
Preliminary Monte Carlo based Inverse Model to Extract Optical
Tissue Properties from Experimental Diffuse Reflectance
Measurements
Coefficients Extraction for Gastrointestinal Dysplasia Detection
S. Pimenta
1
, E. M. S. Castanheira
2
and G. Minas
1
1
Centro Algoritmi, University of Minho, Campus de Azurém, Guimarães, Portugal
2
Centre of Physics (CFUM), University of Minho, Campus de Gualtar, Braga, Portugal
Keywords: Gastrointestinal Cancer, Spectroscopy, Diffuse Reflectance, Monte Carlo Simulations, Absorption
Coefficient, Scattering Coefficient.
Abstract: The ability to detect cancer at its earliest stages, called “dysplasia”, is the key of its successful treatment.
Optical techniques, such as diffuse reflectance and intrinsic fluorescence, may improve the ability to detect
gastrointestinal (GI) dysplasia since they have the potential to provide morphological and biochemical
information of normal and malignant tissues. However, those optical tissue properties can only be provided
if it is possible to extract information from the measured diffuse reflectance and intrinsic fluorescence
signals. This paper presents the implementation and the validation of a preliminary Monte Carlo based
inverse model to extract optical tissue properties, such as the absorption and the scattering coefficients, from
diffuse reflectance experimental measurements in phantoms.
1 INTRODUCTION
The detection of cancer in its initial stage is one of
the major goals of biomedical research.
Gastrointestinal (GI) cancers are usually preceded
by pre-cancerous changes and its early detection,
especially at the dysplasia stage (before
macroscopically visible changes occur on the
tissues), will increase the chances of a successful
treatment to the patient, improving the survival rate
(Georgakoudi et al., 2001, Ell, 2003, Yu et al.,
2008b).
A GI dysplasia is difficult to detect by visual
inspection during endoscopy or colonoscopy, due to
the lack of macroscopically easily visible changes on
the tissues that occur during the early stage of
cancer. Therefore, a large number of biopsies are
performed in order to increase the detection
probability of these invisible lesions (Georgakoudi
et al., 2001, Ell, 2003, Mayinger et al., 2003).
However, biopsies are procedures with sampling
errors (since the sample condition could not be
representative of the tissue malignant stage), high
cost and are uncomfortable to the patients. Finally,
their results are not immediately available, resulting
on a delay of patient’s treatment (Yu et al., 2008b,
Georgakoudi, 2006).
For all these reasons, in recent years, there has
been a strong interest towards the development of
new methods for a more objective detection of the
differences between premalignant and normal GI
tissues. Epithelial tissues acquire altered optical
properties during disease progression, especially due
to the changes on their morphological and
biochemical features. Spectroscopic techniques,
especially diffuse reflectance and intrinsic
fluorescence, have received special attention, due to
their potential to detect small changes in the tissues.
These techniques are based on light-tissues
interactions, allowing the detection of
macroscopically invisible lesions on the tissue
surface, in other words, lesions at the dysplastic
stage (Georgakoudi et al., 2001, Ell, 2003, Yu et al.,
2008b, Georgakoudi, 2006).
Concerning diffuse reflectance measurements, it
is used white light that is directed to the epithelial
tissues where it is absorbed and scattered, and part
of it returns to the tissue’s surface with information
about its optical properties. For the gastrointestinal
tissues, absorption is mainly due to the presence of
114
Pimenta S., Castanheira E. and Minas G..
Preliminary Monte Carlo based Inverse Model to Extract Optical Tissue Properties from Experimental Diffuse Reflectance Measurements - Coefficients
Extraction for Gastrointestinal Dysplasia Detection.
DOI: 10.5220/0004790201140121
In Proceedings of 2nd International Conference on Photonics, Optics and Laser Technology (PHOTOPTICS-2014), pages 114-121
ISBN: 978-989-758-008-6
Copyright
c
2014 SCITEPRESS (Science and Technology Publications, Lda.)

hemoglobin, while scattering is caused by the
collagen fibers present in the connective tissue. An
increase in hemoglobin concentration, related to the
angiogenesis (due to the early cancer progression),
results in a reduction of the diffuse reflectance
signal. Moreover, with cancer progression, the
epithelial thickness increases, which reduce the
quantity of light that reaches the collagen fibers.
Thus, a decrease in scattering also causes a decrease
of diffuse reflectance signal (Georgakoudi et al.,
2001, Yu et al., 2008b, Georgakoudi, 2006).
Regarding intrinsic fluorescence (fluorescence
without diffusion or absorption distortions),
epithelial tissues produce fluorescence when excited
by ultraviolet (UV) or blue-wavelength visible light.
GI tissues have multiple fluorophores related to the
cell structure and metabolism, such as collagen and
NADH, which are known as markers of
pre-cancerous changes. A decrease of collagen and
an increase of NADH could be related to dysplasia
progress, which results in a change of the
fluorescence signal (Georgakoudi et al., 2001, Ell,
2003, Georgakoudi, 2006, Skala et al., 2004).
The intensity and shape of diffuse reflectance
and intrinsic fluorescence spectra are dependent on
the tissues pathological state and, therefore, its
measurement can be used to extract information
about the degree of tissue's malignancy. Several
authors (Georgakoudi et al., 2001, Ell, 2003, Yu et
al., 2008b) have developed prototypes that include
those spectroscopy measurements for an accurate
detection of GI dysplasia. However, their
spectroscopy systems use expensive, complex and
bulky illumination and detection equipments, e.g.,
xenon arc lamps or UV lasers, optical fibers, high
quantum efficiency detectors and spectrographs that
may hamper its integration in several endoscopic
equipments.
Other groups have tried to develop spectroscopy
microsystems (Yu et al., 2008a, Lo et al., 2009).
Such microsystem, due to its small size, could be
used at any screening room. Yu et al. developed a
system to extract optical tissues properties from
diffuse reflectance signal, based on the use of
photodiodes as detectors. However, a
monochromator, a xenon lamp and optical fibers
were still used for illumination, which makes
complex the system miniaturization. Lo et al.
developed a system to measure tissues’ diffuse
reflectance signal, based on the use of LEDs (Light
Emission Diodes), as a light source, and
photodiodes, as light detectors. Despite this great
advance in reducing cost and size, the authors only
used five LEDs, which may not be enough to extract
information for a complete diagnosis of dysplasia.
Thus, the development of a spectroscopy
microsystem on a chip, without the need of regular
optical fibers or spectrometer, that might be used
in-loco, will have a high clinical value and
represents the main innovation of the target project
under this paper. The miniaturized system will be
portable and may be integrated, for example, in
endoscopes or colonoscopes, reducing the
limitations of the existing methods for the early
detection of GI dysplasia.
The microsystem is going to combine the
implementation of two optical techniques – diffuse
reflectance and intrinsic fluorescence – and the
concept of thin-film optical filters deposited on
silicon photodiodes. Miniaturized LEDs will be
incorporated on the chip, featuring illumination
sources for fluorescence and diffuse reflectance
measurements. The model presented in this paper
will extract information of the optical tissue
properties from the diffuse reflectance
measurements.
2 METHODOLOGY
As previously described in detail (Pimenta et al.,
2013), to achieve the objectives described above,
towards the microsystem implementation, the work
is divided in several phases. Then, the main tasks of
the project are presented:
Spectrophotometric measurements in order to
determine the relevant wavelengths/spectral
bands for the detection of GI dysplasia and
determine if the spectroscopic signals are
temperature dependent;
Implementation of mathematical models to
extract optical tissue properties from diffuse
reflectance and fluorescence measured
signals;
Design and fabrication of optical filters based
on Fabry-Perot thin-film optical resonators
and centered in the relevant wavelengths;
Design and fabrication of photodiodes with
improved quantum efficiency at the relevant
spectral bands.
Selection of miniaturized ultraviolet and
white-light LEDs for fluorescence and diffuse
reflectance measurements, respectively;
Microsystem implementation and test.
PreliminaryMonteCarlobasedInverseModeltoExtractOpticalTissuePropertiesfromExperimentalDiffuseReflectance
Measurements-CoefficientsExtractionforGastrointestinalDysplasiaDetection
115
3 WORK DEVELOPED
This section presents the experimental work that has
already been developed by the research team,
towards the spectroscopy microsystem
implementation.
3.1 Validation of the Relevant Spectral
Bands for Diagnosis
Studies performed by D. S. Ferreira (Ferreira et al.,
2011b, Ferreira et al., 2011a) concluded that 16
spectral bands (between 350 nm and 750 nm) would
be an appropriate number of wavelengths for
diagnosis of dysplasia. The authors designed 16
thin-films optical filters centered in 16 spectral
bands, and six of them were fabricated, at
INESC-MN, Lisbon. The validation of the fabricated
optical filters to extract spectroscopic signals was
performed with commercial equipment and
phantoms. As future work, the thin-film optical
filters centered in wavelengths in near-UV/blue and
near infra-red wavelength range will be improved.
3.2 Temperature Dependence of the
Diffuse Reflectance Signal
In order to evaluate the temperature dependence of
the diffuse reflectance signal, experimental
measurements with a set of phantoms were
performed, at four different temperatures: Tr (room
temperature), T1 (37 ºC), T2 (40 ºC) and T3 (42 ºC).
Thus, a set of liquid homogeneous phantoms
were created, with variable concentrations of an
absorber (hemoglobin) and a scatterer (intralipid), in
order to simulate tissues with variable properties.
The hemoglobin (Hb) used is water soluble and was
obtained from Sigma-Aldrich (H0267). The
intralipid used is a 20% emulsion and was also
obtained from Sigma-Aldrich (I141). During the
experimental tests, it was assumed that the
hemoglobin oxygenation was constant.
Table 1 shows the combinations of phantoms
created to the temperature tests. The experimental
measurements were performed in a commercial
UV-Vis-IR spectrophotometer (Shimadzu UV-
3101PC) equipped with integrating sphere.
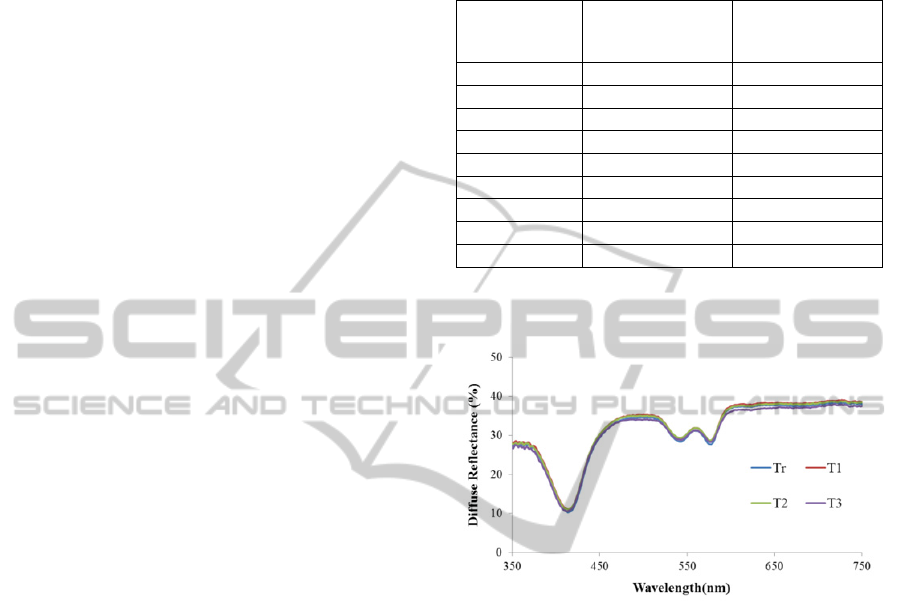
Figure 1 shows the results for the phantom 5. As
it can be seen, the diffuse reflectance signal is
similar for all the temperatures tested. Similar results
were obtained for all the phantoms, which allow
concluding that the temperature of the sample does
not affect the diffuse reflectance signal. Thus, to
extract the optical properties of a tissue, from the
diffuse reflectance signal, it is not necessary to
consider its temperature.
Table 1: Created phantoms for temperature tests.
Phantom
Intralipid mass
concentration
Hb concentration
(mg/mL)
1 0.50 0.25
2 0.50 0.50
3 0.50 1.00
4 1.00 0.25
5 1.00 0.50
6 1.00 1.00
7 2.00 0.25
8 2.00 0.50
9 2.00 1.00
In the future, similar tests will be performed for
the fluorescence signal.
Figure 1: Diffuse reflectance spectra at different
temperatures for phantom 5.
3.3 Implementation of a Model to
Extract Tissue Properties
As previously mentioned, the intensity and shape of
diffuse reflectance and fluorescence spectra are
dependent on the tissues properties and its
measurement can be used to extract information
about the degree of tissues dysplasia. The extraction
of these parameters requires the application of
mathematical models.
3.3.1 Theoretical Considerations
The diffuse reflectance signal is affected by the
absorption and scattering properties of the tissue,
which are defined by absorption and scattering
coefficients (μ
and μ
, respectively). μ
is related
with the concentration of chromophores in the
tissue, for example hemoglobin, while μ
is related
with the size and concentration of scattering
PHOTOPTICS2014-InternationalConferenceonPhotonics,OpticsandLaserTechnology
116

molecules, such as collagen fibers in the epithelial
tissues. Another important concept is the reduced
scattering coefficient, μ
’, which is the μ
with a
corrected factor (anisotropic factor) – equation (1).
is a factor that takes in account the angular
dependence of a scattering event (it can vary from -1
[scattering in the backward direction] to 1 [scattering
in the forward direction]), and its typical values for
epithelial tissues are in the range of 0.7 to 0.95,
across the wavelengths () 750 nm to 350 nm,
respectively (Yu et al., 2008b, Georgakoudi, 2006,
Zhu, 2007).
μ
’ μ
1
(1)
The information carried on the diffuse
reflectance signal is difficult to interpret due to the
presence of combined absorption and scattering
events. So, a change in the diffuse reflectance signal
could be related with a change in absorption and/or
scattering. Quantifying the affectation of each event
on the diffuse reflectance signal, allows the
possibility to extract μ
and μ
that define tissue
optical properties.
Several authors have developed models to extract
the absorption and scattering coefficients of tissues
from diffuse reflectance experimental
measurements. Some models are based on an
approximation of the transport diffuse equation,
developed by Zonios et al. in 1999 (Zonios et al.,
1999). Despite the low computational intensity,
these models are not suitable for a wide wavelength
range of the electromagnetic spectrum.
Other models are based in Monte Carlo
simulations (Bender et al., 2009, Palmer and
Ramanujam, 2006, Yu et al., 2008a) and in a scaling
approach developed by Graaff et al. (Graaff et al.,
1993). Monte Carlo simulations allow the simulation
of light propagation through a medium, such as a
tissue, and based on the optical properties of the
medium (μ
and μ
) obtaining the diffuse reflectance
spectrum. The main advantages of these simulations
are related to the facts that are suitable for a wide
range of absorption and scattering coefficients and
for a wide wavelength range of the electromagnetic
spectrum (UV-Vis to NIR). Moreover, the accuracy
of the model is dependent of the high contrast of the
chromophore extinction coefficient, over the
wavelengths, which happens with hemoglobin
(Palmer and Ramanujam, 2006).
In this paper, it is presented a preliminary inverse
model and its primary validation with liquid
homogeneous phantoms, that allow extracting μ
and μ
from the diffuse reflectance signal, using a
Monte Carlo forward model (freely available
software (Wang and Jacques, 1995) and an equation
that relates the diffuse reflectance with absorption
and scattering coefficients of a tissue, presented in
(Graaff et al., 1993). Equation (2) relates the diffuse
reflectance of a tissue (normalized by the diffuse
reflectance of a reference phantom with predefined
optical properties) at each wavelength – R
λ with
its optical properties as a function of wavelength -
μ
λ
and μ
:
R
λ
N
j
c
λ
c
λ
(2)
where N represents the mean number of interactions
between each photon (that exits the tissue surface)
and the medium. N
j is the portion of
reflected photons after j interactions with the
medium. cλ represents the ratio
, that
define the optical properties of the tissue. Finally,
c
λ represents the ratio
,
,
,
, that
define the optical properties of the reference
phantom. As mentioned above, its diffuse
reflectance (obtained with Monte Carlo forward
model) will be used for normalizing the
experimental diffuse reflectance of the tissue.
Thus, as first step it is necessary to choose a
reference phantom with known optical properties
that will be used in the forward Monte Carlo model
to obtain its diffuse reflectance. It was used a liquid
phantom with the following properties (similar to the
phantom in (Graaff et al., 1993) in order to use the
tabulated values for N and N
j
):
Hemoglobin (absorber) concentration equal to
0.25 mg/mL;
Intralipid (scatterer) mass concentration equal
to 2%, representing collagen fibers.
Based on the knowledge of hemoglobin and
intralipid concentration, it is possible to obtain the
absorption and scattering coefficients that define the
reference phantom as a function of wavelength
(μ
,
λ and μ
,
λ
, respectively).
The absorption coefficient as a function of
wavelength (μ
λ) could be obtained by the
application of the equation (3):
μ
λ
ln10ε
λ
C
(3)
where ε
λ is the extinction coefficient of the
absorber, that defines its capacity to absorb light as a
function of wavelength; C
is the absorber
concentration in the phantom.
The scattering coefficient as a function of
wavelength could be obtained using the Mie theory
for spherical particles, available as free software in
PreliminaryMonteCarlobasedInverseModeltoExtractOpticalTissuePropertiesfromExperimentalDiffuseReflectance
Measurements-CoefficientsExtractionforGastrointestinalDysplasiaDetection
117
(Mätzler, 2002). The source code can be used in
Matlab, given the volume fraction (vf = 0.1, for the
reference phantom), refractive index (n = 1.362
(Ding et al., 2005)) and size (s = 456 nm (Choukeife
and L’Huillier, 1999)) of the intralipid spheres and
the refractive index of the surrounding medium,
water in this case (n = 1.33).
Once the μ
,
λ and μ
,
λ
are known, they
could be used in the Monte Carlo forward model in
order to obtain the diffuse reflectance of the
reference phantom. As mentioned previously, it was
used a free available software (precompiled PC
version) (Wang and Jacques, 1995). However, some
Matlab functions were created in order to introduce
the features of the reference phantom in the model,
according to the software manual available in (Wang
and Jacques, 1995). The parameters used in the
Monte Carlo simulation were: total number of
photons (30000), μ
, μ
, isotropic
scattering (g=0), model dimensions (1cm(radius)
1cm(depth)), phantom refractive index (1.332) and
above/below refractive index medium (1.0 and 1.5,
respectively).
As a second step, it is necessary to use a Matlab
optimization function, lsqcurvefit, with initial
random input solutions for absorption and scattering
coefficients (λ). This function is based on the
least-squares algorithm and its main goal is
iteratively updated λ until the value of modulated
reflectance (equation (1) output) is similar to the
experimental diffuse reflectance that define our
phantom, allowing to extract its optical properties
μ
λ and μ
λ
. It is important to note again that
both diffuse reflectances (modulated and
experimental) are normalized by the diffuse
reflectance of the reference phantom (previously
obtained with Monte Carlo forward model).
3.3.2 Tissue Phantoms
For a preliminary validation of the implemented
model, a set of liquid homogeneous phantoms were
created, with variable concentrations of the absorber,
hemoglobin (0.27 mg/mL, 0.49 mg/mL and
1.03 mg/mL), and with a mass concentration of
intralipid of 0.5%. The diffuse reflectance spectrum
of each phantom was measured between 350 nm and
750 nm, using an UV-Vis-NIR spectrophotometer
equipped with integrating sphere (Shimadzu
UV-3101PC). Moreover, the wavelength-dependent
hemoglobin extinction coefficient was obtained with
the same spectrophotometer.
3.3.3 Validation of Monte Carlo Forward
Model
Once the implemented inverse model uses the Monte
Carlo forward model to obtain the diffuse
reflectance of the reference phantom, it was
performed a validation using the homogeneous
phantoms created, for evaluating the accuracy of the
free available software used (Wang and Jacques,
1995), in the achievement of the diffuse reflectance
of a medium.
Thus, knowing the hemoglobin and intralipid
concentrations, for each created phantom, the optical
properties that define each of them – μ
λ and
μ
λ
– were obtained by the application of
equation (3) and the freely available software for
Mie theory. After that, these properties were used in
a Monte Carlo simulation with the following
parameters: total number of photons (30000), μ
,
μ
, anisotropic scattering (g=0.8), model
dimensions (1cm(radius) 1cm(depth)), phantom
refractive index (1.332) and above/below refractive
index medium (1.0 and 1.5, respectively).
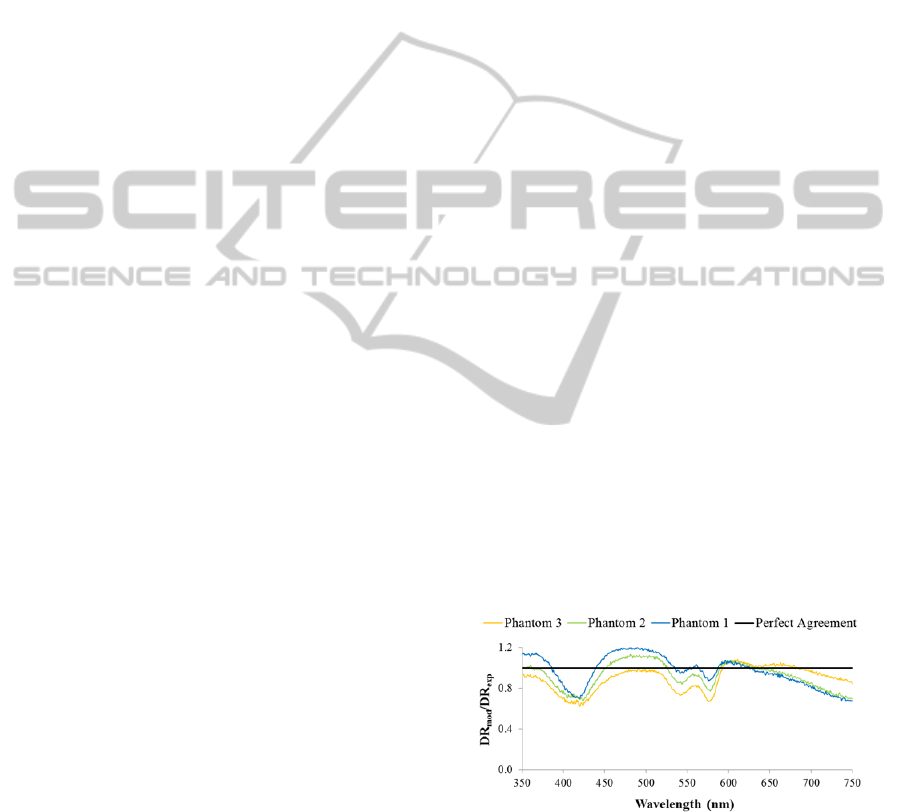
Figure 2 shows the ratio between the modeled
diffuse reflectance (DR
mod
) and the measured diffuse
reflectance (DR
exp
) for each created phantom as a
function of wavelength. Despite the slight deviations
from unity (the perfect agreement), especially in
wavelengths that correspond to the hemoglobin
absorption peaks (420 nm, 540 nm and 580 nm), in
general it can be concluded that there is a good
agreement between the modeled (obtained from
Monte Carlo forward model) and the
experimental/measured diffuse reflectance, since de
ratio between them is close to unity across the
analyzed wavelengths.
Figure 2: Ratio between the modeled diffuse reflectance
(DR
mod
) and the measured diffuse reflectance (DR
exp
)
spectra for each created phantom.
3.3.4 Validation of the Inverse Model
A preliminary validation of the implemented inverse
model was performed using the created liquid
phantoms. Thus, for each phantom, the optimization
PHOTOPTICS2014-InternationalConferenceonPhotonics,OpticsandLaserTechnology
118
function (lsqcurvefit) was repeated several times
with different initial input solutions, for absorption
and scattering coefficients, in order to increase the
probability of extracting a global minimum.
Moreover, the obtained coefficients were
mathematically approximated, in order to ensure that
the values follow the form of the equations that
characterize them.
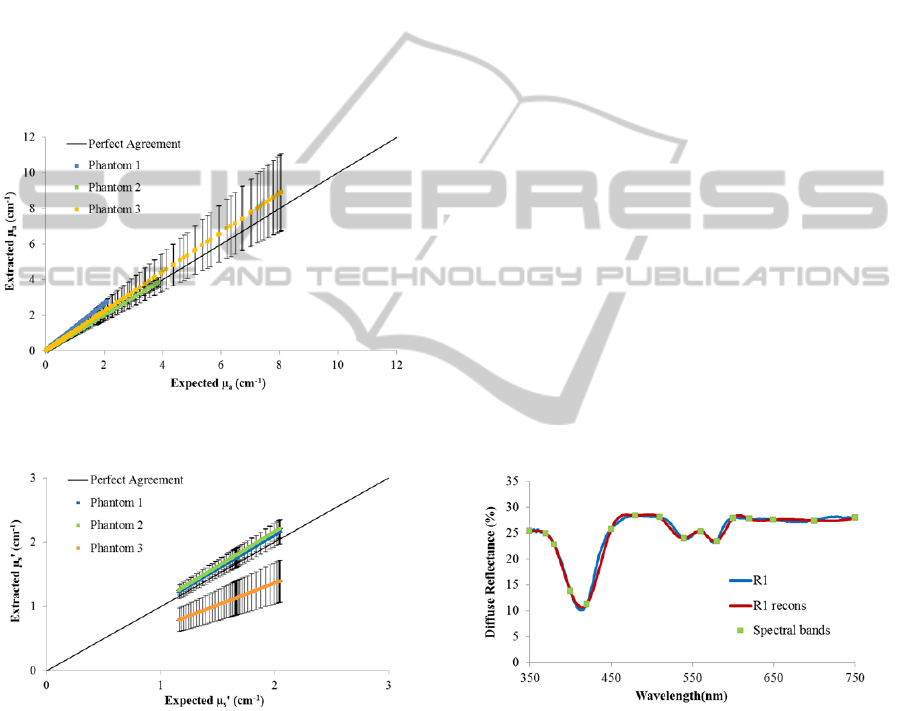
Next, the absorption and scattering coefficients
extracted from the inverse model were compared
with the expected/theoretical coefficients of each
phantom. Figures 3 and 4 show the plots of the
extracted versus expected absorption and reduced
scattering coefficients, respectively, for all
wavelengths and each created phantom.
Figure 3: Extracted versus expected absorption coefficient
(μ
cm
) for each created phantom.
Figure 4: Extracted versus expected reduced scattering
coefficient (μ
′
cm
) for each created phantom.
As it can be seen in figures 3 and 4, the
implemented inverse model allows the extraction of
the absorption and scattering coefficients with low
differences between the expected and extracted
tissue optical properties, especially for the
absorption properties, since the extracted
coefficients are very similar to the expected.
Concerning the reduced scattering coefficient,
phantom 3 has a slight deviation from the expected
result. So, in the future it will be important to
evaluate if this deviation affects the final tissue
diagnostic. If it is significant in the diagnostic result,
the implemented model must be improved in order
to avoid diagnostic errors.
3.3.5 Validation of the Inverse Model with
the 16 Spectral Bands
As previously mentioned, studies performed by D.
S. Ferreira (Ferreira et al., 2011b, Ferreira et al.,
2011a) concluded that 16 spectral bands (350, 370,
380, 400, 420, 450, 480, 510, 540, 560, 580, 600,
620, 650, 700 and 750 nm) would be an appropriate
number of wavelengths for diagnosis of dysplasia.
So, based on the diffuse reflectance values at the
16 spectral validated bands, it was reconstructed the
total diffuse reflectance, for each phantom, using a
Matlab fitting function – spline. The main advantage
of using splines is related with the fact that they can
be used to represent functions over large intervals,
where it would be impractical to use a single
approximating polynomial, since they are smooth
piecewise polynomials.
Figure 5 shows the measured (experimental)
diffuse reflectance spectra (blue curve – R1) and the
reconstructed diffuse reflectance spectra (red curve –
R1 recons) from phantom 1. As mentioned above,
R1 recons are obtained based on the use of the 16
spectral bands previously validated (green points -
Spectral bands) and the spline function.
Figure 5: Diffuse Reflectance from phantom 1:
experimental spectra (R1), reconstructed spectra (R1
recons) and the 16 spectral bands used for reconstruction
(Spectral bands).
Based on the reconstructed spectra of the liquid
created phantoms (1, 2 and 3), a validation of the
implemented inverse model was performed, in order
to compare the results obtained with the original or
experimental spectra. Again, for each phantom, the
optimization function (lsqcurvefit) was repeated
several times with different initial input solutions, in
order to extract the absorption and scattering
PreliminaryMonteCarlobasedInverseModeltoExtractOpticalTissuePropertiesfromExperimentalDiffuseReflectance
Measurements-CoefficientsExtractionforGastrointestinalDysplasiaDetection
119

coefficients. Figures 6 and 7 show the plots of the
extracted versus expected absorption and reduced
scattering coefficients, respectively, for all
wavelengths and for each created phantom.
As it can be seen in figures 6 and 7, the use of
the reconstructed spectra (from the 16 spectral
bands) in the implemented inverse model allows the
extraction of the absorption and scattering
coefficients with low differences between the
expected and extracted tissue optical properties,
especially for the absorption coefficient. As
previously referred, some slight deviations in the
reduced scattering coefficient must be evaluated.
Figure 6: Extracted versus expected absorption coefficient
(μ
cm
), using the reconstructed spectra for each
created phantom.
Figure 7: Extracted versus expected reduced scattering
coefficient (μ
′
cm
), using the reconstructed spectra
for each created phantom.
4 CONCLUSIONS AND FUTURE
GUIDELINES
The work performed until now allow to conclude
that the temperature of a tissue doesn’t affect the
diffuse reflectance signal. Similar tests will be
performed for the fluorescence signal and using
different phantoms.
The absorption and scattering properties of a
tissue could be extracted from the diffuse reflectance
signal, by the implementation of a Monte Carlo
based inverse model. The extracted coefficients
obtained, with a set of phantoms, were similar to the
expected coefficients. Moreover, the implemented
inverse model was validated using the reconstructed
spectra of the phantoms, based only in 16 values of
diffuse reflectance, for each phantom. This
validation will allow the design and fabrication of
optical filters and photodiodes, centered in these 16
spectral bands.
Nevertheless, in the future the model will be
tested with more and different phantoms, for
evaluating its accuracy in the extraction of optical
tissue properties. Moreover, different reference
phantoms will also be used to test their influence in
the accuracy of the model.
Finally, the other tasks described in section 2 are
ongoing towards the full microsystem
implementation.
ACKNOWLEDGEMENTS
This work is funded by FEDER funds through the
"Eixo I do Programa Operacional Fatores de
Competitividade (POFC) QREN, project reference
COMPETE: FCOMP-01-0124-FEDER-020241, and
by FCT- Fundação para a Ciência e a Tecnologia,
project reference PTDC/EBB-EBI/120334/2010.
S. Pimenta thanks the FCT for the
SFRH/BD/87605/2012 PhD grant.
REFERENCES
Bender, J. E., Vishwanath, K., Moore, L. K., Brown, J. Q.,
Chang, V., Palmer, G. M. & Ramanujam, N. 2009. A
Robust Monte Carlo Model For The Extraction Of
Biological Absorption And Scattering In Vivo. IEEE
Transactions On Biomedical Engineering, 56, 960-
968.
Choukeife, J. E. & L’huillier, J. P. 1999. Measurements Of
Scattering Effects Within Tissue-Like Media At Two
Wavelengths Of 632.8 Nm And 680 Nm. Lasers In
Medical Science, 14, 286–296.
Ding, H., Lu, J. Q., Jacobs, K. M. & Hu, X.-H. 2005.
Determination Of Refractive Indices Of Porcine Skin
Tissues And Intralipid At Eight Wavelengths Between
325 And 1557 Nm. Optical Society Of America, 22,
1151-1157.
Ell, C. 2003. Improving Endoscopic Resolution And
Sampling: Fluorescence Techniques. Gut, 52, 30-33.
Ferreira, D. S., Mirkovic, J., Wolffenbuttel, R. F., Correia,
J. H., Feld, M. S. & Minas, G. 2011a. Narrow-Band
Pass Filter Array For Integrated Opto-Electronic
PHOTOPTICS2014-InternationalConferenceonPhotonics,OpticsandLaserTechnology
120

Spectroscopy Detectors To Assess Esophageal Tissue.
Biomedical Optics Express, 2, 1703-1716.
Ferreira, D. S., Pinto, V. C., H.Correia, J. & Minas, G.
2011b. Spectroscopic Detection Of Gastrointestinal
Dysplasia Using Optical Microsensors. Ieee
Transactions On Biomedical Engineering, 58, 2633-
2639.
Georgakoudi, I. 2006. The Color Of Cancer. Journal Of
Luminescence, 119-120, 75-83.
Georgakoudi, I., Jacobson, B. C., Dam, J. V., Backman,
V., Wallace, M. B., Mueller, M. G., Zhang, Q.,
Badizadegan, K., Sun, D., Thomas, G. A., Perelman,
L. T. & Feld, M. S. 2001. Fluorescence, Reflectance,
And Light-Scattering Spectroscopy For Evaluating
Dysplasia In Patients With Barrett’s Esophagus.
Gastroenterology, 120, 1620-1629.
Graaff, R., Koelink, M. H., Mul, F. F. M. D., Zijlstra, W.
G., Dassel, A. C. M. & Aarnoudse, J. G. 1993.
Condensed Monte Carlo Simulations For The
Description Of Light Transport. Applied Optics, 32(4),
426-434.
Lo, J. Y., Yu, B., Fu, H. L., Bender, J. E., Palmer, G. M.,
Kuech, T. F. & Ramanujam, N. 2009. A Strategy For
Quantitative Spectral Imaging Of Tissue Absorption
And Scattering Using Light Emitting Diodes And
Photodiodes. Optics Express, 17, 1372-1384.
Mätzler, C. 2002. Maetzler's Matlab Code For Mie Theory
[Online]. Available: Http://Omlc.Ogi.Edu/Software/
Mie/ [Accessed Fevereiro 2013.
Mayinger, B., Jordan, M., Horner, P., Gerlach, C.,
Muehldorfer, S., Bittorf, B. R., Matzel, K. E.,
Hohenberger, W., Hahn, E. G. & Guenther, K. 2003.
Endoscopic Light-Induced Autofluorescence
Spectroscopy For The Diagnosis Of Colorectal Cancer
And Adenoma. Journal Of Photochemistry And
Photobiology B: Biology, 70, 13-20.
Palmer, G. M. & Ramanujam, N. 2006. Monte Carlo-
Based Inverse Model For Calculating Tissue Optical
Properties. Part I: Theory And Validation On
Synthetic Phantoms. Applied Optics, 45(5), 1062-
1071.
Pimenta, S., Monteiro, T. S., Goncalves, L. M. & Minas,
G. 2013. Spectroscopy And Ph Biosensors For The
Detection Of Gastrointestinal Dysplasia. Published In
Proceedings Of Ieee Embs, 3rd Portuguese
Bioengeneering Meeting, Braga, Portugal, 20-22
February.
Skala, M. C., Palmer, G. M., Zhu, C., Liu, Q., Vrotsos, K.
M., Marshek-Stone, C. L., Gendron-Fitzpatrick, A. &
Ramanujam, N. 2004. An Investigation Of Fiber-Optic
Probe Designs For Optical Spectroscopic Diagnosis
Of Epithelial Pre-Cancers. Lasers In Surgery And
Medicine, 34(1), 25-38.
Wang, L. & Jacques, S. L. 1995. (Mcml) Monte Carlo For
Multi-Layered Media [Online]. Available:
Http://Omlc.Ogi.Edu/Software/Mc/ [Accessed
Fevereiro 2013.
Yu, B., Lo, J. Y., Kuech, T. F., Palmer, G. M., Bender, J.
E. & Ramanujam, N. 2008a. Cost-Effective Diffuse
Reflectance Spectroscopy Device For Quantifying
Tissue Absorption And Scattering In Vivo. Journal Of
Biomedical Optics, 13, 060505.
Yu, C.-C., Lau, C., O'donoghue, G., Mirkovic, J., Mcgee,
S., Galindo, L., Elackattu, A., Stier, E., Grillone, G.,
Badizadegan, K., Dasari, R. R. & Feld, M. S. 2008b.
Quantitative Spectroscopic Imaging For Noninvasive
Early Cancer Detection. Optics Express, 16, 16227-
16239.
Zhu, C. 2007. The Use Of Fluorescence And Diffuse
Reflectance Spectroscopy For Breast Cancer
Diagnosis. Phd, University Of Wisconsin-Madison.
Zonios, G., Perelman, L. T., Backman, V., Manoharan, R.,
Fitzmaurice, M., Dam, J. V. & Feld, M. S. 1999.
Diffuse Reflectance Spectroscopy Of Human
Adenomatous Colon Polyps In Vivo. Applied Optics,
38, 6628-6637.
PreliminaryMonteCarlobasedInverseModeltoExtractOpticalTissuePropertiesfromExperimentalDiffuseReflectance
Measurements-CoefficientsExtractionforGastrointestinalDysplasiaDetection
121
