
Automatic Segmentation of MR Brain Tumor Images using Support
Vector Machine in Combination with Graph Cut
Elisabetta Binaghi
1
, Massimo Omodei
1
, Valentina Pedoia
1
,
Sergio Balbi
2
, Desiree Lattanzi
2
and Emanuele Monti
2
1
Department of Theoretical and Applied Science, Insubria University, Varese, Italy
2
Department of Biotechnologies and Life Sciences, Insubria University, Varese, Italy
Keywords: MRI Segmentation, Brain Tumor Segmentation, Meningioma, Graph Cut, Support Vector Machine.
Abstract: This work focuses the attention on the automatic segmentation of meningioma from multispectral brain
Magnetic Resonance imagery. The Authors address the segmentation task by proposing a fully automatic
method hierarchically structured in two phases. The preliminary unsupervised phase is based on Graph Cut
framework. In the second phase, preliminary segmentation results are refined using a supervised
classification based on Support Vector Machine. The overall segmentation procedure is conceived fully
automatic and tailored to non-volumetric data characterized by poor inter-slice spacing, in an attempt to
facilitate the insertion in clinical practice. The results obtained in this preliminary study are encouraging and
prove that the segmentation benefits from the allied use of Graph Cut and Support Vector Machine
frameworks.
1 INTRODUCTION
Magnetic Resonance (MR) imaging has become an
important tool for the clinical study of brain
pathologies. The high resolution and contrast and
good soft tissue differentiation allow physicians
accurately locate specific pathologies. A precise
segmentation of pathological and healthy tissues
composing the MR image is important for
understanding the pathology, for evaluating the
evolutionary trend, for planning the best surgical
approach or possible alternative solutions.
Automated methods of segmentation represent a
valuable solution by supporting human operators
with varying degrees of automation, in tracing the
boundaries of the different tissue regions and by
automatically providing volumetric computation of
the pathological MRI signal (Clark et al., 1995, Kaus
et al., 2001; Withey and Koles, 2008; Balafar et al.,
2010).
The last 20 years have seen a rapid growth in the
use of semi-automatic or fully automatic
segmentation methods in MR brain tumor studies.
Several techniques have been proposed in studies
distinguished by the type of brain tumor treated, by
the tissue features extracted in different MRI
modalities. The proposed techniques make use of
individual images or multispectral patterns and are
supervised or unsupervised (Gordillo et al. 2013,
Bauer et al. 2013). Despite the sizable achievement
obtained, novel approaches are continuously
investigated to provide robust solutions and fulfil
hard accuracy and reproducibility requirements. In
the last few years Support Vector Machine (SVM)
methods (Vapnik 1995; Schoelkopf and Smola
2002) have shown excellent performances in MRI
segmentation studies aimed at identifying a variety
of neurological conditions (Verma et al. 2008, Bauer
et al., 2013). Recent studies propose the allied use
of SVM and regularisation procedures to introduce
spatial consistency in classification results (Bauer et
al. 2011).
This work focuses the attention on the automatic
segmentation of meningioma from multi-spectral
brain MR imagery. Meningioma is one of the few
benign tumors found in the brain region. Precise
tumor detection contributes to formulate surgical
indications in elderly patients harboring intracranial
meningiomas and supports surgical planning for a
complete surgical resection (Greenberg et al. 1999,
Caroli et al. 2005).
The Authors address the segmentation task by
proposing a fully automatic method hierarchically
structured in two phases. The preliminary
unsupervised phase is based on Graph Cut
152
Binaghi E., Omodei M., Pedoia V., Balbi S., Lattanzi D. and Monti E..
Automatic Segmentation of MR Brain Tumor Images using Support Vector Machine in Combination with Graph Cut.
DOI: 10.5220/0005068501520157
In Proceedings of the International Conference on Neural Computation Theory and Applications (NCTA-2014), pages 152-157
ISBN: 978-989-758-054-3
Copyright
c
2014 SCITEPRESS (Science and Technology Publications, Lda.)

framework (Boykov and Funka-Lea 2006). In the
second phase preliminary segmentation results are
refined using a supervised classification based on
Support Vector Machine (SVM) (Vapnik 1995). The
supervised classification makes use of multichannel
intensities of Post Gadolinium T1-weighted (T1c)
and T2-weighted FLAIR image (T2f). Additional
textural and contextual features are considered and
systematically added during the experimentation in
order to measure their effective contribution. The
proposed hybrid strategy synergically combines the
mutual advantages of the unsupervised and
supervised techniques. The Graph Cut segmentation
takes into account topological constraints and
identify spatially consistent regions subsequently
refined by the SVM which is able to capture
complex multivariate relationships in the data.
The overall segmentation procedure is conceived
fully automatic and tailored to non-volumetric data
characterized by poor inter-slice spacing, in an
attempt to facilitate the insertion in current clinical
practice.
2 THEORETICAL
BACKGROUND
This section briefly outlines the basic concepts of
Graph Cut and SVM adopted in the proposed hybrid
segmentation strategy.
2.1 Graph Cut
Within the graph theoretical approach, the
segmentation problem is modeled in terms of
partitioning a graph into several sub-graphs such that
each of them represents a meaningful object of
interest in the image (Wu and Leahy, 1993; Rother
et al. 2004, Peng and Liu, 2010). Proceeding from
these principles several methods were investigated.
The aim of a volumetric segmentation
problem is to assign to each voxel v ∈ V a label
representing the membership of the voxel to a
specific region Li; in the case of binary
segmentation aimed to subdivide the image into
Object (Obj) and Background (Bkg), the goal is to
find the optimal labeling L = (L
Bkg
;L
Obj
). We
assume that each voxel v has two cost values,
Rv(O
Obj
) and Rv(O
Bkg
), related to Object and
Background labeling respectively. Each pair of
voxels (v,w) belonging to the set N of pairs of 3D
neighboring voxels has a cost B
v;w
. The cost B
v;w
is
related to the type of labeling of the pair of voxels.
The optimal labeling L = (L
Bkg
;L
Obj
) is obtained by
minimizing the following cost function:
LBLRLE
where
Vv
vv
ORLR
Nwv
j
L
i
Lwv
BLB
,
,,
;
ji
ji
j
L
i
L
LL
LL
if 1
if 0
,
R(L) and B(L) are called Regional and Boundary
terms respectively. The application of Graph Cut
requires the identification of object and background
prototypes with which to initialize the overall
segmentation process. Usually this task is
accomplished through an interactive session in
which users manually select seeds on the image.
Recently there have been some proposals
exploiting solutions for automatically initializing
Graph Cut-based segmentations of biomedical
images (Santle et al., 2012).
The present study uses the max-flow/min-cut
algorithm (Boykov and Kolmogorov 2004) as
optimization framework and adopts an automated
initialization procedure based on k-means clustering
algorithm.
2.2 SVM
SVM is a classification algorithm based on kernel
methods (Vapnik 1995; Schoelkopf and Smola
2002) able to map the original parameter vectors
into a higher (possibly infinite) dimensional feature
space through a kernel function. Classes which are
non-linearly separable in the original space can be
linearly separated in the higher dimensional feature
space.
Let {(xi, yi)} a supervised training set of
elements for a two-class classification problem,
with xi X Rn and yi {-1, 1}. Considering
the case of linearly separable data, the solution to
the classification problem consists in the
construction of the decision function
f
w, b
(x) = sgn(g
w, b
(x)) with g
w, b
(x) = w
t
x+ b
that can correctly classify an input pattern x that is
not necessarily from the training set.
SVM classifier defines the hyperplane that
causes the largest separation between the decision
function values for the “borderline” examples from
the two classes. Mathematically, this hyperplane can
be found by minimizing the cost function:
AutomaticSegmentationofMRBrainTumorImagesusingSupportVectorMachineinCombinationwithGraphCut
153

2
2
1
)( WWJ
subject to
1for y 1
1for y 1
i
i
bXW
or
bXW
i
T
i
T
The extension to the nonlinear classification is
based on the function g’= W
T
φ (X ) + b in which
the non liner operator φ (.) is introduced.
In this case the SVM cost function to be
minimized is
2
1
),(
1
2
l
i
i
CWWJ
subject to
1,2,....li ,0 with1
i
ii
t
i
bXwy
Linearly non separable data are analyzed with kernel
functions such as higher order polynomials and
Gaussian Radial Basis Functions (RBF). Suykens
(Suykens et al. 2002) proposed a new formulation of
SVM by adding a least squares (LS) term in the
original formulation of the cost function.
3 FULLY AUTOMATED
MENINGIOMA
SEGMENTATION
The salient aspect of the overall segmentation
strategy is the use of a supervised learning procedure
based on SVM model, able to learn from a set of
labeled image elements the invariant common
properties of the pathological and healthy classes.
The trained classifier automatically assigns labels to
elements never seen during the training phase.
The use of the two MR modalities, T1c and T2f,
is motivated by the fact that each scan depicts
different characteristics of the tissues. The combined
use of the two images allows obtaining higher
discriminant power than just by analyzing one of
them.
The SVM classifier acts as a dichotomizer
receiving in input a multidimensional pattern
including intensities of T1c and T2f MR elements
and contextual/textural features derived from the
two scans respectively. The supervised classification
procedure is built on the top of an unsupervised
unidimensional Graph Cut-based segmentation of
T1c and T2f MR images. The unsupervised stage
facilitates the subsequent supervised task by
identifying an intermediate hybrid class distributed
in a limited area and subsequently subdivided by the
SVM in meningioma and healthy tissue.
Before the segmentation process, T1c and T2f
MR images are co-registered for their combined use
in the analysis and a logarithmic contrast
enhancement is applied in order to enhance the
similarity between the edema and brain tissues in the
T2f image.
3.1 Graph Cut Based Segmentation
Graph Cut segmentation is separately applied to T1c
and T2f images allowing the labeling of intermediate
hybrid regions. The intersection of these initially
identified regions is subsequently analyzed and
classified by the SVM to identify meningioma areas.
The initialization of the Graph Cut segmentation is
automatically accomplished through the use of the k-
means clustering algorithm. The segmentation of
T1c image in three sub-volumes allows to identify a
hybrid region including all the contrast enhanced
tissues: meningioma, vessel and skull tissues.
From the segmentation of T2f image a partition
of the original volume in three regions is also
obtained corresponding to “air”,
“brain/edema/meningioma” and “skull” respectively.
By intersecting the hybrid region originally
identified in T1c with the
“brain/edema/meningioma” region identified in T2f
we obtain a refined region of interest to be presented
in input to the SVM classifier for the identification
of meningioma areas.
3.2 Supervised Multispectral
Classification
The present study considers the following features:
gray scale values from T1c and T2f scans (I
t1c
,I
t2f
),
first order texture features: mean (M) , variance
(Var), skewness (S), kurtosis (K) and entropy (E)
computed on T1c and T2f scans, intensities in
neighborhoods of voxels of both scans (I1-I26) . All
proposed features have been analyzed systematically
in the experimental evaluation phase in order to
determine the combination that is most appropriate
for the classification task (see section 4). The
features have been normalized to have zero mean
and unit variance.
A binary hard categorization is performed by the
SVM classifier that labels co-registered voxels
belonging to the region identified in the
unsupervised phase, as Meningioma (M) and
Healthy tissue (H). The SVM classifier is configured
as soft-margin LS model with Kernel RBF. During
the training phase, the SVM learns an approximation
for the true input–output relationship based on a
given training set of examples constituted by N
NCTA2014-InternationalConferenceonNeuralComputationTheoryandApplications
154

input-output pairs
N,1,i ,,
ii
yx
. The input
pattern
],..[
1
i
m
i
i
ffx
is an m-dimensional feature
vector where m is the number of features considered
in the current configuration, and
HMy
i
,
is a
supervised label denoting the membership in the
meningioma or healthy class.
After the segmentation, if the tumor area presents
necrosis and dishomogeneity, small holes within the
tumor mass classified as healthy tissues may appear .
A morphological procedure is therefore used to
refine the segmented masks making the tumor area
segmented more solid and compact.
4 EXPERIMENTS
The segmentation method was experimented on
multispectral datasets of 10 patients. Each dataset is
composed of T1c and T2f scans. The T1c is acquired
using a 3D sequence characterized by 1 mm
isotropic voxels, the inter-slice spacing of 1 mm and
the slice thickness of 1 mm; the T2f sequence
includes an in-plane resolution between 0,75 and
0,81 mm and slice thickness of 5 mm. The spacing
between slice is 6 mm for cases 1,3,6,7,8,10 and of
6.5 mm for cases 2,4,5,9. Performances were
assessed by adopting a behavioral comparison
strategy in which the masks obtained by the
automated segmentation were compared with the
masks obtained through a manual segmentation of
the T1c images. Manual labeling was performed by
a team of three experts with the support of a slice-
by-slice manual annotator.
The strategy adopted for the definition of a
suitable reference standard starting from
combination of multiple manual segmentations, is
Majory Voting (Heckemann et. al 2006).
4.1 Metrics
MRI segmentation was performed with the purpose
of determining the volume of pathological tissues
and their spatial distribution. The metrics adopted
for the volume estimation error is the normalized
absolute difference in size between reference and
segmented data.
Spatial overlap between reference and automated
maps is measured in terms of Jaccard (J), Precision
and Recall indexes (Bouix et al. 2007).
Common agreement between experts and
automated segmentation is quantified directly by the
Williams’ index. If this index is greater than one for
a given rater, it can be concluded that current rater
agrees with the other raters at least as well as they
agree with each other (Williams 1972).
4.2 Results
A trial and error phase was conducted in which
several configurations of the segmentation procedure
were considered distinguished by different values of
main parameters involved.
A first set of parameters was varied to tune the
Graph Cut model and the LS-SVM classifier. The k-
means algorithm has been used to initialize Graph
Cut segmentation. The value of k parameter was set
equal to three. The k value has been assessed taking
into account the expected MR signal in both the
MRI sequences considered. The value of the
standard deviation (σ) for Gaussian RBF kernel was
chosen as 0.5 in the SVM classifier.
Different configurations of the classification
procedure were also evaluated varying the number
of training examples and the features considered.
The configuration that showed the most balanced
behavior after the trial and error phase is based on
the following vector of features
ft
i
ft
i
ft
i
ft
i
ft
i
ft
i
ft
ct
i
ct
i
ct
i
ct
i
ct
i
ct
i
ct
ftcti
EKSVarMIx
EKSVarMIx
xxx
222222
2
111111
1
21
,,,,,
,,,,,
with,
and used a training set of 10.000 labeled samples
randomly chosen within the ground truth masks.
Table 1: Mean values of Jaccard, Precision, Recall and
Volume Error obtained by performing the leave-one-out
cross-validation (Interpatient) and by training and testing
the classifier on the same dataset (Intrapatient).
Jaccard
Index
Precision Recall
Volume
Error
(%)
Interpatient
Mean 0.867 0.814 0.942 16.087
Std 0.072 0.095 0.081 11.145
Intrapatient
Mean 0.959 0.967 0.991 4.750
Std 0.031 0.031 0.004 3.152
As shown in Table 1, for the intrapatient
analysis, the mean Jaccard coefficient over all 10
patients is 0.959, Precision and Recall have a value
equal to 0,967 and 0,991 respectively; the Volume
Estimation Error is equal to 4,750. The interpatient
analysis has provided a Jaccard cofficient equal to
0,867, Precision and Recall equal to 0,814 and
0,942 respectively and a Volume Estimation Error
equal to 16,087.
In Table 2 the results obtained using the
Williams’ index are listed. Numerical values clearly
AutomaticSegmentationofMRBrainTumorImagesusingSupportVectorMachineinCombinationwithGraphCut
155
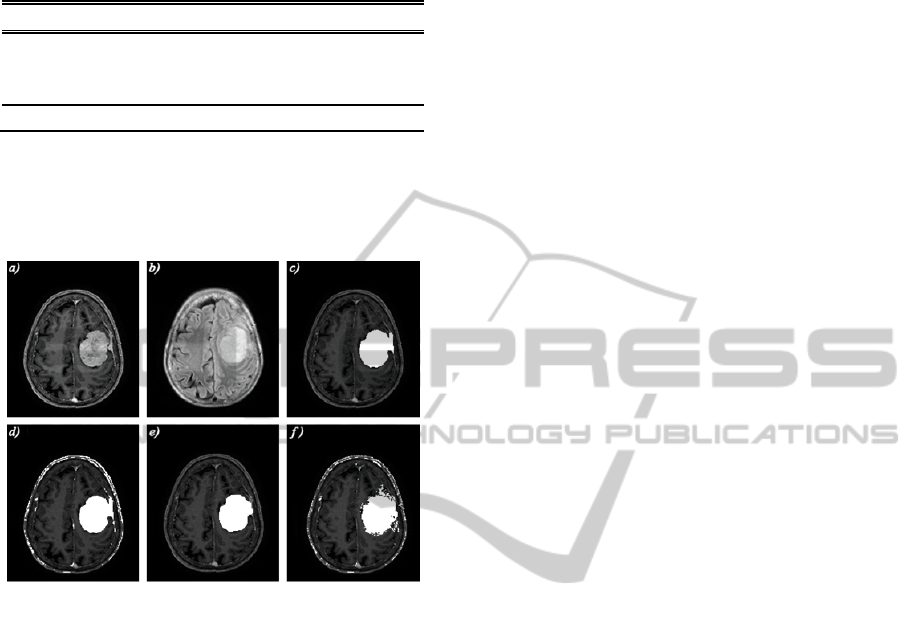
Table 2: Williams’ Indexes obtained by considering the
manual segmentations (E1-E3) and the automatic
segmentation (A) of the 10 cases under study.
W. I. 1 2 3 4 5 6 7 8 9 10
E1 1.0 1.0 1.0 1.0 1.0 0.9 0.9 1.0 0.9 1.0
E2 0.9 0.9 1.0 0.9 0.9 1.0 1.0 1.1 0.9 0.9
E3 0.9 1.0 0.9 1.0 0.9 1.0 1.0 0.7 1.0 1.0
A 1.0 1.0 1.0 1.0 1.0 1.0 1.0 1.1 1.0 1.0
indicate that the automatic segmentation method has
a good behavior with respect to the inter-expert
variability and often it shows a better behavior than
other raters.
Figure 1: Results of one axial slice obtained by the
interpatient analysis. a) Original T1c image, b) Original
T2f image, c) Overlap between original T1c image and the
ground truth mask, d) Overlap between original T1c image
and the intermediate mask obtained by Graph Cut
segmentation, e-f) Overlap between original T1c image
and the final segmentation mask obtained by interpatient
analysis, considering and without considering Graph Cut
stage respectively.
Figure 1 shows an axial slice of one patient and
the results obtained considering and without
considering Graph Cut stage in the overall strategy.
Results are clearly worse when not using the Graph
Cut preliminarily. For the dataset shown in Figure 1,
we obtained a Jaccard coefficient equal to 0.21109
and Volume Error equal to 363.4476 when no Graph
Cut is applied. With the complete procedure, Jaccard
is equal to 0.856 and Volume Error is equal to 7.119.
The mean computation time for the training task is
60 seconds and for the segmentation task performed
by the trained classifier is 159 seconds (without
Graph Cut, 732 s.) on a single CPU running at 2.26
Ghz.
5 CONCLUSIONS
Our objective in this study was to develop a fully
automatic hybrid image segmentation strategy for
meningioma segmentation in Magnetic Resonance
brain images. The supervised segmentation
framework is built on the top of the Graph Cut
algorithm initialized automatically. The strategy was
tested on a preliminary collected data set. The
results prove that the allied use of Support Vector
Machine and Graph Cut produces accurate
segmentation of tumors present in scenarios of
varied complexity. Accuracy results obtained are
encouraging. Future plans contemplate the
acquisition of new data with which to perform a
more significant interpatient analysis and to develop
of a comparative evaluation with other methods.
REFERENCES
Balafar, M., Ramli, A., Saripan, M., Mashohor, S., 2010.
‘Review of brain MRI image segmentation
methods.’Artificial Intelligence Review, 33, 261–274.
Bauer, S., Nolte, L-P., Reyes, M., 2011. Fully automatic
segmentation of brain tumor images using support
vector machine classification in combination with
hierarchical conditional random field regularization.
In: MICCAI Int. Conf. on Medical Image Computing
and Computer Assisted Interventions Berlin:
Springer).
Bauer, S., Wiest, R., Nolte, L-P., Reyes, M., 2013. ‘A
survey of MRI-based medical image analysis for brain
tumor studies.’ Phys. Med. Biol., 58, R97–R129.
Boykov, Y., Kolmogorov, V., 2004. ‘ An experimental
comparison of min-cut/max-flow algorithms for
energy minimization in vision.’ PAMI , 26, 1124- 37.
Boykov, Y., Funka-Lea, G., 2006. ‘ Graph cuts and
efficient n-d image segmentation.’ Int. J. Comput.
Vision, 70, 109-131.
Bouix, S., Martin-Fernandez, M., Ungar, L., Koo,
M.N.M.S., McCarley, R.W., Shenton, M.E., 2007.
‘On evaluating brain tissue classifiers without a
ground truth.’ NeuroImage, 36, 1207–24.
Caroli, M., Locatelli M., Prada F., Beretta F., Martinelli-
Boneschi F., Campanella R., Arienta C., 2005.
‘Surgery for intracranial meningiomas in the elderly: a
clinical–radiological grading system as a predictor of ‘
J Neurosurg, 102, 290–294 .
Clarke, L.P., Velthuizen, R., Camacho, M., Heine, J.,
Vaidyanathan, M., Hall, L., Thatcher, R., Silbiger, M.
S., 1995. ‘ MRI segmentation: methods and
applications.’ Magn Reson Imaging, 13(3), 343–36.
Gordillo, N., Montseny, E, Sobrevilla, P., 2013, ‘State of
the art survey on MRI brain tumor segmentation.’
Magn Reson Imaging, 31(8), 1426-38.
NCTA2014-InternationalConferenceonNeuralComputationTheoryandApplications
156

Greenberg, H., Chandler, W., Sandler, H., 1999. Brain
Tumors, Oxford University Press, Oxford.
Heckemann, R.A., Hajnal, J.V., Aljabar, P., Rueckert, D.,
Hammers, A., 2006. ‘Automatic anatomical brain mri
segmentation combining label propagation and
decision fusion.’ NeuroImage, 33(1), 115 – 126.
Kaus, M., Warfield, S., Nabavi, A., Black, P. M.,
Jolesz, F. A., Kikinis, R., 2001. ‘Automated
segmentation of MRI of brain tumors.’ Radiology,
218, 586–591.
Peng, Y., Liu, R., 2010. Object segmentation based on
watershed and graph cut. In: 3rd International
Congress on Image and Signal Processing (CISP).
Rother, C., Kolmogorov, V., Blake, A., 2004.
‘”grabcut”: interactive foreground extraction using
iterated graph cuts.’ ACM Trans. Graph., 23(3), 309-
314.
Santle, K. Camilus, Govindan ,V. K., 2012. ‘ A Review
on Graph Based Segmentation.’ IJIGSP, 4(5), 1-13.
Schoelkopf, B., Smola, A. 2002. Learning with kernels:
support vector machines, regularization, optimization,
and beyond, MIT Press .
Suykens, J.A.K., Van Gestel, T., De Brabanter, J., De
Moor, B., Vandewalle, J., 2002. Least Squares
Support Vector Machines. World Scientific Publishing
Co., Singapore.
Vapnik, V.N., 1995. The Nature of Statistical Learning
Theory, Springer-Verlag, New York.
Verma, R., Zacharaki, E., Ou, Y., Cai, H., Chawla, S.,
Lee, S., Melhem, E., Wolf, R., Davatzikos, C., 2008.
‘Multiparametric Tissue Characterization of Brain
Neoplasms and Their Recurrence Using Pattern
Classification of MR Images.’ Acad. Radiol., 15(8),
966-977.
Williams, G.W., 1972. ‘Comparing the joint agreement of
several raters with another rater.’ Bio-metrics, 32,
619–627.
Withey, D., Koles, Z., 2008. ‘A review of medical image
segmentation: methods and available software.’ Int. J.
Bioelectromagn, 10(3), 125 -148.
Wu, Z., Leahy, R., 1993. ‘An optimal graph theoretic
approach to data clustering: Theory and its application
to image segmentation.’ IEEE. Trans. on Pattern
Analysis and Machine Intelligence, 15 (11), 1101-13.
AutomaticSegmentationofMRBrainTumorImagesusingSupportVectorMachineinCombinationwithGraphCut
157
