
Blood Propofol Concentrations: On Demand for
Pharmacokinetic/Pharmacodynamics Models and New
Measurement Automatic Technologies
S. Campos
1
, A. Silva
1
, P. G. de Pinho
2
and L. Antunes
1
1
Veterinary Sciences Department, Universidade de Trás-os-Montes e Alto Douro
Quinta de Prados, Apartado 1013, 5001-801 Vila Real, Portugal
2
Laboratory of Toxicology, Department of Biological Sciences, Faculty Pharmacy
Universidade do Porto, Porto, Portugal
Abstract. Propofol is a widespread anaesthetic agent in human medicine. It
allows the practice of total intravenous anaesthesia through the use of
sophisticated delivery systems due to its pharmacokinetic/ pharmacodynamics
properties.
Three New Zealand White rabbits were anaesthetized with three different
infusion rates (70, 100, 130 mg.kg
-1
.h
-1
) in a random order, during thirty
minutes each. Clinical parameters as well as arterial blood samples were
collected at specific time points. Rugloop II Vet software was used to storage
all data and to predict the propofol concentrations during the anaesthetic period.
A correlation analysis was done between real and predicted propofol
concentrations, in each infusion rate.
It was only observed a significant correlation between concentrations during the
lower infusion rate (70 mg.kg
-1
.h
-1
).
There is a lack of automatic devices and pharmacokinetic/pharmacodynamics
models that allow a real-time or predicted measurement of the propofol
concentrations in the patient, for veterinary medicine.
1 Introduction
During the last decades, the growing knowledge about the pharmacokinetic properties
of propofol allowed a continuous remodeling of pooled data, describing together
information of clinical trials and adequate modeling for the specie in which it is used
[6]; [7]. These characteristics are very well documented in humans and, in veterinary
medicine some research groups have already reported pharmacokinetic data for
propofol, especially in the dog and the cat [3]; [5]; [1]; [7].
From a pharmacokinetic point of view, propofol remains the best controllable
intravenous hypnotic, since it has a huge body uptake and a fast elimination due to a
large apparent volume of distribution and a high clearance [6].
Drug delivery systems, based on pharmacokinetic and pharmacodynamic
properties of propofol, were developed allowing a reasonable real-time estimation of
its plasma concentrations, during the anaesthetic period [6]; [9]. This can be equated
with the continuous measurement of end-tidal volatile anaesthetic agent concentration
Campos S., Silva A., G. de Pinho P. and Antunes L..
Blood Propofol Concentrations: On Demand for Pharmacokinetic/Pharmacodynamics Models and New Measurement Automatic Technologies.
DOI: 10.5220/0003880900180023
In Proceedings of the International Workshop on Veterinary Biosignals and Biodevices (VBB-2012), pages 18-23
ISBN: 978-989-8425-94-2
Copyright
c
2012 SCITEPRESS (Science and Technology Publications, Lda.)

that led to the concept of MAC (Minimum Alveolar Concentration) and provides the
best available method to monitor continuous brain concentration, nowadays.
Pharmacodynamic properties of propofol depend on its therapeutic plasma
concentrations. The knowledge of pharmacokinetic models allows a more accurately
prediction of the optimal dosage [9]. The required plasma concentration varies with
the desired pharmacological effect (sedation, induction or maintenance of
anaesthesia), the simultaneous use of other drugs (opioids, muscle relaxants), the type
of operation and the patient’s sensitivity to the drug (age, weight and pre-existing
diseases) [9]. So, the use of propofol demands a continuous titration of the drug
infusion rate to the desired pharmacological end point, however there is an absence of
a clinically useful method for measuring blood propofol concentrations.
In this study, an analysis over real propofol concentrations together with predicted
concentrations was performed using a rabbit model.
2 Methodology
2.1 Animals
All procedures were carried out under personal and project licenses approved by the
national regulatory office (Direcção Geral de Veterinária – DGV). Three healthy male
New Zealand White rabbits, approximately 2 months old, were used.
2.2 Anaesthetic Protocol
Anaesthesia was induced with a dose of 20 mg.kg
-1
propofol (Propofol Lipuro®, B.
Braun Melsungen, Germany) intravenously, using a syringe pump (Asena GH, Alaris
Medical Systems) controlled by the Rugloop II software (developed by Tom DeSmet
(Demed Engineering, Gent, Belgium)) at an infusion rate of 200 ml.h
-1
. Following
blind orotracheal intubation with a cuffed endotracheal tube with 2.5 mm in internal
diameter, propofol started to be administered according to an infusion scheme in
which three infusion rates were used in every animal: each infusion (70, 100 and 130
mg.kg
-1
.h
-1
) was maintained during thirty minutes. The order of the administration
rates chosen for each animal was random.
Monitoring of anaesthesia included cardio-respiratory parameters (heart rate (HR),
mean arterial blood pressure (MABP), arterial blood oxygen (SpO
2
) and respiratory
rate (RR)), temperature (T), clinical evaluation of depth of anaesthesia (DoA) and the
Index of Consciousness (IoC).
Anaesthetic monitoring included cardio-respiratory monitoring provided by a
Datex S/5 Anaesthetic station (Datex Ohmeda, Helsinki, Finland) which included:
pulse-oxymetry and pulse rate monitored with the probe placed in the ear, invasive
mean arterial blood pressure (MBAP), inspired and end-tidal concentrations of
oxygen and carbon dioxide. The animals were under mechanical ventilation with
100% oxygen, with ventilation parameters set to maintain the end-tidal CO
2
(ETCO
2
)
between 35 and 45 mmHg. At the end of the infusion scheme fresh gas flow rate was
19
increased to 5 L.min
-1
of 100% oxygen until the rabbits regained swallowing reflexes
and at this point extubation was performed. Animals were considered recovered from
anaesthesia when they exhibited an alert stance and had regained ambulation and
limbs coordination.
2.3 Data Acquisition
Data were stored using the Rugloop II Vet software that was also used as software to
predict the propofol concentrations at the different time points. This software uses the
Beths’ pharmacokinetic/ pharmacodynamics parameters for propofol in order to
predict its concentration in blood.
2.4 Blood Sampling
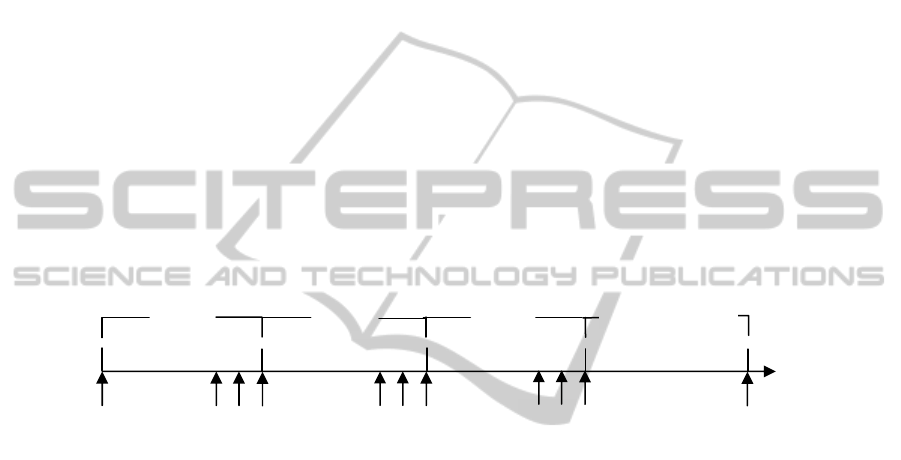
Arterial blood samples were collected before the beginning of anaesthesia and at three
time points in each infusion rate (20, 25 and 30 minutes after the start of the infusion
rate) and in the totally recovered animals (Figure 1). This infusion scheme was
designed to achieve a steady-state, based on pharmacokinetic data of clearance from
Cockshott et al. [3].
Fig. 1. Schematic representation of blood samples (S) collection. S
0
represents the baseline
sample; S
1
, S
2
and S
3
represent the blood sample collection of each infusion rate. S
1
is collected
20 minutes after the beginning of each infusion rate; S
2
is collected 5 minutes after S
1
and S
3
is
collected 5 minutes after S
2
. S
rec
is the blood sample collection from recovered animals.
2.5 Propofol Quantification in Blood Serum
Propofol concentrations were determined by Gas Chromatography/ Ion Trap - Mass
Spectrometry (GC/IT-MS) in rabbit serum. This method was adapted from that
previously described by Guitton et al. 1995, used to quantify propofol in human blood
[4].
2.6 Statistical Analysis
Statistical analyses were performed by using Excel® and GraphPad Prism®
(GraphPad Prism, version 5.00 for Windows, GraphPad Software, San Diego,
California, USA, and www.graphpad.com). A Kolmogorov-Smirnov test [8] was used
to test data for normality.
Recovery
Third infusion rate
First infusion rate
Second infusion rate
S
0
S
1
S
2
S
3
S
1
S
2
S
3
S
1
S
2
S
3
S
rec
30 min. 30 min.
30 min.
Undetermined
20

Pearson and Spearman Rank correlation analysis were used to study the
correlation between real propofol concentrations and the propofol concentrations
calculated by Rugloop II.
3 Results and Discussion
In this study, three rabbits weighing 2.79±0.25 Kg were successfully anaesthetized
with three different propofol infusion rates (70, 100 and 130 mg.kg
-1
.h
-1
) in a
randomized order, during thirty minutes.
Anaesthetic induction was smooth in all animals, without any excitatory
movements and was achieved with a propofol bolus of 20 mg.kg
-1
. Due to the
randomization of the infusion schemes, the same infusions and its duration did not
produce the same propofol plasma concentrations.
The duration of the infusions was stipulated based on pharmacokinetic data of
propofol in rabbits from Cockshott et al. [3]. A ten minute steady-state at the end of
each infusion rate was produced, according to data from these authors. Table 1 shows
the infusion schemes that were made for each rabbit.
Table 1. Propofol infusion schemes order and respective rabbit.
Rabbit(s)
Infusion scheme
(mg.kg
-1
.h
-1
)
1 and 2
70→130→100
3
100→70→130
As it can be observed in the graphs, the propofol concentrations predicted by the
software are underestimated when compared with the real propofol concentrations,
particularly at high infusion rates. This is especially important due to the dose-
dependent effects of anaesthetics that should be precisely measured in the blood and
automatically adjusted to the desired level of anaesthetic depth and hemodynamic
answers of the animal. Besides, it was only observed a very significant correlation
(r=0,833; P=0,0083) between real propofol concentrations and predicted
concentrations during the lowest infusion rate (70 mg.kg
-1
.h
-1
). The other two infusion
rates did not show significant correlations. This indicates that the pharmacokinetic/
pharmacodynamic parameters incorpored in VET Rugloop II are not adequate for
estimation of propofol concentrations when infusion rates above 70 mg.kg
-1
.h
-1
are
used, in rabbits. This can be justified because the pharmacokinetic/
pharmacodynamics parameters from the VET Rugloop II are based on the Beths
model for dogs, which are suited for other species [2].
Despite many advantages over traditional volatile anaesthetic techniques, propofol
total intravenous anaesthesia makes up a small percentage of general anaesthetics
administered. One of the reasons for this is the absence of a clinically useful method
for measuring blood propofol concentrations.
The knowledge of the real propofol plasma concentration achieved at each study
moment may allow a more precise analysis of the effects of propofol on clinical signs
21

R
1
00
S
1
R
1
00
S
2
R
1
00
S
3
R
70
S
1
R
70
S
2
R
70
S
3
R
1
30
S
1
R130
S
2
R130
S
3
0
20
40
60
80
100
120
140
0
10
20
30
40
50
Infusion rate
Real PC
Predicted PC
Rabbit 3
Infusion rate (mL.h
-1
)
Propofol concentration (
μ
g.mL
-1
)
Fig. 2. Graphic representations of real propofol plasma concentration, predicted propofol
concentration and for infusion rate to rabbit 1, 2 and 3. In the x-axis is represented each blood
sample collection (S1- sample 1; S2 – sample 2 and S3 – sample 3) for each propofol infusion
R
70S1
R
70S2
R
70S3
R1
3
0
S
1
R1
3
0
S2
R
1
3
0
S3
R
1
0
0
S1
R
1
0
0
S2
R10
0
S3
0
20
40
60
80
100
120
140
0
10
20
30
40
50
Real PC
Predicted PC
Infusion rate
Rabbit 1
Infusion rate (mL.h
-1
)
Propofol concentration (
μ
g.mL
-1
)
R
70
S1
R
70
S2
R70
S3
R130S
1
R
13
0
S
2
R
13
0
S
3
R
1
00
S
1
R
1
00
S
2
R100S3
0
20
40
60
80
100
120
140
0
10
20
30
40
50
Real PC
Predic ted PC
Infusion rate
Rabbit 2
Infusion rate (mL.h
-1
)
Propofol concentration (
μ
g.mL
-1
)
22

rate (R70 – infusion rate of 70 mg.Kg
-1
.h
-1
; R130 – infusion rate of 130 mg.Kg
-1
.h
-1
and R100 –
infusion rate of 100 mg.Kg
-1
.h
-1
).
of the anaesthetized animals. This is particular important attending to the different
animal species that demand the development of pharmacokinetic/pharmacodynamic
models for each one of them, in order to respond to the individual variance between
species.
In conclusion, regarding the intravenous anaesthesia in the veterinary medicine
there is a huge lack in the offer of devices and pharmacokinetic/pharmacodynamics
models that allow an accurate and real-time measurement (predicted or real) of the
intravenous anaesthetics in the animal, for immediate adjustment of the anaesthetic
plane.
Acknowledgements
This investigation was supported by FEDER funds through the COMPETE Program,
and by national funds from Portuguese Foundation for Science and Technology,
under the project COMPETE: FCOMP-01-0124-FEDER-009497 PTDC/CVT/
099022/2008).and personal grant from FCT: SFRH/BD/72360/2010.
References
1. Beths, T., J. B. Glen, et al. (2001). "Evaluation and optimisation of a target-controlled
infusion system for administering propofol to dogs as part of a total intravenous anaesthetic
technique during dental surgery." Vet Rec 148(7): 198-203.
2. Bras, S., N. Bressan, et al. (2009). "A step towards effect-site target-controlled infusion
with propofol in dogs: a k(e0) for propofol." J Vet Pharmacol Ther 32(2): 182-8.
3. Cockshott, I. D., E. J. Douglas, et al. (1992). "The pharmacokinetics of propofol in
laboratory animals." Xenobiotica 22(3): 369-75.
4. Guitton, J., M. Desage, et al. (1995). "Quantitation of propofol in whole blood by gas
chromatography-mass spectrometry." J Chromatogr B Biomed Appl 669(2): 358-65.
5. Hall, L. W., E. Lagerweij, et al. (1994). "Effect of medetomidine on the pharmacokinetics
of propofol in dogs." Am J Vet Res 55(1): 116-20.
6. Schuttler, J. and H. Ihmsen (2000). "Population pharmacokinetics of propofol: a
multicenter study." Anesthesiology 92(3): 727-38.
7. Sneyd, J. R. (2004). "Recent advances in intravenous anaesthesia." Br J Anaesth 93(5):
725-36.
8. Sokal, R. S. and F. J. Rohlf (1995). Biometry. The Principles and Practice of Statistic in
Biological Reasherch. New York, Freeman: 716-21.
9. White, P. F. (2005). "Intravenous (non-opioid) anesthesia." Seminars in Anesthesia,
Perioperative Medicine and Pain 24: 101-107.
23
