
Stable Measurement of Blood Flow While Running using a Micro
Blood Flowmeter
Wataru Iwasaki
1
, Masaki Nakamura
1
, Takeshi Gotanda
1
, Satoshi Takeuchi
2
, Masutaka Furue
2
,
Eiji Higurashi
3
and Renshi Sawada
1, 4
1
Graduate School of Systemus Life Sciences, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka-city, Japan
2
Graduate School of Medical Sciences, Kyushu University, 3-1-1, Maidashi, Higashi-ku, Fukuoka-city, Japan
3
Research Center for Advanced Science and Technology, The University of Tokyo, 4-6-1,
Komaba, Meguro-ku, Tokyo, Japan
4
Department of Mechanical Engineering, Kyushu University, 744, Motooka, Nishi-ku, Fukuoka-city, Japan
Keywords: Blood Flowmeter, Laser Doppler, Wearable Sensor, MEMS, Exercise.
Abstract: Skin blood flow during exercise has been studied before, with measurements made using laser Doppler
blood flowmeters; however, their use was limited to activities with minimal motion, such as riding bicycle
ergometers, because conventional devices are large and their measurements easily altered by movements of
the optical fiber, rendering them inappropriate for running. We have previously developed a micro
integrated laser Doppler blood flowmeter using microelectromechanical systems (MEMS) technology. The
micro blood flowmeter is wearable and can measure signal stably even while the wearer is moving. We
monitored skin blood flow during running at velocities of 6 km/h, 8 km/h, and 10 km/h, and were successful
in measuring a stable signal under these conditions. We found that at the forehead the skin blood flow
increases and, in contrast, at the fingertip it initially decreases during running. We also found that the level
of these increases and decreases correlated with the running velocity.
1 INTRODUCTION
Recently, more people have started running. The
Sasakawa Sports Foundation reported that the
number of people who run more than twice a week
has doubled in the last ten years in Japan (2010), and
the number of marathon completions is up more than
1.6-fold from ten years ago in the United States
(Mathews et al., 2012). Runners, especially those
with hypertrophie cardiomyopathy, should be aware
of the possibility for harm or even death due to
exercise-induced issues such as cardiopulmonary
arrest (Maron et al., 1996). Therefore it’s necessary
to observe health index of those person while
running.
Many studies on the physiological effects of
exercise have been conducted using laser Doppler
blood flowmeters, which can noninvasively measure
skin blood flow (Kellogg Jr. et al., 1993, Yasuda et
al., 1994. Alonso et al., 1998). Many varieties of
these blood flowmeters have been developed
(Watkins and Holloway, 1978, Wunderlich et al.,
1980), yet they face several challenges: for example,
their large size and the high power consumption
necessary for monitoring blood flow while subjects
run. Furthermore, such instruments are easily
affected by movement or vibration of the optical
fiber in the devices, which is also used as a strain
sensor (Newson et al., 1987). Therefore, laser
Doppler blood flowmeters were primarily used in
experiments having little subject motion, such as
with stationary bicycles, rather than running.
To meet and overcome these challenges, a laser
Doppler blood flowmeter that didn’t require the use
of optical fibers was developed (Mul et al., 1984).
We have also previously applied
microelectromechanical systems (MEMS)
technology to a blood-flow-measuring instrument,
and developed a micro integrated laser Doppler
blood flowmeter which is called MEMS blood flow
sensor (Higurashi et al., 2003). The MEMS blood
flow sensor can measure blood flow while the
subject is moving because it doesn’t use an optical
fiber. We have also developed a probe for the
MEMS blood flow sensor that consumes lower
30
Iwasaki W., Nakamura M., Gotanda T., Takeuchi S., Furue M., Higurashi E. and Sawada R..
Stable Measurement of Blood Flow while Running Using a Micro Blood Flowmeter.
DOI: 10.5220/0004196600300037
In Proceedings of the International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS-2013), pages 30-37
ISBN: 978-989-8565-36-5
Copyright
c
2013 SCITEPRESS (Science and Technology Publications, Lda.)

electric power using a built-in silicon microlens
(Kimura et al., 2010), and have used it to evaluate
dehydration (Nogami et al., 2011), alcohol
consumption (Iwasaki et al., 2012), and systemic
sclerosis (Kido et al., 2007). This probe’s design
enables wafer-level packaging, improving its
fabrication efficiency. Our MEMS blood flow sensor
with this newly developed probe is the world’s
smallest, lightest, and least power-consuming blood
flow sensor.
In the present study, we attempted to measure
blood flow using our developed MEMS blood flow
sensor at the tip of the ring finger, the forehead, and
the earlobe while participants ran. Blood flow was
measured at three different running velocities (6, 8,
and 10km/h) for 30 minutes each on a treadmill. We
evaluated the blood flow at each measurement site
and then compared blood flow with running
velocity.
2 MATERIALS AND METHODS
In this section, we describe the subjects, the MEMS
blood flow sensor, the experimental apparatus, and
the experimental protocol of the study.
Figure 1: Schematic of laser Doppler flowmetry.
2.1 Subjects
The research complied with the ethical standards of
the Human Experimentation Committee of Kyushu
University, and with the 2008 revision of the
Helsinki Declaration. Seven healthy young men
(mean age of 23.4 years old, range 22-26; mean BMI
22.9 kg/m2, range 20.4-29.5) participated in this
study. Subjects were fully informed of the
experimental details and any potential risks before
their participation in the study.
2.2 MEMS Blood Flow Sensor
The MEMS blood flow sensor can measure skin
blood flow noninvasively using the principles of
laser Doppler flowmetry. We explain these
principles in the first subsection, and lay out the
structure of the device in the second, and describe
the components of the blood flow signal in the third.
2.2.1 Principles
Figure 1 illustrates the principles behind laser
Doppler flowmetry. A laser beam with frequency f is
emitted from a light source to the skin and penetrates
it to a certain depth. Some light scatters from static
tissue such as the skin and blood vessels; a small
amount of light is scattered from moving tissue such
as red blood cells. Because of the Doppler effect, the
frequency of the light scattered by the moving
tissues changes slightly. The Doppler-shifted and
non Doppler-shifted light interfere on the photo
diode (PD), which detects the resultant intensity
modulations. The first moment of the power
spectrum of the beat frequency distribution as
obtained using a fast Fourier transform (FFT) is
proportional to the product of the average velocity
and patient-specific concentration of the red blood
cells as they move the body (Bonner and Nossal,
1981).
2.2.2 Structure
The MEMS blood flow sensor contains a probe and
a main body, as shown in Figure 2. The probe has an
optical MEMS chip and first-stage operational
amplifiers, and is attached directly to the subject.
The main sensor body has a digital signal processor
(DSP), a Bluetooth wireless link, display, and
battery. The MEMS blood flow sensor can be run on
a 3.7 V square electric battery, as is used in cell
phones.
Figure 3 shows a schematic of the optical MEMS
chip we developed. We used distributed feedback
laser diode (DFB-LD) as a light source of MEMS
blood flow sensor. Its wavelength is 1310 nm which
easily penetrates human skin. The laser beam
emitted from the DFB-LD is reflected vertically by
the <111> facet etched mirror, focused by the
microlens, and irradiated to the skin. The microlens
decreases coupling loss, a serious problem in
conventional instruments. The laser power coming
from the probe is 2 mW. The laser doesn’t harm to
human body, because it is not perfectly collimated.
Scattered light reenters through the window and is
detected by the PD.
2.2.3 Blood Flow Signal
Figure 4 shows the probes of our MEMS blood flow
sensor and a conventional, optical fiber-type
StableMeasurementofBloodFlowwhileRunningUsingaMicroBloodFlowmeter
31

instrument attached to adjacent fingertips for
comparison, and Figure 5 shows the blood flow
signals measured by each. The hand was waved
from the 10-second mark. It is apparent that the
signal of the fiber-type instrument was greatly
affected, while in contrast, the signal of our MEMS
blood flow sensor was stable.
Figure 2: Photograph of MEMS blood flow sensor.
Figure 3: Schematic of cross-sectional view of the optical
MEMS chip.
Figure 4: Photograph of MEMS blood flow sensor and
fiber-type instrument probes attached to fingertips.
There are three main components of blood flow
waves: basic, cardiac, and reflex waves (Kano et al.,
1993). Basic waves have periods of about ten
seconds, and some researchers have reported that
these large oscillations are modulated by the
sympathetic nervous system or by sympathetic
sympathetic nervous system or by sympathetic
vasomotor tone (Sasano et al., 1999); (Mukae et al.,
2006). Cardiac waves are much smaller and simply
reflect the heartbeat. Reflex waves are a
phenomenon in which peripheral blood flow
temporarily yet markedly decreases when, for
example, the subject takes a deep breath or is
engaging in physical activity. We should note that
reflex waves only appear in palmoplantar areas.
Figure 5: Fingertip blood flow signals measured by the
MEMS blood flow sensor and fiber-type instrument
during hand-waving.
2.3 Experimental Apparatus
Participants ran on Tempo T931 treadmills (Johnson
Health Tech. Co., Ltd., Taiwan). MEMS blood flow
sensors were attached with double-side tape and
medical tape to the tip of the left ring finger, the left
earlobe, and the forehead (using a headband) as
shown in Figure 6. In addition to blood flow, we
measured body temperature, blood pressure, blood
lactate level, and body weight. Body temperature
was measured with an MC-510 ear thermometer
(OMRON HEALTHCARE Co., Ltd., Japan), which
can measure body temperature within one second
and can well perform even while running. Blood
pressure was measured with an upper-arm type
sphygmomanometer.
Figure 6: Photograph of subject with attached MEMS
blood flow sensor.
Blood lactate level was measured with the
Lactate Pro LT-1710 meter (ARKRAY, Inc., Japan),
which can measure blood lactate levels from only 5
BIOSIGNALS2013-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
32
μl of blood. We obtained blood samples using the
Naturalet EZ (ARKRAY, Inc., Japan), a device that
can easily obtain small blood samples with minimal
pain. We measured blood pressure and lactate levels
before and after running, because we couldn’t
measure them while the subject ran. Body weight
was measured before and after the experiment.
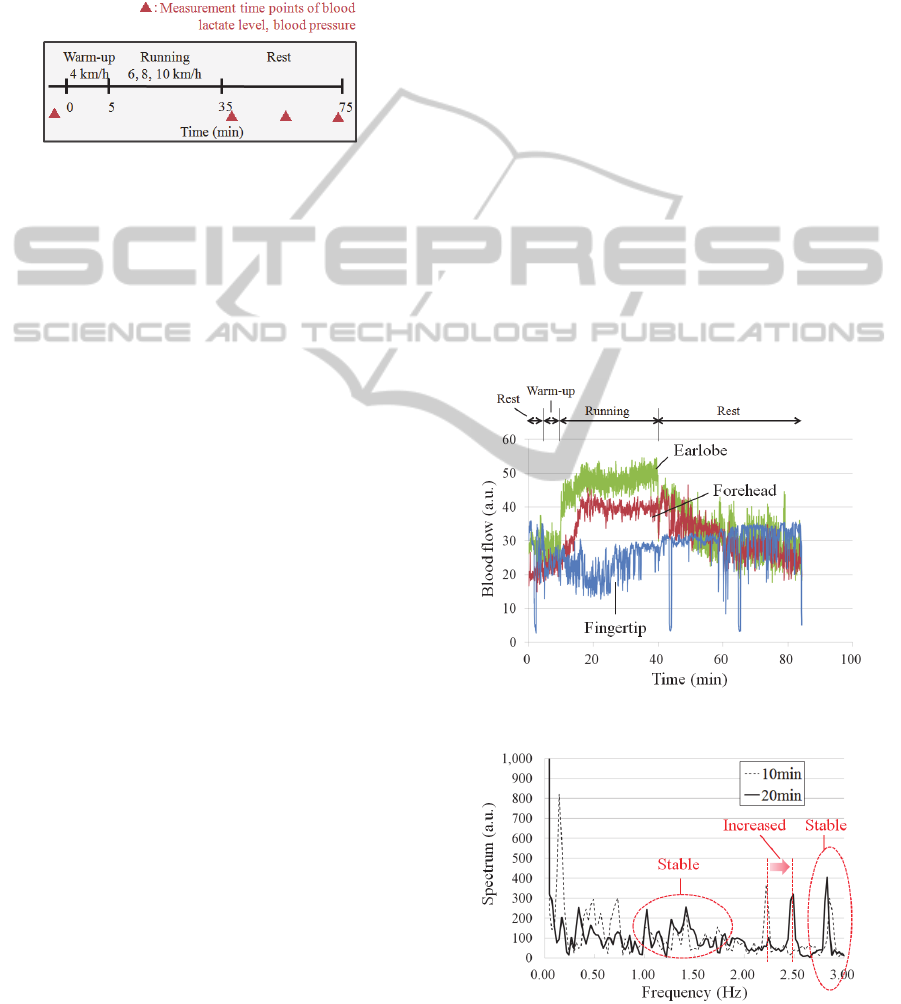
Figure 7: Experimental protocol.
2.4 Experimental Protocol
We measured blood flow at three different running
velocities (6, 8, and 10 km/h) for 30 minutes after
walking for 5 minutes at 4 km/h as a warm-up.
Subjects ran each velocity on different days with
intervals of more than three days between sessions,
to counteract fatigue from the previous experiment.
We observed blood flow when sitting for 40 minutes
after running to observe the recovery of blood flow.
If subjects complained of exhaustion while running,
we stopped the running session but continued to
observe blood flow for 40 minutes.
Figure 7 shows the experimental protocol. At the
beginning of a session, we measured resting blood
flow, body temperature, blood pressure, and blood
lactate levels while the subject sat. Then, the subject
walked on the treadmill at 4 km/h for 5 minutes.
Body temperature was measured 4 minutes after
walking onset. The subject ran at a constant velocity
for 30 minutes after the warm-up. Body temperature
was measured at 5, 10, 15, 20, 25, and 29 minutes of
running. We observed blood flow while sitting for
40 minutes. Body temperature, blood pressure, and
blood lactate levels were all measured just after, 20
minutes after, and 40 minutes after running
concluded.
3 RESULTS
We describe how the stability of blood flow signals
differ among measurement sites in the first
subsection; how heart rate is isolated from the blood
flow signal in the second subsection; and how blood
flow changes with velocity in the final subsection.
3.1 Stabilities of Blood Flow Signals in
each Measurement Site
Figure 8 shows the experimental data for a
representative subject. The blood flow signals
corresponding to sensor location are displayed. The
data was sampled at 50 Hz, and the values displayed
were averaged every 100 points, and plotted at 2-
second intervals for clarity. The three signals had
different values in the resting states because density
of capillary differs across measurement sites and
because of individual variability in MEMS blood
flow sensor calibration. Blood flow at the earlobe
suddenly and greatly increased upon the start of
running, indicating that the sensor is very sensitive
to subject movement at this location. Blood flow at
the forehead gradually increased as the subject
began running and gradually decreased after subject
had stopped. On the other hand, blood flow at the
fingertip decreased at the start of running and
gradually increased once it had ended. There were
many reflex waves observed in fingertip blood flow
during running. We could confirm these changes of
blood flow in all subjects.
Figure 8: Blood flow signals during running.
Figure 9: Spectrum of fingertip blood flow at 10 minutes
and 20 minutes after start of running.
StableMeasurementofBloodFlowwhileRunningUsingaMicroBloodFlowmeter
33
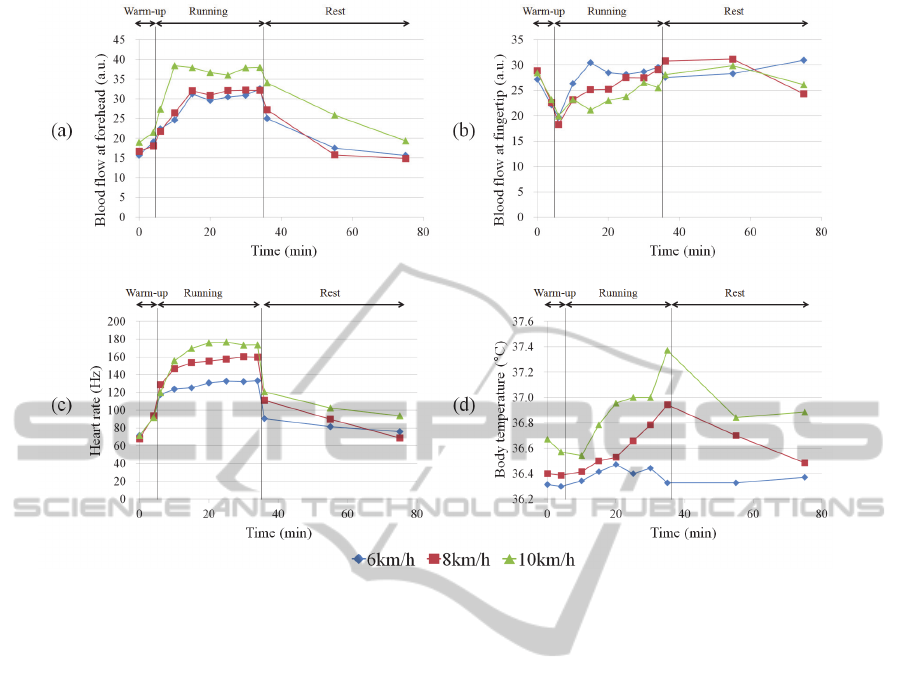
Figure 10: Average changes in blood flow (n=7) at forehead (a) and at fingertip (b), heart rate (c), and body temperature (d)
during the running experiment; standard deviations not shown for clarity.
3.2 Separation of Heart Beat from
Rhythm of Running
When subjects weren’t exercising, we could usually
see a single spectral peak around 1 Hz, which was
caused by heart rate. However we observed some
additional spectrum peaks during running. Figure 9
shows a spectrum distribution of blood flow at the
forehead at 10 and 20 minutes after the
commencement of running at 8 km/h. These spectra
were obtained by performing FFTs on blood flow
signal data lasting 40.96 seconds (2048 points).
Most spectral peaks remained unchanged between
samples, staying at a stable rhythmic frequency due
to running at constant velocity. However, one peak
did shift, suggesting it was tracking heart rate,
increasing during running. We can determine heart
rate by performing a FFT on a power spectrum of
blood flow data, tracking the rhythmic running
peaks as a constant, and observing the incremental
increase of the peak corresponding to heart rate: this
is possible even while running.
3.3 Changes of Blood Flow with
Running Velocity
Mean (n=7) blood flow at the forehead and at the
fingertip, heart rate, and body temperature at each
running velocity are shown in Figures 10(a), 10(b),
10(c), and 10(d), respectively. All subjects
completed the 30 minutes of running in the 6 km/h
and 8 km/h sessions. In the 10 km/h session, one
subject retired after 15 minutes, and another after 20;
all other subjects completed the exercise. Blood flow
at the forehead gradually increased during running
for each velocity, with the 10 km/h session yielding
the highest. On the other hand, blood flow at the
fingertip of each velocity suddenly decreased then
gradually rose during running for each velocity, with
the 10 km/h session yielding the slowest increase.
Mean body temperature rose to over 37.0 °C in the
later stages of the 10 km/h running session.
Systolic and diastolic blood pressure, blood
lactate level, and body weight data are shown in
Table 1. The “Baseline” column contains the pre-
warm-up data, and the “35 min”, “55 min”, and “75
min” columns contain values obtained just after, 20
minutes after, and 40 minutes after running,
respectively. Differences between the baseline value
and the values at each time point were evaluated by
paired t-test, p<0.05 was considered statistically
significant. Systolic blood pressure showed a
statistically significant increase from baseline just
after running for each running velocity. Diastolic
BIOSIGNALS2013-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
34

Table 1: Systolic and diastolic blood pressure, blood lactate level, and body weight at each measurement time.
Baseline 35 min 55 min 75 min
Systolic blood
pressure
(mmHg)
6 km/h 114.6 ± 9.9 136.0 ± 11.3 ** 113.7 ± 10.4 113.6 ± 14.1
8 km/h 118.0 ± 8.6 133.3 ± 12.7 * 115.3 ± 10.5 116.6 ± 7.0
10 km/h 116.7 ± 13.9 147.6 ± 14.3 ** 119.1 ± 16.5 114.1 ± 13.1
Diastolic blood
pressure
(mmHg)
6 km/h 66.4 ± 8.6 74.3 ± 5.6 ** 68.4 ± 7.9 71.7 ± 7.7
8 km/h 67.0 ± 9.1 67.4 ± 7.3 68.6 ± 9.7 70.7 ± 6.0
10 km/h 68.0 ± 8.3 67.7 ± 4.8 66.6 ± 11.2 67.0 ± 8.5
Blood lactate
level
(mmol/L)
6 km/h 3.06 ± 2.09 4.97 ± 4.20 4.07 ± 4.51 2.76 ± 2.08
8 km/h 1.84 ± 0.44 5.23 ± 6.88 3.10 ± 3.50 2.57 ± 2.35
10 km/h 2.09 ± 1.48 7.11 ± 4.46 * 3.60 ± 2.35 2.40 ± 1.23
Body weight
(kg)
6 km/h 67.2 ± 9.1 - - 66.8 ± 9.1 **
8 km/h 68.1 ± 9.6 - - 67.5 ± 9.4 **
10 km/h 65.2 ± 4.7 - - 64.6 ± 4.6 **
Baseline= pre-warm-up; 35 min = just after running; 55 min = 20 minutes after running; 75 min = 40 minutes after running.
Differences between the Baseline values and the values at each time point were evaluated by paired t-test (*: p<0.05, **: p < 0.01).
blood pressure significantly increased from baseline
just after running only for when running velocity
was 6 km/h. Blood lactate level was significantly
higher than baseline only when subjects ran at 10
km/h. Body weight significantly decreased after
running for all velocities due to sweating, with the
dehydration amount reaching almost 1 percent of
body weight.
4 DISCUSSION
In this study, we succeeded in stably measuring
blood flow in running subjects using our previously
developed MEMS blood flow sensor. We found that
blood flow at the fingertips decreases just after
running commences and then gradually increases
during continuous running. Conversely, blood flow
at the forehead and earlobe showed a sudden rise
just after the start of running, gradually increasing
during running. Moreover, we found that blood flow
at the forehead increased the most when subjects ran
at 10 km/h, the only velocity at which mean lactate
level was significantly higher than baseline, and at
which some subjects complained of exhaustion and
retired. These increases in blood flow most likely
function to lower the body temperature, helping with
sweating to release heat. When subjects ran at 10
km/h, although compensatory blood flow increases
were large, body temperature still increased to over
37 °C; and, perhaps because the release of body heat
could not catch up with the exercise-generated
hyperthermia, some subjects couldn’t complete the
full time. In contrast to this, blood flow at the
fingertip actually decreased. Blood flow at fingertip
was likely decreased by neural vasoconstriction, or
because muscle tissue was given priority of blood
supply over peripheral perfusion at the beginning of
running. Blood flow was subsequently increased in
order to lower body temperature.
The MEMS blood flow sensor can measure
blood flow during running, but it also captures noise
caused by running motion. Some spectral waves
with the same frequency as running rhythm were
observed in a power spectrum distribution of blood
flow signal. This noise is likely due to shearing of
the skin caused by the running motion. When such
shearing occurs, blood vessels move relative to the
MEMS blood flow sensor, which then captures the
relative, shifted movement (Figure 11). The blood
flow signal at the earlobe, where sensors are difficult
to affix and the skin is soft and easily sheared, was
strongly affected by motion. In contrast, the blood
flow signal at the fingertip and forehead, where a
solid hold was made possible by medical tape and
headbands, were only slightly affected by the
running rhythm. Even then we could eliminate the
waves by monitoring the spectral pattern change
from moment to moment, and treating the pace as
constant. Therefore, we think the fingertip and the
forehead are suitable measurement sites for blood
StableMeasurementofBloodFlowwhileRunningUsingaMicroBloodFlowmeter
35

flow during running or other activities with
significant motion. We may also even be able to
eliminate the shearing noise at the earlobe, by
filtering it from blood flow signals using a FFT.
Figure 11: Schematic of blood flow measurement when a
shearing occurs.
In conclusion, we have succeeded in stably
measuring blood flow during running using the
MEMS blood flow sensor. The device also captures
the noise caused by running motion; however, we
can eliminate this and obtain the heart rate by
performing a FFT on blood flow power spectra,
cancelling out the steady rhythm of the pace, and
observing for an incremental frequency shift in the
spectrum peak that corresponds to heart rate. The
MEMS blood flow sensor would contribute to
researches in areas like sports science or health
science. Moreover, we found that the extent of the
observed changes to blood flow depend on the
intensity of exercise. These results suggest that the
MEMS blood flow sensor has potential as a new
portable device for detecting the running intensity,
and alerting runners to any dangers from excessive
exercise by detecting substantially increased or
decreased blood flow and heart rate.
ACKNOWLEDGEMENTS
We would like to express our special thanks to the
volunteers who participated in the study. We also
would like to thank Mr. Kazuto Mishima of
Vinciamo Incorporated and Mr. Takaaki Miyahara
of NEXIS Incorporated for the integration of the
MEMS blood flow sensor. This work was partially
supported by research grants from the Ministry of
Education, Culture, Sports, Science and Technology
of Japan.
REFERENCES
Alonso JG., Calbet JAL. and Nielsen B. (1998). Muscle
blood flow is reduced with dehydration during. The
Journal of Physiology, 513(3), 895-905.
doi:10.1111/j.1469-7793.1998.895ba.x
Bonner R. and Nossal R. (1981). Model for laser Doppler
measurement of blood flow in tissue, Applied Optics,
20(12), 2097-2107. doi:10.1364/AO.20.002097.
de Mul FFM., van Spijker J., van der Plas D., Greve J.,
Aarnoudse JG. and Smits TM. (1984), Mini laser-
Doppler (blood) flow monitor with diode laser source
and detection integrated in the probe. Applied Optics,
23(17), 2970-2973. doi:10.1364/AO.23.002970.
Higurashi E., Sawada R. and Ito T. (2003). An integrated
laser Doppler blood flowmeter. Journal of Lightwave
Technology, 21(3), 591-595. doi:10.1109/JLT.2003.
809551.
Iwasaki W., Nogami H., Ito H.Gotanda T., Peng Y.,
Takeuchi S., Furue M., Higurashi E. and Sawada R.
(2012). Useful Method to Monitor the Physiological
Effects of Alcohol Ingestion by Combination of
Micro-integrated Laser Doppler Blood Flow Meter
and Arm-Raising Test. Proceedings of the Institution
of Mechanical Engineers, Part H: Journal of
Engineering in Medicine, 1-6. doi:10.1177/
0954411912452974
Kano T., Shimoda O., Higashi K., Sadanaga M. and
Sakamoto M. (1993). Fundamental patterns and
characteristics of the laser-Doppler skin blood flow
waves recorded from the finger or toe. Journal of
Autonomic Nervous System, 45(3), 191-199.
doi:10.1016/0165-1838(93)90051-U.
Kellogg Jr. DL., Johnson WL., Pergola PE. and Kosiba
WA. (1993). Mechanisms of control of skin blood
flow during prolonged exercise in humans. American
Journal of Physiology. Heart and Circulatory
Physiology, 265(2), H562-H568.
Kido M., Takeuchi S., Hayashida S., Urabe K., Sawada R
and Furue M. (2007). Assessment of abnormal blood
flow and efficacy of treatment in patients with
systemic sclerosis using a newly developed
micowireless laser Doppler flowmeter and arm-raising
test. British Journal of Dermatology, 157, 690-697.
Kimura Y., Goma M., Onoe A., Higurashi E. and Sawada
R. (2010). Integrated Laser Doppler Blood Flowmeter
Designed to Enable Wafer-Level Packaging. IEEE
Transactions on Biomedical Engineering, 57(8), 2026-
2033. doi:10.1109/TBME.2010.2043842.
Maron BJ., Shirani J., Poliac LC., Mathenge R., Roberts
WC. and Mueller FO. (1996). Sudden Death in Young
Competitive Athletes: Clinical, Demographic, and
Pathological Profiles. The Journal of the American
Medical Association, 276(3), 199-204.
doi:10.1001/jama.1996.03540030033028.
Mathews SC., Narotsky DL., Bernholt DL., Vogt M.,
Hsieh YH., Pronovost PJ. and Pham JC. (2012).
Mortality Among Marathon Runners in the United
States, 2000-2009. The American Journal of Sports
Medicine, 40(7), 1495-1500. doi:10.1177/03635465
12444555.
Mukae H., Mochizuki M. and Taniguchi Y. (2006).
BIOSIGNALS2013-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
36

Physiological Analysis of Mood State Using
Spontaneous Rhythmic Oscillations Measured with
Laser Doppler Flowmetry. Japanese Journal of
Physiological Anthropology, 11(2), 81-86.
Newson TP., Obeid A., Wolton RS., Bogget D. and Rolfe
P. (1987). Laser Doppler velocimetry : The problem of
fiber movement artefact. Journal of Biomedical
Engineering, 9(2), 169-172. doi:10.1016/01415425
(87)90030-6
Nogami H., Iwasaki W., Abe T., Kimura Y., Onoe A.,
Higurashi E., Takeuchi S., Kido M., Furue M. and
Sawada R. (2011). Use of a simple arm-raising test
with a portable laser Doppler blood flow meter to
detect dehydration. Proceedings of the Institution of
Mechanical Engineers, Part H: Journal of
Engineering in Medicine, 225(4), 411-419.
doi:10.1243/09544119JEIM727
Sasakawa Sports Foundation. (2010). The 2010 SSF
National Sports-Life Survey. Tokyo: Sasakawa Sports
Fundation.
Sasano H., Hayano J., Tsuda T. and Katsuya, H. (1999).
Effects of sympathetic nerve blockades on low-
frequency oscillations of human earlobe skin blood
flow, Journal of the Autonomic Nervous System,
77(1), 60-67. doi:10.1016/S0165-1838(99)00029-6
Yasuda Y., Yoshizawa M. and Nishino H. (1994). Effect
of Exercise Intensity on the Spectral Properties of Skin
Blood Flow. Japanese Journal of Physiology, 44, 533-
546. doi:10.2170/jjphysiol.44.53.
Watkins D. and Holloway Jr GA. (1978). An instrument to
measure cutaneous blood flow using the Doppler shift
of laser light. IEEE Transactions on Biomedical
Engineering, BME-25(1), 28-33. doi:10.1109/TBME.
1978.326374
Wunderlich RW., Folger RL., Giddon DB. and Ware BR.
(1980). Laser Doppler blood Flow meter and optical
plethysmograph. Review of Scientific Instruments,
51(9), 169-172. doi:10.1063/1.1136413.
StableMeasurementofBloodFlowwhileRunningUsingaMicroBloodFlowmeter
37
