science and Technology Publications, Lda.)

coupling of two coils. The concept is characterized
by extracorporeal coils and permanent magnets
inside the THR acting as loosening sensors. In the
present work, we demonstrate the development of a
custom trigger circuit as well as first results gained
by functional models of a sensor prototype.
2 METHODS
2.1 Principle of the Loosening Sensor
The proposed method is based on the conservation
of momentum including impingement of a magnetic
body on a membrane placed inside the femoral hip
stem (Figure 1). The magnetic body, fixed on a flat
spring, is part of the sensor, which is designed as a
simple mechanical oscillator for impinging in the
middle of a membrane (Ruther, 2010). The magnetic
poles allow oscillations using an external magnetic
field impulse. Thus, motion of the oscillator is
initiated using an external coil.
Figure 1: System concept of the novel sensors for
excitation of the THR inside the hip stem and wireless
detection of implant loosening.
The end velocity of the oscillator after
impingement varies depending on the
osseointegration of the implant. On the one hand, a
well osseous integrated implant leads to lower end
velocities of the oscillator after impingement caused
by the higher energy transfer of the oscillator due to
lower deformation energy. Therefore, the spring
back of the sensor will be lower. On the other hand,
a loosened THR with soft tissue on the external
surface of the membrane results in higher end
velocities caused by higher deformation energy.
Since the oscillator is magnetic, the read out of
the measured data can also be achieved by an
external coil.
2.2 Experimental Setup
In order to demonstrate the functional principle of
our loosening detection approach, a cylindrical setup
composed of non magnetic aluminium was built. A
0.25 mm thick titanium membrane was mounted to
the test device and tightened by screws to ensure
symmetrical oscillations of the membrane
(Figure 2). The oscillator was made of a steel spring
and an attached magnetic neodymium body (1.3T).
Figure 2: Experimental setup for the new loosening
sensor. Left: Fixing device with oscillator, which impinge
on a titanium membrane. Right: Cross section of the setup
with different material layers attached to the membrane.
The influence of different material layers on the
impingement behaviour of the oscillator was also
tested. Thus, artificial bone substitute material
(20 pcf, Sawbones, Malmö, Sweden) with a
thickness of 50 mm was used to simulate full
osseous implant integration. A further setup with
additional gelatine layers of 10 mm and 5 mm
thickness, simulating a loose implant was
investigated (Figure 2). Gelatine was used to
represent collagen and fluid outside the membrane
as in the case of implant loosening. In a final setup,
small gaps were included in the artificial bone in
order to simulate bone defects adjacent to the
membrane (partial osteolysis). Measurements for
each material setup were repeated 20 times.
2.3 Power Supply and Detection
2.3.1 Inductive Unit
The inductive unit is composed of two coils and
shall be placed outside the patients` leg in defined
distances to the oscillators.
In the experimental setup (Figure 3) the
excitation coil made of a ferrite core to concentrate
A NEW METHOD FOR DETECTION OF TOTAL HIP REPLACEMENT LOOSENING - Development and First Results
of a Novel Mechano-acoustical Sensor
71
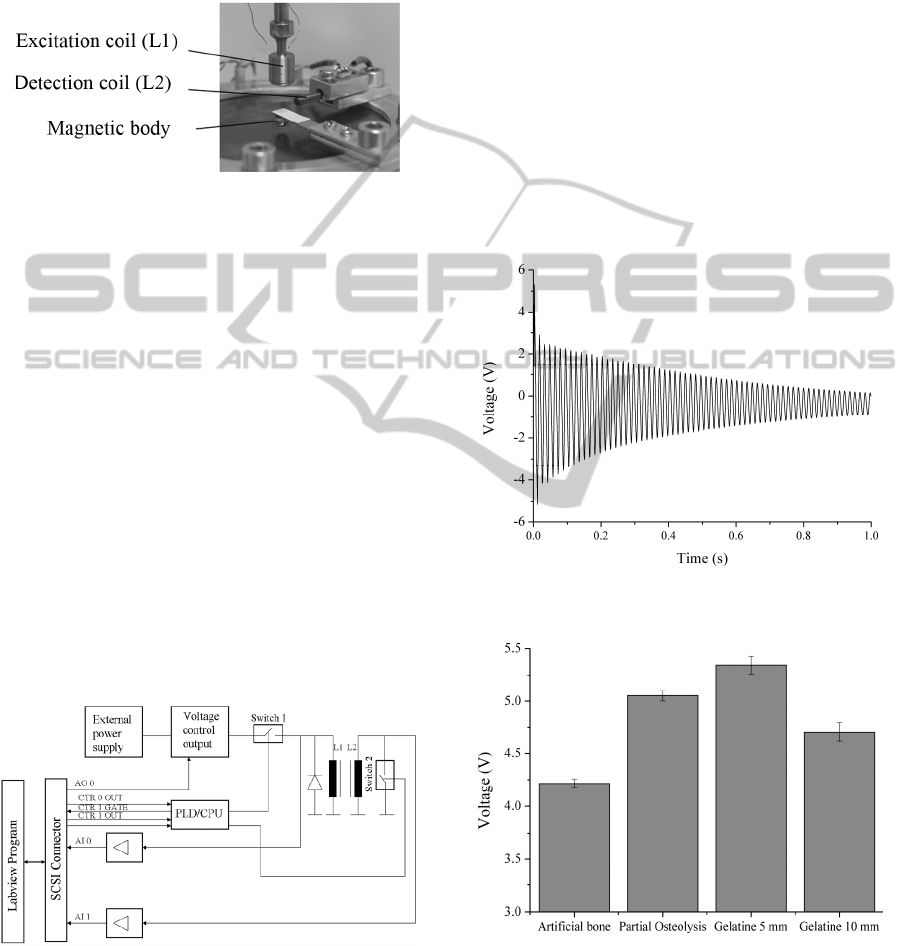
the streamlines of the magnetic field on the
oscillator inside the implant. Ferrite cores allow
faster reduction of the magnetic field than iron cores.
The detection coil included an iron core with a
diameter of 2.3 mm for precise detection of the
oscillator signal.
Figure 3: Arrangement of the excitation and detection
coils around the oscillator.
2.3.2 Trigger circuit
For adequate configuration of the trigger circuit
(Figure 4), it was important that the magnetic field
of the excitation coil (L1) reduces very fast.
Otherwise, the signal of L1 overlaps the signal of the
oscillator. Therefore the external hardware included
a central processing unit (CPU) where the sequence
control could be achieved by different counters,
configured by a custom Labview program (NI 9.0,
TX, USA). Counter 0 defined past which time
counter 1 was closed. Counter 1 determined the time
how long switch 1 was closed. An overflow of
counter 0 resulted in an increasing edge (CTR 0
OUT), thus signalizing that switch 1 could be closed
due to the excitation time and displacement of the
oscillator. Simultaneously, the release of counter 1
was introduced (CTR 1 GATE). The signal for the
overflow of counter 1 was CTR 1 OUT.
Figure 4: Block diagram of the external hardware.
A recovery diode enabled the fast decrease of the
excitation magnetic field. Switch 2 was controlled
by a digital signal defining the time when the
detection coil (L2) could monitor the oscillator
signal without detection of the excitation magnetic
field. The signal of L2 was amplified and evaluated
in the Labview program. Parameters such as the
highest amplitude after impingement and amplitudes
in a frequency spectrum were evaluated.
3 RESULTS
The detection of the oscillation signal of the
loosening sensor resulted in a good differentiation of
the varying material layers at the external side of the
membrane. For evaluation the first amplitude in the
time signal proportional to the highest velocity of
the oscillator after impingement was considered
(Figure 5). The results are presented as mean values
± standard deviation in Figure 6.
Figure 5: Time signal of the oscillation of the loosening
sensor for artificial bone with partial osteolysis.
Figure 6: Results of the amplitude measurements with
different material layers attached to the external side of the
membrane.
Furthermore, the signal in the frequency domain
as spectral analysis with a Fast Fourier
Transformation was investigated (Figure 7). The
BIODEVICES 2011 - International Conference on Biomedical Electronics and Devices
72

first eigen frequency of the oscillator was identified
at 67 Hz. The peak of the second harmonic was at
134 Hz. Comparison of different material layers
revealed decreased amplitudes with a thicker
gelatine layer.
Figure 7: Frequency spectrum of the oscillations of the
loosening sensor.
4 DISCUSSION
With respect to the standard application of total hip
replacements, unsatisfying results in loosening
diagnosis increase the demand for more precise in
vivo techniques. Inductive coupling based on radio
frequency powering to provide energy supply of in
vivo sensors causes problems, especially with regard
to the coupling factor between the two coils. To
circumvent this problem, our approach was to show
the capability of a novel sensor principle using basic
research models.
The trigger circuit to control the excitation and
detection coils allows the identification of the
highest velocity after impingement of the oscillator
on the membrane. This leads to promising
experimental results, which show the usability of the
described in vivo method for detecting total hip
replacement loosening with extracorporeal coils and
a passive internal sensor.
The robustness of the oscillators due to the
simplicity of the assembly guarantees functionality
during intraoperative impaction and sterilization of
the implant. Moreover, the oscillators can be used in
experimental applications to determine the quality of
osseointegration of new coated implant materials.
5 CONCLUSIONS
In the present study, a new measurement method for
in vivo diagnosis of total hip replacement loosening
without inductive coupling based radio frequency
powering was demonstrated. The described
loosening sensors with two coils and a custom
trigger circuit shows results with good prospects in
preliminary tests.
Future work will include the implementation of
the loosening sensor in real implants. Furthermore,
solutions for continuous excitation and therefore the
optimization of the trigger circuit are aspired.
Enhancements of the inductive unit are
designated with respect to air-core coils as detection
coils, which are switched as differential coils for
better erasure of the excitation field during
detection.
ACKNOWLEDGEMENTS
This research project is granted by the German
research foundation (DFG) under Reg.No. KL 2327.
REFERENCES
Temmerman, O. P., Raijmakers, P. G., Berkhof, J.,
Hoekstra, O. S., Teule, G. J., Heyligers, I. C., 2005.
Accuracy of diagnostic imaging techniques in the
diagnosis of aseptic loosening of the femoral
component of a hip prosthesis. In: Journal of Bone
and Joint Surgery, 87, pp. 781-785.
Pastrav, L. C., Jaeques, S. V. N., Jonkers, I., Van der
Perre, G., Mulier, M., 2009. In vivo evaluation of a
vibration analysis technique for the per-operative
monitoring of the fixation of hip prostheses. In:
Journal of Orthopaedic Seurgery and Research. 4, pp.
1-10.
Marschner, U., Jettkant, B., Ruwisch, D., Zhu, Y., Grätz,
H., Fischer W. J., Clasbrummel, B., 2007. FEM
simulation and wireless measurement of hip prosthesis
vibrations for loosening detection. European
Symposium , Technical Aids for Rehabilitation, 25-26
January 2007, Berlin, pp. 87-88.
Puers, R., Catrysse, M., Vandevoorde, G., Collier, R. J.,
Louridas, E., Burny, F., Donkerwolcke, M., Moulart,
F.,1999. A telemetry system for the detection of hip
prosthesis loosening by vibration analysis. In: Sensors
and Actuators, 85, pp. 42-47.
Ruther, C., Ewald, H., Biemann, A., Nierath, H., Bader,
R., Kluess, D., 2010. A New Concept for Non-
Invasive Radiation-Free Detection of Implant
Loosening. 56
th
Annual Meeting of the Orthopaedic
Research Society, New Orleans, USA, 6-9 March
2010, #2413.
A NEW METHOD FOR DETECTION OF TOTAL HIP REPLACEMENT LOOSENING - Development and First Results
of a Novel Mechano-acoustical Sensor
73
