
A BIOLOGICAL NEURAL NETWORK FOR ROBOTIC
CONTROL
Towards a Human Neuroprocessor
José M. Ferrández, Victor Lorente, Javier Garrigós
Departamento de Electrónica, Tecnología de Computadores y Proyectos, Universidad Politécnica de Cartagena, Spain
Eduardo Fernández
Instituto de Bioingeniería, Universidad Miguel Hernández de Elche, CIBER-BBN, Spain
Keywords: Cultured neural network, Induced plasticity, Multielectrode recordings, Robotic control.
Abstract: The main objective of this work is to analyze the computing capabilities of human neuroblastoma cultured
cells and to define stimulation patterns able to modulate the neural activity in response to external stimuli
for controlling an autonomous robot. Multielectrode Arrays Setups have been designed for direct culturing
neural cells over silicon or glass substrates, providing the capability to stimulate and record simultaneously
populations of neural cells. This paper tries to modulate the natural physiologic responses of human neural
cells by tetanic stimulation of the culture. If we are able to modify the selective responses of some cells
with a external pattern stimuli over different time scales, the neuroblastoma-cultured structure could be
trained to process pre-programmed spatio-temporal patterns. We show that the large neuroblastoma
networks developed in cultured MEAs are capable of learning: stablishing numerous and dynamic
connections, with modifiability induced by external stimuli.
1 INTRODUCTION
Using biological nervous systems as conventional
computer elements is a fascinating problem that
permits the hybridation between Neuroscience and
Computer Science. This synergic approach can
provide a deeper understanding of natural perception
and may be used for the design of new computing
devices based on natural computational paradigms.
The brain uses millions of biological processors,
with dynamic structure, slow commutations
compared with silicon circuits, low power
consumption and unsupervised learning. This kind
of computation is more related to perceptual
recognition, due to the natural variance of the
perceptive patterns and the a priori lack of
knowledge about the perceptual domain.
There exist many research approaches based on
mimicking this bioinspired parallel processing, not
only from the algorithm perspective (Anderson and
Rosenfeld, 1998), but also from the silicon circuits
design. These bioinspired approaches are useful for
pattern recognition applications, like computer
vision or robotics, however they are implemented
over serial and artificial silicon processors with fixed
and static structure. A real biological processor with
millions of biological neurons and a huge number of
interconnections, would provide much more
computational power instead of their low transition
rates due to high number of computing elements and
the extraordinary network capability of adaptation
and reconfiguration to unknown environments. This
extraordinary capability is related with natural
unsupervised learning.
Learning is a natural process that needs the creation
and modulation of sets of associations between
stimuli and responses. For understanding the process
of learning, is necessary to define the physiological
mechanisms that support the creation and
modulation of associations and determine the
relation that modulate the configuration between
stimuli and responses associations. These
mechanisms and relation have been studied by many
508
M. Ferrández J., Lorente V., Garrigós J. and Fernández E. (2009).
A BIOLOGICAL NEURAL NETWORK FOR ROBOTIC CONTROL - Towards a Human Neuroprocessor.
In Proceedings of the International Joint Conference on Computational Intelligence, pages 508-513
DOI: 10.5220/0002335305080513
Copyright
c
SciTePress

neurophysiological studies at different levels mainly
in single cell experimentation.
Our learning experiments were performed in neural
cultures containing 120.000 human neuroblastoma
SY-5Y, under the assumption that this kind of cells
are able to respond electrically to external stimuli
and modulate their neural firing by changing the
stimulation parameters. Such cultured
neuroblastoma networks showed dynamical
configurations, beeing able to develop and adapt
functionally in response to external stimuli over a
broad range of configuration patterns. We are
especially interested in analizing if populations of
neuroblastoma cells are able to process and store
information, and if learning can be implemented
over this biological structure.
The main objective of this work is to analyze the
computing capabilities of human neuroblastoma
cultured cells for controlling a robot. Multielectrode
Arrays Setups have been designed for direct
culturing neural cells over silicon or glass substrates,
providing the capability to stimulate and record
simultaneously populations of neural cells . This
paper describes the process of growing human
neuroblastoma cells over MEA substrates and tries
to change the natural physiologic responses of these
cells by external stimulation of the culture provided
by the robot sensors. Modifying the global responses
of some cells with a external pattern stimuli means
adjusting the biological network behaviour due to
changes in synaptic efficiency or long-term
potentiation (LTP). Therefore, the neuroblastoma-
cultured structure could be trained to process pre-
programmed spatio-temporal patterns. In what
follows, we show that the large neuroblastoma
networks developed in cultured MEAs are capable
of learning: stablishing numerous and dynamic
connections, with modifiability induced by external
stimuli.
2 HUMAN NEUROBLASTOMA
CULTURES
The physiological function of neural cells is
modulated by the underlying mechanisms of
adaptation and reconfiguration in response to neural
activity. Hebbian learning describes a basic
mechanism for synaptic plasticity wherein an
increase in synaptic efficacy arises from the
presynaptic cell's repeated and persistent stimulation
of the postsynaptic cell. The theory is commonly
evoked to explain some types of associative learning
in which simultaneous activation of cells leads to
pronounced increases in synaptic strength. The N-
methyl-D-aspartate (NMDA) receptor, a subtype of
the glutamate receptor, has been implicated as
playing a key role in synaptic plasticity in the CNS
(Bading and Greenberg, 1991), where as dopamine
receptors are involved in the regulation of motor and
cognitive behaviors. For most synaptic ion channels,
activation (opening) requires only the binding of
neurotransmitters. However, activation of the
NMDA channel requires two events: binding of
glutamate (a neurotransmitter) and relief of Mg2+
block. NMDA channels are located at the
postsynaptic membrane. When the membrane
potential is at rest, the NMDA channels are blocked
by the Mg2+ ions. If the membrane potential is
depolarized due to excitation of the postsynaptic
neuron, the outward depolarizing field may repel
Mg2+ out of the channel pore. On the other hand,
binding of glutamate may open the gate of NMDA
channels (the gating mechanisms of most ion
channels are not known). In the normal
physiological process, glutamate is released from the
presynaptic terminal when the presynaptic neuron is
excited. Relief of Mg2+ block is due to excitation of
the postsynaptic neuron. Therefore, excitation of
both presynaptic and postsynaptic neurons may open
the NMDA channels, this is closely related with
Hebbian learning.
Another important feature of the NMDA channel is
that it conducts mainly the Ca2+ ion which may
activate various enzymes for synaptic modification,
even nictric oxide has been identified as a relevant
element in synaptic regulation. The enhancement of
synaptic transmission is called the long-term
potentiation (LTP), which involves two parts: the
induction and the maintenance. The induction refers
to the process, which opens NMDA channels for the
entry of Ca2+ ions into the postsynaptic neuron. The
subsequent synaptic modification by Ca2+ ions is
referred to as the maintenance of LTP.
A human neuroblastoma SY5Y cell line, that
express clonal specific human dopamine receptors,
and also NMDA receptors, will be the biological
platform for studying learning in cultured cells.
Neuroblastoma SH-SY5Y cells are known to be
dopaminergc, acetylcholinergic, glutamatergic and
adenosinergic, so in this line they respond to
different neurotransmitters. The cells have very
A BIOLOGICAL NEURAL NETWORK FOR ROBOTIC CONTROL - Towards a Human Neuroprocessor
509

different growth phases, as it can be seen in Figure
1. The cells both propagate via mitosis and
differentiate by extending neurites to the
surrounding area. The dividing cells can form
clusters of cells which are reminders of their
cancerous nature, but chemicals can force the cells
to dendrify and differentiate, in some kind of
neuritic growth.
Figure 1: Human neuroblastoma cells.
As conclusion, neuroblastoma culture cells show
electrophysiological responses similar to standard
neurons, as potential actions generation sensible to
tetrodotoxin (TTX) and acetylcholyn. They have
neurotransmitters synthesis process and are able to
neuritic growth in culture medium.
3 EXPERIMENTAL SETUP
The neuro-physiology setup provides a complete
solution for stimulation, heating, recording, and data
acquisition from 64 channels. The MEA
(microelectrode array) system is intended for
extracellular electrophysiological recordings in vitro
of different applications that include acute brain,
heart, and retina slices; cultured slices; and
dissociated neuronal cell cultures.
The basic components of the proposed system are
shown in Figure 2. These components are:
• A microelectrode array is an arrangement of 60
electrodes that allows the simultaneous
targeting of several sites for extracellular
stimulation and recording. Cell lines or tissue
slices are placed directly on the MEA and can
be cultivated for up to several months. Almost
all excitable or spontaneously active cells and
tissues can be used.
• Raw data from the MEA electrodes are
amplified by MCS filter amplifiers with custom
bandwidth and gain, which are built very small
and compact using SMD (Surface Mounted
Devices) technology. The small-sized amplifier
combines the interface to the MEA probe with
the signal filtering and the amplification of the
signal. The compact design reduces line pick up
and keeps the noise level down. The amplifiers
are mounted over an inverted microscopes.
• The analog input signals are then acquired and
digitized by the MC-Card that is preinstalled on
the data acquisition computer, that supplies the
power for the amplifiers, and the pattern stimuli
to the stimulators.
• The robot sends information about the
environment to the computer using a bluetooth
link. The sensor consists in infrared sensors for
detecting obstacles.
Figure 2: Experimental Setup.
4 METHODS
Human neuroblastoma cultures were produced using
the commercial line SH/SY5Y . Neural cells were
then plated on Micro-Electrode Arrays -MEAs
(MultiChannel Systems, Reutlingen, Germany).
Initially the nitrogen frozen cells, was immersed in a
37 degree bath, and centrifuged at 1000 rpm during
5 minutes. When cells have grown in a uniform
mono-layer process, they are washed three time with
buffer Phosphate-buffered saline (PBS) for keeping
the pH approximately constant. 0,5 per cent trypsin
was added to the solution in order to re-suspend cells
adherent to the cell culture dish wall during the
process of harvesting cells. The cells were kept in
the incubator for 5 minutes and passed through a 40
microm cell strainer (Falcon, Bedford, MA) to
remove large debris. Finally the cells are transferred
to a specific medium in order to inactivate trypsin,
and centrifugated again during 5 minutes at 1000
rpm.
IJCCI 2009 - International Joint Conference on Computational Intelligence
510

For seeding the plate cells are stained with trypan
blue, (because cells that loose their permeability get
colorred with this solution) and counted with a
Neubauer chamber. Finally, 80.000 or 120.000 total
neuroblastoma cells have been placed over the MEA
substrate.
Maintaining cells in culture is essential for studying
their physiological properties. Cell culturing is
dependent on the growth surfaces and cells must
adhere to the electrode substrate in order to establish
the best connection with the electrodes material. For
most cultures coated tissue culture plates are
prerequisite for seeding. The most commonly used
coatings are positively charged polymers. In this
work, the insulation layer (silicon nitride) of some of
the plates was pre-treated with polyethyleneimine
(PEI), showing no advantages compared with no
covered plates.
The neuroblastoma cultures are maintained in a 37
degree humidified incubator with 5 per cent CO2
and 95 per cent O2 with serum-free Neurobasal
medium. Under the aforementioned conditions we
were able to record stable electrophysiological
signals over different days in vitro (Div). The
medium was replaced one-half of the medium every
5 days.
5 RESULTS
The cultured neuroblastoma cells establish synaptic
connections. In Figure 3 it can be seen differentiated
and non-differentiated neuroblastoma cell bodies
growing around the whole electrode population. The
dendritic arborescence is more evident in the
magnification Figure 3 where differentiated neural
cells surround the four electrodes while the rest of
the cells are in their growing process. This Figure
corresponds to 80.000 neuroblastoma cells seeded in
a no-PEI MEA at 2nd day in vitro (div).
The electrophysiological properties of the
neuroblastoma cultures were analized by recording
the spontaneous activity of the network. Time course
of experiments was over 15 days; recordings were
done using two MCS-Meas with two neuroblastoma
cell cultures (but only in one the cells survived till
day 15). In vitro neuroblastoma networks show
spontaneously firing. This firing rates change during
the culture development with marked day
differences and the global rate is closely related to
the age of the network.
Figure 3: Biological neural network over multielectrode
array.
The physiological recordings correspond to
neuroblastoma cultures in the range of 1-7 div. They
show bursting and spiking activity, with usually
negative depolarisations. Figure 4 show the spiking
activity of the neural population with an automatic
detection level for each electrode. This is very
convenient if you have multiple channels for
extracting spikes.
Figure 4: Spontaneus neural activity detected by the
multielectrode array.
The standard deviation of each data trace is used to
estimate its spike threshold. A time interval of 500
ms is used to calculate the standard deviation. By
fixing the factor, by which the standard deviation is
multiplied, the sign of the factor determines whether
the spike detection level is positive or negative, only
values above this will be extracted as spiking
A BIOLOGICAL NEURAL NETWORK FOR ROBOTIC CONTROL - Towards a Human Neuroprocessor
511
activity. A value between -1 and -4 is appropriate for
most applications the threshold was fixed at standard
deviation equal to -3 with respect to the electrode
activity in order to identify spikes embedded in the
noisy signals.
During the neuroblastoma development, a wide
range of population bursting or synchronized
activity has been observed, according to some
studies in neural cultures preparations (Wagenaar,
Pine, and Potter, 2006). The burst usually contains a
large number of spikes at many channels, with
variable duration, from milliseconds to seconds.
5.1 Tetanic Stimulation
Spontaneous activity was recorded for intervals of 3
minutes before stimulation (PRE-data), and the total
number of spikes extracted was counted. The
biphasic stimulus consists in a 10 trains of a 100
anodic-first waveform with 1 Volt amplitude
delivered to all 60 electrodes in order to propagate a
tetanization stimulus to the neuroblastoma culture.
In neurobiology, a tetanic stimulation consists of a
high-frequency sequence of individual stimulations
of a neuron. It is associated with long-term
potentiation, the objective of this work. High-
frequency stimulation causes an increase in
transmitter release called post-tetanic potentiation
(Antonov, Antonova, Kandel, 2003). This
presynaptic event is caused by calcium influx.
Calcium-protein interactions then produce a change
in vesicle exocytosis. Some studies (Jimbo,
Robinson, and Kawana, 1998) use repetitive
stimulation for training neural cultures, achieving
activity potentiatiation or depresion
Once the tetanization stimulus was applied to the
whole population 5 minutes after the stimulation a 3
minutes interval was recorded (POST-data). Only
neuronal signals which had at least a 2:1 signal:noise
ration were valued as "spikes". Again, the total
number of spikes extracted was counted. This
process was made for cultures at 1 day in vitro (div),
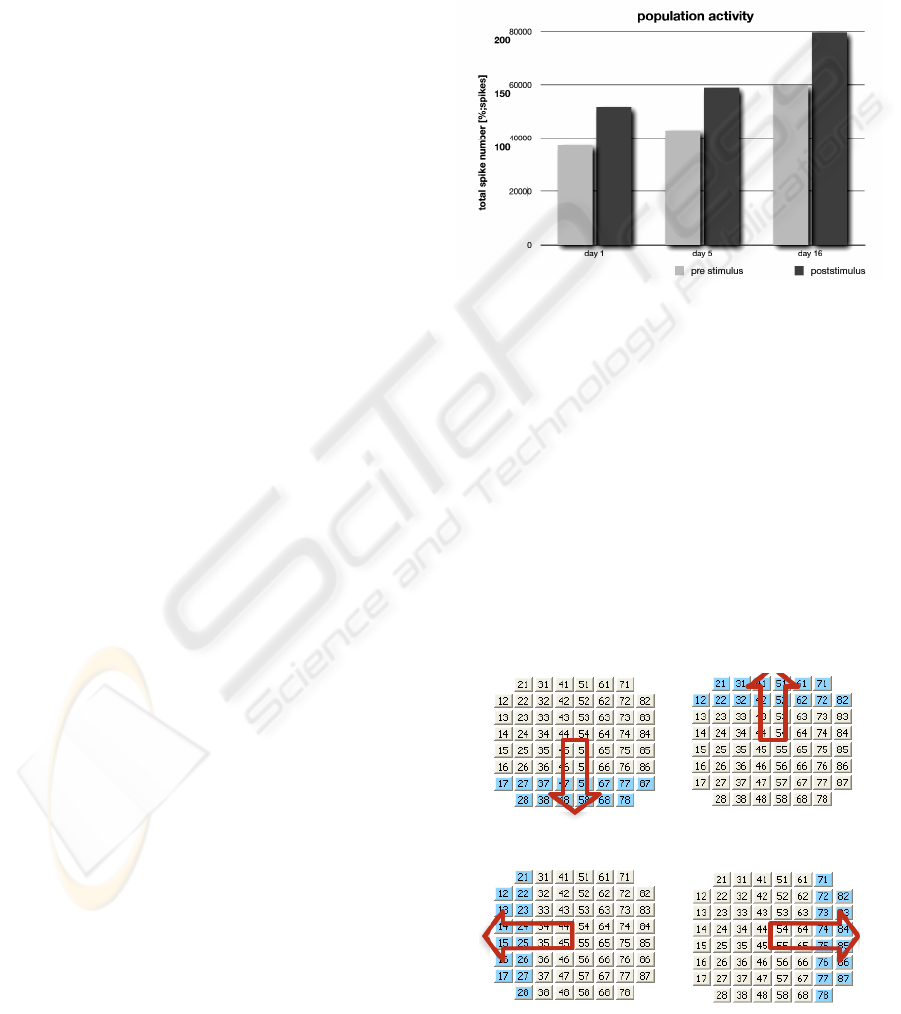
5 div and 16 div. Figure 5 represents the counted
spikes with bar charts for the different recordings.
The conclusion from this Figure is:
1) While the neuroblastoma culture is growing
new connections are created, and the number
of spikes increases as the culture expands
over the MEA.
2) After a tetanic stimulation the cells continue
with their increased spiking rate, providing a
persistent change in the culture behaviour.
When this change in the network response
lasts, these changes can be called learning.
In all the experimentation performed, tetanic
stimulation was applied as training method, and the
electrophysiological properties of the neuroblastoma
culture change, getting a potentiation effect on the
spontaneous firing, modulating in this way the
culture neural activity.
Figure 5: Induced neural activity by tetanization stimuli.
5.2 Robotic Control
For controling the direction of the robot we propose
to compute the vector resulting from neural activity
recorded in the human neuroblastoma culture. This
vector will be provided to the robot in order to guide
his movement. The sensors will detect the obstacles,
and the information will be passed to the computer
in order to induce a selective tetanization of the
biological neural network for changing the resulting
direction vector. In Figure 6, the selected electrodes
for the tetanization are shown in order to selective
induce a persistent change in the biological neural
network behaviour.
Figure 6: Selective electrode tetanisation.
IJCCI 2009 - International Joint Conference on Computational Intelligence
512

When the robot detect an obstacle in his left path, an
stimilation signal will be sent to the system for
tetanizing the right tissue. By tetaniztion the
electrodes of the right part of the array, an increase
in the firing rate of the neural cells that lie in the part
of the culture will be achieved, and the direction
vector will point to the right in this particular case.
We expect to apply some basic Braitenberg
principles to the system in order to study the
biological neural network behaviour induced by a
tetanization learning scheme.
6 DISCUSSION
Learning in cultured neuroblastoma networks by a
stimulation process, without the involvement of a
natural adaptation process to the environment
requires identifying the correct stimuli to provide to
the neurons maintained ex vivo. These
neuroblastoma networks form a large culture
covering the whole electrode array and generating a
rich dendritic configuration. The connectivity can be
modulated by external stimulation as has been
described in many studies, but also the activity of
the network can be modulated with the appropriate
stimulation scheme.
Tetanization consists in high-frequency stimulation
to the culture, in order to cause an increase in
transmitter release called post-tetanic potentiation.
The results illustrate the existence of qualitatively
different responses to stimulation. Our results
indicate the existence of a clear facilitation
mechanism in response to the tetanization stimuli at
different stages of cell development. Since this kind
of stimulation has been used in attempts to induce
plasticity in neuroblastoma, refining some crucial
aspects of the stimulation is still indispensable.
It is very important to adjust the frequency of the
train pulses of the stimulation for suppressing
bursting in the culture. While in vivo networks
suppress bursting naturally with the tissue
development and sensory inputs, ex-vivo cultures
need to reduce this synchronized activity by
adjusting the stimulation parameters. Also, for
superimposing a desired behaviour on the biological
networks it is necessary to stimulate locally some
part of the culture in order to facilitate some parts of
the networks, or achieve some kind of electrical
stimulation that depress the local activity of a
restricted location. With this local potentiation-
inhibition scheme the culture global behaviour could
be controlled.
Future work consists in determine the optimal
stimulation to apply for inducing permanent firing
changes in the culture, and the strategies for
connecting the robot sensors to the stimulation
patterns. These aspects will then constitute the basis
for inducing stable goal-directed plasticity, and
hence for designing new biological neuroprocessors
applied to robotics.
ACKNOWLEDGEMENTS
This work was supported by the Spanish
Government through grants TIN2008-06893-C03,
TEC2006-14186-C02-02 and SAF2008-03694,
Cátedra Bidons Egara, Fundación Séneca
08788/PI/08, CIBER-BBN and by the European
Comission through the project "`NEUROPROBES"'
IST-027017.
REFERENCES
Anderson J.A., and Rosenfeld E., 1998. “Neurocomputing:
Foundations of research'', MIT Press Cambridge, MA,
USA.
Bading H. and Greenberg M.E., 1991. “Stimulation of
protein tyrosine phosphorylation by NMDA receptor
activation '', Science, 253, Issue 5022, pp. 912-914.
Wagenaar, D. A., Pine, J. and Potter, S. M., 2006. “An
extremely rich repertoire of bursting patterns during
the development of cortical cultures.'', BMC Neuro-
science, 7:11.
Antonov I., Antonova I., Kandel E.R., 2003. “Activity-
Dependent Presynaptic Facilitation and Hebbian LTP
Are Both Required and Interact during Classical
Conditioning in Aplysia '', Neuron, Volume 37(1), pp.
135-147.
Jimbo Y., Robinson H.P., Kawana A., 1998. “Strength-
ening of synchronized activity by tetanic stimulation
in cortical cultures: application of planar electrode
arrays'', IEEE transactions on Biomedical
Engineering, 45(11), pp. 1297-1304.
A BIOLOGICAL NEURAL NETWORK FOR ROBOTIC CONTROL - Towards a Human Neuroprocessor
513
