
AUTOMATIC DETECTION OF ATRIAL FIBRILLATION
AND FLUTTER
A Tachogram-based Algorithm for Mobile Devices
Stefanie Kaiser, Malte Kirst and Christophe Kunze
FZI Research Center for Information Technology, Haid-und-Neu-Str. 10-14, Karlsruhe, Germany
Keywords:
Atrial fibrillation, Atrial flutter, Automatic detection, Mobile device, Tachogram, Morphological filters.
Abstract:
Two versions of a new detector for automatic real-time detection of atrial fibrillation and atrial flutter in non-
invasive ECG signals are introduced. The methods are based on beat to beat variability, tachogram analysis
and simple signal filtering. The implementation on mobile devices is made possible due to the low demand on
computing power of the employed analysis procedures. The proposed algorithms correctly identified 436 of
440 five minute episodes of atrial fibrillation or flutter and also correctly identified up to 302 of 342 episodes
of no atrial fibrillation, including normal sinus rhythm as well as other cardiac arrhythmias. These numbers
correspond to a sensitivity of 99.1 % and a specificity of 88.3 %.
1 INTRODUCTION
Atrial fibrillation (AF) is a widely spread disease and
the most frequently diagnosed cardiac arrhythmia in
western countries. Approximately 1–5 % of the popu-
lation in such countries suffer from atrial fibrillation,
with increasing percentages at higher patients’ ages,
reaching an incedence of almost 12 % in male patients
at ages over 85. Due to the rising average age in the
industrial nations and to the ascending commonness
of other established risk-factors, such as hipertension
or overweight, experts expect a doubling of the inci-
dences during the next 50 years.
Whereas atrial fibrillation at itself is not a life-
threatening disease, it entails dangerous secondary
complications, such as embolisms and apoplectic
strokes. Approximately 15 % of all strokes are caused
by atrial fibrillation.
The timely diagnosis of AF proves to be compli-
cated due to several reasons. First, atrial fibrillation
implicates scant perceivable symptoms and is mostly
not noticed by the patients themselves. Second, in
early stages the disease occurs in irregular episodes
with unpredictable times of appearance and durations.
On the other hand, physicians have ever fewer time
spendable on each patient, making it impossible to an-
alyze long-term ECG manually.
Therefore, automatic detection of atrial fibrilla-
tion in electrocardiograms is and will be increasingly
important and necessary during the next decades.
(Heeringa et al., 2006; Ringborg et al., 2008; Hohn-
loser et al., 2005)
2 ATRIAL FIBRILLATION
AND FLUTTER
The healthy heart beats at a regular rhythm with ap-
proximately 60–80 bpm (normal sinus rhythm, NSR),
where the electrical excitation for each beat starts
at the sinus node and subsequently spreads over the
atrium and ventricles.
In contrast, during atrial fibrillation, the vestibules
are stimulated at a frequency of 350–600 activations
per minute, causing a quasi constantly circulating ex-
citation. This condition provokes a dysfunction of the
blood pumping activity in the atrium, creating the risk
of blood accumulation and therefore the risk of em-
bolisms. Also, the constant stimulation of the atrium
does not allow the organized and periodic conduc-
tion of the activation toward the chambers. Rather,
the points in time of the simulation propagation to-
ward the chambers and the so induced heartbeats are
random and the time intervals between two heart-
beats (RR interval) become absolutely irregular and
chaotic.
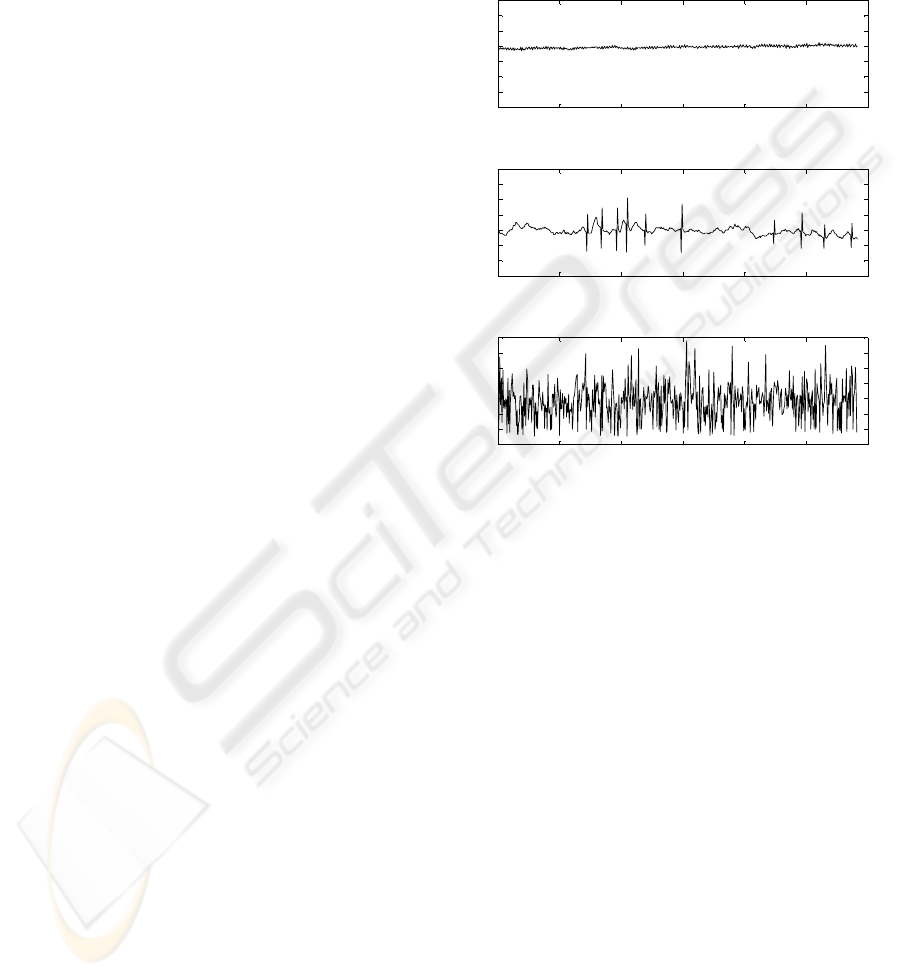
In the ECG, atrial fibrillation is perceptible by
131
Kaiser S., Kirst M. and Kunze C. (2009).
AUTOMATIC DETECTION OF ATRIAL FIBRILLATION AND FLUTTER - A Tachogram-based Algorithm for Mobile Devices.
In Proceedings of the International Conference on Bio-inspired Systems and Signal Processing, pages 131-138
DOI: 10.5220/0001547201310138
Copyright
c
SciTePress

high disparity of the length of the RR intervals –
meaning very irregular heartbeat, the absence of the
p-wave and through a constant baseline fibrillation,
caused by the constant activation of the atrium. Fi-
gure 1 shows the ECG of atrial fibrillation compared
to the ECG of normal sinus rhythm.
(a) Normal Sinus Rhythm (NSR)
(b) Atrial Fibrillation (AF)
Figure 1: Comparison of the morphology of an ECG signal
for normal sinus rhythm and atrial fibrillation.
Atrial flutter (AFLUT) appears as a disease that
shows big similarities to atrial fibrillation, and also
only differs in minor aspects concerning the morphol-
ogy of the ECG and specially concerning the heart
beat rhythm. Due to this reason, in the following
atrial fibrillation (AF) refers both, atrial fibrillation
and atrial flutter.
3 STATE OF THE ART
Automatic detection of atrial fibrillation has been base
of research during many years and several methods
have been developed in this field, predicated on dif-
ferent approaches. A distinction into methods appro-
priate for the detection on invasive ECG signals and
those for detection on non-invasive signals has to be
made, due to the discrepancy of the signal quality, the
signal-to-noise ratio (SNR) and the information con-
tained in the signal.
The two general main approaches investigated for
the detection of AF are the analysis of the signal-
baseline between heart-beats, including baseline vari-
ation, zero-crossing and detection of p-waves (Kim
et al., 1995), and the analysis of the rhythm, including
variance of RR intervals (Logan and Healey, 2005),
density allocation of RR intervals (Tateno and Glass,
2000), analysis with artificial neural networks (Artis
et al., 1991) and analysis in frequency domain (Sadek
and Ropella, 1995), between others. In the field of
mobile devices, detection on non-invasive signals is
required and in this range the approach of rhythm
analysis is the most commonly used.
Furthermore mobile devices demand low comput-
ing power costs. Therefore complicated and computa-
tionally intensive procedures, such as artificial neural
networks or transformation into the frequency domain
should be avoided.
In addition, the length of the analyzed datasets
has to be taken into consideration at the examination
of different algorithms, since the longer the contem-
plated dataset, the easier will the detection of a certain
signal pattern be. However, short analysis sections are
preferable for prompt detection.
4 DATASETS
In the run-up to this work an adequate, statistically
relevant test-database was generated, based on the fol-
lowing four source-databases.
• Physionet MIT-BIH Atrial Fibrillation Database
(Goldberger et al., e 13)
• American Heart Association (AHA) Database
(Goldberger et al., e 13)
• ECG signals recorded at the Institute for Signal
Processing Technology (ITIV), Universit
¨
at Karls-
ruhe (TH), Germany
• ECG signals recorded at the University Hospital
T
¨
ubingen (UKT), Germany
These databases include recordings with normal sinus
rhythm, atrial fibrillation and flutter as well as other
cardiac arrhythmias, such as unifocal and multifocal
premature ventricular contraction (PVC), bigeminy,
trigeminy and quadrageminy, couplets, triplets and
tachycardia.
The created test database consists of 782 five-
minute datasets with representative ECG rhythms,
that were classified into ”atrial fibrillation datasets”
(AF) and ”no atrial fibrillation datasets” (NOAF).
Finally, out of the 782 records 440 were catego-
rized as AF and 342 as NOAF. Out of the latter 142
show normal sinus rhythm or isolated PVC (NSR) and
200 show other strong arrhythmias (OAR).
Table 1: Overview of the final test database.
ECG-Type # Datasets Total Length
AF 440 2200 min
NSR 142 710 min
OAR 200 1000 min
All 782 3910 min
The length of five minutes was chosen as a com-
promise between easier detection on longer entities
and prompt detection on shorter episodes. The final
decision over the contemplation period for detection
was taken in collaboration with physicians of the Uni-
versity Hospital T”ubingen.
BIOSIGNALS 2009 - International Conference on Bio-inspired Systems and Signal Processing
132
In order to obtain a standardized database, as well
as to ensure the possibility of saving other additional
records, such as tachograms, results, etc. along with
the original ECG signals, all 782 ECG records were
converted into the Unisens format (Kirst et al., 2008;
Kirst and Ottenbacher, 2008).
5 ALGORITHM
According to the requirements of a mobile device, an
algorithm was developed that reliably detects atrial
fibrillation on non-invasive ECG signals under adher-
ence of low processing power costs. The proposed
method rests upon the analysis of the rhythm of the
heart beats and is more precisely based on the analy-
sis of the RR interval tachogram.
Furthermore the developed algorithm divides into
two separated detection methods, the PPV-Detector
and the PPV-MF-Detector. Whereas the further con-
stitutes the basic detection algorithm, the PPV-MF-
Detector answers an extension, achieving an improve-
ment of the detection quality.
5.1 ECG Premachining
The tachogram-based analysis requires a premachin-
ing of the ECG signal, consisting of the QRS detec-
tion and the computing of the actual tachogram.
5.1.1 QRS Detection
QRS detection creates a list containing the points in
time of the heartbeats. Numerous QRS-detection-
algorithms have been published. In this work the
Open Source ECG Analysis algorithm has been used
for the QRS detection (Hamilton, 2002).
5.1.2 Tachogram Generation
The tachogram is a heart rate variability signal (HRV),
that considers not only normal heart beats but also
PVC and that measures the beat-to-beat variations in
the heart rate. It shows the RR interval duration be-
tween the actual and the previous beat over the time
of the actual beat.
RR
i
= t(R
i
) − t(R
i−1
) (1)
This means, for each heartbeat the time interval to
the previous heartbeat is calculated. The result cor-
responds to the value of the tachogram at the point in
time of the contemplated heartbeat.
The tachogram then provides information about
the ECG rhythm and its regularity. A regular heart-
beat, such as appears in NSR, will generate a flat
tachogram with an almost constant value. Arrhyth-
mias in the ECG will lead to amplitude varieties and a
heart beat as irregular as it occurs during AF will lead
to a tachogram with an appearance similar to white
noise. Figure 2 shows the tachograms of a NSR-ECG,
a NSR-ECG with PVC and an AF-ECG.
RR-Interval-Duration
Seconds
100500 150 200 250 300
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
(a) Normal Sinus Rhythm (NSR).
RR-Interval-Duration
Seconds
100500 150 200 250 300
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
(b) Normal Sinus Rhythm with PVC.
RR-Interval-Duration
Seconds
100500 150 200 250 300
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
(c) Atrial Fibrillation (AF).
Figure 2: Comparison of the tachograms of different ECG
signals.
5.2 PPV-Detector
The PPV-Detector comprehends the basic detection
method of the proposed algorithm and represents
a fully functional AF detection algorithm by itself.
PPV-Detector stands for ”Peak-to-Peak and Variance
Analysis Detektor”.
Methodically, the PPV-Detector divides the 300
seconds long datasets into 30 equally long 10 second
segments. Each segment is further on analyzed sepa-
rately and individually.
5.2.1 Peak-to-Peak and Variance Reckoning
The peak-to-peak value PP defines the maximum dif-
ference between any two RR intervals in one segment,
and equals the difference between the maximum and
the minimum amplitude inside the examined segment.
It is therefore calculated as
PP(s
n
) = max(RR(s
n
)) − min(RR(s
n
)) n = 1, .., 30,
(2)
AUTOMATIC DETECTION OF ATRIAL FIBRILLATION AND FLUTTER - A Tachogram-based Algorithm for Mobile
Devices
133
where s
n
corresponds to the segment n and RR(s
n
) to
the set of RR interval durations of segment s
n
.
The variance of the set of RR interval durations of
each segment s
n
is calculated by
var(s
n
) =
∑
I
i=1
(RR
i
− mean(s
n
))
2
I − 1
, (3)
mean(s
n
) =
∑
I
i=1
RR
i
i
, (4)
where I corresponds to the amount of RR interval val-
ues RR
i
in each segment s
n
.
5.2.2 PPV-Detector Decision Tree
The datasets are classified as AF and NOAF by us-
ing a decision tree based on threshold comparisons.
This decision tree can be devided into two separate
parts, where the first analyzes the individual seg-
ments, whereas the second classifies the entire dataset
into either AF or NOAF.
In a first step the classification for every single
segment is made. Thereby, for each segment s
n
the
PP(s
n
) is calculated. Each segment with a PP higher
than 0.2 is classified as AF-typical whereas those with
a PP smaller than this are classified as not-AF-typical.
A decision for not-AF-typical leads to the increment
of a counter in order to keep track of the number of
not-AF-typical segments in the dataset. In addition,
only in this case the variance Var of the concerned
segment is calculated and saved in a buffer.
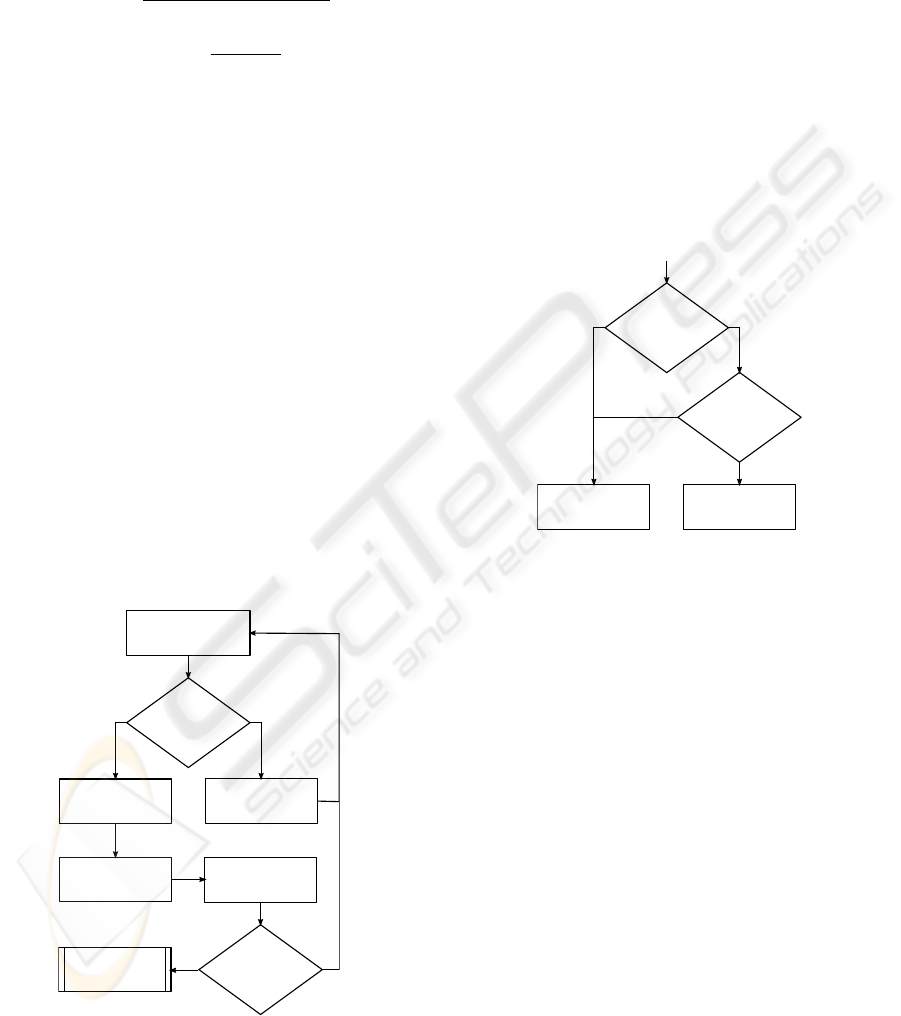
Figure 3 shows this first part of the flow chart for
the PPV-Detector decisions.
n = n+1
analyze segment n
PP(sn) < 0.2?
sn
AF-typical
counter
Not_AF ++
calculate
var(sn)
n = 30?
sn
Not-AF-typical
yes no
yes no
n=n+1
analyse segment n
PP(sn) < 0.2?
sn
AF-typical
Counter
Not_AF ++
calculate
Var(sn)
n =30?
sn
Not-AF-typical
yes
no
yes
no
final dataset
classification
Figure 3: Flowchart for the segment classification.
Once every segment in the dataset has been treated
according to the explained method, a final diagnosis
of the dataset is reached by the second part of the de-
tection algorithm as follows.
If the number of not-AF-typical segments exceeds
10, the dataset is immediately classified as NOAF. If
this is not the case, the buffered variances Var(s
n
) are
taken in consideration and are compared to another
threshold. If more than four of the buffered variances
out of the dataset segments are smaller than 0.00075,
the dataset will again be classified as NOAF.
Only if the number of not-AF-typical segments
is smaller or equal to ten and less than four of the
buffered variance are smaller than the set threshold,
the dataset will be diagnosed as AF.
Figure 4 shows the second part of the flow chart
for the PPV-Detector, in which the final diagnosis de-
cision for the dataset is made.
Counter Not_AF
> 10?
Amount seg.
(Var<0.00075)
>4
NOAF
AF
number of
(var<0.00075)
> 4?
counter Not_AF
> 10?
AF
NOAF
Amount seg.
(var<0.00075)
>4
Counter Not_AF
> 10?
AF
NOAF
Start
End
yes no
yes
no
Figure 4: Flowchart for the final dataset classification.
5.3 PPV-MF-Detector
Whereas the PPV-Detector algorithm consists of a
very simple AF analysis focused on the reckoning
of peak-to-peak values and variances, the PPV-MF-
Detector understands a further analysis, that is based
on the PPV-Detector, but includes a second analysis
helped by morphological filters (MF).
The fundamentals of this method lie in the fact,
that strong structural differences, that do not show
in the analysis of peak-to-peak values and variances,
can be found between tachograms of atrial-fibrillation
ECG signals and those obtained from other strong ar-
rhythmias, such as bigeminal premature ventricular
contractions (bigeminy), trigeminal premature ven-
tricular contractions (trigeminy), quadrageminal pre-
mature ventricular contractions (quadrageminy) or se-
ries of couplets and series of triplets. These structural
differences consist of the existence of repeating mor-
phologies or structures in tachograms of such other
arrhythmias.
Figure 5 clearly shows these differences in
the tachograms of atrial fibrillation compared to
BIOSIGNALS 2009 - International Conference on Bio-inspired Systems and Signal Processing
134
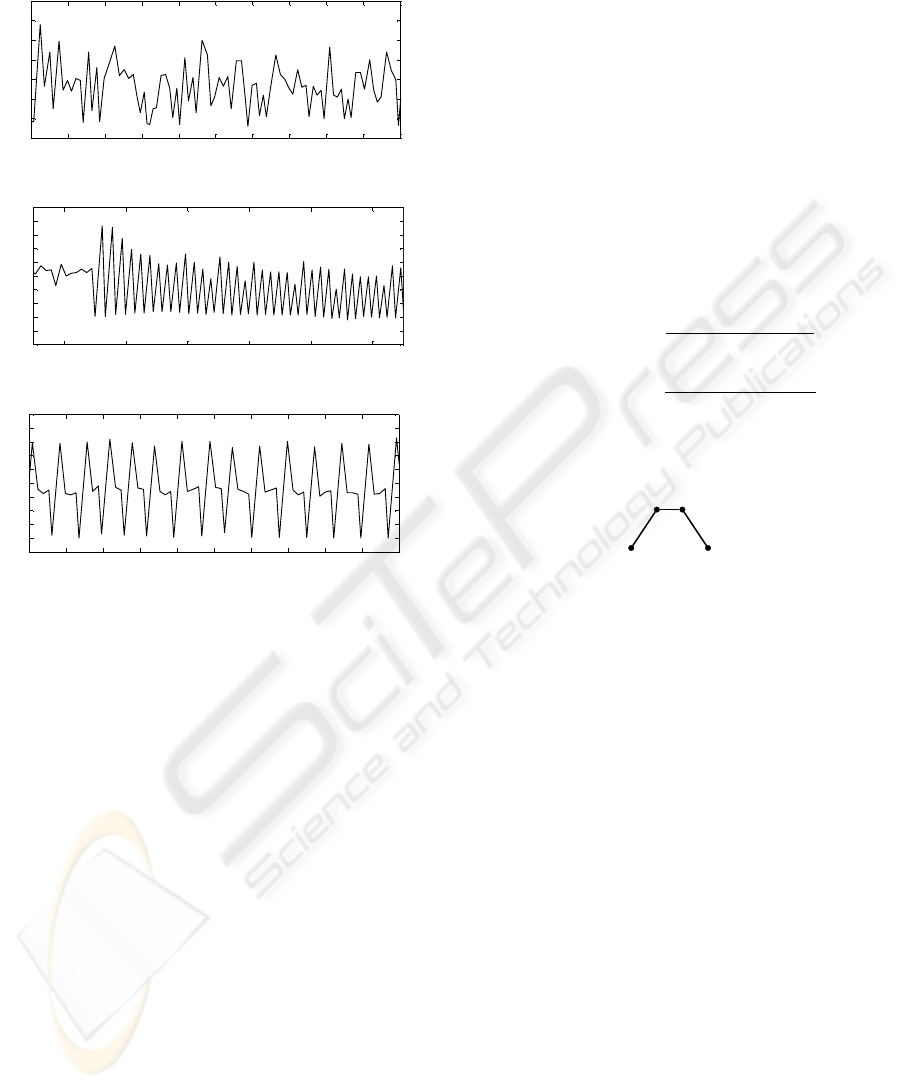
bigeminy and quadrageminy.
0 5 10 15 20 25 30 35 40 45 50
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
Seconds
RR-Interval-Duration
(a) Atrial Fibrillation (AF)
90 100 110 120 130 140
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
1.1
1.2
1.3
Seconds
RR-Interval-Duration
(b) Bigeminal PVC
50 55 60 65 70 75 80 85 90 95 100
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
1.1
1.2
1.3
Seconds
RR-Interval-Duration
(c) Quadrageminal PVC
Figure 5: Comparison of the tachograms of different arhyth-
mias.
The idea of the PPV-MF-Detector is to use mor-
phological filters in order to suppress these repeating
structures in the tachogram. The result of this pro-
cedure is a second, modified tachogram, that is then
again analyzed by a basic PPV-Detector.
5.3.1 Morphological Filtering Process
Morphological filters find their origin in the area of
image processing, where they still today find the most
frequent use. Nevertheless these filters have also
found applications in signal processing, mainly in the
field of noise reduction but also to suppress specific
signal structures.
The basic idea of morphological filtering consists
of the sum or rest of a structuring element with the
signal that is to be filtered, being the two most im-
portant operations Opening and Closing. Morpholog-
ical filtering has been previously applied in the field
of biosignal processing as in (Chu and Delp, 1989).
Since the form and length of the structuring el-
ement of a morphological filter influence the result
of the filtering process in an essential way, an ad-
equate structuring element has to be found for each
tachogram. It has been proved during this work, that
in general a structuring element with a length of four
datapoints and with the morphology of a rectangle-
function is the most adequate choice for this filtering.
The exact values of the 4 points of the structuring ele-
ment are however adapted to each dataset that is to be
analyzed.
In our method, the amplitude of the first and the
fourth point of the structuring element correspond to
the mean value of the 75 lowest points of the original
tachogram, while the amplitude of the second and the
third point of the structuring element correspond the
mean value of the 75 highest points of the original
tachogram.
SE(1) = SE(4) =
∑
75
i=1
min
i
(RR(s
n
))
75
, (5)
SE(2) = SE(3) =
∑
75
i=1
max
i
(RR(s
n
))
75
, (6)
Figure 6 shows the general appearance of form of the
structuring element chosen for the morphological fil-
tering of the tachograms.
Figure 6: Form chosen for the structuring element of the
morphological filter.
5.3.2 Creation of the Alternative Tachogram
The alternative tachogram is created through a two
step procedure.
Previous to the morphological filtering, the mean
value of the original tachogram signal is calculated.
Then, the original tachogram is morphologically
filtered by sequential implementing a closing and an
opening MF operator. For each of these two morpho-
logical operations the same, previously calculated,
structuring element is used.
Figure 7 shows two tachograms, one of
quadrageminy and one of atrial fibrillation in
the uppermost graphs. The output signal of this step
is shown in the second graphs of the same figure.
In a following step, the resulting signal is rested
from the original tachogram, resulting in the ∆-signal.
(Graph 3 in figure 7 for quadrageminy and AF respec-
tively).
Finally, for each signal point at which the ∆-signal
reaches an amplitude higher then 0.3, the value of the
original tachogram is substituted by the mean value
of the tachogram. The result signal of this step consti-
tutes the new, alternative tachogram. This result sig-
nal can be seen in the lowest graph of figure 7. It
AUTOMATIC DETECTION OF ATRIAL FIBRILLATION AND FLUTTER - A Tachogram-based Algorithm for Mobile
Devices
135

0 50 100
0 50 100
0
0.5
1
1.5
0 50 100
0
0.5
1
0 50 100
New Tachogram
Morphologically Filtered Tachogram
Original Tachogram
0.5
1
1.5
0.5
1
1.5
-Signal
0 50 100
0
0.5
1
0 50 100
0
0.5
1
1.5
0 50 100
0
0.5
1
0 50 100
0
0.5
1
Morphologically Filtered Tachogram
Original Tachogram
-Signal
New Tachogram
Quadrageminy Atrial Fibrillation
Figure 7: Output signals of the different steps of the creation of the new tachogram. Quadrageminy on the right side and AF
on the left side.
is clearly observable that the new tachogram for the
quadrigeminy signal shows important disparities with
the original tachogram, appearing now as a very con-
stant signal. On the other hand, the new tachogram of
the AF signal shows relatively very little differences
compared to the original tachogram.
Figure 8 shows the flowchart for the creation of
the alternative tachogram signal.
5.3.3 PPV-MF-Detector Decision Tree
The final MF detection algorithm combines the origi-
nal PPV-Detector with the morphological filtering and
creation of the new, adapted tachogram.
The detector diagnoses the ECG signal in two
steps. In the first step, the original PPV-Detector di-
agnoses the ECG signal. Only if the first diagnosis
is AF, the PPV-MF-Detector continues with the cre-
ation of the alternative tachogram, which is then, once
again, analyzed again by a second, slightly adapted
PPV-Detector.
For the first, initial PPV-Detecor, the standard
PPV-thresholds are to be used. For the second PPV-
Detector, the thresholds have been slightly adapted.
The threshold values for both PPV-Detectors are
listed in table 2. All threshold values have been de-
termined empirically.
Figure 9 shows the decision tree equivalent to the
PPV-MF-Detector.
Tachogram
MF-Closing
MF-Opening
+
_
-Signal
for i = 1 .. length( )
(i) > 0.3?
Tacho_new(i) = mean(RRi)
Tachogram
MF-Closing
MF-Opening
Δ-Signal
Tacho_new(i) = mean(RRi)
for i = 1 .. length(Δ-Signal)
Δ(i) > 0.3?
Σ
+ -
yes
no
Figure 8: Flowchart for the building of the new, adapted
tachogram.
6 RESULTS
An overview of the detection results obtained with the
basic PPV-Detector and for the PPV-MF-Detector is
displayed in table 3. Here, sensitivity (Se) indicates
BIOSIGNALS 2009 - International Conference on Bio-inspired Systems and Signal Processing
136

Table 2: Thresholds for the PPV decisions.
PP(s
n
) # PP Var(s
n
) # Var
basic PPV 0.2 10 0.00075 4
PPV-MF 0.2 10 0.0006 4
PPV-Detector
TH-Set 1
Result = AF?
Build new
tachogram
PPV-Detector
TH-Set 2
Result = AF?
AF
NOAF
PPV with
threshold set 1
result = AF?
build new
tachogram
PPV with
threshold set 2
result = AF?
AF
NOAF
yes no
yes
no
Figure 9: Decision Tree for the final dataset diagnosis for
PPV-MF-Detector.
the portion of AF signals that have been correctly de-
tected as AF, whereas the specificity (Sp) denotes the
percentage of NOAF signals, this is to say NSR sig-
nals and other arrhythmias, that have been correctly
detected as NOAF.
Table 3: Detection qualities for the proposed methods.
Detection Algorithm Sensitivity Specificity
PPV-Detector 99.1 % 80.1 %
PPV-MF-Detector 99.1 % 88.3 %
As table 3 shows, both, the PPV-Detector and the
PPV-MF-Detector reach the same, very high level of
sensitivity. The difference in the qualities of both al-
gorithms rests in the specificity. Here, the PPV-MF-
Detector achieves a notably higher percentage than
the PPV-Detector, at the expense of an increased nec-
essary computing power in relation to the first.
A more precise look at the results for the different
datasets is presented in table 4. It reveals that not only
the sensitivity of the two detector versions is equal,
but also the specificity for normal sinus rhythm sig-
nals is equal and very high for both algorithms. The
results differ only in reference to the specificity for
ECG signals with arrhythmias other than atrial fibril-
lation. The quality increase of the specificity between
the two algorithms in this domain is of 14 %.
Table 4: Detection Qualities for the different ECG signal
conditions.
PPV-Detector
ECG TP FN TN FP Se Sp
All 436 4 274 68 99.1 % 80.1 %
AF 436 4 — — 99.1 % —
NSR — — 137 5 — 96.5 %
OAR — — 137 63 — 68.5 %
PPV-MF-Detector
ECG TP FN TN FP Se Sp
All 436 4 302 40 99.1 % 88.3 %
AF 436 4 — — 99.1 % —
NSR — — 137 5 — 96.5 %
OAR — — 165 35 — 82.5 %
7 CONCLUSIONS
AND DISCUSSION
Within this article, two alternatives of an algorithm
for the detection of atrial fibrillation in ECG signals
have been proposed.
As it can be observed by means of the results
exposed in section 6, the two algorithm reach very
satisfying detection qualities in terms of sensitivity
for atrial fibrillation and specificity of normal sinus
rhythm. On the other hand, the methods differ no-
ticeably in the specificity regarding ECG signals with
strong arrhythmias other than atrial fibrillation. At
the expense of a higher demand on computing power,
the PPV-MF-Detector produces better results than the
PPV-Detector itself.
Both alternatives have been developed focusing on
the intention of detecting episodes of AF in a long
term electrocardiogram, that are to be flagged for the
later revision by a physician and the ultimate diagno-
sis. On the other hand the algorithms have been de-
veloped under the constraints of mobile devices. This
is, in the first place, low processing power. Due to
the characteristics named earlier, each of the two pro-
posed algorithms has different advantages and disad-
vantages, so that the ideal choice depends on the pre-
cise utilization environment.
In summary the two versions of the detector that
have been proposed, provide very high sensitivity be-
ing the algorithms based on very simple basic princi-
ples, such as threshold comparisons and therefore on
very low computing power demands.
AUTOMATIC DETECTION OF ATRIAL FIBRILLATION AND FLUTTER - A Tachogram-based Algorithm for Mobile
Devices
137

8 OUTLOOK
A further improvement of the specificity in the
area of other strong arrhythmias, such as bigeminy,
trigeminy, couplets, etc., may be reached under a
slight increase of computing power demands.
One alternative approach to the detection consists
in the suppression of PVC beats previous to the anal-
ysis with the PPV- and the PPV-MF-Detectors. A new
determination of the thresholds would be necessary in
this case.
Another alternative modification of the proposed
methods would be the analysis of ECG episodes con-
taining a fixed amount of beats instead of a fixed time
period. This would specially simplify the implemen-
tation of the methods on mobile devices due to non-
variable memory allocations.
Further on, the possibility of distinguishing and
diagnosing not only between ”atrial fibrillation” and
”not atrial fibrillation”, but also between the different
other arrhythmias should be taken in consideration.
Another approach in this area could be the intent of
delivering the exact number of PVC beats occurred in
one certain ECG segment.
ACKNOWLEDGEMENTS
This work has been kindly supported by the German
government BMBF project MµGUARD. The authors
thank the University Hospital T
¨
ubingen (UKT) for
their time and support with medical questions and
supply of clinical ECG records. We also want to thank
the Institute for Signal Processing Technology (ITIV),
Universit
¨
at Karlsruhe (TH), Germany for providing
further ECG records.
REFERENCES
Artis, S., Mark, R., and Moody, G. (1991). Detection of
Atrial Fibrillation using Artificial Neural Networks.
IEEE Proceedings on Computers in Cardiology, pages
173–176.
Chu, C.-H. H. and Delp, E. J. (1989). Impulsive noise sup-
pression and background normalization of electrocar-
diogram signals using morphological operators. In
IEEE Transactions on Biomedical Engineering, Vol
36, NO. 2, pages 262–273.
Goldberger, A. L., Amaral, L. A. N., Glass, L., Haus-
dorff, J. M., Ivanov, P. C., Mark, R. G., Mietus,
J. E., Moody, G. B., Peng, C.-K., and Stanley, H. E.
(2000 (June 13)). PhysioBank, PhysioToolkit, and
PhysioNet: Components of a new research resource
for complex physiologic signals. Circulation, 101
(23):e215–e220. Circulation Electronic Pages: http://
circ.ahajournals.org/cgi/content/full/101/23/e215.
Hamilton, P. (2002). Open Source ECG Analysis. IEEE
Computers in Cardiology, pages 101–104.
Heeringa, J., van der Kuip, D., Hofman, A., Kors, J., van
Herpen, G., Stricker, B., Stijnen, T., Lip, G., and Wit-
teman, J. (2006). Prevalence, incidence and lifetime
risk of atrial fibrillation: the Rotterdam study. Eu-
ropace.
Hohnloser, P. D. S., Gr
¨
onefeld, P.-D. D. G., and Israel,
P.-D. D. C. (2005). Prophylaxe und Therapie von
Vorhofflimmern. UNI-MED Verlag, Bremen, London,
Boston, 1. edition edition.
Kim, J., Bocek, J., White, H., Crone, B., Alferness, C., and
Adams, J. (1995). An atrial fibrillation detection al-
gorithm for an implantable atrial defibrillator. pages
169–172.
Kirst, M. and Ottenbacher, J. (2008). Unisens.
http://www.unisens.org.
Kirst, M., Ottenbacher, J., and Nedkov, R. (2008).
UNISENS – Ein universelles Datenformat f
¨
ur Multi-
sensordaten. In Biosignalverarbeitung : Innovationen
bei der Erfassung und Analyse bioelektrischer und
biomagnetischer Signale, pages 106–108.
Logan, B. and Healey, J. (2005). Robust Detection of Arial
Fibrillation for a a Long Term Telemonitoring System.
IEEE Computers for Cardiology, pages 391–394.
Ringborg, A., Nieuwlaat, R., Lindgren, P., Jnsson, B., Fi-
dan, D., Maggioni, A., Lopez-Sendon, J., Stepinska,
J., Cokkinos, D., and Crijns, H. (2008). Costs of atrial
fibrillation in five European countries: results from
the Euro Heart Survey on atrial fibrillation. Europace,
10:403–411.
Sadek, L. E. and Ropella, K. M. (1995). Detection of Atrial
Fibrillation from the Ssurface Electrocardiogram us-
ing Magnitude-Squared Coherence. IEEE Engineer-
ing in Medicine and Biology Society, 1:179–180.
Tateno, K. and Glass, L. (2000). A Method for Detection of
Atrial Fibrillation using RR Intervals. IEEE Comput-
ers for Cardiology, pages 391–394.
BIOSIGNALS 2009 - International Conference on Bio-inspired Systems and Signal Processing
138
