
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC
PATIENTS
Mark Hoogendoorn, Michel C. A. Klein and Nataliya M. Mogles
VU University Amsterdam, Department of Artificial Intelligence, De Boelelaan 1081, Amsterdam, Netherlands
Keywords: Diabetes, Model-based reasoning, Intelligent support.
Abstract: Diabetes Mellitus type I is a disease that forces patients to manage their blood glucose level manually, by
balancing their activities, food intake and insulin dosages. There is a large experience with building
computational models for blood glucose level in diabetic patients, which are primarily used to design the
medication regime for a specific patient. In this paper, the design of an intelligent support application is
presented that uses a standard model for blood glucose level to give patients real-time advice about insulin
and food intake. The advice is based on measurements of blood glucose level, the electronic agenda of a
patient and model-based predictions of the glucose level in the near future. A simulation of the application
is presented that illustrates the feasibility of the system.
1 INTRODUCTION
Diabetes mellitus is a syndrome that is characterized
by dysfunctional metabolism, resulting in too high
blood sugar levels. According to the World Health
Organization, the prevalence of diabetes for all age-
groups worldwide is estimated to be 2.8% in 2000
and 4.4% in 2030. The total number of people with
diabetes is projected to rise from 171 million in 2000
to 366 million in 2030 (Wild et al, 2000).
The glucose level in humans is regulated by a
mechanism that is composed of several interacting
systems, in which the hormone insulin is the very
important, as it decreases the blood glucose level.
There are two types of diabetic patients: in type 1
patients, the high blood glucose levels are caused by
the loss of the insulin-producing cells in the
pancreas, in type 2 patients the body developed a
reduced sensitivity (or even resistance) to insulin.
Diabetes mellitus is currently a chronic disease,
without a cure, and is treated by a combination of
dietary guidelines, exercises and insulin
supplementation. The main challenge for diabetes
patients is to manually keep their blood glucose
level within a safe range, by balancing both their
glucose intake, their physical activities as their
insulin dosages (note that type II patients are not
always treated with insulin). This management is
complicated and a burdensome task for diabetic
patients. Specifically, the following reasons are
mentioned (Wikipedia, 2009):
1. The glucose cycle is a system which is affected
by two factors: entry of glucose into the
bloodstream and also blood levels of insulin to
control its transport out of the bloodstream;
2. As a system, it is sensitive to diet and exercise;
3. It is affected by the need for user anticipation
due to the complicating effects of time delays
between any activity and the respective impact
on the glucose system;
4. Management is highly intrusive and
compliance is an issue, since it relies upon user
lifestyle change and (often) upon regular
sampling and measuring of blood glucose
levels, multiple times a day in many cases;
5. It changes as people grow and develop;
6. It is highly individual.
In this paper, we investigate the options for an
intelligent support system that helps diabetic patients
controlling their blood glucose, exploiting
measurements via sensor devices, information about
activity from electronic agenda’s and model-based
predictions of the blood glucose level. One of the
features of such a system could be automated tuning
of the model parameters to an individual patient.
Together, such a system would ease the management
of diabetes for patients in several aspects. The usage
of a glucose-insulin model automates the assessment
of the interaction of the different factors (complexity
1 & 2 in the list above), the prediction and usage of
98
Hoogendoorn M., Klein M. and Mogles N. (2010).
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC PATIENTS.
In Proceedings of the Third International Conference on Health Informatics, pages 98-105
DOI: 10.5220/0002747600980105
Copyright
c
SciTePress

electronic agenda information automates part of the
anticipation (number 3 in the list), while parameter
tuning allows for personalization and adaptation
over time (complexity 5 & 6).
In the next Section, we describe the main factors
of blood glucose regulation process in the human
body and a mathematical model that is often used for
describing the glucose-insulin interaction. Section 3
sketches the main elements of an intelligent support
system using the glucose model and sensor
measurements. In Section 4 we show a number of
simulations of patients with different activity
patterns and the effect of an intelligent support
system consisting of the components described in
the section before. Finally, Section 5 concludes the
paper.
2 MODELING BLOOD GLUCOSE
LEVEL
Mathematical or computational models of diabetes
type I have been under development for several
decades (see e.g. Bolie, 1961, Ackerman, 1965,
Sorensen, 1985, Puckett, 1992, and Leaning and
Boroujerdi, 1991). Models range from ordinary
systems of differential equations to stochastic
differential equations. Makroglou et al present a nice
overview of the various types of models that exist.
Within the ordinary systems of differential
equations, the model used most frequently is the so-
called minimal model which has been introduced by
Bergman. The development of the model has been
motivated by a desire to model the intravenous
glucose tolerance test. Such models consist of many
parameters that are very specific towards patients.
As a result, parameter estimation techniques have
been proposed that allow the tailoring of the models
towards the patients. In (De Geatano and Arino) the
results of one of such parameter estimation
techniques are shown, namely a quasi-Newton
minimization algorithm.
In this paper, the minimal model as introduced
by Bergman et al (1979; Tololo et al 1980) is
adopted. The model consists of the following three
formulas:
dG(t)/dt = – [p
1
+ X(t)]G(t) + p
1
G
b
(1a)
= – X(t)G(t) + p
1
[G
b
– G(t)] (1b)
dX(t)/dt = – p
2
X(t) + p
3
[I(t) – I
b
]
(2)
dI(t)/dt = p
4
[G(t) – p
5
]
+
t – p
6
[I(t) – I
b
] (3)
In this formula, G(t) is the blood glucose
concentration, I(t) is the blood insulin concentration,
and X(t) is related to the interstitial insulin level, i.e.
the insulin that is at a location where it can actually
effect the glucose uptake by cells. Furthermore,
• G
b
is the subject's baseline glucose level;
• I
b
is the subject's baseline insulin level;
• p
1
is the glucose “mass action” rate constant, i.e.
the insulin-independent rate constant of tissue
glucose uptake;
• p
2
is the rate constant expressing the spontaneous
decrease of tissue glucose uptake ability;
• p
3
is the insulin-dependent increase in tissue
glucose uptake ability;
• p
4
is the rate of pancreatic release of insulin after
glucose intake;
• p
5
is the pancreatic “target” blood sugar level;
• p
6
is the decay rate constant for insulin in plasma;
For patients with diabetes type 1, we assume that the
pancreas does not produce any insulin anymore. In
the model, the effect is that parameters p
4
and I
b
are
zero. Consequently, the insulin level is determined
only by the artificial intake of insulin and the decay,
with I
s
(t)denoting the artificial insulin supply at
certain time points:
dI(t)/dt = – p
6
[I(t)] + I
s
(t) (4)
The minimal model does not take the effect of
physical effort into account. The effect of physical
effort on the insulin and the blood glucose balance is
twofold:
• it increases the insulin use by cells;
• it lowers the glucose concentration during and
after the exercise (Goodyear and Kahn, 1998).
Especially the fact that the glucose concentration is
also lowered after the exercise (up to 24 hours) is an
important factor to take into account. According to
(Derouich and Boutayeb, 2002), the minimal model
extended with the effect of exercises can be
described by the following formulas:
dG(t)/dt = – [1 + q
2
] X(t)G(t) + [p
1
+ q
1
][G
b
– G(t)] (5)
dX(t)/dt = – p
2
X(t) + [p
3
+ q
3
][I(t) – I
b
] (6)
In these formulae, the q-parameters define the effect
of physical activity. They are defined as follows:
• q
1
: the effect of the physical exercise in
accelerating the utilization of glucose by muscles
and the liver.
• q
2
: the effect of the physical exercise in
increasing the muscular and liver sensibility to
the action of the insulin.
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC PATIENTS
99

• q
3
: the effect of the physical exercise in
increasing the utilization of the insulin.
The q
2
variable is also larger then 0 for some time
after the exercise.
3 INTELLIGENT SUPPORT
Currently, diabetes patients design a schema for
insulin intake in consultation with their medical
practitioner. This schema is based on a registration
of regular blood glucose measurements. Patients will
also use their common sense knowledge about the
effect of their activities on their blood glucose level:
for example, if a large meal is consumed, the person
will take a somewhat higher dose of insulin, or if
sporting activities are planned, some additional food
(especially) carbohydrates will be taken. In addition
to this, patients will do regular blood glucose
measurements to verify whether it is still within safe
bounds, and possibly to correct it.
The envisaged intelligent support system will
give advice to a patient on when to take which
amount of insulin or a meal. This advice is based on
a prediction of the blood glucose level using the
most recent measurement and the activities listed in
the electronic agenda. The listed activities influence
the blood glucose level, but also determine the time
points when insulin or a meal can be taken. For
example, in the middle of a sporting activity of
while working, it is not easily possible to take a meal
or insulin. The system could be implemented as an
advanced mobile phone or PDA application. Blood
glucose measurements will ideally be transferred
form the electronic device (see Figure 1) via a
wireless technology such as Bluetooth, but could
also be manually typed in into the application.
Figure 1: Electronic blood glucose meter.
For the prediction of the glucose level, the model as
explained in the previous section is used. The
parameters in the model should be fitted to the
personal characteristics of the patient. For this
fitting, there are quite a number of approaches (De
Geatano and Arino, 2000). In this paper, we assume
that the parameter fitting has been implemented
using one of the described techniques. Our
intelligent support system will use the model with
the fitted parameters and dynamically determine the
amount of insulin to be taken.
The system internally uses a list of activities and
associated values for the q parameters. Each type of
activity can have different parameters. For example,
walking could have different parameters for the
utilization of glucose and insulin than intense
sporting. The activities are read from the agenda,
and from the latest time point of measurement, the
current glucose level is calculated based on the
activities that are undertaken since the last
measurement. In addition, the upcoming activities
are used to predict the blood glucose level at the end
of the next activity that still has to be started. For
example, when a person is currently working and the
next activity will be cycling, the glucose level at the
end of the cycling activity is measured. In case this
measurement is too high, advice is given to take
insulin at the end of the current activity. In case this
measurement is too low, advice is given to take
some food at the end of the current activity. The
amount of insulin or food is dynamically determined
by simulation within the support system. At the end
of the current activity, the patient will get a message,
for example via his mobile phone application, to
take a specific amount of insulin or food before the
next activity.
4 SIMULATION EXPERIMENTS
The model and prediction rules that are used by the
system have been implemented in Matlab. A number
of simulation experiments have been run in this
environment. In these experiments, the activities of a
person during two consecutive days are simulated.
Table 1 gives an overview of the activities and the
time points. Note that the simulation time uses units
of 15 minutes.
For the parameters, values were used that are
found in the literature as realistic values for a
specific person. Specifically, we used the parameters
of subject 2 in (Gaetano and Arino, 2000): p
0
= 100,
p
1
= 0.1, p
2
= 0.2196, p
3
= 0.0064, p
4
= 0, p
5
= 23, p
6
= 0.096, p
7
= 0.5, I
b
= 0, G
b
= 120. Note that p
4
and
I
b
are 0 because we consider a diabetic patient. For
the desired minimum and maximum glucose levels,
we use 80 and 120 mg/dL (Erzen et al, 2000).
HEALTHINF 2010 - International Conference on Health Informatics
100

Figure 2: Blood glucose level of a diabetic patient without insulin intake.
Table 1: List of activities during two days.
Activity Clock
time
Time points in
simulation
Sleeping till 7am 0:00 0 96
Breakfast 7:00 28 124
Cycling / driving 7:30 30 126
Office work 8:15 33 129
Coffee 10:15 41 137
Office work 10:30 42 138
Lunch 12:30 50 146
Office work 13:00 52 148
Tea 16:00 64 160
Office work 16:15 65 161
Cycling / driving 17:00 68 164
Diner 17:45 71 167
Relaxing 18:30 74 170
Intense sporting / no sporting 19:30 78 174
Relaxing 20:30 82 178
Sleeping till 0:00 22:30 90 186
Sleeping till 7am 0:00 96 192
4.1 Blood Glucose Level without
Insulin Intake
The first simulation shows the blood glucose level
for a person that does not produce any insulin
anymore. At the start of the simulation, there is still
a small amount of insulin available (0.5 μU/ml),
however, this dissolve in a few hours. It can be seen
that the blood glucose level will be almost always
too high (see Figure 2).
4.2 Regular Insulin Intake with and
without Physical Activities
In the second set of simulations the glucose level of
a patient that is treated with a schema of regular
insulin intake doses. We assume in the simulation
that insulin is taken just before every meal,
effectively three times per day. The second
assumption is that a patient takes a lower dose of
insulin if physical activities are undertaken during
the day. We did this simulation for two different
scenarios: one with physical activities, in which the
person uses a bicycle to commute and does sporting
in the evening, and one in which the person drives to
his work by car and does no sporting. For the effect
of physical activities on the blood glucose balance,
we used a q
2
value of 0.25 for cycling and 0.5 for
intense sporting. The effect of the duration of the q
2
effect was set to 24 hours. The parameters q
1
and q
3
were both set to 0.25; no empirical values were
available. The effect on the blood glucose level and
the insulin is depicted in Figure 3 and 4.
It can be seen in Figure 4 that the patient takes a
smaller amount of insulin on a regular basis using
his common sense to keep the blood glucose within
safe boundaries in the scenario in which physical
activities are undertaken.
4.3 Prediction of Maximum and
Minimum Glucose Levels
To give a person intelligent support about his insulin
intake, we have to predict the effect of the activities
on the future blood glucose level. We do this by
running a simulation of the future values of the
blood glucose level during the current and next
activity at every time point within the simulation of
the scenarios. For this, we determine the end of the
next activity, take the planned activities into account,
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC PATIENTS
101

Figure 3: Simulation of a diabetic patient that takes regular insulin dose with physical activities.
Figure 4: Simulation of a diabetic patient that takes regular insulin dose without physical activities
Figure 5: The predicted maximum and minimum blood glucose level during the current and next activity.
and simulate the blood glucose level till the end of
the next activity, assuming that no insulin will be
taken. Then, the maximum and minimum levels of
the blood glucose during that period are determined.
Figure 5 illustrates how this would look for the
scenario in which no insulin is taken (i.e. the
scenario depicted in Figure 2; the jigsaw pattern is a
side effect of using a small time step in the
simulation.)
When it is predicted that the maximum glucose
level will become too high (in this case > 120) till
the end of the next activity, the system will run
another simulation to predict the effect of taking a
standard insulin dose at the end of the current
activity. When it is predicted that the minimum
glucose level will become too low (in this case <
80) before the end of the next activity, the system
will run another simulation to predict the effect of
taking a standard meal(e.g. a cup of coffee, a
chocolate bar) at the end of the current activity.
Based on the size of the effect, the optimal dose is
determined. This is done by comparing the required
effect and the achieved effect with the standard dose.
HEALTHINF 2010 - International Conference on Health Informatics
102

Figure 6: Simulation of a patient using the intelligent support system without physical activities.
For example, if a standard dose of insulin reduces
the excess of the predicted level over the maximum
level by 30%, the advised dose is 100/30 = 3.33
times the standard dose.
4.4 Using the Intelligent Support
System for Insulin and Food Intake
We also ran a number of simulations of scenarios in
which patients actually follow the advice of the
intelligent support system. In these scenarios, the
advised dose of insulin is actually taken, and the
effect of this insulin is taken into account in the next
predictions. We show two scenarios: the first one is
the one without physical activities (Figure 6), the
second one with physical activities (Figure 7).
The figures show that the system is able to adapt
automatically to the activities of the person as
registered in his electronic agenda. In the scenario
without physical activities (Figure 6) the person gets
advice to take insulin three times per day with a dose
that corresponds to an insulin level increase of
around 3 μU/ml in the blood. No advice about
additional meals is given: it can be seen in Figure 6
that the glucose intake is standard and corresponds
to 5 time regular meals during the day, such as
breakfast, coffee , lunch, tea and dinner.
In the second scenario (see Figure 7), the advice
is to take insulin three times per day and five
additional meals during the first day and three
times per day insulin and one time an additional meal
during the second day. During the first day of the
second scenario the glucose intake occurs ten
times a day instead of standard five and six times
during the second day.
In both scenarios, the blood glucose level is most
of the time below the advised maximum level and
above the advised minimum level. Moreover, the
advice is always given at appropriate times, i.e.
never in the middle of an activity or during the night.
5 DISCUSSION
In this paper, the working of an intelligent support
system for diabetic patients is presented. The system
is based on the existing technologies like mobile
phones with electronic agenda’s, electronic blood
glucose meters, and mathematical models of the
blood/insulin balance. Keeping the blood glucose
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC PATIENTS
103
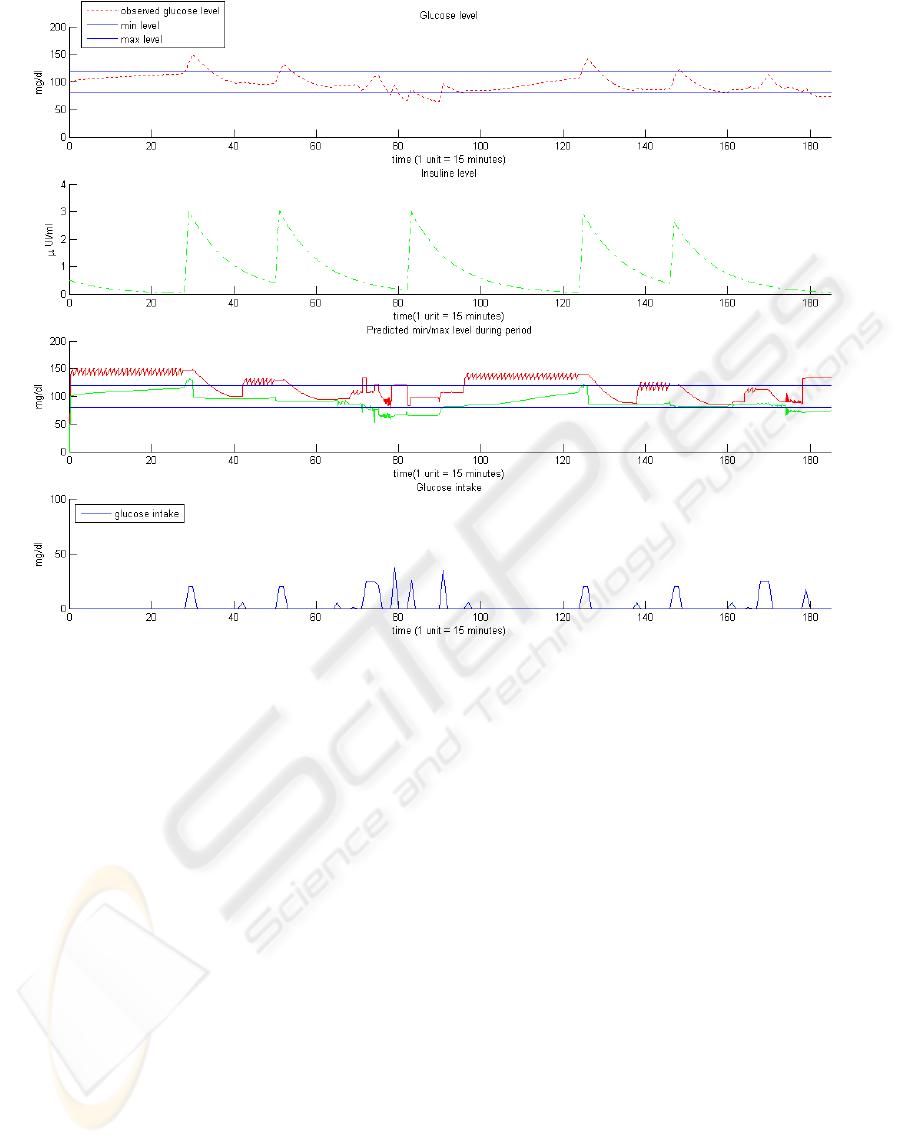
Figure 7: Simulation of a patient using the ambient support system with physical activities.
level within the safe boundaries by balancing insulin
dosages, glucose intake and physical activities is a
complicated task for diabetes patients. The
advantage of this system is that it adapts
automatically to the personal schedule of a patient
and gives concrete advice about insulin and food
intake at appropriate time points if it predicts that the
blood glucose level of the patient will rise above the
higher boundary or drop below the lower boundary at
the end of the next activity. The simulation results
demonstrated that in the scenario with the patient’s
physical activities the intelligent support system
helps better to maintain an appropriate blood glucose
level in comparison with the regular insulin
prescription.
Thus, the system could possible release a bit of
the burden of diabetic patients as it can predict the
effect of the upcoming activities more precisely than
humans. Moreover, the effective blood glucose level
management may minimize the progression of the
disease and reduce the risk of later complications
that accompany this chronic disease (Deutsch,
1994).
There are a number of extensions of this system
imaginable. First of all, one could think of using
continuous, non-invasive blood glucose measuring
techniques. At the moment, those are not yet
available, but it is expected that these become
available in the near future. Second, more specific
rules for restrictions on the advice can be
implemented, for example the minimum of
maximum dose, the minimal time between insulin
doses, the maximum doses per day, etc. These
extensions could make the system even more
realistic and more effective for patients.
REFERENCES
Ackerman, E., Gatewood, L. C., Rosevear, J. W. and
Molnar, G. D., 1965. Model studies of blood-glucose
regulation. Bulletin of Mathematical Biophysics 27:21-
37.
Bergman, R. N., Ider, Y. Z., Bowden, C. R. and Cobelli,
C., 1979. Quantitative estimation of insulin sensitivity.
American Journal of Physiology 236: E667-E677.
Bolie, V. W., 1961. Coefficients of normal blood glucose
regulation. Journal of Applied Physiology 16:783-788.
HEALTHINF 2010 - International Conference on Health Informatics
104

Derouich, M., Boutayeb, A., 2002. The effect of physical
exercise on the dynamics of glucose and insulin.
Journal of Biomechanics 35(7): 911-917.
Deutsch, T., Lehmann, E. D., Carson, E.R., Roudsari,
A.V., Hopkins, K.D., Sönksen, P.H., 1994. Time
series analysis and control of blood glucose levels in
diabetic patients.Computer Methods and Programs in
Biomedicine 41:167–182.
Erzen, F.C., Birol, G., Cinar, A., 2000. "Simulation
Studies on the Dynamics of Diabetes Mellitus," bibe,
pp.231, IEEE International Symposium on Bio-
Informatics and Biomedical Engineering (BIBE'00).
De Gaetano, A. and Arino, O., 2000.Mathematical
modelling of the intravenous glucose tolerance test,
Journal of Mathematical Biology 40: 136-168.
Goodyear, L.J., Kahn, B.B., 1998. Exercise, glucose
transport, and insulin sensitivity, Annual Review of
Medicine 49: 235-261.
Leaning, M.S. and Boroujerdi, M.A., 1991. A system for
compartmental modelling and simulation. Computer
Methods and Programs in Biomedicine 35:71–92.
Puckett, W.R., 1992. Dynamic modelling of diabetes
mellitus. PhD Thesis, University of Wisconsin -
Madison.
Sorensen, J.T., 1985. Physiologic model of glucose
metabolism in men and its use to design and asses
improved insulin therapies for diabetes. PhD Thesis,
MIT.
Tololo, G., Bergman, R. N., Finegood, D. T., Bowden, C.
R. and Cobelli, C., 1980. Quantitative estimation of
beta cell sensitivity to glucose in the intact organism: a
minimal model of insulin kinetics in the dog. Diabetes
29: 979-990.
Wild, S., Roglic, G., Green, A., Sicree, R., King, H., 2004.
Global prevalence of diabetes: estimates for the year
2000 and projections for 2030. Diabetes Care 27 (5):
1047–53.
Wikipedia, 2009. Diabetes management, section
Complexities relating to management,
http://en.wikipedia.org/wiki/Diabetes_management,
retrieved at June 8, 2009.
AN INTELLIGENT SUPPORT SYSTEM FOR DIABETIC PATIENTS
105
