
Stearic Acid Grafted Carboxymethyl Chitosan and Its
Nanoencapsulation of Macadamia Oil
H Wang
1,2
, Z M Yang
2
, S D Li
2,3
, Y Z Xia
4
, B B Wang
4
, C Zhou
2
, C Wang
2
and P
W Li
2, *
1
College of Food Science & Technology, Huazhong Agricultural
University,Wuhan ,430070, China
2
Agricultural Products Processing Research Institute, Chinese Academy of Tropical
Agricultural Sciences, Zhanjiang, 524001, China
3
College of Chemistry and Environment, Guangdong Ocean University, Zhanjiang,
524088, China
4
Collaborative Innovation Center for Marine Biomass Fibers, Materials and Textiles
of Shandong Province, Qingdao University, Qingdao, 266071, China
Corresponding author and e-mail: P W Li,1520102069@qq.com
Abstract. This work is aims to synthesize a novel stearic acid (SA) grafted N,O-
carboxymethyl chitosan (NOCC), and then use it as a wall material to nanoencapsulation
macadamia oil. The grafted copolymer is a amphiphilicity sample. By the catalysis of 1–(3-
dimethyl amino propyl)-3-ethyl carbodiimide hydrochloride (EDC·HCl) and N-hydroxy
succinimide(NHS), we got hydrophilic and hydrophobic NOCC-SA polymers via the
condensation reaction between the carboxyl of arboxymethyl chitosan and the carboxyl of
stearic acid in a water bath. Macadamia oil has significant effect on whitening and spotting,
also as a bioactive compound with health beneficial potential, which are used as safe
additives in food , medicine, cosmetics. However Macadamia oil is sensitive to light, thermal
condition and oxidation. Nanoencapsulation could be an adequate technique to overcome
these challenge. Here, we obtained the analysis by scanning electron microscope and
grainsize analyzer that most nanocapsules sizes ranged from 130.2 to 200.8 nm. To sum up,
this study showed that amphiphilic chitosan were a kind of potential carrier. It could increase
the solubility of hydrophobic substances and make it sustained release by nanoencapsulation.
1. Introduction
Macadamia integrifolia, also known as Australian walnuts, Queensland fruits and macadamia nuts,
belong to evergreen trees and dicots. Among many dried fruits in the world, macadamia integrifolia
have high nutritional and medicinal value, and therefore it have the highest economic value, and
always been regarded as the “king of dried fruit” [1]. The main fatty acid content of macadamia oil is
mainly composed of unsaturated fatty acids such as oleic acid, palmitic acid and stearic acid, and a
small amount of linoleic acid. Among them, macadamia integrifolia contain more than 80% of
unsaturated fatty acids, making macadamia oil with lower blood pressure, blood lipid regulation, and
can be used as an effective additive in food and drugs. However, it can also be used as an effective
Wang, H., Yang, Z., Li, S., Xia, Y., Wang, B., Zhou, C., Wang, C. and Li, P.
Stearic Acid Grafted Carboxymethyl Chitosan and Its Nanoencapsulation of Macadamia Oil.
In Proceedings of the International Workshop on Materials, Chemistry and Engineering (IWMCE 2018), pages 297-301
ISBN: 978-989-758-346-9
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
297

additive for moisturizing, nourishing and whitening products [2]. It can be applied directly to the
basal layer of the skin and very widely used in cosmetics, skin care products. Because of the high
ratio of unsaturated fatty acids, Macadamia oil is extremely susceptible to oxidation, which is
affecting the storage quality and transport of the oil. Therefore, nanoencapsulation technique might
be a suitable solution to solve these problems. In order to protect sensitive ingredients,
nanoencapsulation technology allows “wall material” enveloped these ingredients or “core” materials
from adverse reactions, volatile loss, or nutritional deterioration [3]. In this research, stearic acid
grafted N,O-carboxymethyl chitosan as a amphiphilicity polymer because of its ability to control the
release of sensitive ingredient, and cross-linking to form nanocapsulates. Nanocapsules based on
chitosan and modified starch as a wall material to enveloped Macadamia oil as an active ingredient
by freeze-drying. In their study, the sizes of most nanocapsules ranged from 339.3 to 553.3 nm and
chitosan: Hicap is 1.5: 8.5%, which showed the lowest oil release. In another study, Abreu [4] use
chitosan and cashew gum nanogel for L.sidodies oil encapsulation systems. Few studies have used
amphipathic modified chitosan as a wall material to deliver hydrophobic substances (such as plant
essential oils)[5]. The amphiphilic NOCC-SA polymers were prepared by stearic acid grafted N,O-
carboxymethyl chitosan, which nanocapsulation macadamia oil can improve the biological activity
and utilization of macadamia oil, making it more versatile and greatly increase its economic value.
What’s more amphiphilic NOCC-SA polymers can be a meaningful hydrophobic substances model,
provide new ideas for following related research.
2. Materials and methods
2.1. Materials and reagents
chitosan was purchased from Aladdin Industrial Co. Ltd. (Shanghai,China). The degree of
deacetylation and viscosity of chitosan were determined as 95% and 100-200 mpa.s, respectively.
stearic acid (SA), 1–(3-dimethyl amino propyl)-3-ethyl carbodiimide hydrochloride (EDC·HCl), N-
hydroxy succinimide(NHS) were all purchased from J&K Scientific LTD. (Beijing, China). All
other reagents were of analytical grade.
2.2. Synthesis of N,O-carboxymethyl chitosan (NOCC)
NOCC was prepared according to the previous report by Chen [6] with some modifications. Briefly,
5 g chitosan dispersed and mixed thoroughly in 500 mL of isopropyl alcohol flask at room
temperature. Then, the solution comprising 35 g monochloroacetic acid and 10 mL of 10 M NaOH
was dropwise added to the stirred slurry over a period of 30 min. The alkaline slurry was stirred for
additional 20 min. The resulting mixture was heated at 60
o
C for 3 h. And then, dialyzed against
distilled water with 7000 Da molecular weight cut-off membrane for 48 h. Finally, the dialyzate was
freeze-dried to afford NOCC [7].
2.3. Preparation of stearic acids grafted NOCC
The preparation of NOCC grafted coplymers were preformed by using stearic acid (SA). Briefly, 500
mg NOCC was dissolved in 50 mL of distilled water to make solution I. And then, 500 mg stearic
acids was dissolved in 40 mL of hot ethyl alcohol absolute, and add 1.5 times the molar amount of
EDC·HCl and NHS were stirred at 65°C until completely dissolved to prepare solution II.
Subsequently, Solution II was added to solution I under vigorous stirring conditions. The reaction
was carried out at 60°C for 5 h, dialyzed against distilled water for 48 h, and freeze-dried for 2 d.
IWMCE 2018 - International Workshop on Materials, Chemistry and Engineering
298
O
O
O
OH
OH
NH
2
NH
2
O
HO
HO
n
Chitosan
IPA/NaOH
monochloroacetic acid
O
O
O
OH
OCH
2
COO
-
NH·
NHCH
2
COO
-
O
HO
HO
n
NOCC
O
O
O
OH
OCH
2
COO
-
NHR
NHCH
2
COO
-
O
HO
HO
n
NOCC-SA
stearic acid (SA)
EDC·HCL/NHS
R =
OH
O
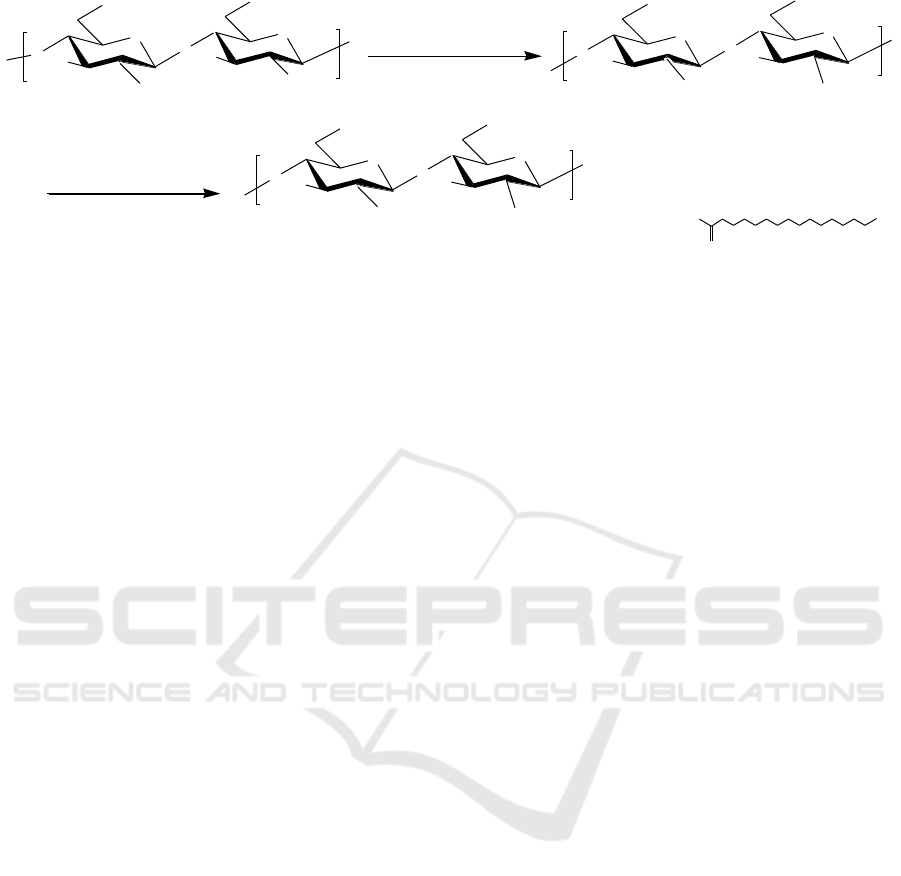
Figure 1. The proposed mechanisms for the synthesis of stearic acids grafted NOCC.
2.4. Preparation of NOCC-SA nanocapsulation Macadamia oil
NOCC-SA nanocapsulation macadamia oil were prepared with slight modification of previous
studies [8-9]. Briefly, 500 mg NOCC-SA was dissolved in 50 mL of distilleded water, after it was
completely dissolved. The macadamia oils was added in a 1:4 ratio (w/w, macadamia oils/ NOCC-
SA solution) to the solution. The resulting mixture flask at room temperature for 1d, and dialyzed
against distilled water with 7000 Da molecular weight cut-off membrane for 48 h. Then, dialysate
was treated three times with probe-type ultrasonic cell crusher: Power 80 W; Pulse on 2.0 s; Pluse off
3.0 s; Time 2 min, over 0.8 μm filter. Finally, the dialyzate was freeze-dried into nanocapsule powder.
2.5. Characterization of the products
Morphology of the freeze-dried nanocapsules was examined to study the surface structures of
powders by scanning electron microscopy (SEM; S-4800, HITACHI, Tokyo, Japan). The samples
were glued onto an adhesive tape mounted on the specimen stub, and particles were covered with
gold-palladium prior to analysis. Preparation a suitable concentration of NOCC-SA macadamia oil
nanocapsules solution, add 2 mL of nanocapsules solution to the cuvette, and measure the
nanocapsules size with a laser particle size analyzer.
3. Results and discussion
3.1. Preparation of phenolic acids grafted NOCC
In this study, stearic acid (SA) grafted NOCC were synthesized by using EDC·HCl and NHS as
catalyst. The possible mechanism for the stearic acid grafted NOCC was proposed in Figure 1. Firstly,
NOCC was synthesized in isopropyl alcohol/NaOH/ monochloroacetic acid system. And then, The
use the EDC·HCl and NHS as catalyst, which can catalyzes the reaction of the carboxyl group on SA
with the amino group on NOCC. During the reaction, EDC·HCl is used to activate the carboxyl
group and NHS is used to assist the reaction. NOCC and SA are linked together by amide bonds. The
obtained NOCC-SA grafted coplymers were amphiphilic samples.
3.2. Morphology
Scanning electron microscope (SEM) is based on the interaction between electrons and substances to
obtain the physical properties and chemical properties of the tested sample. SEM images of the
NOOC-SA macadamia oil nanocapsules are shown in follow Figure 2.By the analysis of represented
by Scanning electron microscope images, we know that the obtained nanocapsules showed a
spherical shape surface without rough.
Stearic Acid Grafted Carboxymethyl Chitosan and Its Nanoencapsulation of Macadamia Oil
299
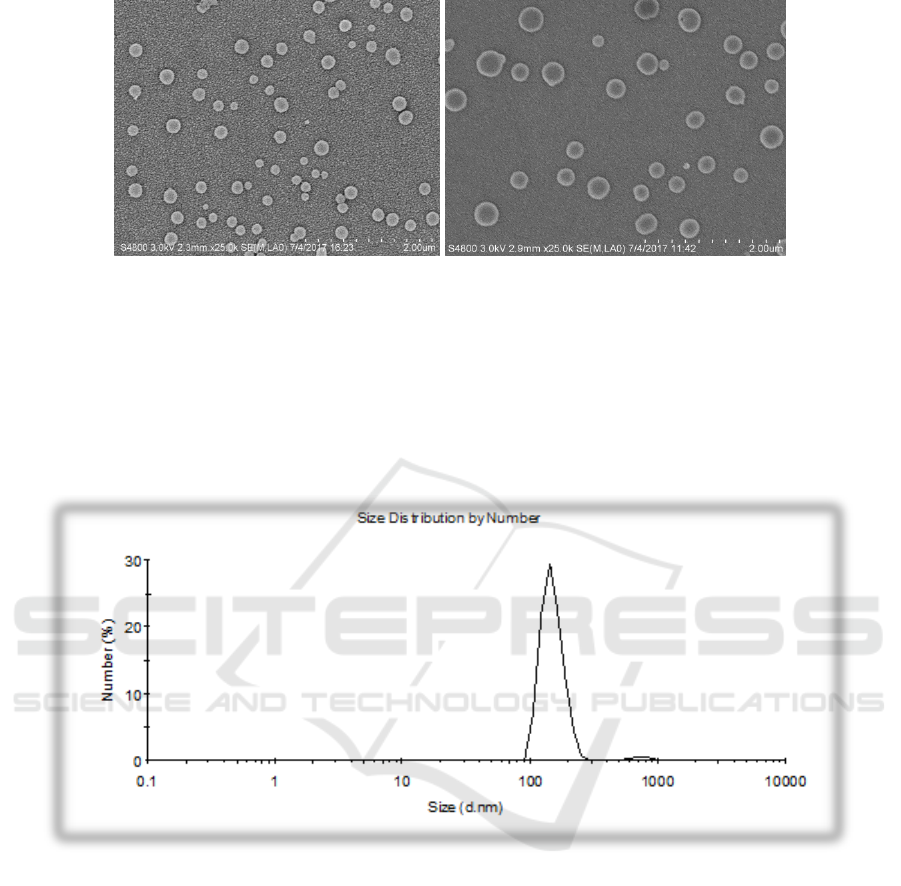
Figure 2. The SEM images of NOCC-SA macadamia oil nanocapsules.
3.3. Particle size
The hydrodynamic size of nanocapsules were determined by Malvern 3000HS (Malvern Instruments
Ltd., UK). In order to quantitatively determine the nanoparticle size of NOOC-SA macadamia oil
nanocapsules, set the detection angle to 90°, 25°C, and λ=532 nm. Most nanocapsules sizes ranged
from 130.2 to 200.8 nm. Particle size analysis chart of NOOC-SA macadamia oil nanocapsules as
shown Figure 3:
Figure 3. The size distribution of NOOC-SA macadamia oil nanocapsules.
4. Conclusions
The preparation, characterization of stearic acids grafted NOCC were investigated in the study.
Results suggested that the stearic acids could be easily grafted into NOCC by using EDC·HCl and
NHS as catalyst. In addition, the nanocapsules showed a Spherical and smooth surface by SEM
images. In conclusion, NOCC-SA is the promising coat material for developing delivery systems for
the hydrophobic substances. The findings obtained from the study suggest that NOCC-SA is
amphiphilicity which had a great potential to improve the stability of macadamia oil for its
utilization in perfumery, food industries, antimicrobial and antiseptic products.
Acknowledgment
This work was supported by the Natural Science Foundation of Guangdong Province (No.
2016A030307003), and the Central Public-interest Scientific Institution Basal Research Fund for
Chinese Academy of Tropical Agricultural Sciences (No.1630122017011, No.1630122017009).
IWMCE 2018 - International Workshop on Materials, Chemistry and Engineering
300

References
[1] Navarro S L B and Rodrigues C E C 2016 Macadamia oil extraction methods and uses for the
defatted meal byproduct J. Trends in Food Science & Technology 54 148-154
[2] Chen Y, Tang H, Liu Y and et al 2016 Preparation and study on the volume phase transition
properties of novel carboxymethyl chitosan grafted polyampholyte superabsorbent
polymers J. Journal of the Taiwan Institute of Chemical Engineers 59 569-77
[3] Liu J, Lu J F, Kan J and et al 2013 Preparation, characterization and antioxidant activity of
phenolic acids grafted carboxymethyl chitosan J. International Journal of Biological
Macromolecules 62(2) 85-93
[4] Abreu F O, Oliveira E F, Paula H C and et al 2012 Chitosan/cashew gum nanogels for
essential oil encapsulation J. Carbohydrate Polymers 89(4) 1277-82
[5] Donsì F, Annunziata M, Sessa M and et al 2011 Nanoencapsulation of essential oils to enhance
their antimicrobial activity in foods J. LWT - Food Science and Technology 44(9) 1908-14
[6] Kusuma H S, Al-Sa’Bani A F and Darmokoesoemo H N 2015 O-Carboxymethyl chitosan: an
innovation in new natural preservative from shrimp shell waste with a nutritional value and
health orientation J. Procedia Food Science 3 35-51
[7] Jung B O, Chung S J and Sang B L 2006 Preparation and characterization of eugenol-grafted
chitosan hydrogels and their antioxidant activities J. Journal of Applied Polymer Science
99(6) 3500–06
[8] Stefan D, Dima C and Iordăchescu G 2015 Encapsulation of Functional Lipophilic Food and
Drug Biocomponents J. Food Engineering Reviews 7(4) 417-438
[9] Bahreini E, Aghaiypour K, Abbasalipourkabir R and et al 2014 Preparation and
nanoencapsulation of l -asparaginase II in chitosan-tripolyphosphate nanoparticles and in
vitro, release study J. Nanoscale Research Letters 9(1) 340
Stearic Acid Grafted Carboxymethyl Chitosan and Its Nanoencapsulation of Macadamia Oil
301
