
Analysis of Glioblastoma’s Peritumoral Brain Zone
Developing of Per-operative Analysis and New Therapeutic Targets
Jean-Michel Lemée
1,2
, Anne Clavreul
1
and Philippe Menei
1,2
1
Department of Neurosurgery, University Hospital of Angers, 4, rue Larrey, 49933, Angers Cedex 09, France
2
INSERM U1066, LUNAM University, 4, rue Larrey, 49933, Angers Cedex 09, France
1 STAGE OF THE RESEARCH
Our research topic is focused on the glioblastoma
(GB), an aggressive primary brain tumor, and
especially the study of its peritumoral brain zone
(PBZ). The aim of our study is to developp per-
operative analysis of GB-PBZ to guide the surgery
and to develop targeted therapies for post-operative
therapeutic management.
During the first part of my PhD, we studied the
heterogeneity of the glioblastoma’s peritumoral
brain zone (GB-PBZ) using the different techniques
used routinely to analyze tumor samples : radiology,
histology, flow cytometry, genomic, transcriptomic,
proteomic and cellular cultures. We identified a
specific profile of the GB-PBZ, distinct from both
the normal brain tissu and the glioblastoma’s
tumoral zone. This research will be the subject of a
publication, currently being drafted.
We also identified in this study several
alterations specific to the GB-PBZ. These alterations
of the GB-PBZ profile will be the subject of further
studies to define whether or not their expression may
be a prognosis or therapeutic response marker, or a
potential target for targeted therapies.
More, the proteomic study of the GB-PBZ
confronted us to a methodological problem: the
absence of a defined control sample for the
proteomic analysis of brain tumor samples. This
methodological problem was solved and was the
subject of a publication (Lemée et al. 2013).
2 OUTLINE OF OBJECTIVES
The second part of my PhD will be focused on two
main subjects: the development of per-operative
analysis of the GB-PBZ to guide the surgical
resection of the tumor, and the molecular analysis of
the GB-PBZ to develop new therapeutic targets for
GB treatment.
3 RESEARCH PROBLEM
The GB is the most frequent and aggressive primary
tumor of the central nervous system with an
incidence of 4.96/100.000/year, a poor prognosis
and the absence of a curative treatment.
One of the leading causes of this poor prognosis
is the systematical recurrence of the tumor,
occurring in 90% of cases from the margin of the
resection’s cavity (Sherriff et al., 2013); (Petrecca et
al., 2013). Few observations have been made on this
peritumoral zone defined by the brain tissues outside
the contrast enhancement on gadolinium T1
weighted MR-scan.
Most of them were post mortem observation and
indicated that microscopic tumor infiltration
extended a considerable distance beyond the low
densities areas on CT scan or the high-intensity
areas on T2 weighted MRI. This may offer a
potential explanation to the tumoral recurrence
around the resection’s cavity (Yamahara et al.,
2010); (Nagashima et al., 1999).
Infiltrating tumor cells have been isolated, by the
culture in a serum-free medium, of biopsy
specimens taken from the brain tissue surrounding
the resection cavity (Glas et al., 2010). These cells
display the alterations typical of GB, but can be
distinguished from tumor cells isolated from the
tumor core on the basis of their distinctive molecular
marker profiles and responses to drug and irradiation
challenges in vitro (Glas et al., 2010 #87).
Using a different type of culture medium, we
have isolated, from the brain tissue surrounding GB,
another unexpected population of cells from the
stroma that we have named glioblastoma associated
stromal cells (GASCs) (Clavreul et al., 2011). These
cells do not display the genomic alterations typical
of GB cells and resemble the cancer-associated
fibroblasts (CAFs) described in the stroma of
carcinomas.
These few studies showed that the peripheral
brain zone, although macroscopically and
54
Lemée J., Clavreul A. and Menei P..
Analysis of Glioblastoma’s Peritumoral Brain Zone - Developing of Per-operative Analysis and New Therapeutic Targets.
Copyright
c
2014 SCITEPRESS (Science and Technology Publications, Lda.)

radiologically considered as normal brain tissue, is
in fact distinct at different levels from both the
tumor zone of glioblastoma (TZ) and the normal
brain (NB). A better understanding of the
characteristics of the PBZ is critical to understand
the mechanisms of GB recurrence, optimize the
quality of the surgical resection and develop new
therapies.
3.1 Per-operative Study of
Glioblastoma’s Peritumoral Brain
Zone
The gross total resection of the GB is an important
prognostic factor of overall survival and tumoral
progression-free survival for the patient (Chaichana,
2013).
The difficulty of achieving a gross total resection
in GB lays in the infiltrative nature of the tumor,
with the presence a small tumoral infiltration in the
surroundings of the tumor. To avoid this we cannot
take security margins around the tumor during the
resection, because of the major risk of aggravating
the already impaired neurological status of the
patient, without a significant benefit for him. In this
condition, a macroscopically complete resection is
the optimal surgical treatment we can propose.
Thus, the development of new techniques of
neurosurgery, or the adaptation of the existing ones,
to allow a fast, easy, accurate and reproducible
analyse of the GB-PBZ during the surgery is
essential to ensure a complete resection of the tumor
without impairing the patient’s neurological status.
Technological advances in this field may be quickly
transferred to clinic and will have a critical impact
on the patients’ therapeutic management, functional
outcome and survival.
3.2 Development of New Therapeutic
Targets
Despite the development of new therapies, the
prognosis of GB remains poor with a mean survival
of 15 months with an optimal therapeutic
management. The identification and the validation of
new therapeutic targets are essential for the future of
adjuvant GB’s therapy in an era of personalized
medicine.
The future of GB therapy will be the
development of targeted therapies for custom-made
treatment after specific tumor profiling. One lead is
the identification of targets for existing targeted
therapies like Herceptin in Her2+ breast cancer or
vemurafenib in B-Raf proto-oncogene V660E muted
lung cancer and melanoma. The second option may
be the identification of specific target for GB and
GB-PBZ allowing the development of new targeted
therapies through the use of lipidic nanocapsules
(LNC). Different chemotherapeutic agents can be
encapsulated in these particles and thus focused
delivery will allow us to increase the dose of
therapeutic agent without increasing the potential
side effects of chemotherapy agents.
4 STATE OF THE ART
4.1 Glioblastoma’s Therapeutic
Management
GB is the most frequent and aggressive primary
tumor of the central nervous system. The
mechanisms of recurrence remain unclear but the
infiltration of the PBZ by GB cells may be
considered as the explanation of the high rate of
local recurrences.
The gold standard for GB’s therapeutic
management is a surgical gross total resection
followed by radiotherapy and chemotherapy with
temozolomide, according to the protocol described
by Stupp (Stupp, 2009). However, even with this
optimal treatment, the prognosis remains poor with a
progression-free survival of 7 months and a mean
survival of 15 months.
4.2 Per-operative Study of the
Peritumoral Brain Zone
As mentioned before, the quality of the surgical
resection of the GB is critical for its therapeutic
management making the realization of per-operative
quality controls of the surgical resection essential.
Actually, the gold standard of GB surgery is to
perform a macroscopically complete resection,
stopping the surgery when no tumoral tissue is left in
the surgical cavity, without taking any security
margin. But if the complete resection of the tumor
jeopardizes the neurological status of the patient, a
subtotal resection is performed instead. Numerous
techniques have been used to attempt to develop a
better per-operative control of the resection’s
quality.
The actual techniques used in per-operative GB-
PBZ study are the per-operative imaging and the
per-operative fluorescence. Per-operative imaging
consist in the realization during the surgery of a
brain scanner using a CT- or a MR-scan to ensure
the complete resection of the tumor. Due to the cost
AnalysisofGlioblastoma'sPeritumoralBrainZone-DevelopingofPer-operativeAnalysisandNewTherapeuticTargets
55

of a dedicated scanner or MRI in the operating
room, this technique is still confidential and only
available in a few centers.
The use of per-operative fluorescence allows to
see with a specific light the tumoral cells stained in
red, after the ingestion of a 5-amino levulinic acid
analog by patients 5 hours prior to surgery. This
simplify the completion of the surgical resection by
showing clearly where are located the tumoral
remnants on the wall of the resection’s cavity. This
technique is still under evaluation in France and is
actually the object of a national clinical trial
(RESECT).
4.3 Therapeutic Targets in GB
Actually, there are no validated or available drugs
targeted therapies available for GB treatment. GBs
are currently under a specific study to identify
potential targets for known and commercially
available targeted therapies in AcSE study.
For now, we didn’t dispose of a specific marker
of GB or GB-PBZ tumoral tissue for target specific
delivery of chemotherapeutic agents.
5 METHODOLOGY
5.1 Per-operative Study of the
Glioblastoma’s Peritumoral Brain
Zone
In the search of new means to study the GB-PBZ,
we will study several techniques routinely used in
medical practice in specialities other than the
Neurosurgery.
5.1.1 Second Harmonic Imaging Microscopy
and Two Photon-excited Fluorescence
First, we decided to study two modern techniques of
microscopy: the Second Harmonic Imaging
Microscopy (SHIM) and the Two Photons-Excited
Fluorescence (TPEF). SHIM is an efficient non-
destructive method to study in vivo tissues without
involving molecular excitation. TPEF is a technique
similar to the immunofluorescence, but less
susceptible to lead to phototoxicity and
photobleaching. SHIM reacts specifically to non-
centrosymmetric molecules such as collagen,
myosin and microtubules. This is an interesting lead
because normal brain cells do not produce collagen
whereas GB cells produce collagen in their
extracellular matrix using it as a support for cell
migration.
TPEF is a living imaging fluorescence that
allows deep study of tissue up to 1mm. TPEF can
excite fluorescent dyes with far less toxicity than the
other fluorescent methods and has a high definition
thank to a strong suppression of background signal
due to infrared photon absorption.
Preliminary Study. In collaboration with the Dr
Denis Gindre, of the Department of Physics of the
University of Angers, we did a preliminary study on
paraffin embedded sections of brain tumor to assess
the feasibility of the study.
The obtained images showed a differential SHIM
signal between the different brain zones, with the
absence of SHIM signal in the central tumoral
necrosis, a strong signal in tumoral zone and an
intermediary signal in the GB-PBZ (Figure 1). These
data are in favour of the continuation of this study
on a wider panel of brain tumor samples.
Adaptation of the Technique. However, before
continuing in scanning the samples, we will have to
solve a problem specific to the brain tumor and the
SHIM, the representativity of the sample.
Indeed, the SHIM and TPEF techniques allow the
scanning of a square of 100x100 μm. This is an
insufficient size to obtain a representative SHIM and
TPEF profile of a GB sample, which is by definition
a heterogeneous tumor with foci of necrosis and
tumoral proliferation.
Dr. Gindre and one of his PhD student are
currently creating a program which will scan random
squares of 100x100 μm of the tumor sample under
different polarization angle to create an mean image
of the SHIM and TPEF signals of the lesion in order
to obtain a representative SHIM and TPEF profiles
of the lesion.
Creating an Atlas. The next objective, when the
acquisition of a representative SHIM and TPEF
profiles of the studied samples will be validated, will
be to create an atlas of the SHIM and TPEF profiles
of the different types of normal brain and brain
tumor samples, using a collection of brains’ paraffin
embedded sections with an histologically confirmed
diagnosis.
The constitution of the atlas of SHIM and TPEF
signals of brain tumors samples will allow us to
assess the samples obtained per-operatively, based
on the profile obtained and its comparison with the
atlas.
Per-operative Testing. The final step will be to
perform extemporaneous analysis of the tumor
samples obtained during surgery to perform an
BIOSTEC2014-DoctoralConsortium
56
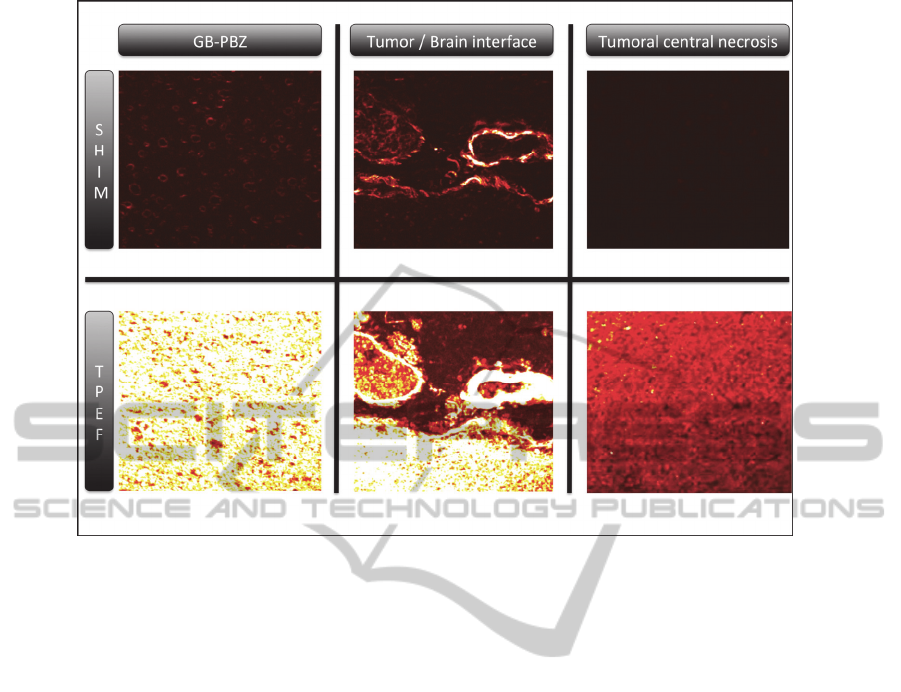
Figure 1: Illustration of the SHIM and TPEF acquisition of different GB area. The peritumoral brain zone from
glioblastoma (GB-PBZ), the interface between florid tumor and peripheral brain and the central necrotic zone of the tumor.
immediate diagnosis of the tumor sample, which can
take up to 10 days with classical pathological
analysis, and also assess samples of the border of the
resection’s cavity to confirm the absence of tumoral
cells and thus the complete resection of the tumor.
5.1.2 Other Potential Leads for
Per-operative Study of the GB-PBZ
We are planning to try the optic coherence
tomography (OCT) to study the 3D conformation
and surface of the resection’s cavity to ensure an
optimal resection of the tumor. This opportunity
comes from the commercialization of smaller,
portable and usable in an operating theatre.
Also, one other possibility, depending on the
identification of specific targets of GB tumoral cells
will be a colorimetric staining of the resection’s
cavity walls with a reagent that will change colour
when in contact with a specific biomarker present in
GB cells.
5.2 Development of New Therapeutic
Targets
5.2.1 Validation of the Proteins
We identified from our multimodal analysis of the
GB-PBZ potential biomarkers suitable as therapeutic
target.
First, we will perform a double validation of the
presence of these proteins by immunochemistry
(IHC) and western blot analysis on the samples from
the patients used for multimodal analysis of the GB-
PBZ, whose profiles are known.
5.2.2 Study of the Biomarkers in Tumor
Samples
After the validation of the proteins, we will assess
the expression of these proteins in a cohort of
patients from the national clinico-biological
collection of GB, coordinated by Pr Menei. Crossing
the data obtained with the epidemiologic
characteristics and the evolution of the patients will
allow us to determine if the presence (or absence) of
these biomarkers have an impact on the survival or
the therapeutic response of patients with a GB.
A first cohort of 50 patients from the clinico-
biological collection and tumors samples will be
analysed using IHC.
If the results are concluding for this first cohort,
the analysis will be extended to the whole
population of the clinico-biological collection and be
done routinely on new patients included in the
national database.
AnalysisofGlioblastoma'sPeritumoralBrainZone-DevelopingofPer-operativeAnalysisandNewTherapeuticTargets
57

5.2.3 Study of the Biomarkers in Blood
Samples
The proteins identified in the PBZ are components
of the brain cells exosomes, and thus can be assayed
in blood samples from patients.
We look forward to this possibility because
blood samples are easy to obtain and allow repetitive
and reproductive measure of biomarkers blood level
in GB patients during their post-surgical evolution
and treatment.
We will assess the presence of these biomarkers
in the peripheral blood of GB patients using an
enzyme-linked immuno-assay (ELISA) technique.
As with the analysis of the tumor samples, a
preliminary study will be performed on a small
cohort of patients from the clinico-biological
collection and will be extended to the whole cohort
if the first results are positives.
5.2.4 Biomarkers as Therapeutic Targets
We will study the potential role of the identified
biomarkers as therapeutic target in GB treatment. A
step next will be to develop monoclonal antibodies
targeted against these proteins to see if the specific
neutralization of one of these proteins can improve
the survival or the therapeutic response of a
population of murine GB model.
If we identify a significant improvement in terms
of survival after GB implantation in a murine model
of the disease, we will begin a human clinical trial.
5.2.5 Biomarkers as Targets for Vectorized
Therapies
One other axis of development will be the use of
vectorized LNC aimed against the proteins identified
in the GB-PBZ. Production of LNC, encapsulation
of chemotherapeutic agents and targeted delivery of
these nanocapsules are a field of expertise of my
laboratory, which possess an international
recognition in this domain.
We aim to produce LNC targeted against the
identified biomarkers to use them as therapeutic
agent against GB. In a first phase, we will produce
LNC containing fluorochromes to assess the
distribution of the LNC in a murine model of GB.
Then, if the results are positive, we will produce
LNC containing a chemotherapeutic agent, and
assess in the same murine model of GB the impact
of the LNC injection in the functional prognosis and
survival of the mice, with always as first aim the
transfer of these researches to the clinical field in
human.
6 EXPECTED OUTCOME
At short-term, during the next year, the results of the
SHIM and TPEF imaging analysis of brain tumor
samples will be the subject of a publication in a high
impact review of Neuro-Oncology. Also the
validation of the biomarkers in the brain tumor and
the peripheral blood samples of the patients will be
the subject of a publication in a high impact review
in Neuro-Oncology.
The use of per-operative OCT or colorimetric
staining of the tumor are planned for the end of my
PhD thesis or will be the subject of my post-doctoral
studies.
The use of the proteins of interest as therapeutic
target or as target for chemotherapy-loaded LNC
will be the subject of further studies in my
laboratory, implicating Pharmacists and Galenists.
REFERENCES
Chaichana KL, Jusue-Torres I, Navarro-Ramirez R, Raza
SM, Pascual-Gallego M, Ibrahim A, Hernandez-
Hermann M, Gomez L, Ye X, Weingart JD, Olivi A,
Blakeley J, Gallia GL, Lim M, Brem H, Quinones-
Hinojosa A. Establishing percent resection and
residual volume thresholds affecting survival and
recurrence for patients with newly diagnosed
intracranial glioblastoma. Neuro Oncol. 2013 Nov 26.
[Epub ahead of print]
Clavreul A, Etcheverry A, Chassevent A, Quillien V,
Avril T, Jourdan ML, Michalak S, François P, Carré
JL, Mosser J; Grand Ouest Glioma Project Network,
Menei P. Isolation of a new cell population in the
glioblastoma microenvironment. J Neurooncol. 2012
Feb;106(3):493-504.
Glas M, Rath BH, Simon M, Reinartz R, Schramme A,
Trageser D, Eisenreich R, Leinhaas A, Keller M,
Schildhaus HU, Garbe S, Steinfarz B, Pietsch T,
Steindler DA, Schramm J, Herrlinger U, Brüstle O,
Scheffler B. Residual tumor cells are unique cellular
targets in glioblastoma. Ann Neurol. 2010
Aug;68(2):264-9.
Lemée JM, Com E, Clavreul A, Avril T, Quillien V, de
Tayrac M, Pineau C, Menei P. Proteomic analysis of
glioblastomas: what is the best brain control sample? J
Proteomics. 2013 Jun 24;85:165-73.
Nagashima G, Suzuki R, Hokaku H, Takahashi M, Miyo
T, Asai J, Nakagawa N, Fujimoto T. Graphic analysis
of microscopic tumor cell infiltration, proliferative
potential, and vascular endothelial growth factor
expression in an autopsy brain with glioblastoma.
Surg Neurol. 1999 Mar;51(3):292-9.
Petrecca K, Guiot MC, Panet-Raymond V, Souhami L.
Failure pattern following complete resection plus
radiotherapy and temozolomide is at the resection
BIOSTEC2014-DoctoralConsortium
58

margin in patients with glioblastoma. J Neurooncol.
2013 Jan;111(1):19-23.
Sherriff J, Tamangani J, Senthil L, Cruickshank G,
Spooner D, Jones B, Brookes C, Sanghera P. Patterns
of relapse in glioblastoma multiforme following
concomitant chemoradiotherapy with temozolomide.
Br J Radiol. 2013 Feb;86(1022):20120414
Stupp R, Hegi ME, Mason WP, van den Bent MJ,
Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A,
Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek
J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P,
Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe
D, Cairncross JG, Mirimanoff RO; European
Organisation for Research and Treatment of Cancer
Brain Tumour and Radiation Oncology Groups;
National Cancer Institute of Canada Clinical Trials
Group. Effects of radiotherapy with concomitant and
adjuvant temozolomide versus radiotherapy alone on
survival in glioblastoma in a randomised phase III
study: 5-year analysis of the EORTC-NCIC trial.
Lancet Oncol. 2009 May;10(5):459-66.
Yamahara T, Numa Y, Oishi T, Kawaguchi T, Seno T,
Asai A, Kawamoto K. Morphological and flow
cytometric analysis of cell infiltration in glioblastoma:
a comparison of autopsy brain and neuroimaging.
Brain Tumor Pathol. 2010 Oct;27(2):81-7.
AnalysisofGlioblastoma'sPeritumoralBrainZone-DevelopingofPer-operativeAnalysisandNewTherapeuticTargets
59
