
PARAFAC CLASSIFICATION OF LAMB CARCASS SOFT
TISSUES IN COMPUTER TOMOGRAPHY (CT) IMAGE STACKS
Jørgen Kongsro
Animalia – Norwegian Meat Research Centre, P.O. Box 396 Økern, N-0513 Oslo, Norway
Keywords: Lamb carcass, Tissue, PARAFAC, Multi-Way Analysis, Computer Tomography, Classification, Image,
Stacks.
Abstract: Computer Tomography is shown to be an efficient and cost-effective tool for classification and
segmentation of soft tissues in animal carcasses. By using 15 fixed anatomical sites based on vertebra
columns, 120 lamb carcasses were CT scanned in Norway during autumn of 2005. Frequency distributions
of CT values (HU [-200,200]) of soft tissues from each image were obtained. This yielded a 3-way data set
(120 samples * 400 CT values * 15 anatomical sites). The classification of the soft tissues was done by
multi way Parallel Factor Analysis (PARAFAC), which resulted in 3 components or soft tissues classified
from the images; fat, marbled and lean muscle tissue.
1 INTRODUCTION
Computer Tomography is based on the attenuation
of X-ray through a body. There is high correlation
between the density of the body or body
components, and the X-ray attenuation measured.
This relationship is used to estimate the body
composition, volume or weight of a biological
sample. The attenuation of X-rays is visualized by
reconstruction of 360
o
rotation of X-ray tube in a CT
tomogram or CT image. Image data from Computer
Tomography can be orientated in different ways.
Single slice tomograms can be handled like 2-way
(rows*columns) data arrays. Stacks of tomograms
from 3D samples are often orientated as multi-way
data arrays (rows*columns*stack). Combining CT
data with other types of data, like MRI etc., can also
yield multi-modal data arrays which can be handled
either in a multi-dimensional fashion or be unfolded
prior to analysis. Unfolding of multi-way data may
sometimes lead to poor estimation and
interpretability of variation between the different
stacks or batches in a multi-way data array.
There are two primary ways to perform
classification. Supervised classification (1), where
classes are known in advance (a priori), and un-
supervised classification (2), where classes are not
known in advance. For classification of soft tissues
from CT images of lamb carcasses, it can be difficult
to obtain solid a priori knowledge or reference data
of classes. Traditionally, reference data has been
collected by using destructive dissection. This
procedure is both expensive and not very accurate
due to differences operators / butchers (Nissen et al.
2006). The accuracy of classification of tissues may
be influenced the accuracy of the reference method
used, i.e. for calibration purposes or detection of
false negatives or positives. By using non-supervised
classification, validation techniques can ensure that
the model works for new data and finds the optimal
or true number of classes in the model. The non-
supervised approach will not be affected by
reference or a priori error.
Parallel Factor Analysis (PARAFAC) is one
method designed to analyze and decompose multi-
way data, and was introduced by Harshman in 1971
for Psychometrics. The PARAFAC method can be
used as a non-supervised classification tool to
classify soft tissues in CT image stacks sampled
from whole lamb carcasses.
The main purpose of this study is to apply
PARAFAC decomposition of multi-way CT image
data array as a classification tool of different lamb
carcass soft tissues.
2 MATERIALS & METHODS
2.1 Sampled Animals
120 lambs from a single Norwegian abattoir were
sampled according to an experimental design from
242
Kongsro J. (2008).
PARAFAC CLASSIFICATION OF LAMB CARCASS SOFT TISSUES IN COMPUTER TOMOGRAPHY (CT) IMAGE STACKS.
In Proceedings of the First International Conference on Bio-inspired Systems and Signal Processing, pages 242-248
DOI: 10.5220/0001062202420248
Copyright
c
SciTePress

August to September in 2005. The design was set up
to cover the variation in all levels of fatness in the
carcasses, and the principle of over-sampling at the
extremes was applied (Engel et al. 2003): The
carcasses were sampled in three groups; low,
intermediate and high level of fatness. Selection was
made using fatness score from the EUROP carcass
grading system for lamb in Norway. Low fatness
equals –2 standard deviations (st.dev.) and below
mean value. High fatness + 2 std and above mean
value (Kirton et al., 1995). Intermediate between
high and low (table 1). 40% of the samples were
selected for each of the groups low and high fatness
and 20% selected for intermediate fatness (Tab. 1),
yielding a 40-20-40 grouping of the designed
samples. In addition, two subsets of equal size (50-
50) were constructed for validation by split-half-
analysis.
Table 1: Sampling and experimental design.
n = 120 Low Mid High
% n % n % n
Design
1
40 48 20 24 40 48
Subset 1
2
38 23 20 12 42 25
Subset 2
2
38 23 20 12 42 25
1 40-20-40 design for sampling
2 Data subsets for Split-half analysis
2.2 Computer Tomography
2.2.1 Settings
The lambs were scanned at the Norwegian
University of Life Sciences using a Siemens
Somaton Emotion CT Scanner. Two persons were
involved in the scanning of lamb carcasses: one
operation the scanner, and the other preparing and
entering the carcasses into the machine. The
capacity of this procedure was approximately 8-10
carcasses per hour. The protocol for CT scanning is
described in Table 2.
Table 2: CT protocol used for scanning of lamb carcasses.
Topogram Sequence
100 mA
130 kV
Slice width: 2.0 mm
Length: 1024 mm
Tube position: AP
Direction: Caudiocranial
Kernel: T80s (sharp)
Window: 256-64
170 mAs
130 kV
Scan time: 0.8s
Slice width: 3 mm
Number of scans: 15
Direction: Caudiocranial
Kernel: B50M
Window: 100-50
Field of view (FOV): 400
2.2.2 Anatomical Sites
Figure 1: Scanning sites CT, lamb carcass.
Fifteen (15) anatomical scanning sites spanning the
entire carcass were selected from a topogram using
spine vertebras as fixing points (Fig. 1). A topogram
is a survey picture produced by the CT-scanner. It is
obtained by fixation of the X-ray tube in the upper
position and moving the object at constant speed
through the gantry. Each colour of the lines
represents anatomical sections of the carcass
(cervical, thoracic, lumbar, sacral and caudal). The
anatomical sites were selected to span the entire
variation of the carcass, but the number of images
was limited due to capacity. High X-ray dose (170
mAs) was selected to increase the resolution of the
tomograms. The anatomical sites collected from the
mid-section of the carcass, were selected using
literature reference sites for grading of lamb
carcasses (Berg et al. 1997;Bruwer et al.
1987;Chandraratne et al. 2006;Chandraratne,
Kulasiri, & Samarasinghe 2007;Cunha et al.
2004;Jones et al. 1992;Kirton et al. 1995). In
addition to literature reference sites, additional sites
on the leg and shoulder were added using spine
vertebras as fixing points.
2.2.3 Import and Pre-processing of Images
The CT scanner generated images in DICOM
format, which is a common medical image format.
The images were imported into MATLAB using the
Image Processing Toolbox routine dicomread.
Figure 2: Raw image, Binary image for arithmetic
extraction and processed image after extraction.
In the raw CT images, the couch material (non-
carcass component) was visible (Fig. 2). This was
removed using arithmetic extraction in MATLAB.
The extraction was performed using image array
PARAFAC CLASSIFICATION OF LAMB CARCASS SOFT TISSUES IN COMPUTER TOMOGRAPHY (CT) IMAGE
STACKS
243

multiplication, subtracting the couch material from
the raw image using a binary image (Fig. 2)
containing zeros and ones to remove the couch area
(zeros) of the image. The lamb area of the image
was now extracted and ready for further analysis
(fig.3).
2.2.4 Frequency Distribution of Pixel Values
(HU)
Figure 3: 15 pre-processed CT images from all scanning
sites, from neck (1) to knee joint of leg (15).
A frequency distribution of the signal intensities
(pixels) was generated for each anatomical site
(Dobrowolski et al., 2004) (Fig. 5) from each of the
pre-processed images (Fig. 5) using the frequency of
Hounsfield Units [HU] in the interval [-200,200].
HU is related to density of biological tissues, where
0 is regarded as the HU of pure water. The interval
of 400 HU is expected to cover the soft animal
tissues (fat and muscle) in the CT images
(Dobrowolski et al. 2004;Romvari et al. 2002). Each
image was represented as a frequency distribution 2-
way array [1 x 400]. For each sample, 15 images
were generated, generating a 3-way array [1 x 400 x
15], giving a [120 x 400 x 15] data array for the
entire samples.
2.3 PARAFAC
PARAFAC is a generalization of Principal
Component Analysis (PCA) to higher order arrays
(Bro 1997). Decomposition of the data array is made
into triads or trilinear components (Fig. 4), but
instead of one score vector and one loading vector as
in bilinear PCA, each component consist of one
score vector and two loading vectors (trilinear).
PARAFAC is regarded as a “strong” multi-way
method utilizing the multi-mode structure for
modelling without unfolding, and providing other
attractive features (Huang et al. 2003).
Figure 4: PARAFAC decomposition of a 3-way data
array.
In this study, PARAFAC is used to decompose
the landscapes of the frequency distributions and
anatomical positions into a number of trilinear
components (f).
∑
=
=
F
f
ijkkfjfifijk
ecbaX
1
The element x
ijk
represent the landscape of
histogram spectra and anatomical positions of the
lamb carcass sample i, frequency distribution j,
anatomical position k. The landscapes are
decomposed into sample scores a
if
, frequency
distribution loadings b
jf
and anatomical position
loadings c
kf
for each factor f or PARAFAC
component f. The residuals e
ijk
, contains variation
not explained by the model.
The PARAFAC components will be estimates of
the CT histogram signals from the individual
chemical components (fat & lean) if the data are
approximately low-rank trilinear and when the
correct number of components is used. If the optimal
case is found, the scores for each of the components
represent the relative content of carcass soft tissues.
The number of components will represent the non-
supervised classes of soft tissues suggested by the
validated PARAFAC model.
PARAFAC models of CT image histogram
landscapes were estimated with 1 to 4 components.
The models were mean-centred since this has proven
to yield the best result and interpretation of the
figures. Since each component is expected to
represent a single peaked frequency distribution (one
local maxima) of a soft tissue, unimodality
constraints was imposed on the model (Johansen et
al. 2006). When calibrating PARAFAC models of
CT images against a dissection reference,
unimodality constraints seemed to yield the most
accurate results (Johansen, Egelandsdal, Røe, Kvaal,
& Aastveit 2006).
BIOSIGNALS 2008 - International Conference on Bio-inspired Systems and Signal Processing
244

The correct number of components was selected
using core consistency and split-half analysis (Bro
1997) as validation tools. The split was done using a
50-50 split of the actual designed (table 1) samples,
estimating independent PARAFAC models for both
split data subsets. Due to the uniqueness of the
PARAFAC model, the same loadings will be
obtained from different samples if the samples
reflect the same CT histogram variables, when the
correct number of components is chosen and enough
data are available in each of the split data subsets
(Andersen & Bro 2003).
All the models were constructed using the
PLS_Toolbox 4.0, August 10, 2006, Copyright
Eigenvector Research, Inc. 1995-2006 for
MATLAB, the Image Processing Toolbox V5.3
(R2006b) for MATLAB and MATLAB 7.3.0.267
(R2006b), August 03, 2006 © 1984-2006The
MathWorks Inc.
3 RESULTS & DISCUSSION
3.1 Landscapes
Figure 5: 3D CT histogram landscape of one sample, raw
data.
From the landscape, a distinctive frequency
distribution that appears between different
anatomical sites is revealed (Fig. 5). There are two
peaks identified as the shoulder site and the leg
anatomical site. These sites are the “muscular” parts
of the carcass (leg muscles), and therefore provide a
high response or histogram intensity (number of
pixels). There are two ridges in the landscapes, one
larger than the other. The large ridge is identified as
the lean tissue, and the small ridge as the fat tissue
part. When comparing very fat animals with very
lean animals, the fat ridge is almost absent in the
very lean animals. These observations will be further
investigated in the PARAFAC analysis.
3.2 PARAFAC
Table 3: PARAFAC diagnostics. Full model (n=120). # of
components, explained variance, core consistency, number
of iterations and computation time (s).
# Expl. Var.
(%)
Core cons. # iter Time
(s)
1 50.06 100 5 6
2 66.36 95 11 12
3 73.18 78 18 20
4 76.80 0 23 28
The results from the PARAFAC models are shown
in Table 3. Three components seem to yield a
consistent model, with relatively low number of
iterations and computation time.
In order to validate the appropriate number of
components in the model, the results from the split-
half analysis is shown in Figure 6. The figure shows
the frequency distribution loadings for 1 to 4
components, were subset 1 has solid lines, and
subset two dashed lines. Due to the uniqueness of
the PARAFAC model, the same loadings should be
obtained if the samples reflect the same CT
histogram and anatomical site pattern when the
optimal number of components is chosen. The solid
and dashed lines seem to be correlated for the 1-, 2-
and 3-component models, while for the 4-component
model; the solid and dashed lines do not correlate.
Thus, the model seems to be valid with 3
components.
The PARAFAC decomposition of the CT
histogram landscapes is shown in Figure 7, were the
raw landscape in Figure 5 is decomposed into three
PARAFAC components. The 1st component seems
to model the fat tissue in the frequency distribution,
and the 2nd seem to model muscle tissue. The 3
rd
component seems to model very lean muscle tissue.
Component 2 and 3 seem to be two types of muscle
tissue, “marbled” or muscle tissue with higher fat
content (# 2) and lean muscle tissue (# 3)
For practical purposes, the PARAFAC models
yields a better understanding of the uniqueness and
nature of the CT value (HU) frequency distribution.
From the images in Figure 8, the PARAFAC
loadings were applied to a test image from a lamb
carcass belly. Loadings above a manually set
baseline (0.02) were selected to ease interpretation
of the test images. The first image from left
represents the total area of soft tissue. The 2
nd
image
PARAFAC CLASSIFICATION OF LAMB CARCASS SOFT TISSUES IN COMPUTER TOMOGRAPHY (CT) IMAGE
STACKS
245

Figure 6: Validation of PARAFAC components (split-half analysis). 1 to 4 component PARAFAC models. 1 –and 2 –
component model (top), 3- and 4- component model (bottom).
Figure 7: PARAFAC decomposition of a 3D CT frequency distribution. 3 components or classes identified. # 1 represent fat
tissue, # 2 muscle tissue with marbling fat and the 3
rd
lean muscle tissue.
5
10
15
-100
0
100
Anatomy
Comp.1
HU
5
10
15
-100
0
100
Anatomy
Comp.2
HU
5
10
15
-100
0
100
Anatomy
Comp.3
HU
BIOSIGNALS 2008 - International Conference on Bio-inspired Systems and Signal Processing
246
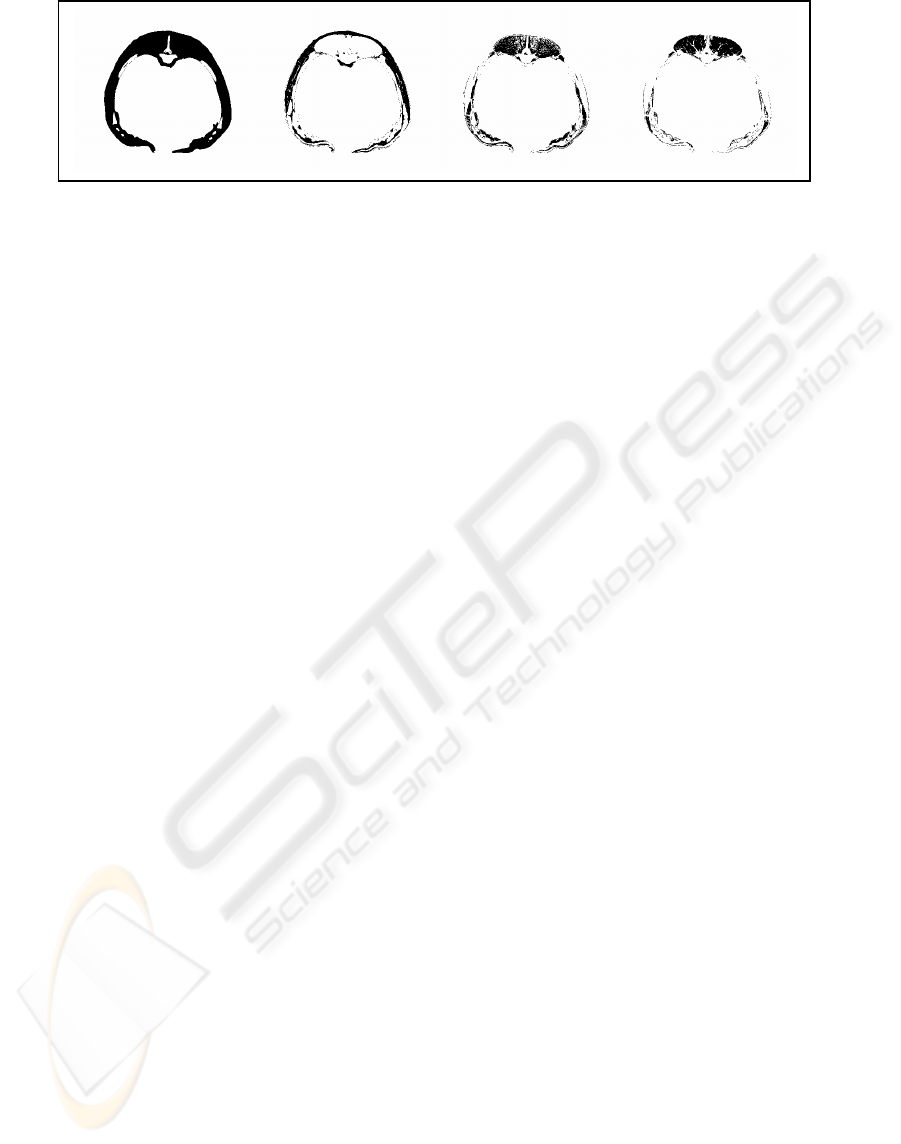
Figure 8; PARAFAC CT value frequency loadings > 0.02 applied on CT image from belly. From left, soft tissue HU= [-
200, 200], # 1, # 2, # 3.
represents component 1, the 3
rd
component 2 and 4
th
(right) component 3. When inspecting the images
visually, # 1 represent fat tissue, # 2 muscle tissue
with marbling fat and the 3
rd
lean muscle tissue.
PARAFAC yields a consistent decomposition of the
3D frequency distribution of the CT images, and
selected 3 unique soft tissue components
representing fat, and two types of muscle tissue.
4 CONCLUSIONS
This paper presents modelling and decomposition of
multi-way array CT image data, using PARAFAC as
a non-supervised classification tool for different
lamb carcass soft tissues. Multi-way modelling
applying PARAFAC did yield sensible interpretation
of the 3D CT value frequency distribution. Three
components or classes of soft tissues were extracted
from the model; fat, marbled and lean muscle.
ACKNOWLEDGEMENTS
This study was sponsored by grant 162188 of the
Research Council of Norway, as part of a Ph.D.
study program. I am grateful for the assistance from
Prof. Rasmus Bro at the Faculty of Life Sciences,
University of Copenhagen.
REFERENCES
Andersen, C. M. & Bro, R. 2003, "Practical aspects of
PARAFAC modeling of fluorescence excitation-
emission data", Journal of Chemometrics, vol. 17, no.
4, pp. 200-215.
Berg, E. P., Neary, M. K., Forrest, J. C., Thomas, D. L., &
Kauffman, R. G. 1997, "Evaluation of Electronic
Technology to Assess Lamb Carcass Composition",
Journal of Animal Science, vol. 75, no. 1997, pp.
2433-2444.
Bro, R. 1997, "PARAFAC. Tutorial and applications",
Chemometrics and Intelligent Laboratory Systems,
vol. 38, no. 2, pp. 149-171.
Bruwer, G. G., Naude, R. T., Dutoit, M. M., Cloete, A., &
Vosloo, W. A. 1987, "An Evaluation of the Lamb and
Mutton Carcass Grading System in the Republic of
South-Africa .2. the Use of Fat Measurements As
Predictors of Carcass Composition", South African
Journal of Animal Science-Suid-Afrikaanse Tydskrif
Vir Veekunde, vol. 17, no. 2, pp. 85-89.
Chandraratne, M. R., Kulasiri, D., Frampton, C.,
Samarasinghe, S., & Bickerstaffe, R. 2006, "Prediction
of lamb carcass grades using features extracted from
lamb chop images", Journal of Food Engineering, vol.
74, no. 1, pp. 116-124.
Chandraratne, M. R., Kulasiri, D., & Samarasinghe, S.
2007, "Classification of lamb carcass using machine
vision: Comparison of statistical and neural network
analyses", Journal of Food Engineering, vol. 82, no. 1,
pp. 26-34.
Cunha, B. C. N., Belk, K. E., Scanga, J. A., LeValley, S.
B., Tatum, J. D., & Smith, G. C. 2004, "Development
and validation of equations utilizing lamb vision
system output to predict lamb carcass fabrication
yields", Journal of Animal Science, vol. 82, no. 7, pp.
2069-2076.
Dobrowolski, A., Romvári, R., Allen, P., Branscheid, W.,
& Horn, P. 2004, "Schlachtkörperwertbestimmung
beim Schwein", Fleischwirtschaft, vol. 3, no. 2004,
pp. 109-112.
Engel, B., Buist, W. G., Walstra, P., Olsen, E. V., &
Daumas, G. 2003, "Accuracy of prediction of
percentage lean meat and authorization of carcass
measurement instruments: adverse effects of incorrect
sampling of carcasses in pig classification", Animal
Science, vol. 76, no. 2003, pp. 199-209.
Huang, J., Wium, H., Qvist, K. B., & Esbensen, K. H.
2003, "Multi-way methods in image analysis-
relationships and applications", Chemometrics and
Intelligent Laboratory Systems, vol. 66, no. 2, pp. 141-158.
Johansen, J., Egelandsdal, B., Røe, M., Kvaal, K., &
Aastveit, A. H. 2006, "Comparison of different
calibration models for prediction of lamb carcass
composition using Computerized Tomography (CT)
imaging", Chemometrics and Intelligent Laboratory
Systems, vol. Submitted.
Jones, S. D. M., Jeremiah, L. E., Tong, A. K. W.,
Robertson, W. M., & Gibson, L. L. 1992, "Estimation
PARAFAC CLASSIFICATION OF LAMB CARCASS SOFT TISSUES IN COMPUTER TOMOGRAPHY (CT) IMAGE
STACKS
247

of Lamb Carcass Composition Using An Electronic
Probe, A Visual Scoring System and Carcass
Measurements", Canadian Journal of Animal Science,
vol. 72, no. 2, pp. 237-244.
Kirton, A. H., Mercer, G. J. K., Duganzich, D. M., &
Uljee, A. E. 1995, "Use of Electronic Probes for
Classifying Lamb Carcasses", Meat Science, vol. 39,
no. 2, pp. 167-176.
Nissen, P. M., Busk, H., Oksama, M., Seynaeve, M.,
Gispert, M., Walstra, P., Hansson, I., & Olsen, E.
2006, "The estimated accuracy of the EU reference
dissection method for pig carcass classification", Meat
Science, vol. 73, no. 1, pp. 22-28.
Romvari, R., Hancz, C., Petrasi, Z., Molnar, T., & Horn,
P. 2002, "Non-invasive measurement of fillet
composition of four freshwater fish species by
computer tomography", Aquaculture International,
vol. 10, no. 3, pp. 231-240.
BIOSIGNALS 2008 - International Conference on Bio-inspired Systems and Signal Processing
248
