
SEED–GROWING HEART SEGMENTATION IN HUMAN
ANGIOGRAMS
Antonio Bravo, Jos´e Clemente
Grupo de Bioingenier´ıa, Decanato de Investigaci´on, Universidad Nacional Experimental del T´achira
San Crist´obal 5001, Venezuela
Rub´en Medina
Grupo de Ingenier´ıa Biom´edica, Facultad de Ingenier´ıa, Universidad de Los Andes, M´erida 5101, Venezuela
Keywords:
Segmentation, Unsupervised clustering, Mean shift, Cardiac images, Human heart, Left ventricle.
Abstract:
In this paper an image segmentation scheme that is based on combinations of a non–parametric technique
and a seed based clustering algorithm is reported. The method has been applied to clinical unsubtracted
angiograms of the human heart. The first step of the method consists in applying a mean shift–based filter in
order to improve the left ventricle cavity information in angiographic images. Second, the initial seed is semi–
automatically generated from the aortic valve manual localization by a specialist. Third, each angiographic
image is segmented using a clustering algorithm that begins with the seed which is grown until image pixels
associated to the left ventricle cavity are clustered. A validation is performed by comparing the estimated
contours with respect to contours manually traced by a cardiologists. From this validation stage the maximum
of the average contour error considering six angiographic sequences (a total of 178 images) is 7.30 %.
1 INTRODUCTION
Image segmentation methods are a prerequisite to ef-
ficiently analyze the cardiac function from image data
acquired by X–Ray angiographic systems during car-
diac exams. Information about the size and shape
of heart cavities during the cardiac cycle could be
extracted. The use of traditional segmenting proce-
dures to cardiac images does not allow to obtain ac-
curate results due to the complexity and variability
of the anatomic shapes. Low contrast, noise, and
non–uniformityof regional intensities are some of the
problems associated with X–ray based cardiac imag-
ing modalities.
Left ventricle (LV) cavity plays a major role in the
heart dynamics since it pumps oxygenated blood to
the entire body. Left ventricle angiograms are ob-
tained from a medical imaging modality based on X–
rays, after the injection of a contrast medium in the
cavities of the heart aiming at enhancing the contrast
with respect to other tissues. Such examination en-
ables the assessment of morphology and function of
the heart. Ventriculographic image analysis requires
a precise description of ventricular shape in order to
quantify the parameters associated with the cardiovas-
cular function (Kennedy et al., 1970) (Rabit, 2000)
or alternatively for performing the visualization of
this anatomical structure (Medina et al., 2004). LV
volumes and ejection fraction (EF) are important in-
dexes for clinical assessments of heart function (Yan
et al., 1978). The accurate description of ventric-
ular function is important, since cardiovascular dis-
ease (CVD) accounts for one third of the deaths in the
world (WHO, 2002).
The general image segmentation problem involves
the division of a dataset into groups of similar ob-
jects. Thus, image segmentation consists in deter-
mining the regions containing pixels that have simi-
lar properties (Jain et al., 1999). There are many dif-
ferent methods for cardiac image segmentation based
on supervised learning methods (Suzuki et al., 2004;
Oost et al., 2006) or unsupervised learning (learning
without teacher) (Sui et al., 2001; Bravo and Med-
ina, 2008) . In general, these methods provided an
accurate representation of ventricular borders, how-
ever, they are not yet fully validated and accepted by
clinicians as a gold standard.
The goal of this research is the developping of
91
Bravo A., Clemente J. and Medina R. (2010).
SEED–GROWING HEART SEGMENTATION IN HUMAN ANGIOGRAMS.
In Proceedings of the International Conference on Computer Vision Theory and Applications, pages 91-96
DOI: 10.5220/0002848900910096
Copyright
c
SciTePress
a left ventricle semi–automatic segmentation method
based on unsupervised clustering. The performance
of the proposed method is quantified by estimating the
difference between the contours obtained by the our
approach with respect to the contours traced by the
cardiologist. The segmentation error is quantified by
using a set of metrics that has been proposed and used
in the literature (Suzuki et al., 2004; Bravo and Med-
ina, 2008). The paper is organized as follows. First,
the complete process that performs the segmentation
of the LV is detailed. Then, experiments carried out
to validate the method are presented, and the results
obtained are discussed. Finally, future developments
are proposed.
2 METHOD
2.1 Dataset
Six mono-plane angiograms acquired at several in-
stants of the cardiac cycle are used to evaluate the
proposed unsupervised clustering method for left ven-
tricle cavity identification. The sequences of ventricu-
lographic images are acquired with a Siemens X–Ray
angiographic system, considering one standard exam-
ination view (right anterior oblique–RAO 30
◦
), with
a field of view (FOV) of 27 cm, a spacing between
pixels of 0.285 mm, a frame rate of 25 images/s and
a total acquisition time of 2 s. The spatial resolution
of the image is 512× 512, with a depth of 8 bits per
pixel.
2.2 Image Enhancement
Non–parametric methods have been widely used in
segmentation problems due to their robustness in sta-
tistical analysis. These methods estimate the data
underlying distributions (unknown density function)
without prior knowledge of the distributions struc-
tures (Fukunaga, 1990).
The mean shift procedure is a non–parametric
technique that does not depend on any geometric
model assumptions (Fukunaga and Hostetler, 1975).
This procedure has been used in image processing
tasks such as filtering and clustering (Comaniciu and
Meer, 2002). The objective of the procedure is to esti-
mate the density gradient based on kernel density esti-
mation method (Parzen window technique). This den-
sity gradient is defined as the direction from a sample
point towards the center of its cluster. The estimated
density gradient (
ˆ
∇f(x)) is used to obtain an estima-
tion (
ˆ
f(x)) of the unknown density function (f(x)).
The main goal of the estimation is to characterize the
modes of the data distribution. These modes are com-
puted by iteratively moving the data according to a
mean shift vector calculated using (1).
m
h
(x) =
1
2
c h
2
ˆ
∇f
h
(x)
ˆ
f
h
(x)
(1)
where h is the bandwidth (smoothing kernel parame-
ter) and c is the normalization constant.
According to (1) the mean shift vector in the point
x is aligned with the local gradient of the estimated
density function. The idea is to track the path leading
x to the stationary point of the estimated density. The
stationary points in the estimated density represent the
modes of the data distribution. If the density function
modes are considered cluster centers, the mean–shift
filtering algorithm shrinks data points towards these
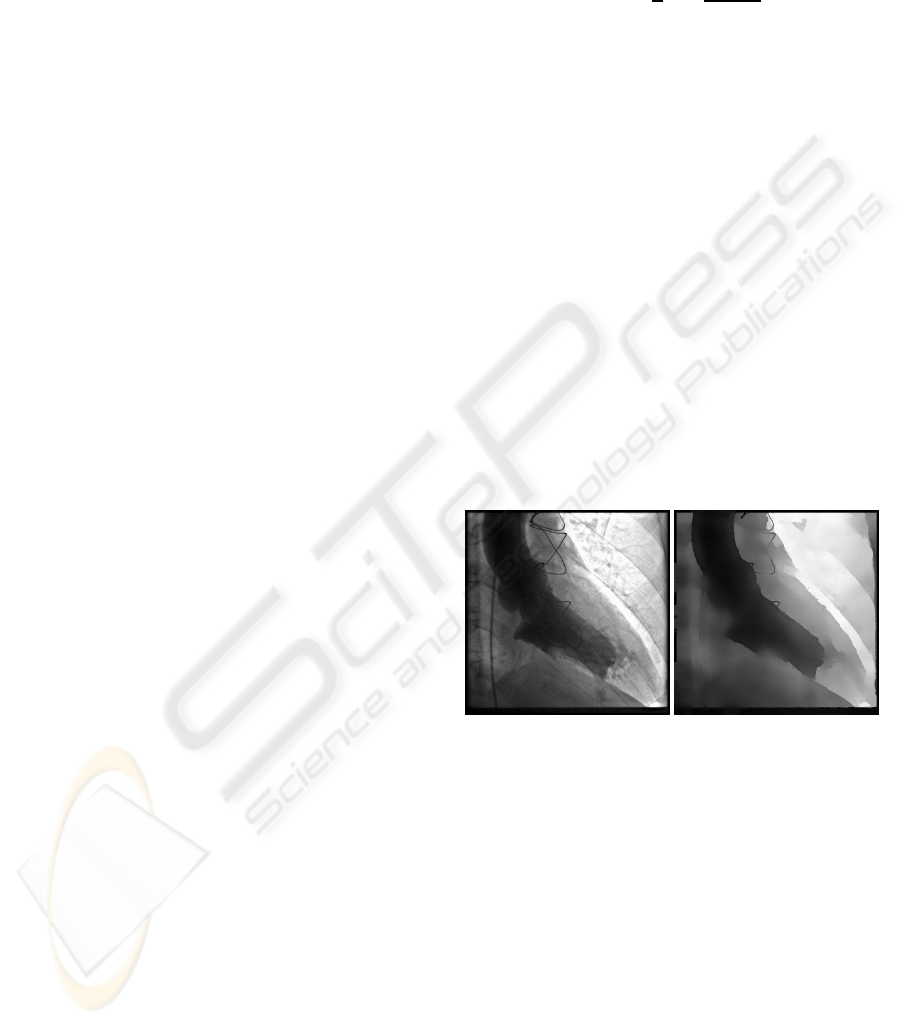
centers. A filtering example is shown in Figure 1.
An end–systole ventriculogram image (RAO view) is
used to illustrate the preprocessing stage. This ven-
triculogram corresponds with the end–systole image
of the patient after a surgical ventricular restoration
(SVR) (Ferrazzi et al., 2006). In the filtered image
(Figure 1.b) is shown that part of the patches of autol-
ogous or artificial material that are placed to close the
defect are removed. Additionally, the left ventricle
cavity information is enhanced.
(a) (b)
Figure 1: Enhancement process. (a) Original image. (b)
Mean shift filtered image.
2.3 Segmentation Stage
A region growing technique is used to segment the
LV. Region growing methods have been used for per-
forming the segmentation of several medical imaging
modalities (Lehmann et al., 2005). The region grow-
ing is usually based on simple linkage, on multiple
connections or centroid based linkage (Haralick and
Shapiro, 1992).
The unsupervised clustering algorithm proposed
in this paper requires a seed point located inside the
region of interest to identify the cardiac cavity. The
seed is established in one angiographic projection
(one time instant of the image sequence).
VISAPP 2010 - International Conference on Computer Vision Theory and Applications
92

2.3.1 Seed Selection Procedure
The seed is used for starting the region–growing seg-
mentation process. This seed is established in the 2–D
image I
t
(t represents the time instant of the angiog-
raphy dataset) according to following procedure:
1. A manual process performed by a cardiologist is
applied to locate the aortic valve sides (VA and
VP).
2. After the aortic valve points are identified, they
are joined starting from the VA point and ending
in the VP point using a straight line. The distance
l between VA and VP points is computed.
3. The midpoint (V
M
) of the line described by VA
and VP points is computed. This midpoint is used
to construct a new line perpendicular to the line
described by VA and VP points.
4. The seed point is the one located at the distance
l measured along the perpendicular line from the
V
M
point. This procedure must guarantee that y
coordinate of the seed point is greater than y coor-
dinate of the V
M
point.
Figure 2 shows the seed selection procedure. A
silhouette of the left ventricle contour is used to illus-
trate the selection procedure.
Figure 2: Seed point selection.
When the image I
t
has been segmented (process
described in section 2.3.2) a binary image
b
I
t
is ob-
tained. In this image, pixels in white represent the
segmented region. From
b
I
t
the seed points neces-
sary to segment the entire angiographic sequence are
estimated. The center of mass of the segmented re-
gion in the image
b
I
t
is calculated and denoted as
r(x, y). The pixel r(x, y) is the new seed to segment
the image I
t+1
.
The procedure based on calculation of the center
of mass, results in a point located very near of the
LV anatomical axis. In consequence, the seed point
is always located inside the target region (inside the
LV).
2.3.2 Region Growing Algorithm
The algorithm is developed using dynamic linked–
lists. The algorithm inputs are the enhanced image
and a binary image with all pixels set to zero (0). The
lists are implemented as a First In First Out (FIFO)
queue. The list is used to store temporarily the pix-
els that fulfill the clustering criterion. The objective is
to develop an iterative algorithm highly efficient with
respect to memory requirements aiming at avoiding
memory overflows. Each node in the list contains the
pixel information: location and gray level intensity.
The first node inserted in the list is the seed pixel. An
overview of the proposed algorithm is shown on the
flowchart in Figure 3. After introducing the seed in
the FIFO list, the algorithm considers the following
steps:
1. The first node of the list is dequeue.
2. The gray level information associated with the an-
alyzed node is compared with pixel intensities in a
8 pixels neighborhood to determine if these neigh-
bor pixels belong or not to the target region. The
pixels of the neighborhood that fulfill the cluster-
ing criterion are inserted at the end of the list and
their values in the binary image are modified to
one (1). The pixels that do not fulfill the condi-
tion are rejected.
3. The algorithm continues with this process while
there are nodes in the list. The algorithm output
is the binary image where pixels values set to one
represent the region of interest.
The uniformity criterion for grouping the pixels is
as follows: pixels are grouped if the difference be-
tween the pixel value in the neighborhood and the in-
tensity of the pixel extracted from the list is below
1
8
of the standard deviation of pixels in the enhanced
image.
2.3.3 Validation
In order to validate the proposed method, the differ-
ence between the estimated LV shape with respect to
a ground truth shape, traced by an expert is quanti-
fied. A methodology to evaluate the LV segmenta-
tion method is considered. The approach proposed
by Suzuki et al. (Suzuki et al., 2004) for evaluating
the performance is incorporated. Suzuki’s quantita-
tive evaluation methodology is based on calculating
two metrics that represent the contour error (E
C
) and
the area error (E
A
). Equations (2) and (3) show the
contour and area errors expressions.
SEED-GROWING HEART SEGMENTATION IN HUMAN ANGIOGRAMS
93

Figure 3: Clustering process.
E
C
=
∑
x,y∈R
E
[a
P
(x, y) ⊕ a
D
(x, y)]
∑
x,y∈R
E
a
D
(x, y)
, (2)
E
A
=
|
∑
x,y∈R
E
a
D
(x, y) −
∑
x,y∈R
E
a
P
(x, y)|
∑
x,y∈R
E
a
D
(x, y)
, (3)
where:
a
D
(x, y) =
1, (x, y) ∈ R
D
0, otherwise
, (4)
a
P
(x, y) =
1, (x, y) ∈ R
P
0, otherwise
, (5)
where R
E
is the region corresponding to the im-
age support, R
D
is the region enclosed by the con-
tour traced by the cardiologist, R
P
is the region en-
closed by the contour obtained by our segmentation
approach, and ⊕ is the exclusive OR operator.
3 RESULTS
Figure 4 shows the results of the segmentation for six
ventriculogramsequences. The contours traced by the
cardiologist are represented by white dashdotted lines
and the contours obtained with our approach are rep-
resented by black dashdotted lines. The first and third
column show the end–systole images while the sec-
ond anf fourth column show the end–diastole images.
Tables 1–3 show a comparison of extracted con-
tours with respect to the contours traced by the car-
diologist. In these tables, the mean, the maximun
(max), the minimun (min) and the standard deviation
(std) for contour and area errors calculated according
to Suzuki’s metrics are shown.
Table 1 shows the errors for 178 images included
in all ventriculographic sequences.
Table 1: Errors obtained for a total of 178 images processed.
min mean max std
E
A
[%] 2.11 3.45 5.71 1.16
E
C
[%] 4.24 7.30 10.38 2.45
Tables 2 and 3 show the errors obtained for end–
diastole and end–systole images, respectively.
Table 2: Contour and area errors for end–diastole images.
min mean max std
E
A
[%] 2.91 4.07 5.23 1.64
E
C
[%] 4.99 6.86 8.72 2.63
The volume for the left ventricle during the car-
diac cycle is estimated using the Area-Length method
(Kennedy et al., 1970) from the contours obtained us-
ing the segmentation method. The correlation values
between the clinical parameters (end–diastole volume
VISAPP 2010 - International Conference on Computer Vision Theory and Applications
94

Figure 4: Results of LV segmentation. Ground truth (white dashdotted line) and estimated contour (black dashdotted line).
Table 3: Contour and area errors for end–systole images.
min mean max std
E
A
[%] 2.19 3.37 4.16 1.07
E
C
[%] 4.24 6.58 9.03 2.40
and end–systole volume) estimated using the LV con-
tours obtained with the proposed method and those
estimated from contours traced by a cardiologist was
determined using the squared correlation coefficient.
The correlation values were 0.9977 for end–diastole
volume and 0.9983 for end–systole volume.
4 DISCUSSION
The approaches recently reported were based on
quantifying only the errors at two cardiac instants:
end–diastole and end–systole. In this research, we
have estimated the segmentation error for all 152 im-
ages in the ventriculographic sequences. The aver-
age contour error obtained using our segmentation
method was 7.30 % which is smaller than the av-
erage error for the end–diastole contour (6.2%) and
end-systole contour (17.1%) reported by Suzuki et
al. (Suzuki et al., 2004). The average contour error
for the end–diastole (4.1%) reported using the semi–
automatic approach presented by Oost et al. (Oost
et al., 2006) is smaller than our value while the error
at end-systole (12.8%) is greater. However, the av-
erage area error (3.45%) obtained by our method is
smaller than the area errors reported in (Suzuki et al.,
2004; Oost et al., 2006).
A strong correlation between the clinical param-
eters (end–diastole volume and end–systole volume)
estimated using the left ventricle contours obtained
with proposed method and those estimated with the
contours traced by cardiologist was determined using
the squared correlation coefficient.
Although our approach has been tested with a
smaller number of sequences of images than the ap-
proaches recently reported, we have considered ven-
triculograms with complex pathologies.
SEED-GROWING HEART SEGMENTATION IN HUMAN ANGIOGRAMS
95

5 CONCLUSIONS
A method for LV image segmentation from an-
giographics sequences was proposed. The pre–
processing stage was based on a mean shift procedure
aimed at performing the smoothing and enhancement
of image contours. A region growing algorithm was
controlled by a seed point located in an image at one
time instant, which was propagated to the rest of in-
stants in order to segment the entire angiographic se-
quence. The comparison was performed based on the
methodology proposed in (Suzuki et al., 2004) which
is also used in (Oost et al., 2006) and (Bravo and Med-
ina, 2008). The validation stage shows that errors are
small. The method allowed to detect LV important
features such as the papillary muscles.
As a future work, a more complete validation is
necessary, including a comparison of estimated pa-
rameters describing the cardiac function with respect
to results obtained using MSCT image modality.
ACKNOWLEDGEMENTS
The authors would like to thank the Investigation
Dean’s Office of Universidad Nacional Experimen-
tal del T´achira and LOCTI grant PR0100401 for their
support to this project.
REFERENCES
Bravo, A. and Medina, R. (2008). An unsupervised clus-
tering framework for automatic segmentation of left
ventricle cavity in human heart angiograms. Comput
Med Imaging Graph., 32(5):396–408.
Comaniciu, D. and Meer, P. (2002). Mean shift: A robust
approach toward feature space analysis. IEEE Trans.
Pattern Anal. Machine Intell., 24(5):603–619.
Ferrazzi, P., Matteucci, M., Merlo, M., Iacovoni, A.,
Rescigno, G., Bottai, M., Parrella, P., Lorini, L.,
Senni, M., and Gavazzi, A. (2006). Surgical ven-
tricular reverse remodeling in severe ischemic dilated
cardiomyopathy: the relevance of the left ventricular
equator as a prognostic factor. J Throac Cardiovasc
Surg, 131(2):357–363.
Fukunaga, K. (1990). Introduction to Statistical Pattern
Recognition. Academic Press Inc, USA.
Fukunaga, K. and Hostetler, L. (1975). The estimation of
the gradient of a density function, with applications
in pattern recognition. IEEE Trans. Inform. Theory,
21(1):32–40.
Haralick, R. and Shapiro, L. (1992). Computer and Robot
Vision, volume I. Addison-Wesley Publishing Com-
pany, USA.
Jain, A., Murty, M., and Flynn, P. (1999). Data clustering:
a review. ACM Comp. Surv, 31(3):264–323.
Kennedy, J., Trenholme, S., Kaiser, I., and Wash, S. (1970).
Left ventricular volume and mass from single–plane
cineangiocardiogram. A comparison of anteroposte-
rior and right anterior oblique methods. Am Heart J,
80(3):343–352.
Lehmann, T. M., Beier, D., Thies, C., and Seidl, T.
(2005). Segmentation of medical images combining
local, regional, global, and hierarchical distances into
a bottom-up region merging scheme. In Proceedings
of SPIE, volume 5747, pages 546–555.
Medina, R., Garreau, M., Toro, J., Coatrieux, J. L., and
Jugo, D. (2004). Three–dimensional reconstruction
of left ventricle from two angiographic views: An ev-
idence combination approach. IEEE Transactions on
Systems, Man, and Cybernetics—Part A: Systems and
Humans, 34(3):359–370.
Oost, E., Koning, G., Sonka, M., Oemrawsingh, P. V.,
Reiber, J. H. C., and Lelieveldt, B. P. F. (2006). Auto-
mated contour detection in X–ray left ventricular an-
giograms using multiview active appearance models
and dynamic programming. IEEE Trans. Med. Imag.,
25(9):1158–1171.
Rabit, O. (2000). Quantitative analysis of cardiac function.
In Bankman, I. N., editor, Handbook of Medical Imag-
ing: Processing and Analysis, pages 359–374. Aca-
demic Press, San Diego.
Sui, L., Haralick, R., and Sheehan, F. (2001). A knowledge–
based boundary delineation system for contrast ven-
triculograms. IEEE Trans. Inform. Technol. Biomed.,
5(2):116–132.
Suzuki, K., Horiba, I., Sugie, N., and Nanki, M. (2004). Ex-
traction of left ventricular contours from left ventricu-
lograms by means of a neural edge detector. IEEE
Trans. Med. Imag., 23(3):330–339.
WHO (2002). Reducing risk and promoting healthy life.
The World Health Report 2002 Geneva, World Health
Organization.
Yan, S., Lamberto, B., Vladir, M., and Harry, G. (1978).
From Cardiac Catheterization Data to hemodynamic
Parameters. F. A. Davis Company, USA.
VISAPP 2010 - International Conference on Computer Vision Theory and Applications
96
