
The Origin of Artificial Kinetic
Spectroscopy and Its Application
Ilya Fine
OrSense Ltd., Bergman 2, 76705 Rehovot, Israel
Abstract. Intentionally creating local blood flow cessation at different body sites initi-
ates the time dependent optical response. Specifically, applying circumferential over
systolic pressure at the finger base creates a very stable post-occlusion optical signal
(POS). Geometrically independent features of light absorption and scattering of the
blood are extracted from POS by evaluating the parametric slope (PS). It was shown
that PS, being determined from suitable pairs of wavelengths, correlates with arterial
blood oxygen saturation. Other PS pairs exhibited a strong correlation with blood he-
moglobin. The time dependent signal was simulated in vitro by using the red blood cells
(RBC) aggregation process and was shown to resemble the main features of in vivo
POS
.
1 Introduction
The non-invasive technique for measuring various blood parameters has become very
popular since it eliminates the withdrawal of a blood sample from a patient. Optical
technique utilizes the detection of light response from different locations on the pa-
tient’s body. A commonly used device of this kind, such as the pulse-oximeter, origi-
nates from photoplethysmography (PP). In PP, the light response experiences variable
attenuation due to the pulsation of arterial blood.
Recently, a new approach [1,2], was established, known as Occlusion Spectroscopy,
as an additional method to obtain blood related dynamic spectral characteristics. In
Occlusion Spectroscopy over-systolic pressure is applied to the fleshy tissue to create
a state of temporary blood flows cessation at the measurement site. To optimize this
signal, the PP volumetric affect has to be suppressed.
Once blood flow cessation is established, the optical characteristics start to change
dramatically, changing the detected dynamic signal by about 30%, and sometimes
even by 60%, in comparison to the pulsatile signal that fluctuates only about 2-3%.
The time and spectral kinetic features of the occlusion spectroscopy signal can be
simulated in vitro, by using the RBC aggregation process [5,6].
Our goal was twofold: a) to create a better understanding of POS and b) to elaborate
appropriate algorithmic approaches for utilization in practical applications.
Fine I. (2005).
The Origin of Artificial Kinetic Spectroscopy and Its Application.
In Proceedings of the 1st International Workshop on Biosignal Processing and Classification, pages 110-120
DOI: 10.5220/0001192001100120
Copyright
c
SciTePress

2 Measurement and control system
The Occlusion Spectroscopy device is a microprocessor-based instrument comprised
of the measuring, controlling and display circuitry, the pneumatic system and the
hand-held sensor probe.
The probe optoelectronics consists of 10-segmented light sources in the spectral range
of 610 to 945 nm and a photodetector, which is synchronized and sequentially acti-
vated to generate the required spectroscopy.
The probe housing contains the controlled pneumatic cuff for temporary finger-base
blood occlusion.
3 Hand-held Probe
3.1 Fingertip type probe
The preferred site for pulse-oximetry is the fingertip. Naturally, it is the first choice
for the occlusion spectroscopy investigation. Two air pressure lines are used to ma-
nipulate the signal. The primary line creates the blood occlusion (flow cessation) and
the secondary line controls a varying pressure, which is applied independently at the
optical measurement site.
3.2 Ring – like probe
A second option to control the POS is the ring shape probe configuration. A single
pressurizing line is served for both the optimization of pre-occlusion condition and for
creating over systolic occlusion pressure to initiate POS.
A B
Fig. 1. Probe and monitor overview. A Fingertip probe B “Ring” probe
4 Typical Forms of Post-Occlusion Signals
Two different modes of POS were investigated – “direct” and modulated
111

4.1 Non-modulated occlusion signals (Direct)
The measurement procedure includes two stages. The initial stage begins by applying
moderate under systolic pressure on the testing site (fingertip) in order to deplete the
tissue from venous blood to a standard volume. The second stage involves applying
occluded pressure and opto measurement. Keeping the occluded volume of blood and
tissue unchanged is essential to guarantee a stable evolvement of a POS signal. Typi-
cal transmission and reflection POS are shown in Fig. 2 and Fig. 3, respectively.
02468
0.76
0.78
0.80
0.82
0.84
0.86
0.88
0.90
0.92
0.94
0.96
0.98
1.00
1.02
Occlusion location: Finger base
Measurement location: Finger tip
815nm
613nm
Tranmsission (A. U.)
Time (in sec)
Fig. 2. Typical post occlusion transmission signal measured at fingertip location.
02468
0.82
0.84
0.86
0.88
0.90
0.92
0.94
0.96
0.98
1.00
1.02
Occlusion location: Finger base
Measurement location: Finger base
815nm
613nm
Reflection (R.U)
Time (in sec)
Fig. 3. Typical post occlusion reflection signal measured at finger base.
112

4.2 Modulated Occlusion Mode (MOM)
Creating perturbations of the occluded media with the secondary pressure wave dur-
ing the blood flow cessation facilitates occlusion spectroscopy. This mode of signal
modulation, or Modulated Occlusion Mode (MOM), enables the generation of optical
changes induced by blood volume perturbation. For the “fingertip” probe, the primary
pressure cuff is kept over-systolic, while the secondary cuff is modulated [Fig. 4]. For
the “Ring” probe, both occlusion and modulated pressure coincide in one finger-base
cuff while the modulation is superimposed on the over systolic pressure [Fig 5].
0 5 10 15 20 25 30 35
0.65
0.70
0.75
0.80
0.85
0.90
0.95
1.00
1.05
Occlusion location: Finger base
Measurement location: Finger tip
Start of Modulation Signal
Start of Occlusion
810nm
613nm
Transmission (A.U.)
Time (in sec)
Fig. 4. Typical post MOM signal measured at fingertip. Over systolic pressure of 200mm Hg is
applied at finger base. Pressure modulation (0 – 30 mm Hg) is applied on the fingertip
0 5 10 15 20 25 30 35
0.60
0.65
0.70
0.75
0.80
0.85
0.90
0.95
1.00
1.05
Start of occlusion
Occlusion location: Finger base
Measurement location: Finger base
810nm
613nm
Transmission (A.U.)
Time (in sec)
Fig. 5. Typical MOM transmission signal measured at finger base site. Over systolic pressure is
modulated between 200 – 230 mm Hg
5 Parametric Slope
Occlusion initiates the time dependent optical signal. Some optical properties of
blood, related to its biochemical content, are not time dependent (in scale of seconds).
In order to link the time dependent signal with the sought blood parameter, there is a
113
need to eliminate time dependent characteristics. This can be achieved using signals at
a pre-determined wavelength as a reference for the rest of the signals.
5.1 Basic Properties of the Parametric Slope
The definition of the parameter slope (PS) for the two signals
1
λ
I
(t) and
2
λ
I
(t) being
measured at two wavelengths,
2,1
λ
λ
is [2,3]:
tILn
tILn
PS
∂∂
∂∂
=
/)(
)/)((
2
1
λ
λ
(1)
where
2
λ
I
is a reference signal. In our analysis we used a signal at 876nm as a refer-
ence signal. PS can be calculated at any stage of POS.
The physical meaning of PS is quite similar to the parameter called Gamma, widely
used in pulse-oximetry. Gamma for the pulsatile signal is defined as [7]:
)(
))((
)(
))((
2
2
1
1
λ
λ
λ
λ
I
I
I
I
Gamma
∇∇
=
(2)
where
)(
λ
I∇
is fluctuation of pulsatile component of the optical response.
The typical spectral structure of PS was calculated from 2000 measurements per-
formed on 270 patients [Fig. 6]. This spectral structure resembles HbO
2
absorption,
which is less affected by blood perfusion and finger size.
600 650 700 750 800 850 900 950
0.2
0.4
0.6
0.8
1.0
1.2
1.4
1.6
1.8
Reference 876nm
Transmission
Parametric Slope (PS)
Wavelength (nm)
Fig. 6. Average PS for transmission signal as a function of wavelengths, obtained from the
“Ring” probes, at the first 2 seconds of measurement
5.2 Time Dynamic Structure of Parametric Slope
The absence of time dependency of PS is well founded only for the initial part of the
occlusion signal. Due to the course of occlusion evolvement, the values of PS’s are
changed with different rates. Delta PS is defined as a difference between the PS at t=8
sec, and PS at t=0, from starting of over-systolic pressure application [Fig.7].
114

600 650 700 750 800 850 900 950
-0.8
-0.7
-0.6
-0.5
-0.4
-0.3
-0.2
-0.1
0.0
0.1
0.2
0.3
Reference 876nm
Transmission
Delta (Parametric Slope)
Wavelength (nm)
Fig. 7. Average Delta PS for transmission signal as a function of wavelengths
6 In Vitro Measurements
The main features of POS can be simulated in vitro, assuming that the RBC aggrega-
tion mechanism is the driving force behind the optical signal. There are a variety of in
vitro configurations where the optical signal changes in association with RBC aggre-
gation [5,6]. In our case, the sample of RBC suspension mixed with 1% of Dextran
was pumped into a reservoir where two pipes were connected to the specially de-
signed rigid glass cuvette of 1.mm thickness. The cessation of blood flow initiated
RBC aggregation. In the first version of the cuvette (open space cuvette), the internal
space volume was available for the blood motion, while in the second version (tissue-
like), the cuvette was filled with 50 microns of glass fibers, leaving only 30% space
available for the blood motion. The motivation to design the tissue like model was to
observe the optical signal behavior in proximity of one dimensional aggregation sce-
nario (like in small blood vessels) and to create a DC scattering environment, resem-
bling the scattering behavior of in-vivo tissue. In both cases the signal was observed
at two stages, before blood flow cessation and after it. The pulse-like variations of
transmission signals at the first stage (Fig. 8) coincide with the peristaltic pump ac-
tion.
6.1 Open Space Blood model
The transmission changes measured in the open space cuvette [Fig.8] reveals signifi-
cant transient changes near the point of blood flow cessation. The characteristic rise
time and relative changes of the signal reasonably resembles the behavior of in vivo
POS.
115

0 5 10 15 20
0.70
0.75
0.80
0.85
0.90
0.95
1.00
Start of aggregation process
In vitro
Open space cuvette
810nm
613nm
Transmission (A.U.)
Time (in sec)
Fig. 8. Transmission signal before and after blood flow cessation. RBC suspension. Hematocrit
40% with 1% of Dextran. Glass cuvette (0.3mm thickness).
6.2 Tissue-like cuvette modeling
In the case of tissue-like cuvette, the appearance of a transient peak is negligible
[Fig.9]. The aggregation related signal is expressed in terms of PS by using expres-
sion (1). The graphs of PS’s and delta PS, as a function of light source (LED’s) wave-
lengths, for the tissue-like cuvette in-vitro signals are shown in Fig.10 and Fig. 11,
respectively.
05101520
0.55
0.60
0.65
0.70
0.75
0.80
0.85
0.90
0.95
1.00
1.05
Blood flow cessation
In vitro measurement: Blood aggregation dynamics
810nm
613nm
Transmission (A.U.)
Time
Fig. 9. Transmission signal before and after blood flow cessation. RBC suspension Hematocrit
40% with 1% of Dextran Glass cuvette (4mm thickness). 75% of cuvette volume were packed
by 50-micron fibers
.
116

550 600 650 700 750 800 850 900
0.5
1.0
1.5
2.0
2.5
3.0
In vitro aggregation
Parametric Slope (PS)
W a velen gth (nm )
Fig. 10. Parametric Slope as a function of wavelength, immediately after beginning of blood
flow cessation session in tissue-like cuvette
600650700750800850900
-0.15
-0.10
-0.05
0.00
0.05
0.10
In vitro
Transmission
Delta Parametric Slope
Wavelength (nm )
Fig. 11. Changes of Parametric Slope during 15 seconds of blood flow cessation obtained in
vitro in tissue-like cuvette
7 Clinical Applications
Post Occlusion optical signals depend on RBC absorption and scattering. Light ab-
sorption of the blood in visible and near infrared optical region is dominated by oxy-
hemoglobin (HbO
2
), hemoglobin (Hb) and it’s derivatives.
7.1 Non-pulse Oximetry
We performed an experiment in which POS was measured by the “Ring” probe. The
pre-occlusion pulsatile signals were measured and stored as well. This has been done
concurrently with conventional pulse oximeter readings (Datex Ohmeda 3900). Five
volunteers participated in the experiment. Each one participated in two independent
measurement sessions. In the first session, the volunteer breathed normally, and in the
second session, a facemask was applied supplying 100% oxygen. The correspondence
between PS (for the pair 670nm, 876nm) and Gamma with SpO
2
reading of the pulse
oximeter (R=0.8) is presented in Fig 12. The comparison between PS and SpO
2
[Fig.13] shows significant correlation (R=0.82).
117

93 94 95 96 97 98 99
0.45
0.50
0.55
0.60
0.65
0.70
Gam m a - for pulse
PS - for occusion
Parametric Slope, Gamma
SpO2
Fig. 12. PS and Gamma for 670nm and 876nm versus SpO
2
measured by oximeter
0.50 0.55 0.60 0.65 0.70
0.52
0.56
0.60
0.64
0.68
0.72
O cclu sion ve rsu s p ulse
(The same patients)
PS (670nm vs. 870nm)
Gamma (670nm, 876nm)
Fig. 13. Correlation between the Parametric Slopes obtained from the occlusions versus
Gamma values of pulsatile component of same patients
7.2 Hemoglobin
After approval by an Ethics Committee (IRB), and receipt of informed consents, a
group of 188 subjects from a hematology ward was studied. A “Finger- base probe”
was used to obtain POS. A standard lab blood analyzer measured the values of refer-
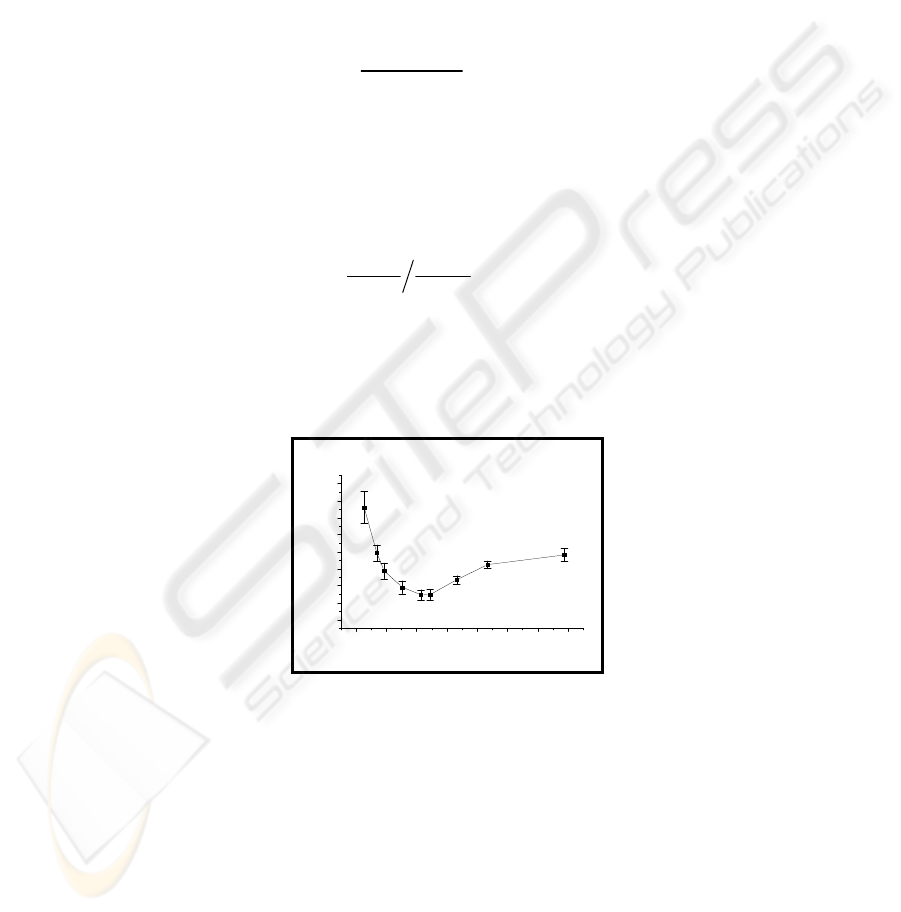
ence Hb. Hb and PS values were averaged over 70 points [Fig. 14].
118

8 9 10 11 12 13 14 15 16
0.4
0.5
0.6
0.7
0.8
0.9
1.0
1.1
1.2
1.3
1.4
1.5
1.6
1.7
1.8
1.9
2.0
2.1
2.2
2.3
610nm
633nm
646nm
673nm
701nm
718nm
760nm
812nm
940nm
PS (Parametric Slope)
Hemaglobin (g/dl)
Fig. 14. Average PS’s for transmission POS as a function of hemoglobin
8 Discussion
Two testing approaches were combined and investigated, i.e.1) blood occlusion plus
applying pressure modulation (superimposed) 2) stabilizing the measurement by re-
ducing the volumetric deviations at the opto-measurement site. This in turn resulted in
a strong blood related optical signal. The physiological origin of this post-occlusion
signal (POS) could be one of the following: local blood or tissue volume variations, or
alteration of light scattering of the blood as a result of RBC aggregation phenomena.
The mutually supporting experimental facts contribute to comprehend the underlying
nature of POS signal; a) Non-dimensional Parametric Slope (PS), extracted from
POS, exhibits a strong correlation with arterial blood oxygen saturation. b) The PS,
being determined for the pair 613nm and 876nm correlates with blood Hb. c) The
wavelength dependant structure of PS resembles HbO
2
absorption.
All these facts lead to an unequivocal conclusion that the major component contribut-
ing to POS is optical changes associated with arterial blood.
Another very important fact is that the kinetic behavior of a post-occlusion signal is
barely dependent on the location of the application of the over systolic pressure. How-
ever, the volumetric change is almost entirely pressure dependent. These facts signify
that the POS is driven by internal kinetics of RBC aggregation. Another important
feature of the POS signal is expressed in terms of PS changes. Very similar PS time
dependency was observed both in vivo and in vitro aggregation induced measure-
ments. Regarding the volumetric mechanism in vitro, there was observed no notice-
able PS vs. thickness dependency. This fact excludes volumetric mechanisms from
being materially responsible for PS changes in vivo.
In summary, we conclude that POS is initiated by erythrocyte aggregation. From the
practical point of view, it was shown that POS is useful for determining different
biochemical parameters of blood, including oxygen saturation and Hb level.
119

References
1. Fine, I., Fikhte, B., Shvartsman, LD.: RBC aggregation assisted light transmission through
blood and occlusion oximetry. Proc. SPIE, Vol. 4162, (2000) 130-140
2. Fine, I., Fikhte, B., Shvartsman, LD.:Occlusion spectroscopy as a new paradigm for non-
invasive blood measurements. Proc. SPIE, Vol. 4263, (2001) 122-130
3. Shvartsman, L.D., Fine, I. Optical transmission of blood: Effect of erythrocyte aggregation.
IEEE trans. on Biom. Eng., Vol. 50, (2003) 1020-1026
4. Finarov, A., Kleinman Y., Fine I.: Optical device for non-invasive measurement of blood
related signals utilizing a finger holder. (2001) US Patent 6,213,952B1
5. Priezzhev A.V., Ryaboshapka O.M., Firsov N.N., Sirko I.V.: Aggregation and disaggrega-
tion of erythrocytes in whole blood: Study by backscattering technique. Journal of Biomed.
Optics, Vol. 4, (1999), 76-84
6. Barshtein G., Wagnblum D., Edgar S.: Kinetics of linear rouleaux formation studied by
visual monitoring of red cell dynamic organization. Biophysical Journal, Vol. 78, (2000)
2470-2474
7. Kamat V.: Pulse oximetry. Indian J. Anaesth., Vol. 46, (2002) 261-268
120
