
TOWARDS UNOBTRUSIVE AUTOMATED SLEEP STAGE
CLASSIFICATION
Polysomnography using Electrodes on the Face
Igor J. Berezhnoy, Gert-Jan de Vries, Tim Weysen, Jonce Dimov and Gary Garcia-Molina
Philips Research Laboratories, High Tech Campus 34, 5656 AE Eindhoven, The Netherlands
Keywords:
Sleep, Automated Scoring, Comfortable Polysomnography.
Abstract:
Although sleep stage annotation (SSA) is historically known from clinical practice and typically performed
by a certified expert on the basis of visual examination of polysomnography (PSG) signals. Automatic SSA
has emerged as a tool to assist sleep experts and to accelerate the analysis of PSG data. New advances in
signal processing and sensor technology start to enable the application of SSA in home solutions as well.
In today’s busy lives, sleep plays a central role and good quality sleep helps us to deal with the stress of
everyday life. Being able to enhance sleep quality thus is a major opportunity to help people in reducing the
influence of stress on their live, health and wellbeing. The advent of consumer products aimed at enhancing
the sleep experience has propelled the need for home sleep monitoring and inducing solutions which can i)
provide automatic SSA using sensors that interfere minimally with the sleep process and ii) provide sleep
stage information in real-time in order to be suitable for closed-loop sleep inducing solutions. In this paper,
we examine two possible alternatives for unobtrusive sleep monitoring. The first one uses respiratory, cardiac
and wrist actigraphy signals while the second one relies on Facial PSG electrodes positioned on the facial area
which allow for unobtrusive and comfortable sensors arrangements.
1 INTRODUCTION
Sleep stage scoring in clinical practice results from
manual visual scoring on the basis of Polysomnogra-
phy (PSG) data. Typical PSG setups require affixing
electrodes at various locations of the patient’s body to
capture a number of physiological signals including
electroencephalogram (EEG), cardiac activity, respi-
ratory effort, ocular activity (EOG), and chin myo-
graphic activity (EMG). Consumer applications rely-
ing on sleep monitoring do not require the same level
of accuracy as clinical practice. In addition, the level
of obtrusiveness of traditional clinical PSG setups is
unacceptable in the consumer domain.
In this paper, we consider two minimally obtru-
sive sleep monitoring approaches. The first approach,
which we refer to as RHA, is based on the monitoring
of respiratory effort, cardiac signals, and wrist actig-
raphy. The second approach, which we refer to as
Facial PSG, is based on the measurement of signals
from two electrodes positioned on a single side of the
subjects’s face.
To establish a reference for our study, in Table 1
we present average agreement scores between two
sleep stage scoring approaches i) clinical sleep scor-
ing (performed by trained human experts and scoring
according to criteria of (Rechtschaffen and A.Kales,
1968), and ii) an automated scoring techniques. Both
of their approaches use data of the full PSG setup
(EEG, EOG and EMG).
Table 1 shows the level of agreement between
raters in terms of i) Cohen’s Kappa statistics (sec-
ond column) and ii) percentage of agreement (third
column). It is important to note that the reported
values in Table 1 were not obtained from the same
dataset. They originate from various studies: i) av-
erage agreement between two independent sleep ex-
perts (row two) observing the same PSG data has been
reported in (Virkkala, 2005), ii) comparison of au-
tomated techniques has been performed on the data
recorded in this study.
Table 1: Sleep stages classification agreement figures.
Cohen’s Kappa Agreement(%)
Human expert(PSG) vs. Hu-
man expert (PSG)
0.80 86.00
Automated technique A
(PSG) vs. Automated
technique B (PSG)
0.69 85.48
487
J. Berezhnoy I., de Vries G., Weysen T., Dimov J. and Garcia-Molina G..
TOWARDS UNOBTRUSIVE AUTOMATED SLEEP STAGE CLASSIFICATION - Polysomnography using Electrodes on the Face.
DOI: 10.5220/0003890104870492
In Proceedings of the International Conference on Health Informatics (BSSS-2012), pages 487-492
ISBN: 978-989-8425-88-1
Copyright
c
2012 SCITEPRESS (Science and Technology Publications, Lda.)

From the agreement figures shown in the Ta-
ble 1 we can conclude that agreement between human
raters is significantly higher than that of automated
techniques with expert annotation despite operating
on the same kind of input data (PSG).
In this paper we compare performances of the
RHA and Facial PSG approaches on the task of auto-
mated epoch bases sleep state annotation. This paper
is organized as follows. Section 2 describes the exper-
imental setup. In Section 3 we present the signal pro-
cessing and machine learning methods that were used
in this study. The results are discussed in Section 4.
Finally Section 5 summarizes the main conclusions of
this work and proposes future research directions.
2 EXPERIMENT DESCRIPTION
This study considers two data sets: First and Second,
acquired in two separate studies. The First dataset in-
cludes respiratory effort, cardiac, and acti-graphy sig-
nals. The RHA approach was tested on the First data
set.
The Second dataset was used for validation of the
Facial PSG approach which had as a major objective
to show that sleep state estimation can be performed
with electrodes applied on a single side of the face
(for which we used EOG-Left placed above the eye
and mastoid reference). Further details on the datasets
are provided in the next sections.
2.1 FirstDataset
The First data set contains overnight PSG recordings
of six young healthy volunteers (mean age 27 y.o., 4
males and 2 females). In a screening phase, selec-
tion of participants was based on absence of subjec-
tive sleep complaints and regular sleep/wake patterns.
Participants entered the sleep laboratory at 21.00h
and were prepared for PSG measurements. Lights
were turned off at around 23.00h. The waking up sig-
nal was given around 7 o’clock.
Sleep recordings and analysis Polysomnographic
sleep recordings were obtained during all sleep
episodes with a digital recorder, and included EEG
(F3/A2, F4/A1, C3/A2, C4/A1, O1/A2, O2/A1)
sampled at 100Hz, electrooculogram (EOG) sam-
pled at 100Hz, electrocardiogram (ECG) sampled
at 500Hz and chin electromyogram (EMG) sam-
pled at 200Hz. Respiratory effort was measured
with chest and abdominal respiratory effort belts at
10Hz. Obtained PSG recording were scored into
sleep stages using 30s epochs according to standard
criteria (Rechtschaffen and A.Kales, 1968) by the
Alice Philips Resperonics system and further proof
checked by human experts.
2.2 SecondDataset
The Second data set contains overnight PSG record-
ings of six young healthy volunteers (mean age 27, 4
males and 2 females). They signed a consent form. In
a screening phase, selection of participants was based
on absence of subjective sleep complaints and regu-
larity of sleep/wake patterns. Screening was based
on two questionnaires: the Sleep Disorders Question-
naire (SDQ) (Douglass et al., 1994) and the Pittsburgh
Sleep Quality Index (PSQI) (Buysse et al., 1989). All
selected participants scored within the normal range
of the PSQI. Moreover, the participant should not
score higher than the cutoff scores on the subscales
narcolepsy, apnea, restless legs, and psychiatry of the
SDQ (Douglass et al., 1994).
Participants entered the sleep laboratory at 21.00h
and were prepared for Polysomnographic measure-
ments. Lights were turned off at around 0.00h. The
waking up signal was given around 7 o’clock.
Polysomnographic sleep recordings were ob-
tained during all sleep episodes with a digital
recorder (Vitaport-3, TEMEC Instruments, Kerkrade,
Netherlands), and included EEG (F3/A2, F4/A1,
C3/A2, C4/A1, O1/A2, O2/A1) obtained with the
Sleep BraiNet system (Jordan NeuroScience, San
Bernardino, CA), electrooculogram (EOG), electro-
cardiogram (ECG) and chin electromyogram (EMG).
Respiratory effort was measured with Pro-Tech chest
and abdominal respiratory effort belts (Pittsburgh,
USA). The signals were recorded at a sampling fre-
quency of 256Hz. Obtained PSG recording were
scored by the Siesta Group’s software system - ”Som-
nolyzer” and further proof checked by Siesta’s ex-
perts.
3 METHODS
3.1 Feature Extraction
In the following two subsections we describe the
data preprocessing and feature extraction methodol-
ogy used for both the RHA and the Facial PSG ap-
proaches.
3.1.1 RHA Features
The raw respiration signal was first lowpass fil-
tered (cut-off 0.5Hz) and then analyzed for individ-
ual breaths. Based on a localized min/max filter, lo-
HEALTHINF 2012 - International Conference on Health Informatics
488

Table 2: List of features in the RHA approach, extracted from respiration, heart and wrist actigraphy signals.
Feature number Heart rate features: Description
1 Mean Average heart rate
2 Median Median heart rate
3 Standard deviation Standard deviation of the heart rate
4 Gradient Inclanation coefficient, shows whether heart rate goes up or down within the epoch
5 Variability Heart rate variability
Actigraphy features:
6 Amount of motion over time Scalar with values between 0 and 1, shows relative amount of motion detected
Respiration frequency features:
7 Mean Average respiration rate
8 Median Median respiration rate
9 Standard deviation Standard deviation of the respiration rate
10 Gradient Inclanation coeficiant, shows whether respiration rate goes up or down within the epoch
Respiration amplitude features:
11 Mean Average of amplitudes of respiration cycles
12 Median Median of amplitudes of respiration cycles
13 Standard deviation Standard deviation of amplitudes of respiration cycles
14 Gradient Inclination coefficient, shows whether amplitude goes up or down within the epoch
cal minima and maxima were detected. When found
in the right order, they characterize a single breath.
Based on the distribution of identified breath ampli-
tudes in a signal, breaths which were too small or too
large (outliers) were removed.
In a similar manner, the ECG signal is lowpass fil-
tered (cut-off 5 Hz) and de-trended. Individual heart
beats are detected using pattern matching. Again, out-
lier removal is applied and the resulting signal is a se-
quence of inter beat intervals (IBIs), which has been
transformed into (instantaneous) heart rate (in bpm)
by taking its reciprocal and multiplying by 60. The
wrist actigraphy signal has been low-passed using a
running average filter (5x30 seconds epochs in size)
and further normalized on a unit interval.
Figure 1: Respiration rate, heart rate and actigraphy signals
as they change over the night, plotted along the two hypno-
grams from two experts. Colors in the lower graph represent
sleep states.
Sleep is normally scored (Rechtschaffen and
A.Kales, 1968) in non-overlapping 30-second long in-
tervals (epochs). Thus, features of respiration, heart
and actigraphy signals are calculated on a per-epoch
basis. Table 2 lists the complete set of features ex-
tracted from the signals.
3.1.2 Features in the Facial PSG Approach
The raw signal used for feature extraction in the Fa-
cial PSG approach was recorded by electrodes placed
at the following three standardized locations:(1) upper
left eye (EOG Left), (2) left mastoid bone (reference
A1) and (3) ground electrode at the neck of the partic-
ipant. Given this setup for signal extraction the signal
recorded at A1 channel was subtracted from the sig-
nal of the EOG L channel. Furthermore, to estimate
the power spectral density of each epoch, we applied
Welch’s method (Welch, 1967). Figure 2 shows re-
sults of the Welch’s method where the color repre-
sents the power at a certain frequency (top plot).
To facilitate the visual interpretation of the rela-
tion between the Welch’s power plot(features) and
the reference scoring (labels), the bottom plot in the
figure shows corresponding hypnogram and the mid-
dle plot shows power plot but specifically for low
frequencies which correspond to deeper sleep (a.k.a.
slow wave sleep, SWS). As it can be easily seen,
peaks of power in SWS plot correspond to N3 sleep
stages of the hypnogram.
For the machine learning part of the Facial
PSG approach input-output pairs were constructed in
the following manner: for each epoch, a power spec-
trum vector was computed and coupled with a sleep
stage label. This resulted in about 800 input-output
pairs per subject (corresponding to 7 hours of sleep).
TOWARDS UNOBTRUSIVE AUTOMATED SLEEP STAGE CLASSIFICATION - Polysomnography using Electrodes
on the Face
489
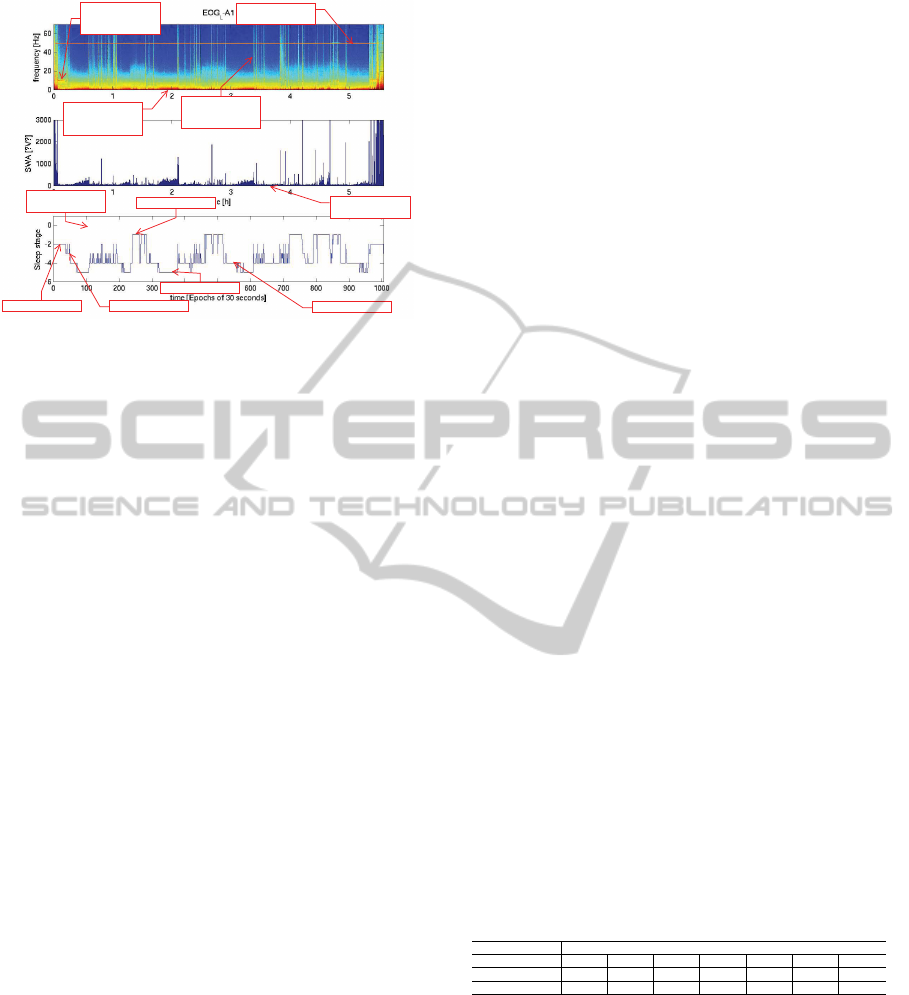
50Hz power line
artifact
High density of low
frequency, deeper
sleep
stripes are
motions, either
eyes or body
only low frequency
power plot
PSG based,
Hypnogram
-1 -REM
-2 wake
-3 N1 stage
-4 N2 stage
-5 N3 stage
A
lfa (11Hz), eyes
closed, but not
asleep yet
Figure 2: Facial PSG approach, features and labels (from
top-to bottom): (1) Signal power vs. frequency over
time, (2) Low frequency(deeper sleep) power over time,
(3) hypnogram plot.
3.2 Classification
Robust Soft Learning Vector Quantization (RSLVQ)
is one of many LVQ variants, originally developed
by Kohonen (Kohonen, 1995). This family of ma-
chine learning algorithms has been applied to classi-
fication problems in many fields (Centre, 2002) and
is characterized by its computational efficacy. LVQ
is a method of prototype-based, multi-class classi-
fication, where each class is represented by one or
more prototypes. A prototype is defined as a point
in the N-dimensional feature space with an accom-
panying class label, and trained by sequential han-
dling of training data. Each time a training sample is
presented, the closest prototypes with correct and in-
correct label are pulled towards or pushed away from
the training sample, respectively. When training pro-
gresses, the prototypes will progressively better rep-
resent the classes. When applied to unseen data, clas-
sification is performed by returning the label of the
closest prototype. Usually Euclidean distance is used
as distance measure.
In a recent study (Witoelar et al., 2010), the per-
formance of several LVQ variants in a controlled envi-
ronment was analyzed. The (relative) robustness and
convergent properties (i.e., insensitivity to overtrain-
ing) motivated our choice for RSLVQ, as proposed
in (Seo and Obermayer, 2003). In this ’soft’ version of
LVQ the magnitude of displacement of prototypes in
each training step is relative to their distance from the
training sample. This method makes an assumption
on the distribution of data samples around the pro-
totypes, which we chose to be Gaussian with equal
variances (for each prototype). The total distribution
of data from a single class therefore is assumed to be
a mixture of Gaussian distributions.
3.3 Performance Measurement
In order to allow in depth comparisons of the two
techniques we present classification results of both
experiments in the shape of confusion matrixes.
Essentially confusion matrix contains three widely
known (in classification tasks assessments) compari-
son entities: (1) confusion matrix, (2) percentage of
agreement and (3) Cohen’s Kappa agreement coef-
ficient. The confusion matrix can be used for de-
tailed assessment of classifier’s performance in terms
of which classes are often mistaken for what other
classes. Furthermore they allow calculation of a base-
line performance based on just class priors. Since we
were mostly interested in overall performance assess-
ment, in section 4 for each cycle of the cross valida-
tion scheme we only present it’s outcome with two
values: (1) percentage of agreement and (2) Cohen’s
Kappa coefficient.
4 RESULTS AND DISCUSSION
This section presents the results obtained by two
sleep monitoring approaches, namely Facial PSG and
RHA. Section 4.1 reports the results obtained with Fa-
cial PSG while Section 4.2 reports the results obtained
with the RHA approach. Both subsections contain
tables presenting percentages of agreement and Co-
hen’s Kappa coefficients per cross validation run, as
well as overall agreement matrices.
4.1 Facial PSG Results
Table 3 shows Cophen’s Kappa and percentage of
agreement figures per run of the cross-validation
scheme. The last column contains average values.
Table 3: Facial PSG approach results, per cross validation
run (per subject).
Subjects (Second study)
1 2 3 4 5 6 mean
Agreement % 76.20 71.43 71.46 64.71 80.91 82.07 74.46
Cohen’s Kappa 0.66 0.58 0.62 0.50 0.69 0.74 0.63
Table 4 shows the overall agreement matrix that
contains: confusion matrix (in bold), percentage of
agreement, Cohen’s Kappa coefficient, positive pre-
dictive values (PPV) and sensitivity of the classifier
per class.
From table 4 it can be seen that the overall perfor-
mance significantly exceeds random guessing, which
is 1989/6292 = 31.61%. Furthermore it can be seen
the largest number of confusions is for actual wake
epochs being (falsely) recognized as light sleep. Ac-
tually, the classifier is falsely biased towards light
HEALTHINF 2012 - International Conference on Health Informatics
490
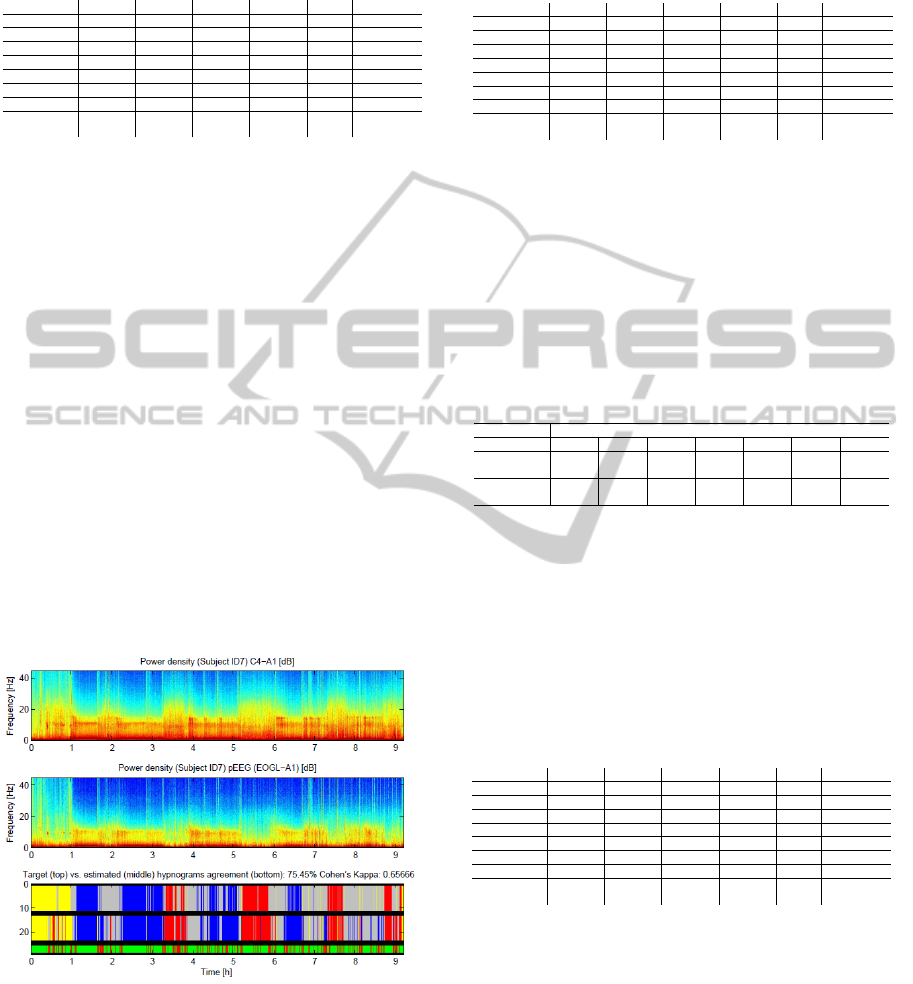
Table 4: Facial PSG approach overall results. Confusion
matrix (in bold), percentage of agreement, Cohen’s Kappa
coefficient, positive predictive values (PPV) and sensitivity
of the classifier per class.
Overall Wake Light Deep Rem Sum Sensitivity
Wake 1206 42 8 42 1298 92.91%
Light 320 1931 456 430 3137 61.56%
Deep 22 102 735 15 874 84.10%
Rem 26 118 16 796 956 83.26%
Sum 1574 2193 1215 1283 6265
PPV 76.62% 88.05% 60.49% 62.04%
Agreement 74.51%
Cohen’s
Kappa
0.64317
sleep, as it classifies half of the total number of epochs
as light sleep (i.e, 3173/6292 = 50.43%), resulting in
a low sensitivity (52.66%) for that class.
In addition to numerical representation of the
classification figure 3 shows both input data (Facial
PSG spectrum; middle plot) and graphical representa-
tion of the hypnograms both target and estimated (bot-
tom plot). The top plot of the figure shows the power
spectrum of C4-A1 PSG channel, which served as an
input for an additional experiment we conducted. The
essence of the experiment was in substituting Facial
PSG signal with C4-A1 signal. Given the fact that
C4 electrode of the PSG setup is mounted close to
the brain and subsequently has a stronger signal, our
assumption was to observe gain in classification per-
formance. However, this experiment proved an op-
posite effect. Despite better signal to noise ratio, a
significant drop in performance of the classifier ap-
peared. This may indicate that when it comes to a
single channel PSG, electrode-positions in the Facial
PSG are better suited for sleep stage estimation.
Figure 3: Results and input data (spectrum) visualization
for subject ID7.
Table 5 shows overall performance matrix for C4-
A1 channel. From this table we may see (when
compared to Table 4) that Cohen’s Kappa statistics
lowered by 0.0662 and percentage of agreement by
6.55%.
Table 5: Facial PSG approach overall results on C4-A1
channel. Confusion matrix (in bold), percentage of agree-
ment, Cohen’s Kappa coefficient, positive predictive values
(PPV) and sensitivity of the classifier per class.
Overall Wake Light Deep Rem Sum Sensitivity
Wake 1199 18 15 66 1298 92.37%
Light 649 1263 799 426 3137 40.26%
Deep 44 97 729 4 874 83.41%
Rem 36 10 241 669 956 69.98%
Sum 1928 1388 1784 1165 6265
PPV 62.19% 90.99% 40.86% 57.42%
Agreement 61.61%
Cohen’s
Kappa
0.49303
4.2 RHA , - Respiration, Heart and
Actigraphy Signals for Hypnogram
Estimation
Table 6 shows Cohen’s Kappa and percentage of
agreement figures per run of the cross-validation
scheme. The last column contains average values.
Table 6: RHA , - respiration, heart and actigraphy features
approach results, per cross validation run (per subject).
Subjects (Boston data set)
1 2 3 4 5 6 mean
Agreement
%
48.37 33.62 27.87 32.33 43.74 13.55 33.25
Cohen’s
Kappa
0.26 0.09 0.06 0.09 0.25 0.32 0.18
Table 7 shows the overall agreement matrix that
contains: confusion matrix (in bold), percentage of
agreement, Cohen’s Kappa coefficient, positive pre-
dictive values (PPV) and sensitivity of the classifier
per class.
Table 7: RHA , - respiration, heart and actigraphy features
approach overall results. Confusion matrix(in bold), per-
centage of agreement, Cohen’s Kappa coefficient, positive
predictive values (PPV) and sensitivity of the classifier per
class.
Overall Wake Light Deep Rem Sum Sensitivity
Wake 247 10 8 73 338 73.08%
Light 596 761 723 1020 3100 24.55%
Deep 118 158 370 254 900 41.11%
Rem 221 241 53 368 883 41.68%
Sum 1182 1170 1154 1715 5221
PPV 20.90% 65.04% 32.06% 21.46%
Agreement 33.44%
Cohen’s
Kappa
0.12265
Table 8 shows the agreement figures presented
earlier in table 1 along with agreement figures
achieved by RHA and Facial PSG approaches. From
these figures we notice that the Facial PSG approach
is superior compared to the RHA one in both per-
centages of agreement and Cohen’s Kappa coefficient
numbers. Figures of the RHA approach shows a very
low performance of the classifies when based on res-
piration, heart and actigraphy features. It can be seen
that the overall performance is very close to random
guessing, which is 1715/5221 = 32.85%. Again, the
TOWARDS UNOBTRUSIVE AUTOMATED SLEEP STAGE CLASSIFICATION - Polysomnography using Electrodes
on the Face
491

Table 8: Sleep stages classification agreement figures.
Cohen’s Kappa Agreement(%)
Human expert(PSG) vs. Human ex-
pert (PSG)
0.80 86.00
Automated technique A(PSG) vs.
Automated technique B(PSG)
0.69 85.48
Automated technique(PSG) vs. Fa-
cial PSG
0.64 74.51
Automated technique(PSG) vs.
RHA
0.12 33.44
classifier is falsely biased towards light sleep, as it
classifies most of the total number of epochs as light
sleep (i.e, 3100/5221 = 59.38%), resulting in a very
low sensitivity (24.55%) for that class.
5 CONCLUSIONS
In our study we were not able to find significant corre-
spondence at individual level(cross subjects) between
PSG based sleep stages estimated by experts and the
features we extracted in the RHA approach. This con-
clusion only holds for this paper’s particular combi-
nation of features and classifier. A separate study on
separability involving at least larger number of sub-
jects is required in order to strengthen this conclusion.
In contrast to the RHA, classification, results ob-
tained on features extracted in the Facial PSG ap-
proach look very promising, the good performance
of the classifier indicate good separability of differ-
ent sleep states manifested in Facial PSG features. In
the current study, we employed only ”simple” (low-
capacity, epoch based) classifier that already showed a
performance of 74.51% agreement and 0.64 Cohen’s
Kappa. Performance wise these results position the
Facial PSG approach next to full PSG based auto-
mated techniques (see Table 1). Further improvement
of classification performance can be expected when
order and transition probabilities between sleep stages
are taken into account.
In addition to very good performance indicators
the Facial PSG is also much less obtrusive compared
to a full PSG setup. The full PSG setup employs 8+
channels resulting in 12+ electrodes mounted on the
skull of a subject, whereas Facial PSG uses only one
channel (3 electrodes) mounted on a single side of the
face allowing subject to have more natural sleeping
position(s) and what is even more important more nat-
ural sleep. Given its obvious advantage of being more
comfortable the Facial PSG approach makes sleep
studies less labor intensive and consequently more ac-
cessible, also making multiple night studies more re-
alistic which will enable research to better understand
the complex mechanisms involved in humans sleep.
REFERENCES
Buysse, D., Reynolds-III, C., Monk, T., Berman, S., and
Kupfer, D. (1989). Pittsburgh sleep quality index: A
new instrument for psychiatric practice and research.
Psychiatric Research, 28(2):193–213.
Centre, N. N. R. (2002). Bibliography on the self-
organizing maps (som) and learning vector quantiza-
tion (lvq).
Douglass, A., Bornstein, R., Ninomurcia, G., Keenan, S.,
Miles, L., and Zarcone, V. (1994). The Sleep Dis-
orders Questionnaire-I - Creation and Multivariate
Structure of SDQ. Sleep, 17(2):160–167.
Kohonen, T. (1995). Learning vector quantization, The
handbook of brain theory and neural networks. MIT
Press, Cambridge, MA.
Rechtschaffen, A. and A.Kales (1968). A manual of stan-
dardized terminology, techniques and scoring system
for sleep stages of human subjects. U. S. National In-
stitute of Neurological Diseases and Blindness, Neu-
rological Information Network, Bethesda, Md.,.
Seo, S. and Obermayer, K. (2003). Soft learning vector
quantizatio. Neural Computation, 15:1589–1604.
Virkkala, J. (2005). Automatic Sleep Stage Classification
Using Electro-oculography. PhD thesis, Faculty of
Computing and Electrical Engineering. Tampere Uni-
versity of Technology.
Welch, P. (1967). The use of fast fourier transform for the
estimation of power spectra: A method based on time
averaging over short, modified periodograms. IEEE
Transactions on Audio Electroacoustics, AU-15:7073.
Witoelar, A. W., Ghosh, A., de Vries, J. J. G., Hammer, B.,
and Biehl, M. (2010). Window-based example selec-
tion in learning vector quantization. Neural Computa-
tion, 22(11):2924–2961.
HEALTHINF 2012 - International Conference on Health Informatics
492
