
Sketching 2D Vessels and Vascular Diseases with Integrated Blood Flow
Patrick Saalfeld
1
, Alexandra Baer
1
, Uta Preim
2
, Bernhard Preim
1
and Kai Lawonn
1
1
Department of Simulation and Graphics, Otto-von-Guericke University, Magdeburg, Germany
2
Department for Radiology, Municipal Hospital Magdeburg, Magdeburg, Germany
Keywords:
Sketch-based Interface, Vascular Diseases, Computational Fluid Dynamics, Patient Education.
Abstract:
In this paper, we present a sketch-based interface, which allows medical doctors to illustrate different vascu-
lar diseases and treatment methods as well as fluid behavior. With this sketching interface, we provide the
physician with an effective tool to illustrate different medical cases, which is important in the complex field
of vascular diseases with respect to patient education. We use techniques from sketch-based interfaces and
GPU-based computational fluid dynamics by considering usability aspects. We provide a concept and a pro-
totypical implementation whose usability is tested with quantitative and qualitative methods. Additionally, we
interviewed a physician to assess the benefits of the tool with respect to patient education.
1 INTRODUCTION
Vascular diseases are a complex field with a large
amount of deaths comprising 31% worldwide (see
(Mendis et al., 2011)) and a big impact on economics
about e 196 billion in Europe (see (European Heart
Network, 2012)). The treatment of such a disease
leads to discussion about prevention, diagnosis, and
therapy not only with colleagues, but also with pa-
tients. Most important, the patient education gives the
patient an insight into the complexity and risk of the
possible outcome. An informed patient can benefit in
various aspects from comprehensive patient education
(see (Keulers, 2008)):
• they need less medication,
• the time of treatment may be reduced,
• they are more active in dealing with their diseases
and act more responsible, and
• they are more independent from their treating
physician.
Therefore, a tool that supports physicians to discuss
and communicate vascular diseases is very useful for
patient education. In this paper, we present a program
that helps the physician to easily create different vas-
cular diseases and to show various treatment options
that may help the patient in understanding their dis-
ease. This tool is inspired by the work of Zhu et al.
(Zhu et al., 2011) about sketching tubular shapes and
simulating liquids. Their method has several limita-
tions with respect to sketching vascular structures and
diseases. Furthermore, it is not optimized for tablet
devices, which could be integrated well in the process
of patient education. The physicians need an effec-
tive tool to sketch patient-specific cases and to illus-
trate the different treatment options for educating the
patient, see Figure 1. Hence, we make the following
contributions:
• We present appropriate methods to sketch vessels,
vascular diseases, and treatment options, which
are usable by medical doctors and understandable
by patients.
• We introduce a plausible representation of blood
flow in real time.
2 MEDICAL BACKGROUND
This work focuses on the vascular diseases of arteries,
which are vessels transporting blood from the heart to
the peripheral capillary of the body. A common rea-
son of those diseases is arteriosclerosis, which leads
to a hardening of vessels by deposition of blood fat,
thrombi, and lime (Kutikhin et al., 2013). This depo-
sition affects vessels in two problematic ways:
1. A weakening of the vessel wall.
2. A narrowing of the vessel (stenosis) up to a com-
plete occlusion.
The weakening can lead to a dilation of the vessels
that may result in an aneurysm. A possible outcome
379
Saalfeld P., Baer A., Preim U., Preim B. and Lawonn K..
Sketching 2D Vessels and Vascular Diseases with Integrated Blood Flow.
DOI: 10.5220/0005196203790390
In Proceedings of the 10th International Conference on Computer Graphics Theory and Applications (GRAPP-2015), pages 379-390
ISBN: 978-989-758-087-1
Copyright
c
2015 SCITEPRESS (Science and Technology Publications, Lda.)

(a) (b) (c)
(d) (e) (f)
Figure 1: Illustration of blood flow before and after a stenting treatment. First, the physician sketches a vessel with an
aneurysm (a). Afterwards, he manipulates and visualizes the underlying fluid simulation to illustrate the blood flow (b-c). The
consequences resulting from the stent treatment (d-e) are illustrated with another color (f).
might be a clot formation inside the aneurysm or a
rupture. A stenosis can lead to an undersupply of in-
volved structures or can cause a clot formation. An-
other dangerous effect is an increased risk of rup-
ture of an aneurysm in cerebral areas (Neugebauer
et al., 2010) with a mortality rate of 40-60% (Bed-
erson et al., 1994). In Figure 2 an aneurysm and
a stenosis are illustrated. For the medical treatment
of such vascular diseases, the physician can use sev-
eral methods, see Wong et al. (Wong et al., 2011)
for an overview. The choice of the treatment depends
on parameters such as location, anatomical access or
pathology. In particular, this work focuses on clipping
as an example of an extravascular treatment method as
well as coiling and stenting as examples of intravas-
cular methods.
The clipping procedure, e.g., for treating a cere-
bral aneurysm, starts with a craniotomy to disclose
the aneurysm. Afterwards, a titanium clip is placed
across the aneurysm neck to stop the blood from en-
tering into the aneurysm (Gasteiger, 2014). Coiling is
performed by entering an artery from the inside. The
coil, a small titanium wire, is used to fill the aneurysm
and to induce a thrombus formation (Teitelbaum et al.,
1994). The stenting method can be used to treat both
stenosis and aneurysms through an intravascular ap-
proach. Similar to the coiling procedure, a catheter is
moved to the affected position from the inside. After-
wards, the stent is expanded and forces the vessel to
expand to treat the stenosis. For treating aneurysms,
stents can help to support the involved vessel during,
e.g., a coiling procedure.
Further descriptions of different forms of vascular
diseases, their treatments, and possibilities for visual-
ization and exploration are mentioned by (Gasteiger,
2014). A historical overview of different treatment
(a) (b)
Figure 2: Illustrations of a saccular aneurysm (a), which
is the most common type, and a stenosis (b) caused by a
narrowed vessel.
options can be found in the work of (Wong et al.,
2011).
3 RELATED WORK
This work involves two main topics: computational
hemodynamics and Sketch-based Interfaces (SBIs), a
form of user interface (UI), which deals with sketch-
ing. The following parts name related work for the
latter topics.
3.1 Computational Hemodynamics
To simulate the behavior of blood, it is necessary to
imitate a non-Newtonian fluid (a fluid with varying
viscosity). Furthermore, in terms of fluid dynamics,
blood is compressible and inhomogeneous. Such a
simulation is not only complicated, but also expensive
regarding the calculation time (Bridson, 2008). We
consider blood as an incompressible, homogeneous
Newtonian fluid to achieve a real time simulation. Ex-
amples for methods to calculate non-Newtonian fluids
can be found in (Ciarlet et al., 2011) and (Ferziger and
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
380

Peri
´
c, 2002). To describe the state of a fluid, there are
two possibilities: the Lagrangian (a particle-based)
and the Eulerian (a grid-based) description. We use
the Eulerian description because the grid-based char-
acter is well suited to be calculated with fragment
shaders on the GPU. Examples for the Lagrangian de-
scription can be found in (M
¨
uller et al., 2004; Qin
et al., 2010). Both deal with particle-based simula-
tion of blood flow in vessels to support surgeons in
virtual surgery scenarios.
3.2 Sketch-Based Interfaces
The usefulness of SBIs to communicate ideas and
concepts is described among others in (Jorge and
Samavati, 2011). The communication of complex is-
sues is possible without the necessity to draw pre-
cisely and accurately. This advantage, however, is a
disadvantage for the computer, which tries to interpret
the sketch. As a consequence, several works deal with
the interpretation of sketches, see (Jorge and Sama-
vati, 2011) for an overview and examples. The foun-
dations of were laid by Ivan Sutherland (Sutherland,
1963) with the program Sketchpad. Sketch-based in-
terfaces can be seen as a part of post-WIMP (win-
dows, icons, menus, and pointers) UIs because the
sketching is performed with direct input, e.g., a pen or
touch (Preim and Dachselt, pear). So the pointer com-
ponent of the WIMP paradigm is no longer necessary
(van Dam, 1997). (Xu et al., 2002) and (Naya et al.,
2007) showed the advantage of reality-based inter-
faces (such as SBIs) by comparing WIMP-based in-
teraction with reality-based interaction and presented
two findings: first, the users preferred the sketch-
based approach and second they were more efficient
with it. SBIs contain three processing steps: resam-
pling of the input data, beautification, and recogni-
tion. This paper only deals with the resampling of the
input data. Examples for the second step (beautifica-
tion) can be found in (Igarashi et al., 1997). There,
line segments were analyzed according to geometric
constraints such as perpendicularity and parallelism.
After that, the program recommends different options
of how to interpret the lines. The user chooses an
option by clicking on it. The processing step recog-
nition describes a procedure where the sketch is com-
pared with an internal representation of symbols. The
similarity is expressed with a parameter. If this pa-
rameter exceeds a value, the sketch is interpreted as
the compared symbol (Jorge and Samavati, 2011). A
simple way to integrate recognition in an application
is described with the 1
¢
Recognizer (Herold and Sta-
hovich, 2012).
An important use case of SBIs is geometric mod-
eling, i.e., the creation of 3D structures. An example
of an SBI for modeling (SBIM) medical structures is
(Pihuit et al., 2010). There, methods are described
to sketch and model branching vessels. To maintain
the sketch-based look of 3D models, it is possible to
visualize them with non-photorealistic rendering. A
comparison of different rendering styles in the med-
ical domain can be found in (Lawonn et al., 2014b).
Contrary to SBIM, the following work addresses the
creation of 2D vessels.
4 METHODS
Our sketch-based interface consists of different con-
cepts to create vessels with interior blood flow based
on intuitive sketch-based gestures. In the following,
we describe the methods and the implementation.
4.1 Blood Flow Simulation
We use the Eulerian fluid simulation, which is de-
scribed in (Stam, 1999; Bridson, 2008; Harris, 2004).
For the fluid simulation, we use the Navier-Stokes
equations for incompressible and homogenous fluid,
which are based on Newton’s second law of fluid mo-
tion:
∂u
∂t
= −(u · ∇)u −
1
ρ
∇p + v∇
2
u + F, (1)
and
∇ · u = 0. (2)
Equation 1 (momentum equation) describes the be-
havior of the velocity vector field u under influ-
ences like advection, diffusion, pressure, and external
forces, which will be described in more detail in the
following. Equation 2 (continuity equation) ensures
the incompressibility by defining u as a divergence-
free vector field.
To achieve a real time fluid simulation, the grid
size is an important factor. While a small grid size ac-
celerates the calculation time, details like whirls may
be lost. Another problem are obstacles. To enable the
blood flow to react on these, it is necessary to model
them in the simulation grid. This is achieved by mark-
ing grid cells as occupied. So the grid resolution also
affects the possible level of detail of the obstacles. To
allow a high spatial resolution of grid cells, the sim-
ulation is performed on the GPU. For the GPU-based
calculation, we used the fragment shader similar to
(Harris, 2004).
To solve the equations for the differential oper-
ators, the finite difference method is used. Further-
more, Equation 1 needs to be split up in single terms,
which are calculated separately.
Sketching2DVesselsandVascularDiseaseswithIntegratedBloodFlow
381
External
Force
External
Force
Advection
Advection
Diffusion
Diffusion
Pressure
Calculation
Pressure
Calculation
Pressure Gradient
Subtraction
Pressure Gradient
Subtraction
Fix
Boundary
Fix
Boundary
Fix
Boundary
Fix
Boundary
Fix
Boundary
Fix
Boundary
Fix
Boundary
Fix
Boundary
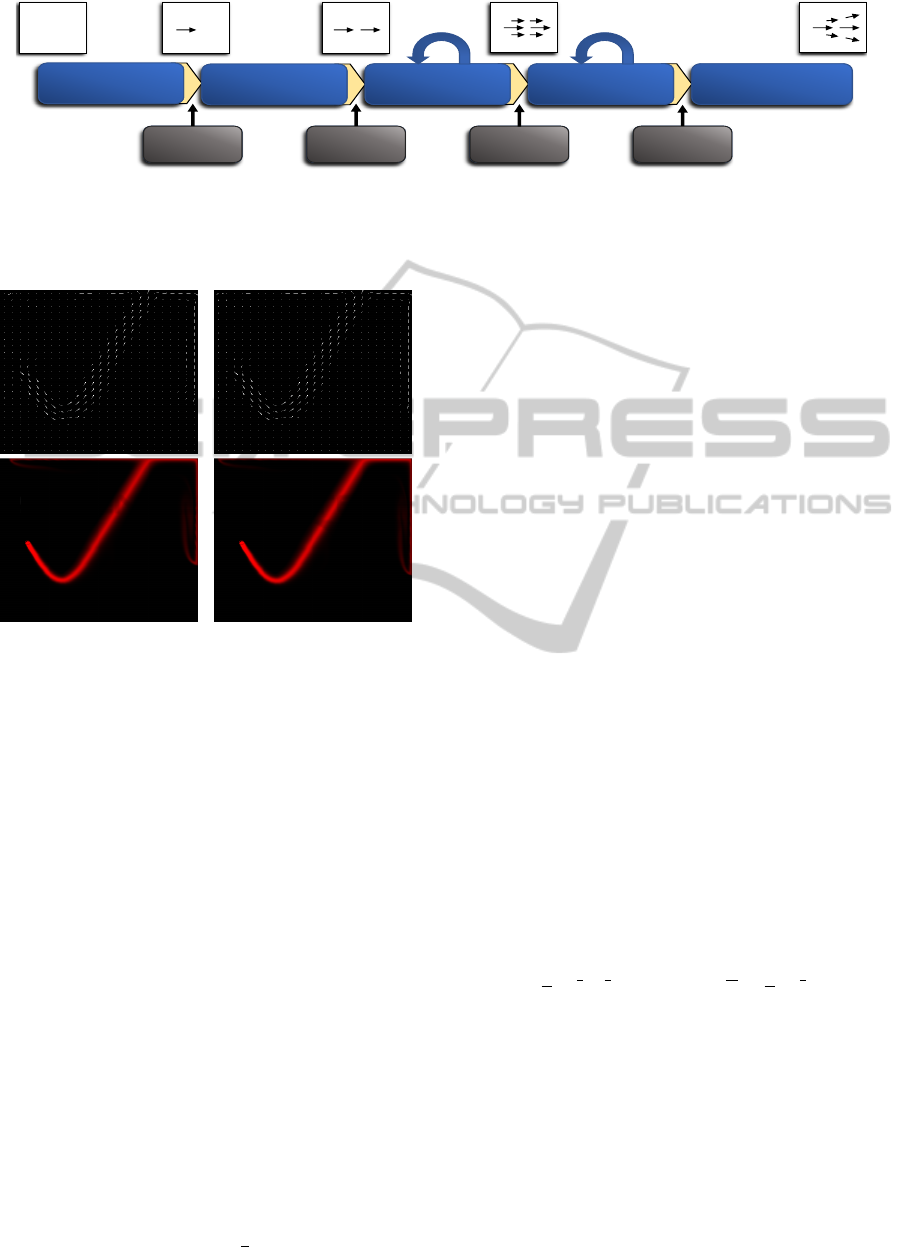
Figure 3: Overview of the calculation pipeline for the fluid simulation including the influence to the underlying vector field.
Each step is realized with a separate fragment shader program. The steps provided with self-referencing arrows show the
steps which are calculated with an adjustable amount of iterations. Here, a trade-off arises between accuracy (high amount of
iterations) and calculation time (low amount of iterations).
(a) (b)
Figure 4: Comparison of the Euler (a) and Runge-Kutta 4
(b) integration methods with a step size of 0.3. Both meth-
ods are visualized with a modified arrow plot visualization
(top) and a scalar field visualization (bottom), which looks
similar to a streakline visualization through the continu-
ous placement of ink during the simulation. Since the dif-
ferences between both methods are small, the faster Euler
method is used.
An overview of the calculation steps is illustrated
in Figure 3. In the following, we describe the mathe-
matical terms and their effects in the fluid simulation.
The first term −(u · ∇)u describes the self-
advection of the fluid, which is the process of mov-
ing the velocity itself through the fluid. Here, the
self-advection is realized with semi-Lagrangian ad-
vection (Stam, 1999). Mostly, fluid simulations use
the Runge-Kutta method (Butcher, 1996) for the in-
tegration, which is less error-prone than the simpler
and faster Euler method. Interestingly, as illustrated
in Figure 4, the differences between both methods are
small, due to the small step size used in the applica-
tion. Therefore, we used the Euler method to decrease
the calculation time.
The pressure, a force that gradually spreads from
regions with high to regions with low pressure, is de-
scribed with the second term
1
ρ
∇p. The factor ρ is
a constant to describe the density. Futhermore, this
term also ensures the incompressibility of the veloc-
ity vector field, and thus simultaneously ensures the
Equation 2. To achieve this, the term is calculated at
the end. For a description of the derivation, see (Brid-
son, 2008). To calculate this term, it is necessary to
solve a Poisson equation. This is accomplished by the
Jacobi approach because it can be mapped directly
to GPU facilities. For a discussion of different ap-
proaches, see (Kr
¨
uger and Westermann, 2003).
The third term v∇
2
u expresses the physical pro-
cess of diffusion, i.e., the property of mixing materi-
als without external forces. Here, v is a constant that
describes the viscosity. The calculation of the diffu-
sion also requires the solution of a Poisson equation.
A disadvantage of diffusion is the resulting smoothing
effect on the applied vector field, which causes a loss
of details. Therefore, the choice whether diffusion is
applied is left to the user.
The last term F describes external forces, which
allows the user to influence the simulation dynami-
cally. Normally, such forces are steady, so the influ-
ence of the force to fluid is constant over time. This
approach would not reproduce the pulsating character
caused by the contractions of the heart. This pulsa-
tion can be imitated by applying a factor to the force,
which changes over time. Therefore, we approximate
the function measured by an electrocardiogram with
the following formula:
f (x) = −
3
4
e
−
1
2
(−
1
2
+x)
2
+ 1, 12e
−
x
2
2
−
1
4
e
−
1
2
(1+x)
2
+ 1.
(3)
This equation was determined by combining three
Gaussian functions with varying heights and widths.
In Figure 5, the difference between a constant and a
pulsating force is illustrated.
Finally, boundary conditions are used to simu-
late the behavior of the fluid at the vessel wall and
the boundary of the draw area. These conditions
are necessary for the velocity vector field and for
the scalar field, describing the pressure. For the ve-
locity vector field, a Dirichlet boundary condition is
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
382

Figure 5: In (a) a constant force is applied to the vector
field. In (b) a heartbeat-like function is applied as a factor,
which mimics a more realistic behavior. The corresponding
functions are plotted at the left bottom.
used, which states that the velocity drops to zero at
boundaries. For the scalar field, a Neumann con-
dition is used, which states that the derivatives are
zero. To calculate the derivative, the normal of the
boundary is necessary. The normals of the top, right,
bottom, and left image boundaries are defined as
(0, −1), (−1, 0), (0, 1), and (1, 0), respectively. For
determining the normals of arbitrary boundaries, we
use the neighborhood of the obstacle. That means that
for each grid cell, which is an obstacle, eight neigh-
bors are analyzed. Depending on the state of these
neighbors (obstacle or no obstacle) the normal is ap-
proximated to one of eight possible directions. This
approach is described in more detail in (Wu et al.,
2004).
4.2 Blood Flow Visualization
To illustrate the unsteady vector field that represents
the blood flow, we implemented two visualization
concepts: (1) a direct and (2) a sparse flow visual-
ization, see Figure 4. The direct visualization is a
modified line plot on which fans are drawn on an ad-
justable grid. Fans are used because they facilitate a
fast realization of the flow direction for the user.
Especially for patients, a scientific visualization
method for vector fields may be inappropriate. Thus,
an additional method is used. It aims to be easily un-
derstandable to visualize the behavior of blood in ar-
eas such as aneurysms and stenoses in a descriptive
way. The used scalar field visualization is inspired by
the idea to place colored ink in the vector field (also
known as dye injection). By diffusion and advection,
this ink is transported through the vector field. The
amount of ink is color-coded with a black-to-hue scale
with different colors. This allows using multiple col-
ors, e.g., to show how blood mixes in an aneurysm
before and after a treatment, see Figure 1.
The colors are taken from the Lab color space,
which allows choosing colors that are roughly per-
ceptually linearized regarding hue and brightness. To
determine different colors, the approach of (Glaßer
et al., 2014) is used. If the ink is placed continu-
ously over time, the visualization technique is similar
to streaklines. In contrast to streaklines, the placed
ink is not connected, but if the amount of placed ink
is high enough, there is the impression of connectiv-
ity. A difficult task by sparse visualization techniques,
namely to identify suitable seed point positions, is left
to the user, i.e., the physician or the patient.
4.3 Sketching
The obtained data from the input device inherits
noise, which is caused by the conversion from the
analog signal to a digital signal (quantization) as well
as the imprecise input from the user. Especially the
quantization, also depending on the sampling rate,
reduces input information during fast input move-
ments from the user. To remove the resulting noise
and obtain equidistant input information, the received
data of the input device is resampled and smoothed.
These steps are commonly applied after the user fin-
ishes drawing, which causes abrupt changes in the
sketch. To avoid these sudden changes, we use on-
the-fly methods that are applied during sketching.
This presents challenges regarding real time capabil-
ity. Ideally, the user did not even realize these steps.
First, the resampling should reduce the obtained sam-
ple points. To achieve this, we use a simple strategy:
we ignore all points that are too close to the last ac-
cepted point. We use the Euclidean distance to mea-
sure the distance of two points, see Figure 6.
A disadvantage of this approach is that it is prone
to draw zigzag lines. This can be neglected, because
vessels, vascular diseases, stents, and coils usually do
not have those shapes. To smooth the accepted sam-
ple points, a local Gaussian filter similar to (Taubin,
1995) is used. More precisely, the 1-neighborhood of
the accepted points is used to adjust the points with a
Gaussian distribution. The filter is only applied to the
pre-last point, which was accepted to prevent the line
segments from shrinking, see Figure 6.
5 APPLICATION
The application should be as flexible as possible re-
garding the used direct input device. More precisely,
it should be possible to control the application with
touch and pen-based input. This implies the follow-
ing limitations:
• Special functions from pen-based input devices
like pressure sensitivity or additional buttons are
Sketching2DVesselsandVascularDiseaseswithIntegratedBloodFlow
383

not supported.
• Multitouch input is not supported.
Through these limitations, the used concept is theo-
retically usable for a mouse with a left button only.
Furthermore, the application is designed according
to major usability criteria, such as ”suitability for the
task”, or ”self-descriptiveness”. The evaluation of the
application revealed possibilities for improvements,
which were partially implemented. Improvements
based on the quantitative part, the qualitative part,
or the interview, are marked and discussed. The
application can be downloaded under wwwisg.cs.uni-
magdeburg.de/∼patrick/application/SketchingVessel
s.zip).
5.1 Sketch Vessels and Vascular
Diseases
Mainly, drawing a vessel requires two lines represent-
ing the border of the vessel. Unfortunately, drawing
each line separately leads to strong variations in the
resulting vessel structures. Therefore, we use a cre-
ate tool that creates both lines simultaneously by us-
ing the sketched path as the center line and drawing
the vessel wall around it. The general advantage of
this process is that uniform vessels can be drawn eas-
ier and faster. We fix the width of the vessel to sim-
plify the application. In contrast to (Zhu et al., 2011),
where it is possible to draw vessels under and over al-
ready existing ones through a 2,5D sketching canvas,
this work limits the sketching area to a 2D canvas.
This decision is reasoned by considering what could
happen if the user sketches over an already created
vessel. Besides the possibility to draw the vessel over
or under the existing ones, it is possible to merge the
new vessel with the old ones. This offers the possibil-
ity to create more complex structures like branching
vessels and aneurysms without changing the drawing
1
2
1
2
3
(a)
�
1
�
2
(b)
Figure 6: (a) The new point p
2
has exceeded a specific dis-
tance to p
1
and thus, is sampled. The points o
1
- o
4
are
too close and thereby omitted. (b) shows the smoothing ap-
proach. In the moment p
2
is sampled, the pre-last point p
1
is smoothed. By sampling the pre-last point, a shrinking of
the line is prevented.
Figure 7: As illustrated, the merging behavior can be used to
draw a vessel containing an aneurysm by simply sketching
on top of the previous drawing.
mode. The merge behavior is illustrated in Figure 7
and is realized with an already implemented polyline-
based functionality in the used framework.
Additionally to the possibility to draw aneurysms,
the application offers a possibility to draw stenoses.
A cut tool is used to allow the user to create irregu-
lar non-symmetrical stenoses in a consistent sketch-
based way. To prevent problems during cutting, e.g.,
ambiguity (see (Heckel et al., 2013)), the user does
not only see his sketched contour, which is used for
the cutting process. The start and end point of the
sketched contour are connected, and thus span a cut-
ting area to support the user. This area is substracted
from the existing vessels (see Figure 8).
This tool does not only allow the creation of
stenoses; in combination with the create tool, the user
has a generic sketching tool, which allows the cre-
ation of any 2D structure under the usage of only two
different modes. To support the medical expert dur-
ing the sketching process, it is possible to load im-
ages in the background. The physician can use this
function to load a slice of patient-specific MRI or CT
data, which contains, e.g., the vessel structures of the
patient. In addition frequently used vessel structures,
such as the Circle of Willis, can be loaded (see Figure
9).
During the evaluation, some participants suggest
a possibility to load vessel structures. This could
Figure 8: The cut tool is used to remove structures from the
already sketched vessel to create a stenosis.
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
384

Figure 9: The medical doctor has loaded a background im-
age of the Circle of Willis which supports the sketching pro-
cess.
help because it allows the physician to not only
load standardized structures, but also patient-specific
data. The functionality is implemented by loading
a monochrome black-and-white image. Every black
pixel is interpreted as a vessel and every white pixel
as free space, where blood is able to flow. This ap-
proach has, however, a disadvantage. It is only possi-
ble to show bloodflow in the loaded vessel structure,
but not to use the other tools, e.g., cutting or treating
the vessel. This disadvantage can be avoided by im-
plementing an object-based save and load mechanism
instead of an image-based, which could be added in
the future.
5.2 Manipulate and Visualize Blood
Flow
As described in Section 4.1, the flow can be manip-
ulated with a force term, which is represented with a
2D vector field. To allow the user to influence this
field in a flexible and easy way, a direction tool is
used, which also works in a sketch-based way. Af-
ter the direction tool is selected, the user can sketch
lines, that are represented by arrows directing in the
draw direction. This arrow represents the influenced
force on the vector field.
To transfer the sketched arrow to the vector field,
it would be obvious to manipulate only the vectors
directly under the sketched arrow. This behavior is
not desirable, because it only allows to manipulate the
flow in small areas. Instead, the force is applied in
a region around the sketched arrow. The size of this
region is adjustable and is initially set to the width of a
vessel. To achieve a natural effect, the force is slightly
decreased at the border of the region by applying a
Gaussian distribution, resulting in a strong force at
the center and a weak force at the border.
To visualize the flow, the user can use a dye tool
to place ink (blood) in the fluid region. A circle of
ink is placed by just tipping on the canvas or drawing
over an area. The ink is interpreted as a source of in-
finite amount of ink, which is added in every render
frame. The width of the ink area is adjustable, but set
to the vessel width to allow the user to fill a vessel
with ink in a fast way. Under the assumption that the
physician wants to show the mixing behavior of fluid
by repetitive using of the dye tool, a new color is used
after each usage of the dye tool with the approach ex-
plained in Section 4.2.
5.3 Treating Options
To show the patient how to treat aneurysms and
stenoses, we implemented the following treatment
methods:
• coiling,
• clipping,
• and stenting.
First, we implemented the coiling tool such that the
user should fill out the aneurysm. This coil was illus-
trated with a line, which occurred during the drawing
process. Generating the coil with this approach is not
applicable, since it takes too much effort and is too
time-consuming, which was stated by the participants
in our evaluation. Therefore, we implemented another
approach to improve the coil placement.
First, an area is sketched, e.g., on the vessel wall
of an aneurysm. After the user raises the pen, every
sample point analyzes its neighborhood with a spe-
cific adjustable distance. This is performed by send-
ing eight rays in a circular manner, starting at ev-
ery sample point. Significantly less rays would result
in an inaccurate vessel wall detection and more rays
would not improve the result. We test every ray for
intersecting the vessel wall. If more than one ray col-
lides with the wall, the ray with the shortest distance
is chosen. After that, the corresponding sample point
is placed on the intersection point of the ray. If no
collision occurs, the point keeps the current position.
The underlying grid cells are than treated as obstacles
and thus, the blood flow changes dynamically corre-
sponding to the drawn coil. Figure 10 illustrates this
algorithm.
The clipping method was inspired by the line
drawing method used in Sketchpad (Sutherland,
1963). The point where the user starts drawing rep-
resents the first point of the clip. As long as the user
draws with the pen, the current pen position repre-
sents the end point of the clip. These two points are
connected with a dashed line to give the user a pre-
view of the clipping result. After the user finishes
drawing and raises the pen, the clip is placed and the
blood flow simulation is affected by it.
Sketching2DVesselsandVascularDiseaseswithIntegratedBloodFlow
385

The stent placement algorithm is inspired by the
real treatment. Here, a balloon catheter is inflated
to dilate the stent in the vessel. We use this inflat-
ing process for providing a stent placement algorithm.
The user draws a line in the center of the vessel with
the stenting tool, which represents the position of the
balloon catheter. After the user finishes the sketch-
ing process, the application calculates the dilation of
the stent. The difficulty to achieve this behavior is to
dilate the stent in the relevant vessel, but not to en-
ter the aneurysm. Since the structure is not differen-
tiated semantically (i.e., in vessels and aneurysms),
the described behavior must be achieved in another
way. To accomplish this, the surrounding region of
the sketched stent is analyzed. The algorithm is de-
scribed in more detail in Figure 11. This method is
more robust according to various input lines. The best
results regarding visual aspects are achieved by draw-
ing a line, which is as close as possible to the center of
the vessel. Inaccurate lines may lead to penetration of
the stent inside the aneurysm. A disadvantage of the
described method is that it depends on four control
points obtained through the start and the end point. If
(a) (b)
(c) (d)
(e) (f)
Figure 10: (a) - (f) shows the algorithm to calculate the coil
area. In (a) the physician sketches imprecise over the vessel
wall of the aneurysm. (b) shows the captured sample points
as well as the rays, which are sent in a circular manner from
each sample point. In (c) all rays that collide with the ves-
sel wall are highlighted, from which those with the shortest
distance are chosen (d). The sample points are moved to the
intersection point or remain on their position if no intersec-
tion occurred (e). The adjusted sample points build the new
area for the coil, which lies precisely on the vessel wall.
(a) (b)
(c) (d)
(e) (f)
Figure 11: (a) - (f) illustrate the algorithm to calculate the
stent dilation. The user input is illustrated as a sketched
line in (a). After the user finishes the stent, the normals
of the start and end point are determined (b). The normals
have the length of the vessel width. All four normals are
tested if they intersect the vessel wall. If not, the stent is
discarded. Otherwise, the distance l is determined, which is
the longest length of the normal start point to its intersection
point. Now, for every point of the sketched line, the normals
with length l are determined (c). All of these normals are
tested for intersections with the vessel wall. If they inter-
sect, they are shortened to the intersection point, otherwise
they keep the length l (d). The achieved end points of the
normals are connected (e) and form the border of the stent,
visualized in (f).
not all control points could be determined, e.g., if the
vessel is too wide, not all normals are calculated and
so the stent will not be placed. Similar to the coil-
ing and clipping method, the grid cells under the stent
are marked as occupied and thus, influence the fluid
simulation.
5.4 Edit, Delete and Copy Objects
The possibility to edit and delete objects such as stents
and coils, is an important aspect regarding the con-
trollability of the application. This is confirmed by
the evaluation (see Section 6.1) in which the partic-
ipants stated that a functionality to delete or edit ob-
jects would be useful. To realize this, it is necessary to
implement a method to select already created objects.
Since a requirement of the application is to control
it with a pen without any further buttons, a suitable
method is necessary. Besides the possibility of ges-
tures or a double tap of the pen, the implemented ap-
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
386

proach is to press and hold the pen on the relevant ob-
ject. Afterwards, the corresponding object is deleted.
The possibility to edit objects, which is currently not
implemented, can be realized with the same approach.
To provide the physician with an easy and fast
possibility to illustrate more treatment options on the
same vessel structures, a copy tool is added to the ap-
plication. It allows to select an area, copy its content
and paste it at another place. With this possibility, the
patient can see different behaviors of the bloodflow,
depending on the treatment option. This can help un-
derstanding why a specific treatment is chosen or why
another is not possible.
5.5 User Interface
The structure of the user interface results from two
considerations:
• The grid used for the fluid simulation is quadratic
and
• adirect input device is used.
The first consideration implies that the canvas in
which the user sketches is also quadratic. Through the
horizontal format of current displays this means that
there is potential free space on the sides of the canvas.
This space is used for the menus. In detail, the space
is divided in a left and a right region and used for a se-
mantic differentiation of the functionality. On the left
side, menus and buttons are placed, which are used to
create and manipulate vessels, to sketch treatment op-
tions, and to visualize and control blood flow. On the
right side, there are temporary menus to control simu-
lation parameters such as the number of iterations for
solving or to activate the diffusion process. The sec-
ond consideration (using a direct input device) leads
to the following requirements with respect to interac-
tion. To achieve an easy interaction, all buttons have a
bigger size and there are no sub-menus. Furthermore,
classical WIMP input elements like spin boxes were
omitted, since exact inputs would be hard with, e.g.,
a pen. In Figure 12, a screenshot of the user interface
is shown.
6 EVALUATION
The evaluation is divided into two parts: in the first
part, we used qualitative and quantitative methods for
assessing the usability of the prototype. In the second
part, we interviewed a physician to compare the pro-
cedure of typical patient education (with hand drawn
sketches) with the prototype and investigate advan-
tages and disadvantages.
Figure 12: Screenshot of the prototype. Buttons (1-2) are
used to create and edit vessels, (3-4) are used to manipulate
and visualize blood flow, (5-7) are used to sketch treatment
options, (8) is used to select the copy tool, (9) is used to load
a background image to assist the sketching or load vessel
structures out of a monochrome image and (10) is used to
reset the whole canvas. On the right side the functionality
to influence the fluid simulation in different ways is shown.
6.1 Usability
The qualitative part of the study was performed with
the think aloud method, where the participants com-
ment their activities while solving a problem. It is
helpful to give insights into the misunderstandings of
the participants as well as to understand how the par-
ticipants predict the behavior of the prototype. This
method gives hints for further refinements. The quan-
titative part of the evaluation was conducted with a
questionnaire. The questions are inspired by a ques-
tionnaire for ergonomic principles from ISO 9241-
110 (suitability for learning, suitability for the task,
self-descriptiveness, conformity with user expecta-
tions, controllability, and error tolerance). The single
questions were categorized in the different principles
and could be rated with a 7-point Likert scale.
The evaluation started with a short introduction
of the prototype on a SMARTBoard, a 70” screen
which allows pen interaction. Here, all features were
demonstrated and we asked to think-aloud and noted
the spoken comments of the participants. Initially, the
participants were asked to perform several tasks that
were handed out in written form. For example, they
should draw a vessel with a trifurcation; then, they
should use the cut tool to change the vessel to a bi-
furcation. Furthermore, they were asked to draw an
aneurysm and treat it with the clipping, coiling, and
stenting tool. Finally, they should create and visualize
the blood flow in a specific direction. Afterwards, the
participants were asked to fill out the questionnaire.
Sketching2DVesselsandVascularDiseaseswithIntegratedBloodFlow
387
+++
++
+
-/+
-
--
---
Figure 13: This figure shows the average results of the ques-
tionnaire for each category of the usability principles.
The evaluation was conducted with 14 researchers
with medical visualization knowledge. We had three
female and eleven male participants, aged from 25 to
44 with an average of 31 years. The participants are
experienced computer users with an experience of 20
years on average (minimum: 14, maximum: 30). Ten
participants were experienced with pen interaction. It
took about 15 to 20 minutes to fill out the question-
naire. The statements of the participants are denoted
with [P#].
Think-Aloud Method: Mostly, the participants were
satisfied with the prototype: ”it is possible to create
vessels and aneurysms according to my own ideas”
[P13]. Regarding the different tools, the majority of
the participants had no problems using them. For ex-
ample, it was stated that the cut tool ”is more precise
than conventional eraser tools” [P6]. Unfortunately,
the cut tool leads to misunderstandings during the first
use. This was caused by different expectations, e.g.,
some participants thought that it can be used as a con-
ventional eraser. After some practice, the functional-
ity was understood and the participants rated this tool
as positive. Another example of a positively rated tool
is the stenting tool. Especially the automatic expand-
ing behavior was noted as useful. In summary, the
participants were satisfied with the prototype. Fur-
thermore, they were also pleased with the alignment
of the tools, as it supports the typical workflow (gen-
erate vessel, treatment, and visualize blood flow). In
general, they stated that this prototype allows a fast
and easy generation of vessels with simulated blood
flow.
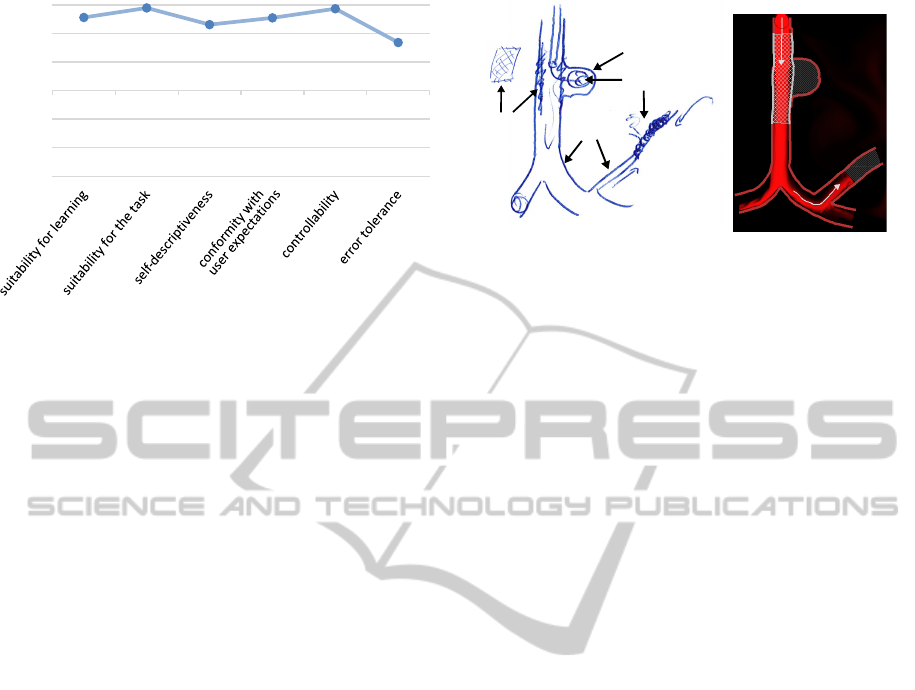
Questionnaire: The results of the questionnaire were
determined by calculating the average of every an-
swer. For this, we assigned the symbols of the 7-Point
Likert scale (- - - to + + +) to the values from -3 to 3.
Figure 13 depicts the average for every category. The
category error tolerance has the lowest rates. Here,
Aneurysm
Vessel
Coil
Stent
(a) (b)
Figure 14: On the left side is the hand sketch of a physi-
cian with additional labels. On the right side are the same
structures made with the application, which the physician
assessed clearer and more understandable.
the users were asked if the effort to correct an inad-
vertently drawn error is significantly high. Mostly, the
participants stated that it should be possible to delete
drawn objects and the effort is high to manually cor-
rect them by redrawing. Thus, we added the possibil-
ity to delete drawn objects like stents and coils indi-
vidually, see Section 5.4. However, with an average
of 1.68, this category was rated well. In summary, the
ratings of all categories were positive and the partic-
ipants were satisfied with the functional range. For
refinement, we used the results of the evaluation and
improved the prototype according to the suggestions
of the participants.
6.2 Structured Interview
We performed a structured interview of about 20 min-
utes with a physician, which has 12 years of experi-
ence in the field of vascular diseases and patient edu-
cation. First, we asked the physician to explain typical
patient education with an example and outline the typ-
ical education procedure. During typical patient edu-
cation, the patient receives textual and image-based
templates with respect to his disease. Additionally,
the physician explains the intervention verbally and
supports this with hand-drawn sketches (see Figure
14). These can help to illustrate specific medical cases
and answer individual questions from the patient. The
description of the physician revealed the following
disadvantages of the hand-drawn sketches:
• To correct errors, the physician draws over the ex-
isting sketch, which results in a cluttered image.
• The blood flow and the implications of a treatment
are usually not drawn, because it is hard to illus-
trate these and could lead to a confusing image.
• The physician only uses one color to draw all
structures, which makes it difficult to distinguish
between different elements.
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
388

• Some patients have problems to understand the
sketch, because they find it difficult to imagine
diseases and treatment options.
After that, we explained the prototype and asked to
perform the patient education again using our appli-
cation. Thereby, we let the physician compare his
hand-drawn images with sketches from the prototype
which showed the following:
• By using different colors, symmetrical, consis-
tent and uniform structures (e.g., for the vessels
or stents), the result is more clear, descriptive and
plastic than the hand sketching, and thereby, the
perception is supported.
• Due to the illustration of blood flow, the ves-
sel structures, the effects of different intervention
methods and possible complications are more un-
derstandable and imaginable.
Furthermore, the physician stated additional advan-
tages of the prototype, e.g., due to the clearer resulting
images, other persons, who were not involved in the
sketching process, are able to understand the sketch.
This is difficult with hand sketches, because of their
cluttering nature. The physician rated the tool as easy
to learn and use, which matches the results of our us-
ability evaluation.
7 CONCLUSIONS AND FUTURE
WORK
This work provided a concept and a prototypical im-
plementation to sketch vessels, vascular diseases, and
treatment methods. It is possible to interactively cre-
ate and manipulate blood flow, which adapts itself in
real time to change the sketch.
The quality and usability of the prototype was
assessed with qualitative and quantitative methods.
The positive feedback of the evaluation indicates that
the proposed concept and prototype are suitable for
sketching vascular structures and treatment options.
The structured interview with the physician revealed
further benefits compared to hand-drawn sketches and
confirmed the idea to improve patient education and
intelligibility by integrating animated blood flow.
A limitation of this work is the representation of
the vessels and blood flow in 2D. The representa-
tion of vessels and fluids in 3D is a challenge with
respect to visualization and interpretation (Lawonn
et al., 2014a). An advantage of the 2D representation
is its easier intelligibility as well as a faster calcula-
tion of details, which is important to achieve a real
time simulation.
For the future, we want to investigate how the de-
scribed concept and prototype can be used for collab-
oration between physicians. This collaboration can
happen at different places where each medical doctor
interacts with an instance of the application. The dif-
ferent instances could be connected and mirror the in-
put from one place to another. Additionally, the com-
munication could be supported with voice chat and
webcams. Another way of collaboration is possible
with only one application, where several physicians
are sketching at the same time in front of a big screen
and discuss a medical case.
Further analysis could investigate the possibility
to lift the whole concept to the third dimension. This
leads to challenges and questions regarding a real time
3D fluid simulation and visualization as well as cre-
ating the vessels. For 3D interaction, there are in-
put and output devices which are more suited than
pen or touch interaction. By lifting the application in
the third dimension, the behavior of blood in vessels
could be simulated more accurately, and thus, make
the application more relevant for scenarios like oper-
ation planning and training.
REFERENCES
Bederson, J. B., Connolly, E. S., Batjer, H. H., Dacey,
R. G., Dion, J. E., Diringer, M. N., Duldner, J. E.,
Harbaugh, R. E., Patel, A. B., and Rosenwasser, R. H.
(1994). Guidelines for the management of aneurys-
mal subarachnoid hemorrhage a statement for health-
care professionals from a special Writing Group of the
Stroke Council, American Heart Association. Stroke,
40(3):994–1025.
Bridson, R. (2008). Fluid Simulation for Computer Graph-
ics. A K Peters/CRC Press.
Butcher, J. C. (1996). A History of Runge-Kutta Methods.
Appl. Numer. Math., 20(3):247–260.
Ciarlet, P., Glowinski, R., and Lions, J. (2011). Numerical
Methods for Non-Newtonian Fluids: Special Volume.
Handbook of Numerical Analysis Handbook of Nu-
merical Analysis. Elsevier.
European Heart Network (2012). European cardiovascu-
lar disease statistics. http://www.ehnheart.org/cvd-
statistics.html. last visited: 18 June 2014.
Ferziger, J. H. and Peri
´
c, M. (2002). Compressible flow. In
Proc. of Computational Methods for Fluid Dynamics,
pages 309–328. Springer Berlin Heidelberg.
Gasteiger, R. (2014). Visual Exploration of Cardiovascular
Hemodynamics. PhD thesis, Otto-von-Guericke Uni-
versity Magdeburg.
Glaßer, S., Lawonn, K., and Preim, B. (2014). Visualization
of 3D Cluster Results for Medical Tomographic Image
Data. In Proc. of Conference on Computer Graph-
ics Theory and Applications (VISIGRAPP/GRAPP),
pages 169–176.
Sketching2DVesselsandVascularDiseaseswithIntegratedBloodFlow
389

Harris, M. J. (2004). GPU GEMS Chapter 38, Fast Fluid
Dynamics Simulation on the GPU. Pearson Higher
Education.
Heckel, F., Moltz, J. H., Tietjen, C., and Hahn, H. K. (2013).
Sketch-based editing tools for tumour segmentation
in 3d medical images. Computer Graphics Forum,
32(8):144–157.
Herold, J. and Stahovich, T. F. (2012). The 1
¢
; Recognizer:
A Fast, Accurate, and Easy-to-implement Handwrit-
ten Gesture Recognition Technique. In Proc. of the
International Symposium on Sketch-Based Interfaces
and Modeling, SBIM ’12, pages 39–46. Eurographics
Association.
Igarashi, T., Matsuoka, S., Kawachiya, S., and Tanaka, H.
(1997). Interactive beautification: A technique for
rapid geometric design. In Proc. of ACM Symposium
on User Interface Software and Technology, pages
105–114.
Jorge, J. and Samavati, F. (2011). Sketch-based Interfaces
and Modeling. Springer London.
Keulers, B. (2008). Computer-based Patient Education: Its
Potential in General and Plastic Surgery. PhD thesis,
University Nijmegen.
Kr
¨
uger, J. and Westermann, R. (2003). Linear Algebra Op-
erators for GPU Implementation of Numerical Algo-
rithms. ACM Trans. Graph., 22(3):908–916.
Kutikhin, A., Brusina, E., and Yuzhalin, A. E. (2013).
A Hypothesis of Virus-Driven Atherosclerosis. In
Viruses and Atherosclerosis, volume 4 of Springer-
Briefs in Immunology, pages 1–3. Springer New York.
Lawonn, K., Gasteiger, R., and Preim, B. (2014a). Adaptive
Surface Visualization of Vessels with Animated Blood
Flow. Computer Graphics Forum.
Lawonn, K., Saalfeld, P., and Preim, B. (2014b). Illustra-
tive Visualization of Endoscopic Views. In Bildverar-
beitung f
¨
ur die Medizin (BVM), pages 276–281.
Mendis, S., Puska, P., Norrving, B., Organization, W. H.,
Federation, W. H., and Organization, W. S. (2011).
Global Atlas on Cardiovascular Disease Prevention
and Control. Nonserial Publications Series. World
Health Organization in collaboration with the World
Heart Federation and the World Stroke Organization.
M
¨
uller, M., Schirm, S., and Teschner, M. (2004). Interac-
tive Blood Simulation for Virtual Surgery Based on
Smoothed Particle Hydrodynamics. Technol. Health
Care, 12(1):25–31.
Naya, F., Contero, M., Aleixos, N., and Company, P. (2007).
ParSketch: A Sketch-based Interface for a 2D Para-
metric Geometry Editor. In Proc. of the 12th Inter-
national Conference on Human-computer Interaction:
Interaction Platforms and Techniques, HCI’07, pages
115–124. Springer-Verlag.
Neugebauer, M., Diehl, V., Skalej, M., and Preim, B.
(2010). Geometric Reconstruction of the Ostium of
Cerebral Aneurysms. In Proc. of VMV 2010 - Vision,
Modeling, Visualization, pages 307–314.
Pihuit, A., Cani, M.-P., and Palombi, O. (2010). Sketch-
based Modeling of Vascular Systems: A First Step
Towards Interactive Teaching of Anatomy. In Proc.
of the Seventh Sketch-Based Interfaces and Modeling
Symposium, SBIM ’10, pages 151–158.
Preim, B. and Dachselt, R. (2015, to appear). Interaktive
Systeme: User Interface Engineering, 3D-Interaktion,
Natural User Interfaces, volume 2. Springer.
Qin, J., Pang, W.-M., Nguyen, B. P., Ni, D., and Chui, C.-K.
(2010). Particle-based Simulation of Blood Flow and
Vessel Wall Interactions in Virtual Surgery. In Proc.
of the Symposium on Information and Communication
Technology, SoICT ’10, pages 128–133.
Stam, J. (1999). Stable fluids. In Proc. of the 26th An-
nual Conference on Computer Graphics and Interac-
tive Techniques, SIGGRAPH ’99, pages 121–128.
Sutherland, I. E. (1963). Sketchpad, A Man-Machine
Graphical Communication System. Outstanding Dis-
sertations in the Computer Sciences. Garland Publish-
ing, New York.
Taubin, G. (1995). Curve and Surface Smoothing Without
Shrinkage. In Proc. of the Fifth International Con-
ference on Computer Vision, ICCV ’95, pages 852–.
IEEE Computer Society.
Teitelbaum, G. P., Higashida, R. T., Halbach, V. V., Larsen,
D. W., McDougall, C. G., Dowd, C. F., and Hieshima,
G. B. (1994). Flow-directed Use of Electrolytically
Detachable Platinum Embolization Coils. Journal
of Vascular and Interventional Radiology, 5(3):453 –
456.
van Dam, A. (1997). Post-WIMP User Interfaces. Commun.
ACM, 40(2):63–67.
Wong, G. K., Tan, H.-B., Kwan, M. C., Ng, R. Y., Yu, S. C.,
Zhu, X.-L., and Poon, W.-S. (2011). Evolution of in-
tracranial aneurysm treatment: From Hunterian liga-
tion to the flow diverter. Surgical Practice, 15(1):16–
20.
Wu, E., Liu, Y., and Liu, X. (2004). An Improved Study
of Real-time Fluid Simulation on GPU: Research Ar-
ticles. Comput. Animat. Virtual Worlds, 15(3-4):139–
146.
Xu, X., Liu, W., Jin, X., and A, Z. S. (2002). Sketch-based
User Interface for Creative Tasks. In Proc. of 5th Asia
Pacific Conference on Computer Human Interaction,
pages 560–570.
Zhu, B., Iwata, M., Haraguchi, R., Ashihara, T., Umetani,
N., Igarashi, T., and Nakazawa, K. (2011). Sketch-
based Dynamic Illustration of Fluid Systems. In Proc.
of the 2011 SIGGRAPH Asia Conference, SA ’11,
pages 134:1–134:8. ACM.
GRAPP2015-InternationalConferenceonComputerGraphicsTheoryandApplications
390
