
An Ultrasonography Assisted Robotic HIFU Ablation Experimental
System
Ching Shiow Tseng
1
, Ja How Syu
1
, Chi Yu An
1
and Chih-Ju Chang
1,2
1
Department of Mechanical Engineering, National Central University, Jhongli, Taiwan
2
Department of Neurosurgery, Cathay General Hospital, Taipei, Taiwan
Keywords: High Intensity Focused Ultrasound, Ultrasound, Robotic, Image-guided.
Abstract: In recent years, noninvasive thermal treatment by using High Intensity Focused Ultrasound (HIFU) has
high potential in tumor treatment. The goal of this research is to develop an ultrasonography assisted
robotic HIFU ablation system for tumor treatment. The system integrates the technologies of ultrasound
image assisted guidance, robotic positioning control, and HIFU treatment planning. With the assistance of
ultrasound image guidance technology, the tumor size and location can be determined from ultrasound
images and the robot can be controlled to position the HIFU probe to focus on the target tumor. An
experiment of using mountain-typed template to verify the positioning accuracy of the ultrasonography
assisted robotic HIFU ablation system has been done. The results show that the average positioning error is
1.06mm with a standard deviation 0.25, which is feasible for tumor therapy.
1 INTRODUCTION
Liver tumor (cancer) is a common disease. Early
diagnosis and treatment of liver disease is very
important measures to avoid worsening. Except
biochemical tests such as GOT/GOP or α-globulin,
ultrasound scanning is usually adopted for first-line
screening and diagnosis. If the disease needs further
treatment, tissue biopsy, percutaneous ethanol
injection, or RF burning will be usually done under
ultrasound guidance. For serious cases, open or
minimally invasive liver dissection treatment will be
necessary. However, all of them are invasive
treatments. In recent years, noninvasive High
Intensity Focused Ultrasound (HIFU) thermal
treatment has demonstrated high potential in tumor
treatment (Martinez, et al., 2012). The physical
principle of this interventional approach is to apply
focused ultrasound waves to the tumor tissue such
that the heating of the tissue causes its necrosis (Seo,
et al., 2010). Since tumors are typically much larger
than the size of HIFU focal point, treatment of the
entire volume of tumor is not suitable for hand-held
HIFU transducer. Most of the research is conducted
with the assistance of robot arm (Masamune, et al.,
2013, Chauhan, 2008, Qiu, et al. 2009). Eventually,
it is quite difficult to assess the quality of this
non-invasive therapy, there is a dire need for a high
accuracy system supporting in planning, conduction,
and monitoring of such treatments. This research is
aimed to study and develop an ultrasound image
assisted robotic HIFU ablation system for tumor
treatment (Qiu, et al., 2009]. With the assistance of
ultrasound image guidance technology, ultrasound
images are used to determine tumor size and
location and the robot is controlled accordingly to
position the focus point of a HIFU probe to the
target position for thermal ablation of the tumor.
2 THE ULTRASONOGRAPHY
ASSISTED ROBOTIC HIFU
SYSTEM
As shown in Figure 1, the ultrasonography assisted
robotic HIFU system integrates the ultrasound
imaging system (ALOKA, Prosound Alpha 6), the
HIFU ablation system (Sonic H-106 probe with
Instek, GFG-8255 signal generator and AR,
150A100B power amplifier), the robotic arm
(YAMAHA, YK400XG), the optic tracker (Northern
Digital, Polaris Spectra), and a notebook (Dell,
M4500) into a system.
109
Tseng C., Syu J., An C. and Chang C..
An Ultrasonography Assisted Robotic HIFU Ablation Experimental System.
DOI: 10.5220/0005207601090114
In Proceedings of the International Conference on Biomedical Electronics and Devices (BIODEVICES-2015), pages 109-114
ISBN: 978-989-758-071-0
Copyright
c
2015 SCITEPRESS (Science and Technology Publications, Lda.)
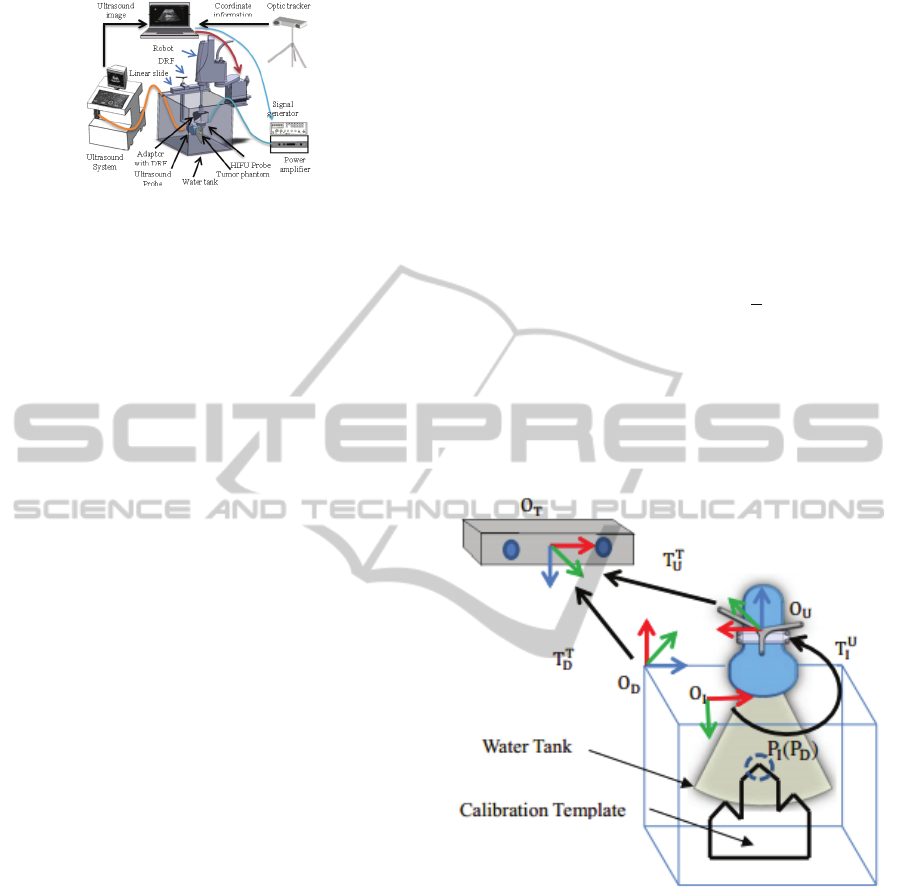
Figure 1: The ultrasonography assisted robotic HIFU
system.
The ultrasound probe scans the tumor phantom to
obtain the location of the tumor. The movement of
the ultrasound probe is controlled by the
motor-driven linear slide and detected by the optic
tracker through the DRF (Dynamic Reference Frame,
a tool with three IR reflective marker spheres),
which is a reference coordinate frame tracked by the
optic tracker. Through coordination transformation
described below, the position of the tumor phantom
relative to the ultrasound image frame can be
transferred and represented by the robot frame. The
robot is thus able to bring the focus point of the
HIFU probe to aim at the tumor phantom. The signal
generator and power amplifier are used to enable the
HIFU probe to generate high-intensity sound power
for thermal therapy.
3 COORDINATE
TRANSFORMTION BETWEEN
THE OPTIC TRACKER AND
THE ULTRASOUND IMAGE
FRAMES
Figure 2 illustrates an experimental system for
determining the coordinate transformation matrix
T
between the ultrasound probe frame (O
U
) and the
ultrasound image frame (O
I)
. A mountain-typed
calibration template with three plates is fixed on the
bottom of the water tank while a DRF (O
D
) is also
mounted on the upper corner of the water tank. The
position (P
D
) of the target point P (Figure 3) relative
to the tank DRF frame (D) is calibrated prior to the
experiment. A DRF (O
U
) is also attached on the
ultrasound probe for position tracking of the probe.
As shown in Figure 3, the middle plate of the
calibration template is scanned by the ultrasound
probe and the image coordinate (P
I
) of the target
point P is determined from the ultrasound image.
The position of the target point P relative to the optic
tracker frame can be expressed through either the
tank DRF frame or the ultrasound probe frame as
shown in equation (1).
T
P
=T
T
P
(1)
where I represents ultrasound image frame
U represents ultrasound probe frame
T represents optic tracker frame
D represents the tank DRF frame
T
, T
, P
and P
are known.
The transformation matrix T
can be
determined by bringing the tracker and image
coordinates of the target point P at three or more
positions, P
i
(P
、P
), i=1, 2,…N, N>3 into equation
(1) and solved by optimization method such as the
least square algorithm. After the transformation
matrix T
has been determined, the coordinates of
any target tumor detected by ultrasound scan (Figure
5) can be transferred and expressed relative to the
optic tracker frame as described by equation (2).
P
=T
T
P
(2)
Figure 2: The coordinate transformation among all frames
for the registration of the optic tracker and the ultrasound
image.
The calibration template used for the registration
of the optic tracker frame and the ultrasound image
frame is shown in Figure 3. Since ultrasound scan
beam has a slice thickness (elevational direction), it
is necessary to determine the middle plane of the
slice so that the following positioning calibration
will be more precise. Therefore, a three-layer
template is designed to make sure that the ultrasound
scan is correctly located on the middle plate which
will have brighter or clear boundary images than the
other two plates.
BIODEVICES2015-InternationalConferenceonBiomedicalElectronicsandDevices
110

Figure 3: The calibration template and the boundary image
of the middle plate, which is clearer than those of the other
two plates.
4 COORDINATE
TRANSFORMATION
BETWEEN THE ROBOT AND
THE ULTRASOUND SYSTEM
Figure 4 shows the coordinate transformation
relationship between the optic tracker and the robot.
A tracking device mounted with a DRF (coordinate
frame E) and a pin of 10cm in length (tip point P
represents the focus point of the HIFU transducer) is
designed and mounted at the end effector of the
robot. A DRF is fixed on the robot base and used to
define the world coordinate frame W in case the
optic tracker is moved during the experiment. The
robot coordinate frame is defined as frame R. The
transformation matrix T
and T
can be
determined directly by the optic tracker. The
transformation matrix T
is to be solved so that the
coordinates relative to the optic tracker frame can be
transformed relative to the robot frame. In other
words, the coordinates of any target point
determined by the ultrasound scan can be
transformed to those relative to the robot frame
through the optic tracker.
The position of the origin of the coordinate frame
E, O
E
, can be described relative to the coordinate
frame W as below.
O
=T
T
O
(3)
If the robot is manipulated to move around, the
coordinates of point O
E
relative to the coordinate
frames R and W are calculated by the robot
controller and equation (3) respectively. Therefore,
the transformation matrix T
between the robot
and optic tracker can be determined by equation (4).
O
=T
O
(4)
Because both O
and O
are not square matrices
(4x1), we use least mean square algorithm to solve
T
.
T
=O
O
O
O
(5)
After completion of the registrations between the
ultrasound image and the optic tracker and between
the optic tracker and the robot, the coordinates of the
target tumor scanned and detected by the ultrasound
system can be transformed and represented by the
robot frame. The transformation is defined by Eq. (6)
and illustrated by Figure 5.
P
=T
T
T
T
P
(6)
where P
: Image coordinate of the target
tumor
Figure 4: The coordinate transformation between the optic
tracker and the robot.
Figure 5 also shows that the HIFU transducer has
been mounted to the end effector of the robot for
HIFU thermal treatment.
Figure 5: The coordinate transformation between the
tumor P
I
and the robot.
AnUltrasonographyAssistedRoboticHIFUAblationExperimentalSystem
111

The procedures of the HIFU thermal ablation of the
ultrasonography assisted robotic HIFU system is
described in the flow chart of Figure 6.
Figure 6: The procedure of thermal ablation of the robotic
HIFU system.
5 EXPERIMENT AND
DISCUSSION
5.1 Position Measure of the Target
An experiment has been conducted to verify the
position measure error through the coordinate
transformation between the ultrasound image and
the optic tracker frames. The mountain-type
template was seated in depth of 3cm, 7cm and 12cm.
The template in each depth was scanned three times
by the
ultrasound probe. The positioning error is
defined as the difference between the image
coordinate of the target point after coordinate
transformation and the coordinate measured directly
by the optic tracker. The average positioning error of
the three peak points of the template in depth of 3cm,
7cm and 12cm are 1.49mm, 1.46mm and 2.15mm
respectively. Table 1 listed the experiment data of
the case in 7cm depth.
5.2 Positioning of the Robot Arm
The robot was commanded to move around to ten
positions to calculate the transformation matrix
T
by Eq. (5). After that, the calibration template of
Figure 3 seated in depth 7cm was scanned by the
ultrasound. Then the robot was command to move
the pinpoint P (Figure 4) of the rod to the three peak
points of the template as shown in Figure 7. The
distance errors between the peak points and the
pinpoint P are listed in Table 2. The positioning error
is 1.06±0.25mm.
Figure 7: The pinpoint of the rod positions to the peak
point of the calibration template.
Table 1: Distance error of the phantom in 7cm depth.
No. of
point
Coordinate of
the target point
No. of
Image
Coordinates of
the guided pinpoint
Distance error
x y z x y z
1 38.9 27.9 -89.5
I 39.1 28.0 -90.5 -0.24
II 39.1 28.0 -90.7 -0.17
III 39.5 27.8 -90.5 -0.62
2 18.3 14.0 -87.7
I 18.0 13.6 -88.1 0.27
II 17.4 13.8 -88.5 0.83
III 18.1 13.9 -88.1 0.18
3 13.7 -0.1 -104.5
I 14.0 -0.5 -105.4 -0.28
II 14.2 -0.2 -105.5 -0.46
III 14.0 -0.0 -105.8 -0.29
Max error 1.38 Average error 1.01 Standard deviation 0.26
BIODEVICES2015-InternationalConferenceonBiomedicalElectronicsandDevices
112

Table 2: Positioning error of the robot arm.
No. of
point
Coordinate of
the target point
No. of
Image
Coordinate of
the guided pinpoint
Distance error
x y z x y z
1 38.9 27.9 -89.8
I 39.2 27.8 -90.6 1.04
II 39.2 27.9 -90.6 1.10
III 39.6 27.7 -90.5 1.16
2 18.3 14.0 -87.7
I 18.1 13.5 -88.2 0.79
II 17.5 13.7 -88.5 1.19
III 18.2 13.8 -88.1 0.50
3 13.7 -0.1 -104.5
I 14.0 -0.6 -105.5 1.16
II 14.2 -0.4 -105.6 1.23
III 14.0 -0.1 -105.9 1.40
Max error 1.4 Average error 1.06 Standard deviation 0.25
Table 3: Positioning error of HIFU thermal ablation.
No. of
points
Position of the target (mm) Position of the ablation (mm) Distance error
X Y Z X Y Z
1 -71.6 248.6 79.7 -71.4 248.2 80.0 0.5
2 -70.6 289.2 81.2 -72.6 288.6 81.1 2.1
3 -112.0 248.4 79.63 -111.0 248.1 79.7 1.0
4 -109.0 289.5 81.24 -108.0 288.7 80.3 1.6
5.3 Positioning of HIFU Ablation
The ultrasonography assisted robotic HIFU
treatment experiment was conducted by
commanding the robot to move HIFU focus point to
ablate the four corner points of a phantom, which
was detected by ultrasound images. Figure 8 shows
the HIFU focus point can be positioned to the target
(corner) points for thermal ablation. The average
positioning error is 1.3± 0.8mm and the distance
error of each corner point is listed in Table 3.
Figure: 8: Positioning experiment of the HIFU thermal
ablation.
6 CONCLUSIONS
This study proposes an ultrasonography assisted
robotic HIFU system for thermal ablation of tumor.
The position coordinates of targets determined by
the ultrasound image are transformed to the robot
coordinate frames so that the robot can move the
HIFU probe to focus on the targets. The experiment
results show that positioning errors of the robotic
HIFU system is accurate enough for thermal ablation
treatment of tumor tissue. Since robots have great
dynamic response in motion, it is highly possible to
apply the robotic HIFU system to treat live tumors in
the future, which requires the compensation of
movement due to respiration.
ACKNOWLEDGEMENTS
The authors would like to thank for the financial
support from Ministry of Science and Technology,
Taiwan, under the contract of NSC 101-2221-E-008-
020-MY3.
The locations of thermal ablation
The corner points of the phantom
AnUltrasonographyAssistedRoboticHIFUAblationExperimentalSystem
113

REFERENCES
Masamune, K., Kurima, I., Kuwana, K., et al., 2013, HIFU
Positioning Robot for Less-Invasive Fetal Treatment,
Procedia CIRP, vol. 5, , pp. 286-289.
Chauhan, S., 2008, FUSBOTs: Image-guided Robotic
Systems for Focused Ultrasound Surgery, Medical
Robotics, Vanja Bozovic, ISBN 978-3-902613-18-9,
I-Tech Education and Publishing, Vienna, Austriap,
pp.526.
Chauhan, S. Tan, M.T., Seet, G., et al., 2009, Minimally
invasive robotic HIFU neurosurgical applications, in
38th Annual Symposium of the Ultrasonic Industry
Association (UIA), , Vancouver, BC, pp. 1-5.
Qiu, Z. Gao, J., Cochran, S., et al., 2009, The development
of therapeutic ultrasound with assistance of robotic
manipulator,” Proc IEEE Eng Med Biol Soc, pp.
733-736.
Seo, J., Koizumi, N., Yoshinaka, N., et al., 2010,
Three-dimensional computer-controlled acoustic
pressure scanning and quantification of focused
ultrasound, IEEE Trans Ultrason Ferroelectr Freq
Control, vol. 57, pp. 883-891.
Martinez, R., Vera, A. and Leija, L., 2012, High-intensity
focused ultrasound thermal mapping by using a
thermocouple embedded in a tissue-mimicking
material, Electrical Engineering, Computing Science
and Automatic Control (CCE), 9th International
Conference, pp. 1-4.
Soneson, J.E., 2009, A User-Friendly Software Package
for HIFU Simulation, AIP Conference Proceedings,
Vol. 1113 Issue 1, p165.
Chauhan, S., 2008, Image-guided Robotic Systems for
FUS (Focused Ultrasound Surgery), Vanja Bozovic,
ISBN 978-3-902613-18-9, I-Tech Education and
Publishing, Vienna, Austriap, pp.526.
Qiu, Z., Gao, J., Cochran, S., et al. 2009, The development
of therapeutic ultrasound with assistance of robotic
manipulator, Proc IEEE Eng Med Biol Soc, pp.
733-736.
Hill, C.R., Rivens, L., Vaughan, M.G., et al., 1994, Lesion
development in focused ultrasound surgery: A
general model, Ultrasound in Medicine & Biology,
vol. 20, pp. 259-269.
Takegami, K., Kaneko, Y., Watanabe, T., et al., 2004,
Polyacrylamide gel containing egg white as new
model for irradiation experiments using focused
ultrasound," Ultrasound Med Biol, vol. 30, pp.
1419-1422.
Tung, Y.S., Liu, H.L., Wu, C.C., et al., 2006,
Contrast-agent enhanced ultrasound thermal
ablation," Ultrasound Med Biol, vol. 32, pp.
1103-1110.
Luo, H., Shen, G., and Chen, Y., 2009, Treatment Planning
of Scanning Time and Path for Phased High-Intensity
Focused Ultrasound Surgery, in Biomedical
Engineering and Informatics, BMEI '09. , pp. 1-4.
Vaezy, S., Shi, X., Martin, R.W., et al., 2001, Real-time
visualization of high-intensity focused ultrasound
treatment using ultrasound imaging, Ultrasound in
Medicine & Biology, vol. 27, pp. 33-42.
Sakuma, I., Takai, Y., Kobayashi, E., et al., 2002,
Navigation of High Intensity Focused Ultrasound
Applicator with an Integrated Three-Dimensional
Ultrasound Imaging System, MICCAI, 5th
International Conference, vol. 2489, pp. 133-139.
BIODEVICES2015-InternationalConferenceonBiomedicalElectronicsandDevices
114
