
Automated Arteriole and Venule Recognition in Retinal Images using
Ensemble Classification
M. M. Fraz
1
, A. R. Rudnicka
2
, C. G. Owen
2
, D. P. Strachan
2
and S. A. Barman
1
1
School of Computing and Information Systems, Faculty of Science Engineering and Computing,
Kingston University London, London, U.K
2
Division of Population Health Sciences and Education, St. George’s, University of London, London, U.K
Keywords: Medical Image Analysis, Retinal Image Processing, Artery Vein Classification, Ensemble Learning.
Abstract: The shape and size of retinal vessels have been prospectively associated with cardiovascular outcomes in
adult life, and with cardiovascular precursors in early life, suggesting life course patterning of vascular
development. However, the shape and size of arterioles and venules may show similar or opposing
associations with disease precursors / outcomes. Hence accurate detection of vessel type is important when
considering cardio-metabolic influences on vascular health. This paper presents an automated method of
identifying arterioles and venules, based on colour features using the ensemble classifier of boot strapped
decision trees. The classifier utilizes pixel based features, vessel profile based features and vessel segment
based features from both RGB and HIS colour spaces. To the best of our knowledge, the decision trees
based ensemble classifier has been used for the first time for arteriole/venule classification. The
classification is performed across the entire image, including the optic disc. The methodology is evaluated
on 3149 vessel segments from 40 colour fundus images acquired from an adult population based study in
the UK (EPIC Norfolk), resulting in 83% detection rate. This methodology can be further developed into an
automated system for measurement of arterio-venous ratio and quantification of arterio-venous nicking in
retinal images, which may be of use in identifying those at high risk of cardiovascular events, in need of
early intervention.
1 INTRODUCTION
With the development of digital imaging and
computational efficiency, image processing, analysis
and modeling techniques are increasingly used in all
fields of medical sciences, particularly in
ophthalmology (Abràmoff, Garvin et al. 2010).
Automated detection of micro-vascular disease such
as diabetic retinopathy in the retinal image using
digital image analysis methods has huge potential
benefits in screening programs for early detection of
disease (Fraz, Remagnino et al. 2012). The blood
vessel structure in retinal images is unique in the
sense that it is the only part of the blood circulation
system that can be directly observed non-invasively,
can be easily imaged using Fundus cameras.
Morphological characteristics of retinal blood
vessels (particularly width) have been prospectively
associated with cardiovascular outcomes in adult life
(Wong, Klein et al. 2001), and with cardio-
metabolic risk factors in early life (Owen, Rudnicka
et al. 2011). Associations between retinal vessel
morphology and disease precursors / outcomes may
be similar or opposing for arterioles and venules. For
instance, hypertension and atherosclerosis may have
different effects in retinal arterioles and venuels,
resulting in a decreased arteriole to venule width
ratio (AVR) (Jack J. Kanski and Brad Bowling
2011). Retinal arteriovenous nicking, a
pathognomonic sign of hypertension, is another
retinal feature worthy of study, characterized by a
decrease in the venular calibre at both sides of an
artery-vein crossing (Jack J. Kanski and Brad
Bowling 2011). However, more subtle changes in
arteriole / venular morphology may be an early
physio-maker of vascular health, which might
predict those at high risk of disease in middle and
later life. However, identifying small changes in
retinal arterioles and venules is a difficult task to
perform manually, as it is subjective, open to
measurement error, and time consuming, limiting its
use in large population based studies. Automated
194
M. Fraz M., R. Rudincka A., G. Owen C., P. Strachan D. and A. Barman S..
Automated Arteriole and Venule Recognition in Retinal Images using Ensemble Classification.
DOI: 10.5220/0004733701940202
In Proceedings of the 9th International Conference on Computer Vision Theory and Applications (VISAPP-2014), pages 194-202
ISBN: 978-989-758-009-3
Copyright
c
2014 SCITEPRESS (Science and Technology Publications, Lda.)

segregation of retinal arterioles and venules could be
used to assist with this task, which would be a pre-
requisite for the development of a computer assisted
tool for use in large populations to identify those at
high risk of disease.
The appearance of arterioles and venules in
retinal images are similar. The general assumption is
that there is a difference in the colour and size of the
venules and arterioles; the later one appears to be
thinner, brighter and present more frequently with a
central light reflex. However, there are some
challenges in building a robust vessel classification
system. There is intra-image and intra-subject
variance in the blood colour. The size and colour of
similar blood vessels changes as they move away
from the optic disc. In the periphery vessels become
so this they are almost indistinguishable. The
context based features may also fail at these
locations due to vessel crossings and branching. In
addition, the curved shape of the retina and non-
uniform illumination add complexity to the
automated vessel classification task.
A number of methods have been reported in
literature for retinal arteriole/venule (a/v)
classification, which can be divided into two broad
categories; automated and semi-automated methods.
In automated methods (Niemeijer, Xiayu et al. 2011;
Huang, Zhang et al. 2012; Dashtbozorg, Mendonca
et al. 2013; Nguyen, Bhuiyan et al. 2013), the vessel
centerline pixels forming the vascular skeleton are
extracted from the segmented vascular tree, followed
by the calculation of various distinguishable features
for each centerline pixel and finally each pixel is
assigned as an arteriole or venule by a classifier. In
semi-automated methods (Rothaus, Jiang et al. 2009;
Vázquez, Cancela et al. 2013), the initial pixels on
the main vessels are marked as arteriole or venule by
an expert, and then these labels are propagated
across the vascular network through vessel tracking
using the structural characteristics and connectivity
information.
Grisan’s method (Grisan and Ruggeri 2003) was
amongst the first to propose automatic a/v
separation. The main idea was to divide the optic
disc centered images into four quadrants with the
assumption that each quadrant will contain
approximately the same number of arterioles and
venules with significant differences in the features.
The variance of the red channel and mean of the hue
are used as vessel features, fuzzy clustering is
applied to each partition independently. In another
method (Saez, González-Vázquez et al. 2012), the
quadrants are rotated in steps of 20 degrees with the
aim of fulfilling the assumption that each quadrant
should contain at least one venule and one arteriole.
K-Means clustering is used to classify the vessels in
two concentric circumferences around the optic disc.
The quadrant-wise classification enforces a
condition to have at least one arteriole and one
venule per quadrant and it seems more suitable for
optic disc centered images rather than macula
centered images. Also, basic K-Means clustering is
sensitive to the initialization and may often become
stuck at a local optimal.
In this paper we have presented an automated
method for retinal a/v classification utilizing an
ensemble classifer of boot strapped decision trees.
The classifier based on the boot strapped decision
trees is a classic ensemble classifier, which has been
broadly applied in many application areas of image
analysis (Fraz, Remagnino et al. 2012), but has not
been extensively utilized for retinal vessel
classification. To our knowledge, this is the first use
of a decision trees based ensemble method for a/v
classification. An important feature of the bagged
ensemble is that the classification accuracy can be
estimated during the training phase, without
supplying the classifier with test data. Moreover, the
importance of each feature in classification can also
be predicted during the training phase, which helps
in identifying the most relevant features used in a/v
classification thus automatically reducing the
dimensionality of the feature vector and boosting
computational speed. The method is validated on 40
macula centered fundus photographs acquired from
20 middle-aged and elderly adults examined as part
of the latest phase of the European Investigation into
Cancer in Norfolk study (EPIC-Norfolk 2013). The
classification is not only performed near the optic
disc but across the entire image. The proposed
method achieves a high classification rate without
increasing the training samples or adding many
features.
The organization of the paper is as follows.
Section 2, presents the methodology for automated
segmentation of retinal blood vessels. Next, the
vessel classification methodology is explained in
section 3. Section 4 presents the validation
methodology and experimental results. Finally, the
discussion and conclusions are given in Section 5.
2 THE METHODOLOGY
The vascular network is segmented from the
coloured retinal image and the vascular skeleton
consisting of centerline pixels is constructed. Vessel
segments are generated by search and removal of
AutomatedArterioleandVenuleRecognitioninRetinalImagesusingEnsembleClassification
195
bifurcations crossing points. For each centerline
pixel in the vessel segment, the feature vector is
computed using pixel based features, profile based
features and vessel segment based features of the
RGB and HSI colour spaces, and finally each
centerline pixel is assigned an artery or vein label by
a decision tree based ensemble classifer.
2.1
Retinal Vessel Segmentation
The retinal vasculature is composed of arterioles and
venules, appearing as piecewise linear features, with
variation in width and their branches visible within
the retinal image (Fraz, Barman et al. 2012). We
have computed a measure of vessel-ness for each
pixel in the retinal image by combining a multi-scale
line detection which is calculated from the inverted
green plane of the coloured retinal images. The
average pixel intensity is measured along lines of a
particular length passing through the pixel under
consideration at 12 different orientations spaced by
15 degrees each. The line with the highest average
pixel intensity is selected. The line strength of a
pixel is calculated by computing the difference in
the average grey values of a square sub-window
centred at the target pixel with the average intensity
of the selected line. This concept was first
introduced by (Ricci and Perfetti 2007) and has also
been employed elsewhere (Fraz, Remagnino et al.
2012). We have used a generalized multi-scale line
detector (Nguyen, Bhuiyan et al. 2012), which uses a
variable length of aligned lines in a fixed square sub-
window, for calculating the line strength measures
for the pixels in the images containing a central
vessel reflex. In the line strength image (LSI), each
value corresponds to the confidence measure of each
pixel to be a part of the vessel or not. The LSI, as
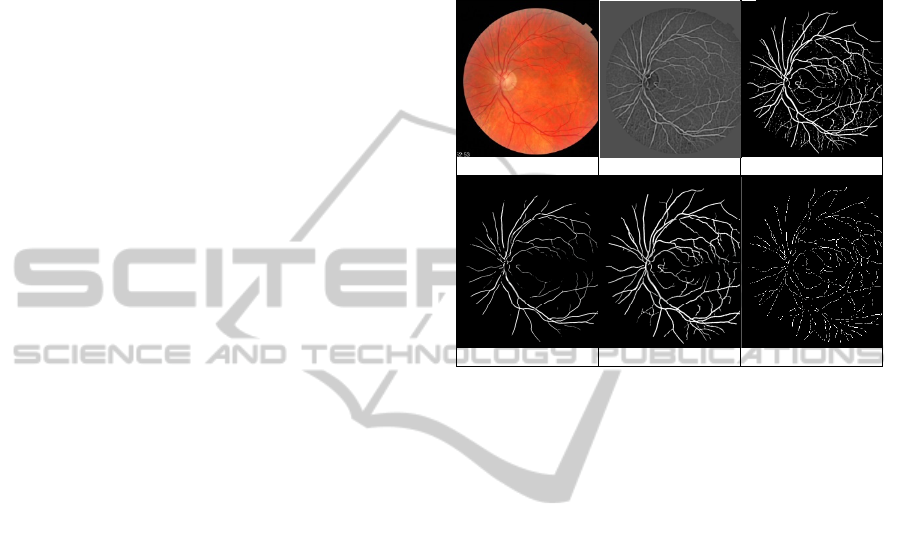
illustrated in Figure 1(b), is often considered as a
greyscale image, where bright pixels indicate a
higher probability of being a vessel pixel.
In order to obtain a vessel binary segmentation, a
hysteresis thresholding based morphological
reconstruction is applied. Hysteresis thresholding
employs a bi-threshold procedure such that the
intensity image is thresholded for two ranges of grey
values, one being included in the other. The image is
first segmented by a narrow threshold range which
concedes only high confidence object pixels and
thus also contains many false negatives. This image
is termed a marker image. The mask image is
generated by applying a wide threshold range to the
greyscale image. These threshold values are derived
from the intensity histogram of the non-null pixels;
each one of these thresholds; T
1
for the marker
image and T
2
for the mask image, is defined as the
highest intensity value such that the number of
pixels with intensities above this limit is greater or
equal to a predefined percentage. This percentage
value is empirically selected for T
1
and T
2
as 90%
and 95% respectively.
(a) (b) (c)
(d) (e) (f)
Figure 1: (a) Coloured retinal image, (b) Line strength
image, (c) Marker Image, (d) Mask Image, (e) Segmented
vascular, (f) Extracted Vessel segments.
The image is first segmented by a narrow
threshold range which concedes only high
confidence object pixels and thus also contains many
false negatives. This image is termed a marker
image. The mask image is generated by applying a
wide threshold range to the greyscale image. These
threshold values are derived from the intensity
histogram of the non-null pixels; each one of these
thresholds; T
1
for the marker image and T
2
for the
mask image, is defined as the highest intensity value
such that the number of pixels with intensities above
this limit is greater or equal to a predefined
percentage. This percentage value is empirically
selected for T
1
and T
2
as 90% and 95% respectively.
The marker image is used as a seed for the
morphological reconstruction using the mask image.
Figure 1(c-e) shows the marker, mask and
segmented vessels image respectively.
2.2
Vessel Segment Extraction
The skeletonization is applied to the binary
segmented vasculature image which reduces all the
vessels to a single centreline one pixel wide. The
bifurcation points and crossing points are detected in
the vessel centreline image by counting the
neighbourhood of each pixel. The vessel centreline
VISAPP2014-InternationalConferenceonComputerVisionTheoryandApplications
196

image is scanned to analyse the neighbours of each
pixel for eight-connectivity. This way, the
intersection number,
(, )In x y
is calculated for each
pixel
(, )
p
xy
of the centreline image, as shown in
equation (2).
8
1
(, ) 0.5* | (, ) (, )|
ii
i
I
nxy N xy N xy
(1)
where
(, )
i
Nxy
are the neighbours of the
analysed point,
(, )
p
xy
.
According to its intersection number In
each
pixel p(x,y) will be marked as vessel end point if
In(x,y)=1, vessel internal point if In(x,y)=2,
bifurcation if In(x,y)=3 and crossover if In(x,y)= 4
The detected bifurcation points and crossing
points are deleted from the vessel centreline image
and the retinal vasculature is cut into the vessel
segments as shown in Figure 1(f).
2.3 Image Normalization and Shade
Correction
The absolute colour of the blood in the vessels varies
between the images and across the subjects. To
overcome this variability some authors (Grisan and
Ruggeri 2003; Saez, González-Vázquez et al. 2012)
have used a quadrant based approach which is
computationally intense, also it enforces a condition
to have at least one arteriole and one venule per
quadrant, and it seems more suitable for optic disc
centered images rather than macula centered images.
We have used a simple linear transformation for
shade correction and variability normalization. For
this purpose, the estimate of the background o is
obtained by applying a filtering operation with a
large arithmetic mean kernel. The size of the filter
kernel is not a critical parameter as long as it is large
enough to ensure the blurred image contains no
visible structures such as vessels. In this work, we
use a 121×121 pixel size kernel. Then the difference
between the morphologically opened image I
o
and
the estimated background I
BE
is then computed for
each pixel to obtain a normalized image I
n
.
(, ) (, ) (, )
nOBE
Ixy I xy I xy
(2)
Likewise, when the fluctuation in background
intensity of retinal images is examined, there can be
significant variation in intensities between images
due to different illumination conditions in the
acquisition process. Therefore, a shade corrected
image is obtained by applying a global
transformation with the purpose of reducing the
intensity variation and contrast enhancement. For
this purpose, the pixel intensities are modified
according to the following global linear
transformation function,
0, ( , ) 0
( , ) 1, ( , ) 1
(, ),
n
Hn
if I x y
Ixy ifIxy
p
x y otherwise
_
(, ) (, ) 0.5
n MAX PIXEL
pxy I xy val
(3)
where, I
H
(x,y) is the homogenized image, I
n
(x,y)
is the normalized image shown in (7), val
MAX_PIXEL
is
the intensity value presenting the highest number of
pixels in the normalized image I
n
(x,y). The pixels
with intensity value equal to val
MAX_PIXEL
belong to
the background of the retinal image. This global
transformation will set them to 0.5 and will
standardize the intensity around this value of those
background pixels with different illumination
conditions. In order to find differences between
arterioles and venules, we have analysed RGB and
HIS colour spaces. Figure 2 shows the red, green
and blue components of the image shown in Figure
1(a) along with respective shade corrected images.
(a) (b) (c)
(d) (e) (f)
Figure 2: The colour spaces; (a-c) Red, Green and Blue
channels of RGB. (d-f) the shade corrected images of Red
green and blue channels respectively.
2.4 Feature Extraction
In previous work (Fraz, Remagnino et al. 2013), we
have computed the local orientation angle of vessel
segment and the width of vessel for each centreline
pixel. Based on this information we have extracted
the vessel profile for each centreline pixel. The
vessel profile is perpendicular to vessel direction,
and the length of vessel profile is equal to the vessel
width. The centreline, edges and the vessel segment
AutomatedArterioleandVenuleRecognitioninRetinalImagesusingEnsembleClassification
197

profiles are shown in Figure 3(a). We define three
types of features extracted from the vessel segments
for each centreline pixel: pixel based features,
profile based features and vessel segment based
features.
The pixel based features are the centreline pixel
intensity values taken from the respective colour
channel. The profile based features are the mean and
variance of the intensity values across a vessel
profile for each centreline. The vessel segment based
features are calculated in two ways for each
centreline pixel from the respective colour channel.
First, the mean and variance of the pixel intensities
are calculated for the entire vessel segment. Second,
the relatively longer vessel segments are divided into
smaller vessel parts of length of almost 50 pixels,
and then the mean and variance of the pixel
intensities are calculated with in these vessel . We
have tried and tested the different size of vessel sub-
segments ranging from 15 to 75 pixels and achieve
the best results using 50 pixels.
Figure 3(b) illustrates a vessel segment where
the edges are marked with blue lines and the
centreline pixels (Ci) are shown with green circles
on the black line. The vessel profile (Pi) consists of
the pixels in the line drawn perpendicular to the
local vessel direction, equal to the approximate
vessel width, and is shown in white. VPi are the
vessel segment parts. Table 1 shows the complete set
of features extracted for each centreline pixel.
Figure 3: (a) Vessel profiles; (b) Vessel Features for A/V
classification.
The feature set (fv) can be summarized as
follows.
( ) [ ]
() ( [ ])
() ( [ ])
() ( [ (, )])
( ) ( [ ( , )])
()
ii
ii
ii
ii
ii
fv n X C C vessel segment
fv n X P P vessel segment
fv n X P P vessel segment
fv n X VP x y VP vessel segment
fv n X VP x y VP vessel segment
fv n
( [(, )]) (, )
() ( [(, )]) (, )
XIxy pixelsxy invesselsegment
f
v n X I x y pixels x y in vessel segment
Table 1: Complete set of features extracted for each
centreline pixel.
No. Feature description Type
1-6
The centreline pixel intensity values from each
component of RGB and HSI colour space
Pixel
based
7-12
Mean of pixel intensities of vessel part VP
i
from
each colour component of RGB and HSI.
Vessel segment
part based
f
ea
t
u
r
es
13-18
Standard deviation of pixel intensities of vessel
part VP
i
from each colour component of RGB and
HSI.
19-22
Min and Max values of pixel intensities of vessel
segment part VP
i
from each R and G component
23-25
Mean of pixel intensities of vessel profiles P
i
from
each colour component of RGB
Vessel profile
based features
26-31
Standard deviation of pixel intensities of vessel
profiles P
i
from each colour component of RGB
and HSI.
32-35
Min and Max values of pixel intensities of vessel
profiles P
i
from R and G component.
36-41
Mean of pixel intensities of complete vessel
segment from each component of RGB and HSI.
Vessel segment
based features
42-47
Standard deviation of pixel intensities of complete
vessel segment from each colour component of
RGB and HSI.
48-51
Min and Max values of pixel intensities of
complete vessel segment from each R and G
component from RGB.
where, n is the number of features, X[C
i
] is the value
of the colour channel X at point C
i
in the image, X ɛ
{R,G,B,H,S,I}. For instance, R[C
i
] is the value of
the centreline pixels in the R colour component,
µ(G[P
i
]) is the mean of the vessel profile pixels in
the green channel.
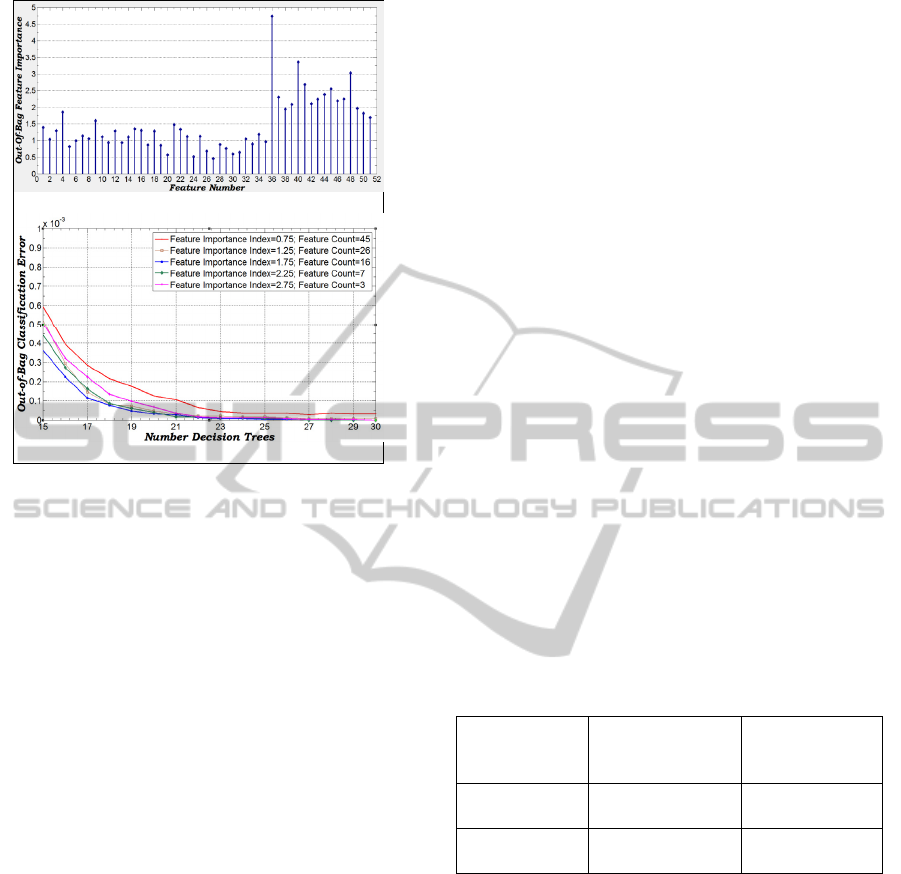
Figure 4(a) shows the graphs for feature
importance index calculated from classifiers created
with 30 decision trees and trained with 15000
training samples. Based on the information shown in
the graph, five sets of features have been created by
selecting those features, where the importance index
is more than the following values {0.75, 1.0, 1.5,
2.0,2.75}. Five classifiers have been trained with the
reduced feature sets and the out-of-bag (OOB)
classification error is computed for each of the
classifiers and is plotted in Figure 4(b). It has been
observed that the performance of the classifier
trained with 16 most significant features is best
among all; therefore 16-D is utilized for vessel
classification.
2.5 Vessel Labelling
The final labelling of vessel pixels is obtained by
employing supervised classification with an
ensemble classifier of bagged decision trees. We
assume that all the pixels in the vessel segment are
either in an arteriole or venule. The classification of
V
P
i
V
P
i+
VISAPP2014-InternationalConferenceonComputerVisionTheoryandApplications
198
(a)
(b)
Figure 4: (a) Feature Importance Index, (b) Out-of-Bag
classification error with reduced feature sets.
the retinal vessel is a two class classification
problem where each pixel in the image either
belongs to an artery (C
A
) or to a vein (C
V
).
In Ensemble classification (Polikar 2006),
multiple classifiers or models are tactically
generated and combined in order to give the solution
to a machine learning problem; with the goal of
obtaining better predictive performance than could
be obtained from any of the constituent classifiers /
models. This process is used to abbreviate the
likelihood of inadequate or unfortunate selection
while improving the classification or prediction
performance of the classifier. We use this strategy
instinctively in our day to day activities, where we
consider the opinion from several experts, evaluate
and merge their recommendations for establishing a
well optimized and well-versed conclusion. In the
same manner, the ensemble methods utilize multiple
classifiers/models to accomplish gain in
classification performance by mixing/aggregating
the outcomes from several weak learners into one
high-class classifier, with the goal of reducing the
variance and amplifying the confidence in the
decision. In the present work, the decision trees have
been used as the component classifier of the
ensemble system and which is created by employing
boot strapped aggregation
Let us consider a set of observations “x
n
” from
the feature vector with a known class label “y” as a
training set, where y ∈{C
A
,C
V
}. The objective is to
predict the class label “y” for the given observations.
The classifier assigns soft labels to the centreline
pixel labels, which can be regarded as a vote for the
label of the complete vessel segment, and the mean
of these votes is assigned as the label for the entire
vessel segment.
3 RESULTS
3.1
Evaluation Criteria and
Performance Measures
We have tested this methodology on retinal images
obtained from EPIC Norfolk study (EPIC-Norfolk
2013). The dataset contains 40 macula centered
retinal images from both of the eyes of 20 study
participants, with the vessel types manually labelled
by trained observers. The images were captured with
non-mydratic fundus cameras and saved in 24-bit
JPEG format with a resolution of 3000x2000 pixels.
The performance measure are obtained for the
centreline pixels in the entire image and evaluated
separately for arterioles and venules. For each of the
vessel type, the True Positives (TP), False Positives
(FP), True Negatives (TN) and False Negatives (FN)
are calculated in the same way as in (Saez,
González-Vázquez et al. 2012), and tabulated in
Table 2.
Table 2: Vessel classification (Class ∈ {a,v}).
Observer Identify
pixel ∈ Class
Observer
identify pixel
∉Class
System Identify
pixel ∈ Class
True Positive
(TP
Clas
s
)
False Positive
(FP
Class
)
System Identify
pixel ∉ Class
False Negative
(FN
Class
)
True Negative
(TN
Class
)
The algorithm is evaluated in terms of Detection
Rate / Sensitivity (SN
a|v
), Specificity (SP
a|v
),
Classification Accuracy (ACC
a|v
), Classification
Error Rate (CER
a|v
), Positive Predictive Value
(PPV
a|v
), Negative Predictive Value (NPV
a|v
) and the
Positive and Negative Likelihood Ratios (PLR
a|v
and
NLR
a|v
). The ACC
a|v
is measured by the ratio of the
total number of correctly classified pixels (sum of
true positives and true negatives) by the number of
pixels under consideration in the image. SN
a|v
reflects the ability of an algorithm to detect the true
positives. SP
a|v
measures the proportion of negatives
that are correctly identified. PPV
a|v
or precession rate
gives the proportion of vessel pixels with correctly
identified positive test results and NPV
a|v
is the
proportion of vessel pixels with negative test results
AutomatedArterioleandVenuleRecognitioninRetinalImagesusingEnsembleClassification
199
that are correctly identified. The predictive values
depends on the percentage of a/v in the retina
(prevalence), therefore the likelihood ratios (PLR
a|v
and NLR
a|v
) are also computed which are not
dependent on prevalence. These metrics are
illustrated in Table 3, based on the terms defined in
Table 2.
Table 3: Performance metrics for vessel classification.
Measure Description
SN
a|v
TP
a|v
/(TP
a|v
+FN
a|v
)
SP
a|v
TN
a|v
/(TN
a|v
+FP
a|v
)
ACC
a|v
(TP
a|v
+TN
a|v
)/(TP
a|v
+FP
a|v
+TN
a|v
+FN
a|v
)
CER
a|v
(FP
a|v
+FN
a|v
) / (TP
a|v
+FP
a|v
+TN
a|v
+FN
a|v
)
PPV
a|v
TP
a|v
/(TP
a|v
+FP
a|v
)
NPV
a|v
TN
a|v
/ (TN
a|v
+FN
a|v
)
PLR
a|v
SN
a|v
/ (1- Sp
a|v
)
NLR
a|v
(1-SN
a|v
)/ SP
a|v
3.2 Experimental Results
We have analysed the methodology by using 3149
vessel segments from 40 colour fundus images from
20 EPIC Norfolk participants. The algorithm is
evaluated by using a two-fold validation
methodology. The images of the right eye are
assigned to set S
1
and the left eye images are
allocated to the set S
2
. The classifier is then trained
on S
1
and tested on S
2
, followed by training on S
2
and testing on S
1
. The performance metrics are
computed separately for arterioles and venules and
presented in Table 4.
Table 4: Vessel classification performance metrics.
Measure Arterioles Venules
Tested
on S1
Tested
on S2
Tested
on S1
Tested
on S2
SN
a|v
0.9067 0.8795 0.7658 0.7652
SP
a|v
0.7658 0.7836 0.9067 0.8804
ACC
a|v
0.8387 0.8298 0.8327 0.8261
CER
a|v
0.1612 0.1701 0.1672 0.1738
PPV
a|v
0.8162 0.8170 0.8757 0.8513
NPV
a|v
0.8757 0.8513 0.8162 0.8170
PLR
a|v
3.6183 5.7391 7.9255 8.8678
NLR
a|v
0.1261 0.1623 0.2763 0.2505
The similarity in the performance metrics
obtained for the sets S
1
and S
2
indicates the
repeatability of the methodology in classification of
vessels. The sensitivity in the set S
1
for the arteries is
0.9067 and for the veins is 0.7658. This in turn
indicates that the probability of incorrect
classification (i.e., false positives) for arterioles and
venules is 9.33% and 23.42% respectively. Some
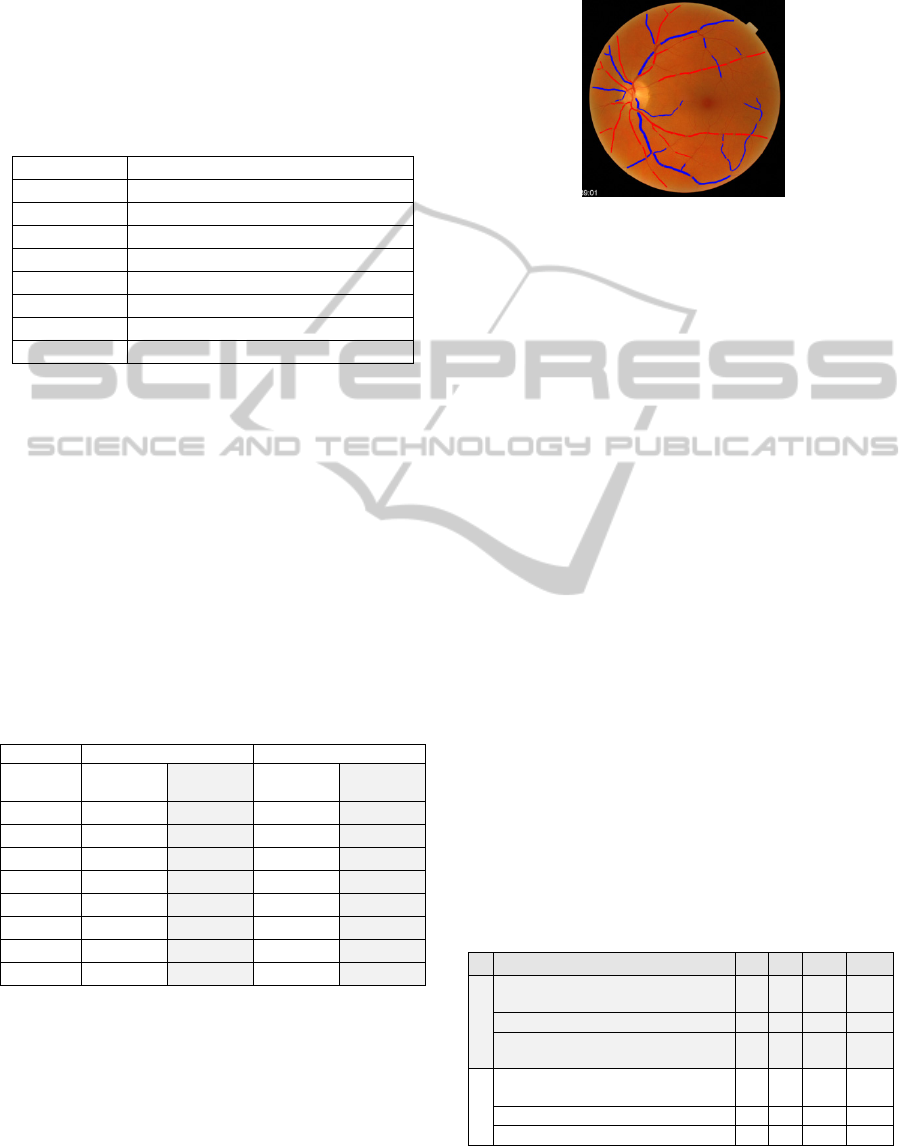
results of the presented A/V classification
methodology are illustrated in Figure 5.
Figure 5: A/V classification result.
A comparison of the proposed method’s
performance metrics with the recently published
methods is shown in Table 5. The sensitivity of
proposed method in identifying arterioles is higher
than the other methods, which suggests that our
method has lower probability of incorrect
classification. The higher specificity of our
algorithm in identifying veins also indicates fewer
false positives compared with other methods. We
have analysed 40 coloured fundus images compared
to 35, 58 and 35 images analysed in (Grisan and
Ruggeri 2003), (Saez, González-Vázquez et al.
2012) and (Relan, MacGillivray et al. 2013),
respectively. Also the resolution of our images are
3000 x 2000 pixels which is greater than 1300x1000
and 786x567 used in previous studies (Grisan and
Ruggeri 2003) (Saez, González-Vázquez et al.
2012). It should be noted that the resolution of our
test images is higher than those used to test other
methods, so the performance metrics for our method
may differ with a different image-set. The
classification accuracy of the method is dependent
upon the vessel segmentation results and the
extraction of vessel centreline pixels. Moreover, the
choice of a different classifier, feature set and retinal
image zone is likely to have an impact of the
method’s performance.
Table 5: Results of our method compared with recently
published methods.
Method SN SP PPV NPV
Arteries
(Saez, González-Vázquez et al.
2012)
0.78 0.89 0.88 0.80
(Relan, MacGillivray et al. 2013) 0.81 0.89 0.90 0.80
Proposed Method 0.90 0.76 0.81 0.87
Veins
(Saez, González-Vázquez et al.
2012)
0.87 0.79 0.81 0.86
(Relan, MacGillivray et al. 2013) 0.76 0.95 0.94 0.83
Proposed Method 0.76 0.90 0.87 0.81
VISAPP2014-InternationalConferenceonComputerVisionTheoryandApplications
200

4 CONCLUSIONS
An automated method for A/V classification in
retinal vasculature based on colour features utilizing
the ensemble classifer of boot strapped decision
trees is presented. To the best of our knowledge, this
is the first time the decision trees based ensemble
classifier has been used for A/V classification.
An application of image processing algorithms
for computer assisted analysis of digital fundus
images offers a number of advantages over a manual
system, including fast, timely and reliable
quantification of abnormalities. The presented
methodology will be incorporated in to a software
package QUARTZ (QUantitative Analysis of
Retinal vessel Topology and siZe). The QUARTZ
software will assist in examining arterio-venous
morphological associations with cardiovascular risk
factors and outcomes in large population based
studies, furthering our understanding of the vascular
changes / consequences associated with the
development of disease.
In future we aim to extend the QUARTZ
software to incorporate the analysis of other retinal
vessel features pathognomonic of cardiovascular
disease, including measurement of arterio-venous
ratio, identification of venous beading and
quantification of arterio-venous nicking.
ACKNOWLEDGEMENTS
The authors would like to thank Professor Paul
Foster and the European Investigation into Cancer in
Norfolk (EPIC Norfolk) study for providing the
retinal images used in this analysis. The EPIC
Norfolk study is supported by grants from the
Medical Research Council, Cancer Research UK and
Research into Ageing.
REFERENCES
Abràmoff, M. D., M. K. Garvin, et al. (2010). "Retinal
Imaging and Image Analysis." Biomedical
Engineering, IEEE Reviews in 3: 169-208.
Dashtbozorg, B., A. M. Mendonca, et al. (2013). "An
Automatic Graph-based Approach for Artery/Vein
Classification in Retinal Images." Image Processing,
IEEE Transactions on PP(99): 1-1.
EPIC-Norfolk. (2013). "European Prospective
Investigation of Cancer (EPIC)." Retrieved September,
2013, from http://www.srl.cam.ac.uk/epic/.
Fraz, M. M., S. A. Barman, et al. (2012). "An approach to
localize the retinal blood vessels using bit planes and
centerline detection." Computer methods and
programs in biomedicine 108(2): 600-616.
Fraz, M. M., P. Remagnino, et al. (2013). "Quantification
of blood vessel calibre in retinal images of multi-
ethnic school children using a model based approach."
Computerized Medical Imaging and Graphics 37(1):
60-72.
Fraz, M. M., P. Remagnino, et al. (2012). "Blood vessel
segmentation methodologies in retinal images – A
survey." Computer methods and programs in
biomedicine 108(1): 407-433.
Fraz, M. M., P. Remagnino, et al. (2012). "An Ensemble
Classification-Based Approach Applied to Retinal
Blood Vessel Segmentation." Biomedical Engineering,
IEEE Transactions on 59(9): 2538-2548.
Grisan, E. and A. Ruggeri (2003). A divide et impera
strategy for automatic classification of retinal vessels
into arteries and veins. Engineering in Medicine and
Biology Society, 2003. Proceedings of the 25th
Annual International Conference of the IEEE.
Huang, Y., J. Zhang, et al. (2012). "An automated
computational framework for retinal vascular network
labeling and branching order analysis." Microvascular
Research 84(2): 169-177.
Jack J. Kanski and Brad Bowling (2011). Clinical
Ophthalmology: A Systematic Approach. London,
Elsevier Health Sciences (UK).
Nguyen, U., A. Bhuiyan, et al. (2013). "An Automated
Method for Retinal Arteriovenous Nicking
Quantification from Colour Fundus Images."
Biomedical Engineering, IEEE Transactions on
PP(99): 1-1.
Nguyen, U. T. V., A. Bhuiyan, et al. (2012). "An effective
retinal blood vessel segmentation method using multi-
scale line detection." Pattern Recognition(0).
Niemeijer, M., X. Xiayu, et al. (2011). "Automated
Measurement of the Arteriolar-to-Venular Width Ratio
in Digital Color Fundus Photographs." Medical
Imaging, IEEE Transactions on 30(11): 1941-1950.
Owen, C. G., A. R. Rudnicka, et al. (2011). "Retinal
Arteriolar Tortuosity and Cardiovascular Risk Factors
in a Multi-Ethnic Population Study of 10-Year-Old
Children; the Child Heart and Health Study in England
(CHASE)." Arteriosclerosis, Thrombosis, and
Vascular Biology 31(8): 1933-1938.
Polikar, R. (2006). "Ensemble Based Systems in Decision
Making." IEEE Circuits and Systems Magazine 6(3):
21-45.
Relan, D., T. MacGillivray, et al. (2013). Retinal vessel
classification: sorting arteries and veins. 35th Annual
International Conference of the IEEE EMBS
Engineering in Medicine and Biology Society
(EMBC), Osaka, Japan, IEEE.
Ricci, E. and R. Perfetti (2007). "Retinal Blood Vessel
Segmentation Using Line Operators and Support
Vector Classification." Medical Imaging, IEEE
Transactions on 26(10): 1357-1365.
Rothaus, K., X. Jiang, et al. (2009). "Separation of the
retinal vascular graph in arteries and veins based upon
AutomatedArterioleandVenuleRecognitioninRetinalImagesusingEnsembleClassification
201

structural knowledge." Image and Vision Computing
27(7): 864-875.
Saez, M., S. González-Vázquez, et al. (2012).
"Development of an automated system to classify
retinal vessels into arteries and veins." Computer
methods and programs in biomedicine 108(1): 367-
376.
Vázquez, S. G., B. Cancela, et al. (2013). "Improving
retinal artery and vein classification by means of a
minimal path approach." Machine Vision and
Applications 24(5): 919-930.
Wong, T. Y., R. Klein, et al. (2001). "Retinal
microvascular abnormalities and incident stroke: the
Atherosclerosis Risk in Communities Study." The
Lancet 358(9288): 1134-1140.
VISAPP2014-InternationalConferenceonComputerVisionTheoryandApplications
202
