
INTELLIGENT EXPANDABLE STRUCTURES BASED ON THE
IMPROVED ACTIVATION OF SHAPE-MEMORY POLYMERS
Andrés Díaz Lantada, Pilar Lafont Morgado, Julio Muñoz-García, José Luis Muñoz Sanz
Javier Echavarri Otero and Juan Manuel Munoz-Guijosa
Grupo de Investigación en Ingeniería de Máquinas, E.T.S.I. Industriales, Universidad Politécnica de Madrid
c/ José Gutiérrez Abascal, nº 2, 28006, Madrid, Spain
Keywords: Shape-Memory Polymers (SMPs), Biodevices, Expandable structures, Improved activation.
Abstract: Shape-memory polymers are active materials with thermomechanical coupling and a high capability to
recover from high levels of deformation, which, combined with their low cost and density has favoured the
appearance of numerous applications, particularly those linked to the Medical Industry. In many cases, these
materials are of medical standard, which increases the chances of obtaining biocompatible devices.
In the last decade enormous progress has been made on many areas, regarding these materials, such as
synthesis, characterization, activation, prototyping and others, aimed at improving their applicability.
However, various spheres of action require additional in depth research to promote the production start-up
of various shape-memory polymer-based devices that have had laboratory validation. One of these areas of
improvement is linked to the activation systems of SMPs.
This work sets outs the possibility of obtaining a more homogeneous heating processes for an optimal
activation of the “shape-memory effect”, which promotes the geometric changes of such devices. These
improvements are based on the development of net-shaped SMP structures to which silver thread is knitted
for subsequent activation through Joule heating. First prototypes and trials are explained in detail, as well as
the possible biomedical applications of this concept.
1 INTRODUCTION TO
SHAPE-MEMORY POLYMERS
(SMPS)
Shape-memory polymers (SMPs) are materials that
show a mechanical response to external stimuli,
usually to changes of temperature. When these
materials are heated above their “activation”
temperature, there is a radical change from rigid
polymer to an elastic state that will allow
deformations of up to 400%. If the material is cooled
down after manipulation it retains the shape
imposed; the said structure is “frozen” and returns to
a rigid but “non-equilibrium” state. If the material is
again heated above its vitreous transition
temperature or “activation temperature” it recovers
its initial non-deformed state. The cycle can be
repeated numerous times without degrading the
polymer and most suppliers can formulate different
materials with activation temperatures ranging from
–30 ºC to 260 ºC, depending on the application
required. Of all the polymers developed that show
shape-memory properties, those most worthy of
mention due to their applicability are epoxy resins,
polyurethane resins, cross-linked polyethilene,
styrene-butadiene copolymers and other
formulations (Lendlein, Kelch, 2002, Liu, 2007).
They are therefore active materials that present
thermomechanical coupling and a high capability for
recovery from deformation, (much greater than that
shown by shape-memory metal alloys), which
combined with their lower density and cost has
favoured the appearance of numerous applications.
Their properties permit applications for the
development of sensing devices and actuators,
especially for the aeronautics, automobile and
medical industry.
Figure 1 shows a scheme of the training and
actuation process of SMP structures.
240
Díaz Lantada A., Lafont Morgado P., Muñoz-García J., Luis Muñoz Sanz J., Echavarri Otero J. and Manuel Munoz-Guijosa J. (2010).
INTELLIGENT EXPANDABLE STRUCTURES BASED ON THE IMPROVED ACTIVATION OF SHAPE-MEMORY POLYMERS.
In Proceedings of the Third International Conference on Biomedical Electronics and Devices, pages 240-245
DOI: 10.5220/0002766602400245
Copyright
c
SciTePress

Figure 1: Training process of the shape-memory effect and
recovery of permanent form.
2 POTENTIAL FOR BIODEVICES
2.1 Some Advantages
As polymers, SMPs can be easily conformed into
different complex shapes and their properties
designed or adapted to specific applications and can
also be integrated with other microelectromechanical
sensors (MEMS) to produce “intelligent”
bioactuators and biodevices.
Compared to other shape-memory alloys used in
numerous medical devices, SMPs show a far greater
capability for changing their geometry during
activation. They are also much cheaper to synthesise
and their large scale mass production costs are
reduced by using technologies such as injection
moulding. All this makes them very versatile active
materials with a high potential for industry, provided
they overcome some of the limitations set out in the
following sections.
2.2 Proposed Devices
Bellow are explained some specific proposals for
developing medical devices based on the use of
shape-memory polymers, most of which have
undergone in vitro laboratory testing. After
undergoing in vitro testing and meeting the
requirements for official approval, in some cases
their commercialisation is subject to their attaining
the goals described at the end of this paper.
Self-expanding Stents. Like the stent designed by
Boston Scientific Corporation using the polymer
from CRG Industries known as “Veriflex” under its
trade-name, to treat the problems arising when the
arteries become narrow or obstructed and also for
removing obstructions from other “tube-shaped”
body parts, like the uretheres and the bronchial
tubes. The stent is inserted in its temporary form
(reduced) and the body’s own heat causes it to dilate
and become attached to the artery. They may be
used to replace stents based on shape-memory alloys
such as Nitinol, once the appropriate
biocompatibility studies have been carried out.
Developments of self-expanding stents have also
been carried out by using injected polyurethane
(Wache, 2003).
Intelligent Sutures. Like those developed at the
Forschungszentrum in Karlsruhe by Lendlein’s team
and at the M.I.T. by Langer’s team, which have a
temporary linear shape and a permanent shape in the
form of a knot, with the change in geometry being
activated by the body’s own temperature. They have
numerous applications in minimally invasive surgery
and, as they are biodegradable, they have additional
advantages over the use of textile sutures and metal
clips (Lendlein, Kelch, Langer, 2002, 2005).
Thrombectomy Devices. With the recent discovery
that the thermal effect of shape-memory can be
activated by a laser, part of whose energy is
absorbed by the polymer, devices with special
geometries have been proposed for removing clots
(Wilson, 2006). The polymer is shaped in a spiral
mould and then heated and stretched to give it its
temporary shape. When the laser light passes
through the polymer, the shape-memory effect is
activated and the device recovers its spiral shape
trapping the clot which can then be removed.
Active Catheters. By using shape-memory
polymers for the distal point of catheters together
with a subsequent activation of the memory effect
by laser light or body heat, different drugs and
antitumoral agents can be released. The presence of
an active catheter point can also help reach zones
that are difficult to access in minimally invasive
surgery tasks (Yackaki, 2007).
Drug Release Devices. If biodegradable shape-
memory polymers are used for implantable medical
devices, drug supply reservoirs can be incorporated
into the device itself. After implant, the polymer
begins to be absorbed by the organism and the drug
is released. Patents have been taken out in this
respect for self-expanding coronary stents or intra-
urethral stents (Boston Scientific Co. and Surmodics
Inc.). The possibility of obtaining temporary
geometries with micro-reservoirs for drug storage
has also been studied. The drugs would then be
INTELLIGENT EXPANDABLE STRUCTURES BASED ON THE IMPROVED ACTIVATION OF SHAPE-MEMORY
POLYMERS
241

released on activation of the shape-memory effect by
body heat (Gall, 2004).
Active Annuloplasty Rings. Aimed at obtaining a
progressive postoperative treatment of mitral
insufficiency, they are based on the use of a
polymeric ring with heating resistances distributed
around the inside to activate the shape-memory
effect by Joule effect. This activation must allow the
cross section of the mitral ring to be gradually
reduced and, therefore, the mitral insufficiency
improved.
3 CHALLENGES RELATED TO
THE ACTIVATION PROCESS
One aspect where most progress has been made in
the last years is the activation of the memory effect
by various methods, especially:
Joule Effect Activation. Based on distributing
heating resistances at the core of the polymer where
the passing of an electric current generates the
necessary heat.
Light or Laser Activation. Based on projecting a
laser through a shape-memory material with a
similar absorption frequency to that of the laser
used, which produces heating (Lendlein, 2005,
Wilson, 2006)
Magnetic Activation. Based on heating by
induction of magnetic or metallic microparticles,
distributed at the core of the polymer while it is
being conformed to its shape (Buckley, 2006).
However, the biocompatibility of the associated
devices needs to be further optimised.
Support Technologies. Progress in the field of
wireless communications means that devices can
now be remotely activated, which is promoting the
appearance of new active implantable biodevices.
One of the main problems of using Joule effect
activation lies in the fact that distributing punctual
heating resistances among the polymer leads usually
to a decrease of mechanical resistance and to
important temperature differences along the
structure during activation.
On the other hand, “light / laser activation” is
normally limited to tubular / linear structures and
“magnetic activation” usually implies problems due
to the important magnetic fields needed for heating.
We propose and explain in the following
chapters some improvements for SMP activation,
obtained by substituting punctual heating resistances
by silver-cloth thread knitted along the structure and
used as single Joule effect heating element.
4 IMPROVING ACTIVATION:
DISTRIBUTING HEAT
As previously stated our objective is to obtain SMP
structures with an improved distribution of heating
element(s) for a better controlled and more
homogeneous activation of “shape-memory effect”.
Figure 2 shows the prototype of a net-shaped
SMP structure with the heating silver-cloth thread
already knitted to it. Such thread was acquired from
the MUTR (www.mutr.co.uk) special materials
facilities.
For manufacturing the prototypes (directly from
3D CAD files with part geometry) a laser
stereolithography “SLA-3500” machine was used to
polymerise a 3D Systems epoxy resin sold under the
trade name of Accura 60. A total electrical resistance
of around 40 Ω was measured between ends of the
thread, which proves to be an acceptable value for
activating the whole structure through Joule effect
heating.
Figure 2: Prototype of net-shaped SMP structure with
knitted silver-cloth thread for improving activation.
(Resistance around 40 Ω).
BIODEVICES 2010 - International Conference on Biomedical Electronics and Devices
242

Figure 3: Temporary form obtained through heating,
imposing a 90º deformation between extremes of the
structure and finally cooling down.
Once the prototypes had been fabricated, and the
heating thread knitted to the structure, the “in vitro”
heating was carried out to verify the designed
heating system and show that it is possible to exceed
the glass transition temperature that leads to the
“shape-memory effect” being activated.
This was controlled using a thermographic IR
camera marketed under the name of “Flyr Systems
Thermacam E300” with its accompanying
“Thermacam Reporter 8.0” analytical software
which enables the thermographs taken to be more
exactly and thoroughly analysed.
A remarkable fact for the trials is to verify that
no short-circuit appears during the training process,
which limits the attainable size reduction. When
bending and reducing structure size during training,
the heating thread can separate slightly from the
polymeric material and promote such problems.
Therefore, fixing points for the heating element
have to be taken into account from the design stage,
so as to improve final performance. Another option
consists of totally embedding the heating element
within the polymeric structure during manufacture,
but that leads to additional problems when casting or
injecting into the moulds.
Figure 4: Recovery process by distributed heating. (View
using IR thermography). Total recovery time Æ 20
seconds. Images taken every 5 seconds (image
corresponding to t=0 not included).
INTELLIGENT EXPANDABLE STRUCTURES BASED ON THE IMPROVED ACTIVATION OF SHAPE-MEMORY
POLYMERS
243
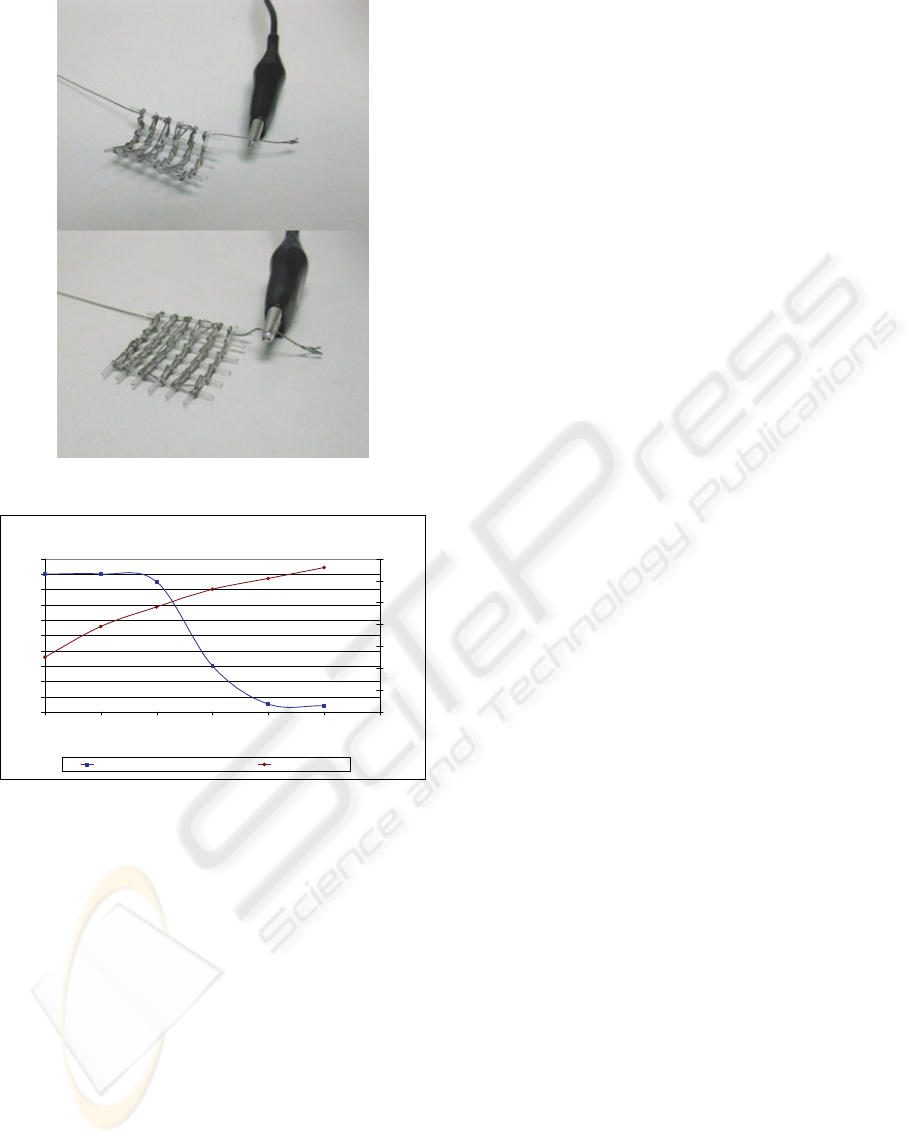
Figure 5: Recovery process by distributed heating.
Recovery of SMP net-shaped structure with improved activation
0
10
20
30
40
50
60
70
80
90
100
0 5 10 15 20 25 30
Time (s)
Bending angle (º)
0
10
20
30
40
50
60
70
Temperature (ºC)
Recover
y
rate of protot
y
pe Temperature
Figure 6: Recovery process (from 90º to 0º) as a function
of time and temperature.
The results show a remarkable homogeneous
heating of the net-shaped structure and the
possibility of going beyond the glass transition
temperature of the polymer, so as to activate the
“shape-memory effect” and the subsequent
geometrical changes.
Additionally, as Figure 5 shows, it is important
to mention that the presence of the silver-cloth
thread for Joule heating does not limit the “shape-
memory effect training” capability or the ability to
recover its original shape through subsequent
heating above glass transition temperature. The
mismatch between original shape “A
0
” and final
shape “A
f
” after activation fulfills the relation:
[A
0
- A
f
] / A
0
< 4%.
5 IMPROVING RESULTS AND
CONCLUSIONS
While the new capabilities brought by these
materials give rise to expectations that many medical
devices will become more effective, considerably
great effort still needs to be put into research and
development, so as to obtain robust and effective
actuators based on these materials.
Some advances linked to obtaining a more
homogeneous heating for improving activation have
been explained. First prototypes of net-shaped SMP
structures with silver-cloth thread knitted to them for
Joule activation have helped to validate the proposal.
It should be mentioned that although the
activation temperature of the materials used of
around 50-55 ºC could not give rise to safe
intracorporeal devices, there are shape-memory
polymers whose activation temperatures are closer
to human body temperature which could be
subjected to a process similar to the one described
here.
The choice of this material was influenced by its
availability in the UPM’s Product Development
Laboratory and by its good processing properties, as
well as its suitability for prototype manufacturing
directly from 3D CAD models, through “laser
stereolithography”. Future work will be focused to
the search and application of SMP formulations with
improved biocompatibility and more suitable
activation temperatures.
Additionally, once having obtained the
prototypes in end materials, different surface
deposits can be used by means of physical or
chemical steam deposition technologies (particularly
the new DLC “diamond like carbon”) coatings, as
well as textile coatings to ensure the
biocompatibility of the device and facilitate its
implantation.
At the same time other medical grade metals and
alloys will be studied as heating coils, so as to obtain
final more appropriate prototypes for “in vitro”
trials. The use of such “in vitro” trials, together with
results from simulations, will help to prepare “in
vivo” studies in a more confident way.
Non-medical applications of this kind of “SMP-
based devices” or “SMP-based smart structures”,
with similar or more complex geometries, can also
be promoted by the possibilities supplied by
distributed threads for Joule activation. Infrared (IR)
thermography proves to be a useful tool for
validating the capabilities of new concept prototypes
and as a support for design tasks.
BIODEVICES 2010 - International Conference on Biomedical Electronics and Devices
244

REFERENCES
Lendlein, A., Kelch, S., 2002. Shape-Memory Polymers.
Angewandte Chemie International.
Lendlein, A., Kelch, S., 2005. Shape-Memory Polymers.
Encyclopedia of Materials: Science and Technology.
Liu, C., Mather, P., 2007. Review of progress in Shape-
Memory Polymers. Journal of Materials Chemistry.
Wache, H., 2003. Development of a polymer stent with
shape-memory effect as a drug delivery system.
Journal of Materials Science – Materials in Medicine.
Lendlein, A., Kelch, S., 2005. Shape-Memory Polymers as
Stimuli-sensitive Implant Materials. Clinical
Hemorheology and Microcirculation.
Lendlein, A., Langer, R., 2002. Biodegradable, Elastic
Shape-Memory Polymers for Potential Biomedical
Applications. Science.
Wilson,T., et al., 2006. Shape-memory Polymer
Therapeutic Devices for Stroke. Lawrence Livermore
National Laboratory.
Small, W., et al., 2005. Laser-activated Shape-Memory
Polymer Intravascular Thrombectomy Device. Optics
Express.
Yakacki, C.M. et al., 2007. Unconstrained Recovery of
Shape-Memory Polymers Networks for
Cardiovascular Applications. Biomaterials.
Yakacki, C.M. et al., 2008. Deformation Limits in Shape-
Memory Polymers. Advance Engineering Materials.
Gall, K.; Kreiner, P. et al., 2004. Shape-memory Polymers
for MEMS Systems. Journal of
Microelechtromechanical Systems.
Díaz Lantada, A., Lafont, P. et al., 2008. Treatment of
Mitral Valve Insufficiency by Shape-Memory Polymer
Based Active Annuloplasty. Biodevices 2008 –
International Conference onBiomedical Electronics
and Devices. INSTICC Press.
Lafont, P., Díaz Lantada et al., 2006. Patent Document
P200603149: Sistema activo de anuloplastia para
tratamiento de la insuficiencia mitral y otras patologías
cardiovasculares. Oficina Española de Patentes y
Marcas.
Bellin, I., et al., 2006. Polymeric Triple-Shape Materials.
Proceedings of the National Academy of Sciences.
Volk, B. et al., 2005. Characterization of Shape-memory
Polymers. NASA Langley Research Centre. Texas
A&M University.
Tobushi, H. et al., 2008. Shape Recovery and
Irrecoverable Strain Control in Polyurethane Shape-
Memory Polymer. Science and Technology of
Advanced Materials.
Liu, C. and Mather, P., 2002. Thermomechanical
Characterization of a Tailored Series of Shape-
memory Polymers. Journal of Applied Medical
Polymers.
Liu, Y. et al., 2003. Thermomechanical Recovery
Couplings of Shape-memory Polymers in Flexure.
Smart Materials and Structures.
Huang, W. and Lee, C., 2006. Thermomechanical
Behaviour of a Polyurethane Shape-memory Polymer
Foam. Journal of Intelligent Material Systems and
Structures.
Liu, Y. et al., 2006. Thermomechanics of shape-memory
polymers: Uniaxial experiments and constitutive
modelling. Int. Journal of Plasticity.
Yakacki, C.M. et al., 2007. Unconstrained recovery of
shape-memory polymer networks for cardiovascular
applications. Biomaterials.
Harrysson, O., et al., 2007, Custom-designed orthopaedic
implants evaluated using FEM analysis of patient
computed tomography data. BMC Musculoskeletal
Disorders.
Paumier, G., et al., 2008. Thermoresponsive Polymer-
Based Microdevice for Nano-Liquid Chromatography.
Biodevices 2008 – Int. Conference on Biomedical
Electronics and Devices. INSTICC Press.
Lendlein, A., et al., 2005. Light-induced shape-memory
polymers. Nature.
Buckley, P., et al., 2006. Inductively Heated Shape-
memory Polymer for the Magnetic Actuation of
Medical Devices. IEEE Transactions on Biomedical
Engineering.
Conti, S., et al., 2007. Modelling and Simulation of
Magnetic Shape-Memory Polymer Composites.
Journal of Mechanics and Physics of Solids.
Yang, B., et al., 2004. On the effects of moisture in a
polyurethane shape-memory polymer. Smart Materials
and Structures.
Yakacki, C.M. et al., 2008. Cytoxicity and
Thermomechanical Behaviour of Biomedical Shape-
Memory Polymer Networks Post-sterilization.
Biomedical Materials.
Cabanlit, M., et al., 2007, Polyurethane Shape-Memory
Polymers Demonstrate Functional Biocompatibility In
Vitro. Macromolecular Bioscience.
Sokolowsky, W., et al., 2007, Medical Applications of
Shape-memory Polymers. Biomedical Materials.
Bar-Cohen, Y., 2006. Artificial Muscles using
Electroactive Polymers (EAP): Capabilities,
Challenges and Potential. SPIE Press.
INTELLIGENT EXPANDABLE STRUCTURES BASED ON THE IMPROVED ACTIVATION OF SHAPE-MEMORY
POLYMERS
245
