
A Comparison Between a Deterministic, Compartmental Model and an
Individual Based-stochastic Model for Simulating the Transmission
Dynamics of Pandemic Influenza
Hung-Jui Chang
1
, Jen-Hsiang Chuang
2
, Tsurng-Chen Chern
1
, Mart Stein
3
, Richard Coker
4
, Da-Wei
Wang
1
, Tsan-sheng Hsu
1
1
Institute of Information Science, Academia Sinica, Taipei, Taiwan
2
Epidemic Intelligence Center, Centers for Disease Control, Taipei, Taiwan
3
National Institute for Public Health and the Environment (RIVM), Bilthoven, The Netherlands
4
Communicable Diseases Policy Research Group, London School of Hygiene and Tropical Medicine, Bangkok, Thailand
Keywords:
Agent-based Model, Equation-based Model, Model Comparison, Parameter Calibration.
Abstract:
Simulation models are often used in the research area of epidemiology to understand characteristics of disease
outbreaks. As a result, they are used by authorities to better design intervention methods and to better plan
the allocation of medical resources. Previous work make use of many different types of simulation models
with an agent-based model, e.g., Taiwan simulation system, and an equation-based model, e.g., AsiaFluCap
simulation system, being the two most popular ones. Some comparison studies has been attempted in the past
to understand the limits, efficiency, and usability of some model. However, there was little studies to justify
why one model is used instead of the other. In this paper, instead of studying the two most popular models one
by one, we try to do a comparative study between these two most popular ones. By observing that one model
can outperform the other in some cases, and vice versa, we hence study conditions that which one should be
used. Furthermore, previous studies show little results in the issue of allocating medical resources. Our paper
studies and compares the two models using medical resources allocation as one of our primary concerns. As a
conclusion, we come out with a general guideline to help model designers to pick one that fits the given needs
better.
1 INTRODUCTION
Simulation models are often used in the research
area of epidemiology estimating the characteristics of
a specified disease outbreak, such as outbreak day,
peak day and prevalence rate (Diekmann and Heester-
beek, 2000). Simulation results can support govern-
ments in designing intervention methods to prevent
the spread of diseases (Tsai et al., 2010) or to estimate
the amount of medical resources needed (Krumkamp
et al., 2011; Rudge et al., 2012; Stein et al., 2012).
There are many different types of simulation mod-
els, such as homogeneous (Diekmann and Heester-
beek, 2000; Krumkamp et al., 2011) or heteroge-
neous (Garnett, 2002; Keeling and Danon, 2009;
Lunelli et al., 2009), deterministic (Diekmann and
Heesterbeek, 2000; Keeling and Danon, 2009) or
stochastic (Britton and Lindenstrand, 2009; Lunelli
et al., 2009), equation-based (Diekmann and Heester-
beek, 2000; Garnett, 2002) or agent-based (Berger,
2001; Davidsson, 2002; Macal and North, 2005;
Moss and Davidsson, 2001; Parker et al., 2003). The
major difference between homogeneous and hetero-
geneous model is a homogeneous model treats all the
people exactly the same way but in a heterogeneous
model, people are partitioned into different groups
according to their characteristic. In a deterministic
model, the number of newly infected people is always
the same for a given number of susceptible people and
infectious people. But in a stochastic model, the num-
ber of newly infected people is often different as this
number is chosen based on a random distribution. An
equation-based model is the one that utilizes numer-
ical time-stepping procedures to simulate the behav-
iors over time. And an agent-based models focuses on
the actions and the interactions among autonomous
agents.
586
Chang H., Chuang J., Chern T., Stein M., Coker R., Wang D. and Hsu T..
A Comparison Between a Deterministic, Compartmental Model and an Individual Based-stochastic Model for Simulating the Transmission Dynamics of
Pandemic Influenza.
DOI: 10.5220/0005040905860594
In Proceedings of the 4th International Conference on Simulation and Modeling Methodologies, Technologies and Applications (SIMULTECH-2014),
pages 586-594
ISBN: 978-989-758-038-3
Copyright
c
2014 SCITEPRESS (Science and Technology Publications, Lda.)

In 2009, the novel influenza H1N1 spread from
Mexico and caused over 18,000 deaths worldwide.
1
The AsiaFluCap project (AFC) (AsiaFluCap, 2009)
has focused on the medical resources issue in the
southeast Asia where infectious diseases similar to
H1N1 may break out. AFC has built a homogeneous
deterministic equation-based model (the AsiaFluCap
Simulation, AFC model) to simulate the disease trans-
mission behavior. This simulator has the same ba-
sis as the SEIR model described in Krumkamp et
al.(Rudge et al., 2012; Stein et al., 2012). In the
same time, Institute of Information Science (IIS)
of Academia Sinica cooperates with CDC in Tai-
wan (Tsai et al., 2010) has developed a heteroge-
neous individual based model (the Taiwan simulation
model, TW model) to simulate the disease transmis-
sion behaviors in order to design effective interven-
tion policies. Although the AFC model and the TW
model are different in design, they both model the
same disease namely pandemic influenza. Since the
two models modeled the same disease, a natural ques-
tion raised: How different are these two models and
how well does each model describe the same disease?
Previous studies have mentioned the relation-
ship between agent-based models and equation-based
models (Ajelli et al., 2010; Bobashev et al., 2007;
Connell et al., 2009; Rahmandad and Sterman, 2008).
These works focused on comparing the simulation re-
sults between the compartment model and individual-
based model (Connell et al., 2009; Rahmandad and
Sterman, 2008), trying to hybrid these two ap-
proaches (Bobashev et al., 2007) and giving the prin-
ciple of which models should be used based on the
policy designed (Rahmandad and Sterman, 2008), but
none of them focused on how to calibrate different
model by calibrating the parameters.
In this paper, we construct a series of methods to
calibrate parameters between these two models. In
order to compare and analyze the simulation results of
these two models, we use the same parameters setting
after calibration. We also give a guideline in choosing
a simulation model that best fits a user’s needs.
2 METHODS
2.1 AsiaFluCap Simulation Model
The AFC model (AsiaFluCap, 2009) is a homoge-
neous and deterministic model which is base on the
1
Pandemic (H1N1) 2009 - update
112, published by WHO and available at
http://www.who.int/csr/don/2010
08 06/en/index.html
fundamental SIR model with three additional com-
partments, Prophylaxis (P), Exposed (E) and Asymp-
tomatic (A). The detail of the model is described in
the supporting text.
In this model, the setting of parameters is the same
for all people. The number of people being transited
from one compartment to the next compartment in
each time slice are given with differential equations.
For example, if the latency between E and A is x days
and the time slice is y steps per day then in every time
slice 1/(x× y) of the people in E are transited to A.
2.2 Taiwan Simulation Model
The TW model (Tsai et al., 2010) is a stochastic and
heterogeneous model. Each individual has his own
personal attributes such as age, gender, contact prob-
ability and daily activities. People with the same daily
actives are called in the same mixing group. The dura-
tion that an infected individual stays in a disease state
is selected according to a probability distribution and
individuals may have different disease courses.
2.2.1 Social Structure
In the TW model, the age of a person is the major fac-
tor that affects his behaviors such as daily activities
and contacts. The social structure basically follows
the one given in (Tsai et al., 2010). There are five age
groups in the TW model, namely c
0
(0-4 years), c
1
(5-
18 years), a
0
(19-29 years), a
1
(30-64 years) and a
2
(above 64 years). At the beginning of the simulation,
the age and gender of each person are stochastically
generated based on the demographic distributions ac-
cording to the real census data.
2
There are ten mixing groups in the TW model,
household (HH), household cluster (CL), neighbor-
hood (NB), community (CM), daycare center (DC),
play group (PG), elementary school (ES), middle
school (MS), high school (HS) and work group (WG).
Each household contains 1 to 7 people. The age, gen-
der and number of people in each household are also
generated according to the Taiwan census data. A
household cluster contains four households. A neigh-
borhood contains about 500 people, and a community
is formed by four neighborhoodsi.e., about 2000 peo-
ple. Each community has one high school, one mid-
dle school and two elementary schools. Each high
school and each middle school are shared by all four
2
2000 Taiwan census data published by Directorate-
General of Budget, Accounting and Statistics, Execu-
tive Yuan, Republic of China (Taiwan), available at
http://eng.stat.gov.tw/lp.asp?CtNode=1627&
CtUnit=777&BaseDSD=7&mp=5
AComparisonBetweenaDeterministic,CompartmentalModelandanIndividualBased-stochasticModelforSimulating
theTransmissionDynamicsofPandemicInfluenza
587

neighborhoods in the same community. Two neigh-
borhoods share one elementary school. Each neigh-
borhood has several daycare centers, playing groups
and working groups. The numbers of those groups
depend on the number of people in those groups in
that neighborhood.
Each day is divided into two periods, the day time
period (DP) and the night time period (NP). Each pe-
riod has 12 hours. In DP, the young children (c
0
) ei-
ther go to the daycare center or stay close to home in
a playing group with an equal probability. The school
age children (c
1
) go to schools, either ES, MS or HS,
depending on their ages with preset drop out rates.
The working adults (a
0
and a
1
) go to work in DP
with a given unemployment rate. All the elder adults
(a
2
), the drop out students and the unemployed adults
stay at home during DP. In NP, people stay at home
within the correspondinghousehold cluster, neighbor-
hood and community.
2.2.2 Disease Transmission Model
Disease transmission behavior is mainly controlled
by two epidemiological parameters: contact proba-
bility (CP) and transmission probability (P
trans
). Con-
tact probabilities are given for people of specific age
group in a social mixing group, i.e., people in the
same age group and same mixing group have the same
contact probability. In any time period, a susceptible
person may be infected by an infectious person in the
same mixing group.
The TW model has four main states S, E, I and
R. Each state comprises several compartments. There
are two different compartments in E, namely Latency
(L) and incubation (In). For all people in E, only peo-
ple in In can infect others. I also contains two dif-
ferent compartments, namely asymptomatic (I
a
) and
symptomatic (I
s
). People in any compartment are
transited to the next compartment after one DP and
one NP, and they will be transited to either Recover
(R) or Death (D) after then.
3 CALIBRATION
We now provide a case study of the calibration be-
tween the AFC model and the TW model. We will
first group parameters according to they are used in
only one model or in both models. For the parameters
used in both models, they are grouped according to
their representation.
3.1 General Descriptions of the
Parameters
In this subsection, parameters are classified accord-
ing to their attributes. The first step is to distinguish
between shared parameters and individual parame-
ters. Shared parameters are those used by both mod-
els such as the transmission probability, the total num-
ber of people in the region. Individual parameters are
those only used by either one model. In the TW model
individual parameters are personal information such
as age, gender, household structure etc. Since the in-
dividual parameters only appear in one model, in the
calibration phase we only focus on the shared param-
eters. The shared parameters can also be classified
into three groups according to the possible range of
their values: fixed-value, fixed-probability and proba-
bility distribution. The tree structure of the parameter
groups is shown in Figure 1.
Parameters
Shared parameter Individual parameter
Fix-value
Fix-probability Probability distribution
Figure 1: Parameter usage and type of representation.
Shared parameters may use the same representa-
tion method, to calibrate them we simply assign the
same value. For shared parameters that belong to dif-
ferent groups in different models, we need to design
a method to transliterate their values.
Before we introduce how to calibrate shared pa-
rameters between the AFC model and the TW model,
we first describe the different types of parameters and
how to group the parameters.
3.2 Individual Parameters
Individual parameters are those only used by either
one model. They describe details about the nature
of the disease and the human behavior. For exam-
ple, the compartment expose only appears in the AFC
model and the corresponding parameter also only ap-
pears in the AFC model. Similarly, the TW model
uses mixing group to describe the social structure, so
the corresponding parameters which denotes the dis-
tribution of household structure and the distribution
of age only appear in the TW model.
3.3 Shared Parameters
Shared parameters are those used in both models.
They usually refer to the natural history of disease
SIMULTECH2014-4thInternationalConferenceonSimulationandModelingMethodologies,Technologiesand
Applications
588

Table 1: Distribution of onset days.
Onset days 3 4 5 6
P
days
0.3 0.4 0.2 0.1
Table 2: Distribution of latency and incubation days.
Incubation (Latency) days 1 (1) 2 (1) 3 (2)
P
Latency,Incubation
= P
Latency
0.3 0.5 0.2
and the human behavior. For example, the number
of days of each state of disease and the transmission
probability and the efficacy of vaccination belong to
the disease natural history. And the reduction of so-
cial contact due to government intervention measures
and the availability of medical resources belong to the
human behavior.
Parameters represented as fixed values include the
parameters such as the total number of people, the
number of simulation days and the amount of avail-
able medical resources.
A parameter represented as a fixed probability
means the probability of a particular event will hap-
pen e.g., the transmission probability of two people
have made an effective contact. Another example is
the probability that a patient becomes symptomatic or
asymptomatic.
Parameter represented as a probability distribu-
tion contains two parts: its sample distribution and
the probability space. For example in the section of
Taiwan Simulation Model, the number of infectious
days are 3-6 days with the corresponding probability
of 0.3, 0.4, 0.2 and 0.1 respectively. See Table 1 for an
illustration. Another example is shown in Table 2 to
illustrate the numbers of latency and incubation days
in the TW model.
3.4 Methods of Calibration
For each shared parameter, we first decide whether
it belongs to the same group in the two models. We
reassign an equal value if they do. If the parameter
belongs to different groups, we design a formula to
transliterate the ‘values’ between the two models.
We first consider how to transliterate between a
fixed-value parameter in one model and a probability-
distribution one in another model. For example, the
number of incubation days is 1 day in the AFC model,
and it is from 1 to 3 days with probabilities 0.3, 0.5
and 0.2 respectively, in the TW model. To calculated
the expected number of incubation days in the TW
model, we simply sum the product of the number of
days and the corresponding probability, that is:
∑
D× Pr{Incubation is D days}. (1)
The expected number of incubation days is 1× 0.3+
2× 0.5 + 3 × 0.2 = 1.9, and the expected number of
onset days is 3 × 0.3 + 4× 0.4 + 2 × 0.5 + 6× 0.1 =
4.1. See Table 1 and Table 2 for details.
Now we consider the case of transliterating from
an expected value parameter in one model to a prob-
ability distribution in another model. We use either a
scaling or a geometric distributed methods.
In scaling method, the value of probability events
of a given distribution is multiplied with the ra-
tio of the given expected value over the expected
value of the given probability distribution. Consider
a distribution has expected number D
E
and sample
space D
1
,D
2
,...,D
n
with corresponding probability
P
1
,P
2
,...,P
n
respectively. If we want to scale this dis-
tribution’s expected value to D
′
E
, we first calculate the
ratio RATIO = D
′
E
/D
E
. Next, we multiply each sam-
ple space D
i
with RATIO and we have a new distri-
bution with expected value D
′
E
= D
E
× RATIO. For
example, the number of latency and incubation days
in the AFC model is 1.0 and the expected latency and
incubation days in the TW model is 1.9. We can scale
down the value of the probability distribution D
latency
in the TW model with 1.0/1.9 = 0.526.
In the above case, if we treat the number of days
as the expected number of a simple probability distri-
bution, then we can reconstruct the distribution by fit-
ting a probability distribution to that expected value.
In the AFC model, each period of the disease course
is described as a geometric distribution. For example,
the number of latency day is 1.0, and the number of
time interval in a day is 15, then in each time interval,
1/15 of the people in E transit to A. Then we can cal-
culate the corresponding probability of the number of
day that a person remain in the same compartment by
P(t) = p(1− p)
t−1
(2)
where t is the number of time intervals and p is the
probability that person transits to the next compart-
ment. For example, the time interval in the TW model
is 2 and the latency period is 1.0 in the AFC parameter
setting, then 1.0/2 = 0.5 of the people transit from E
to A in one time interval, and the corresponding prob-
ability of people remain in the E is 0.5, 0.25 and 0.125
with 0.5, 1.0 and 1.5 day respectively.
3.5 Experiment design
In order to observe the outputs of the two models
under different data settings, we design two experi-
ments. One simulation comprises three major parts,
the simulation model, either the AFC model or the
TW model, the parameter setting, either the AFC data
setting or the TW data setting, and the size of regions.
AComparisonBetweenaDeterministic,CompartmentalModelandanIndividualBased-stochasticModelforSimulating
theTransmissionDynamicsofPandemicInfluenza
589

Table 3: Experiment scenarios for basic experiment.
Model Regions Size Parameter Setting
AFC Large AFC-setting
AFC Large TW-setting
AFC Small AFC-setting
AFC Small TW-setting
TW Large AFC-setting
TW Large TW-setting
TW Small AFC-setting
TW Small TW-setting
Table 4: Experiment scenarios for small region experiment.
Model Region Size Parameter Setting
TW 1. ..100 AFC-setting
TW 1. ..100 TW-setting
The experiment scenarios is detailed in Table 3 and
Table 4.
In the first experiment, we examine the behaviors
of both models using different sizes of regions. We
denote the whole Taiwan as the large region and 1 CM
as the small region. For each model we run the sim-
ulation twice in both region sizes with the TW data
setting and the AFC data setting respectively. Note
that the AFC data setting has longer periods of states
in the disease course, the number of days one patient
is hospitalized is 12 days and the number of day one
patient uses a ventilation is 13 days, and also has a
larger number of expected contacts, 7.0, than that of
using the TW data setting, 4.19.
Since the prevalence of the AFC model is not af-
fected by changing the region size, and the TW model
is sensitive to the size of the region, the second exper-
iment focuses on observing the behavior of the TW
model with different sizes of regions. This experi-
ment uses both the AFC and the TW data settings as
parameter settings. The size of the region, i.e., the
number of CM variates from 1 to 100.
4 EXPERIMENTAL RESULTS
In this section, we introduce the experiment environ-
ments for implementing the AFC model and the TW
model. We show the data settings and the correspond-
ing experiment results.
The TW model is implemented using C++ (Tsai
et al., 2010) and the AFC model is implemented using
an Excel spreadsheet (Krumkamp et al., 2011). The
experiments were run on a server with dual Intel Xeon
X5482, quad-cores, 3.20 GHz CPU and 64GB DDR3
memory. Since TW is an agent-based simulation, for
each simulation of TW model we run 30 times and
take the average.
The first experiment includes eight scenarios for
the combination of the AFC model and the TW
model, the AFC setting and the TW setting, and the
large region (entire Taiwan) and the small region (1
CM). The experiment results are shown in Figure 2
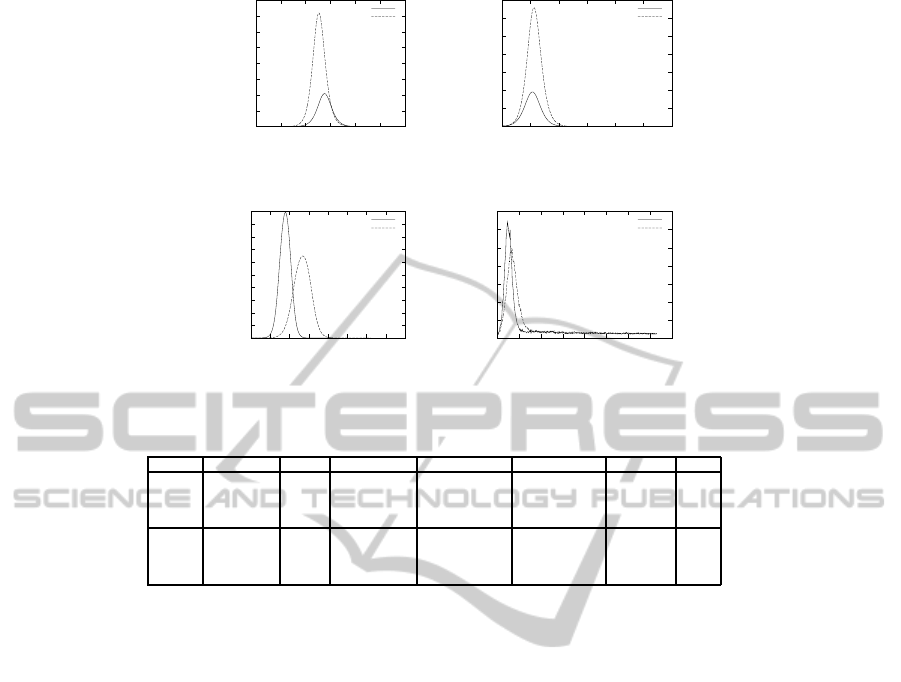
and the detail values are shown in Table 5. Figure 2
shows the number of daily cases with different size
of regions and different parameter settings. The
first three columns in Table 5 denote the model used,
the data settings and the size of each region, respec-
tively. The three columns followed are the prevalence
of symptomatic cases, asymptomatic cases, and their
total. The last two columns denote the peak day and
the R
0
of each experiment. The peak day is the day
with the maximum average number of people that are
currently symptomatic. We first calculate the average
number of symptomatic cases of all 30 simulations
and then choose the day with the largest number of
symptomatic cases as the peak day.
The basic reproduction number R
0
is derived for
each model. The AFC model calculates R
0
by multi-
plying the number of contacts with the transmission
probability and the number of days of the disease
course (Krumkamp et al., 2011).
The TW model calculates R
0
using a formula that
is similar to the one used in calculating the expected
number of contacts (Tsai et al., 2010). In each simu-
lation, we sample 2,000 individuals, i.e., the number
of people in 1 CM. We then calculate the expected
number of peoples that can be infected by each of the
sampled individuals. We calculate the probability that
one susceptible is infected by the indicated infectious
person minus the probability that this susceptible in-
dividual is not infected by that infectious person in all
of the mixing groups they shared.
Table 6: Simulation result in TW model with different num-
bers of CM.
AFC TW
CM All(%) Sym(%) Peak Day All(%) Sym(%) Peak Day
1 74.09 56.88 24 79.70 59.73 31
2 62.61 46.75 29 66.37 47.36 38
3 56.87 41.52 31 61.70 43.18 45
4 56.01 40.58 33 59.22 41.19 46
5 53.65 38.49 35 56.54 39.16 47
6 52.34 37.50 35 55.20 37.92 50
7 51.34 36.60 37 54.83 37.58 53
8 50.22 35.77 35 53.65 36.65 53
9 49.94 35.40 39 53.87 36.81 55
10 49.38 35.07 38 52.65 35.89 55
20 48.89 34.58 40 51.39 34.76 63
30 48.77 34.49 39 50.38 33.99 66
40 47.27 33.11 48 50.01 33.68 70
50 47.11 33.02 48 50.18 33.74 72
60 47.08 32.94 49 50.09 33.68 72
70 46.77 32.73 53 49.76 33.43 75
80 46.52 32.50 50 49.79 33.45 77
90 46.58 32.58 54 49.56 33.23 77
100 46.66 32.64 53 49.46 33.18 79
Taiwan 46.25 32.29 86 49.39 33.09 130
SIMULTECH2014-4thInternationalConferenceonSimulationandModelingMethodologies,Technologiesand
Applications
590
0
200000
400000
600000
800000
1e+06
1.2e+06
1.4e+06
1.6e+06
0 50 100 150 200 250 300
Number of Cases
Day
AFC-parameter
TW-parameter
(a)
0
20
40
60
80
100
120
140
0 50 100 150 200 250 300
Number of Cases
Day
AFC-parameter
TW-parameter
(b)
0
20000
40000
60000
80000
100000
120000
140000
160000
180000
200000
0 50 100 150 200 250 300 350 400
Nuber of Cases
Day
AFC-parameter
TW-parameter
(c)
0
5
10
15
20
25
30
35
0 50 100 150 200 250 300 350 400
Nuber of Cases
Day
AFC-parameter
TW-parameter
(d)
Figure 2: Basic experiments with two models, two parameter settings and different size of regions.
Table 5: Basic experiments with two models, two parameter settings and different size of regions.
Model Parameters Size Symptomatic Asymptomatic Prevalence All Peak Day R
0
AFC AFC large 31.32% 13.43% 44.75% 137 1.32
AFC small 31.41% 13.44% 44.85% 52 1.32
TW large 48.84% 24.42% 73.26% 125 1.78
TW small 48.71% 24.33% 73.04% 55 1.78
TW AFC large 32.29% 13.97% 46.25% 90 1.38
AFC small 56.88% 17.22% 74.09% 24 1.36
TW large 33.09% 16.30% 49.39% 136 1.43
TW small 59.28% 20.20% 79.48% 34 1.41
5 DISCUSSION
5.1 The Baseline Experiment
The first observation is that the prevalence is not af-
fected by the region size in the AFC model and is af-
fected by the region size in the TW model. In the AFC
model, the region size affects the prevalence only
slightly, namely 0.1% for the AFC data setting and
0.2% for the TW data setting. In the TW model, ex-
periments using a large region size have much lower
prevalence than that of using a small region size. It is
46.25%, greatly reduced from 76.25%, for the AFC
data setting and from 79.48% down to 49.39% for
TW data setting.
Peak day in a large region setting is later than that
of the setting for a small region even using the same
model. When using the AFC model with the AFC
data setting, the peaks day for a large region setting
and a small region setting are 137 and 52 respectively,
and when using the AFC model with the TW data set-
ting, the peak day for a large region setting and a small
region setting are 125 and 55 respectively. When us-
ing the TW model with the AFC data setting, the peak
day for a large region setting and small region setting
are 90 and 24 respectively, and when using the TW
model with the TW data setting, the peaks day for
large region setting and small region setting are 136
and 34 respectively.
R
0
of the AFC data setting is almost identical us-
ing all 4 different settings, namely 1.32, 1.32, 1.38
and 1.36, but R
0
of the TW data setting deviates a lot,
namely 1.78, 1.78, 1.43 and 1.41.
The experiment results showthe prevalenceis sen-
sitive to the region size in the TW model and is not
sensitive in the AFC model with either data settings.
This is because AFC model is homogeneous,i.e., con-
tact probability is the same for all individuals. The
TW model is, on the other hand, heterogeneous based
on demographic statistics for the entire nation, i.e.,
an individual with in a given mixing group and has a
distinct contact probability with another individual in
another mixing group.
Both the AFC and TW data settings have a larger
prevalence in the TW model when using a smaller
region setting, and a smaller prevalence when using
a larger region setting. The TW model has similar
behaviors when regions of similar size are used even
when the values of other parameter are different.
Another important factor is that worker flow is not
considered in the AFC model but may affect the ac-
curacy of simulation if there are a large number of
people commuting. Worker flow is the only way two
AComparisonBetweenaDeterministic,CompartmentalModelandanIndividualBased-stochasticModelforSimulating
theTransmissionDynamicsofPandemicInfluenza
591

people in different communities may have a chance
to contact in the TW model. It takes more time for
the disease to spread to other CM when the number
of CM’s increases. This factor affects the peak day
and the prevalence in the TW model.
Although the AFC model is homogeneous, when
the size of regions increases, the peak day also de-
lays. This is because when the initial condition is the
same, the increasing rate of newly infected people re-
mains the same. As the number of susceptible people
increases, more time is needed to infect the same per-
centage of people.
The values of R
0
are similar for AFC model and
the TW model using the AFC data setting, but are
quite different when using the TW data setting. The
main reason is the disease courses used in the two
models are different. The TW model assumes that
asymptomatic patients and patients in the incubation
period only have half of the original transmission
probability. But in the AFC model, the transmission
probability is the same in all states. If we apply the
formula in the AFC model for the TW data setting and
replace the transmission probability for the asymp-
tomatic case and patients in incubation with half of
the original probability, then R
0
is 1.40. This value is
close to the R
0
values in the TW model using the TW
data setting which are 1.41 and 1.43, respectively.
5.2 The Small Region Experiments
This experiment focuses on the behavior of the TW
model using the AFC data setting and the TW data
setting when the size of regions is small.
The prevalence decreases as the region size in-
creases. The AFC data setting has a prevalence of
74.09% when the number of CM’s is 1, and becomes
46.66% when the number of CM’s is 100. The TW
data setting has a prevalence of 79.70% when the
number of CM’s is 1, and 49.46% when the number
of CM’s is 100. In the case of having 100 CM’s, the
prevalence is very close to that of the simulation result
with the entire nation.
The peak day of using a larger region setting is
later than the peak day of using a smaller region set-
ting. Although the peak day does not increase mono-
tonically as the number of CM’s increases, the peak
day of the AFC data setting increases from 24 to 53
and the peak day of the TW data setting increases
from 31 to 79 as the number of CM’s increases from
1 to 100.
Note there is no direct relationship between the re-
gion size and the expected number of contacts. How-
ever, when the number of CM’s increases, the disease
needs more time to transmit to other CM’s due to the
worker flow being that the only way for people to
travel between CM’s. Since the worker flow describe
the probability that a worker lives in a particular town
and works in other town, it only assigns workers to
the level of a town not to the level of a CM. The prob-
ability of a CM in the same town being chosen as a
worker works is uniformly distributed over all CM in
this town. As the number of CM’s in a town increases,
the probability of patients working in some indicated
CM decreases. Hence it is harder to spread over the
whole town and therefor the prevalence decreases.
Due to reasons similar to the above, as the num-
ber of CM’s increases, the disease needs more time
to spread from the original CM where the index cases
are to all other CM’s. Hence the peak day is further
delayed as the number of CM’s increases.
6 CONCLUSION
To analyze the trade-off between various simulation
models and establish a guideline for model selec-
tion, we first introduced two different real instances,
namely the AFC model and the TW model. We
grouped the parameters used and introduced proce-
dures to transliterate parameters. We also described
procedure to perform calibration. We ran both mod-
els with the data settings calibrated between them. We
then analyzed the simulation results and gave the ad-
vantages and disadvantages of these models and pro-
vided a principle of model selection.
As a by-product of our study, we conclude the fol-
lowing recommendations when there is a choice be-
tween using a aggregative model and an agent based
one to study the same disease. According to the anal-
ysis result, we should select the model depending on
user requirements. Generally, if we want to build a
simulation model to observe the high-level disease
transmission behavior, to get a immediate simulation
results, or we have only a limited information about
the disease course, e.g., the average time of each du-
ration, we should choose the AFC model since it can
be built using relatively little information. If we want
to study how the heterogeneous social structure, e.g.,
schools and day care centers, affects the disease trans-
mission behavior, to predict the effect of interven-
tion policy on an individual basis, or to observe the
spatio-temporal spreading behaviors of the disease,
we should choose the TW model since it is geographic
based and can easily contain detail individual infor-
mation.
SIMULTECH2014-4thInternationalConferenceonSimulationandModelingMethodologies,Technologiesand
Applications
592

ACKNOWLEDGMENTS
The AsiaFluCap project is coordinated by the London
School of Hygiene and Tropical Medicine with col-
laborators from the Hamburg University of Applied
Sciences (HAW), Netherlands National Institute for
Public Health and the Environment (RIVM), Inter-
national Health Policy Programme -Thailand (IHPP),
Taiwan Centers for Disease Control, University of In-
donesia Faculty of Public Health, Vietnam Ministry
of Science and Technology, Vietnam Military Med-
ical University, Lao PDR National Emerging Infec-
tious Diseases Coordination Office, Lao PDR Univer-
sity of Health Sciences, Mahidol University Faculty
of Tropical Medicine, Cambodia Department of Com-
municable Disease Control, and Cambodia National
Institute of Public Health. We are grateful to the many
collaborators within this project consortium for their
contribution towards resource characterization, data
collection, and discussions at consortium meetings.
These include: Ly Khunbunn Narann and Chau Dara-
pheak (Cambodia); Sandi Iljanto, Noviyanti Liana
Dewi, Kamaluddin Latief, Amir Suudi, Lilis Much-
lisoh (Indonesia); Nyphonh Chanthakoummane, Sing
Menorath and Rattanaxay Phetsouvanh (Lao PDR);
Yu-Chen Hsu, Yi-Ta Yang, SteveKuo (Taiwan); Porn-
thip Chompook, Jongkol Lertiendumrong and Viroj
Tangcharoensathien (Thailand); Le Minh Sat and La
Thanh Nhan (Vietnam). Excellent administrativesup-
port from Nicola Lord and Wasamon Sabaiwan is
greatly appreciated. We also thanks for James W.
Rudge for revising this paper.
REFERENCES
Ajelli, M., Gonc¸alves, B., Balcan, D., Colizza, V., Hu,
H., J. J Ramasco, S. M., and Vespignani, A. (2010).
Comparing large-scale computational approaches to
epidemic modeling: Agent-based versus structured
metapopulation models. BMC Infectious Diseases 10,
190 (2010).
AsiaFluCap (2009). The AsiaFluCap Simulator.
http://www.cdprg.org/asiaflucap-simulator.php.
Berger, T. (2001). Agent-based spatial models applied to
agriculture: a simulation tool for technology diffu-
sion, resource use changes and policy analysis. Agri-
cultural Economics, 25(2-3):245 – 260.
Bobashev, G. V., Goedecke, D. M., Yu, F., and Epstein,
J. M. (2007). A hybrid epidemic model: combining
the advantages of agent-based and equation-based ap-
proaches. In WSC ’07: Proceedings of the 39th con-
ference on Winter simulation, pages 1532–1537, Pis-
cataway, NJ, USA. IEEE Press.
Britton, T. and Lindenstrand, D. (2009). Epidemic mod-
elling: aspects where stochasticity matters. Mathe-
matical Biosciences, 222(2):109–116.
Connell, R., Dawson, P., and Skvortsov, A. (2009). Com-
parison of an agent-based model of disease propaga-
tion with the generalised SIR epidemic model. Sci-
ence And Technology, 5(3):1–22.
Davidsson, P. (2002). Agent based social simulation: A
computer science view. J. Artificial Societies and So-
cial Simulation, 5(1).
Diekmann, O. and Heesterbeek, J. (2000). Mathematical
epidemiology of infectious diseases: model building,
analysis, and interpretation. Wiley series in mathe-
matical and computational biology. John Wiley.
Garnett, G. P. (2002). An introduction to mathematical
models in sexually transmitted disease epidemiology.
Sex Transm Infect, 78(1):7–12.
Keeling, M. J. and Danon, L. (2009). Mathematical mod-
elling of infectious diseases. British Medical Bulletin,
92(1):33–42.
Krumkamp, R., Kretzschmar, M., Rudge, J. W., Ahmad, A.,
Hanvoravongchai, P., Westenhoefer, J., STEIN, M.,
Putthasri, W., and Coker, R. (2011). Health service
resource needs for pandemic influenza in developing
countries: a linked transmission dynamics, interven-
tions and resource demand model. Epidemiology and
Infection, 139:59–67.
Lunelli, A., Pugliese, A., and Rizzo, C. (2009). Epidemic
patch models applied to pandemic influenza: contact
matrix, stochasticity, robustness of predictions. Math-
ematical Biosciences, 220(1):24–33.
Macal, C. M. and North, M. J. (2005). Tutorial on agent-
based modeling and simulation. In Winter Simulation
Conference, pages 2–15.
Moss, S. and Davidsson, P., editors (2001). Multi-Agent-
Based Simulation, Second International Workshop,
MABS 2000, Boston, MA, USA, July, 2000, Revised
and Additional Papers, volume 1979 of Lecture Notes
in Computer Science. Springer.
Parker, D. C., Manson, S. M., Janssen, M. A., Hoffmann,
M. J., and Deadman, P. (2003). Multi-agent systems
for the simulation of land-use and land-cover change:
A review. Annals of the Association of American Ge-
ographers, 93(2):314337.
Rahmandad, H. and Sterman, J. (2008). Heterogeneity and
network structure in the dynamics of diffusion: Com-
paring agent-based and differential equation models.
Management Science, 54:998–1014.
Rudge, J. W., Hanvoravongchai, P., Krumkamp, R., Chavez,
I., Adisasmito, W., Chau, P. N., Phommasak, B., Put-
thasri, W., Shih, C.-S., Stein, M., Timen, A., Touch,
S., Reintjes, R., Coker, R., and on behalf of the Asi-
aFluCap Project Consortium (2012). Health system
resource gaps and associated mortality from pandemic
influenza across six asian territories. PLoS ONE,
7(2):e31800.
Stein, M., Rudge, J., Coker, R., Weijden, C., Krumkamp,
R., Hanvoravongchai, P., Chavez, I., Putthasri, W.,
Phommasack, B., Adisasmito, W., Touch, S., Sat, L.,
Hsu, Y.-C., Kretzschmar, M., and Timen, A. (2012).
Development of a resource modelling tool to sup-
port decision makers in pandemic influenza prepared-
ness: The asiaflucap simulator. BMC Public Health,
12(1):870.
AComparisonBetweenaDeterministic,CompartmentalModelandanIndividualBased-stochasticModelforSimulating
theTransmissionDynamicsofPandemicInfluenza
593

Tsai, M., Chern, T., Chuang, J., Hsueh, C., Kuo, H., Liau,
C., Riley, S., Shen, B., Wang, D., Shen, C., and Hsu, T.
(2010). Efficient simulation of the spatial transmission
dynamics of influenza. PloS ONE, 5(11):1–8.
SIMULTECH2014-4thInternationalConferenceonSimulationandModelingMethodologies,Technologiesand
Applications
594
