
Application of a MEMS Blood Flowmeter for Power Spectrum
Analysis of Heart Rate Variability
Terukazu Akiyama
1
, Tatsuya Miyazaki
1
, Hiroki Ito
1
, Hirofumi Nogami
2
and Renshi Sawada
1,2
1
Graduate School of Systems Life Sciences, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka-city, Japan
2
Department of Mechanical Engineering, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka-city, Japan
Keywords: MEMS Blood Flowmeter, Heart Rate Variability, Autonomic Nervous System.
Abstract: We investigated the possibility of applying a MEMS blood flowmeter to heart rate variability (HRV)
analysis. We conducted simultaneous measurements of HRV by electrocardiogram and MEMS blood
flowmeter. TPP for the MEMS blood flowmeter was defined as the interval between peaks, which were
designated as where the first-order differential of the signal changes from negative to positive. TRR (i.e., the
R-R interval of the electrocardiogram) and TPP were compared by regression analysis. Autonomic indices
transformed by power spectrum analysis were also compared by regression analysis. Fast Fourier transform
(FFT) and maximum entropy method (MEM) were employed in the frequency analysis. By FFT analysis,
the coefficient of determination for the regression between LF%, HF%, and LF/HF derived by TRR versus
TPP was 0.8781, 0.8781, and 0.8946, respectively. By MEM analysis, the coefficient of determination for
the regression between LF%, HF%, and LF/HF derived by TRR versus TPP was 0.9649, 0.8026, and
0.9181, respectively. These high correlations suggest that the TPP of the MEMS blood flowmeter is a
reliable metric that can be utilized in applications of HRV analysis.
1 INTRODUCTION
The autonomic nervous system plays key roles
throughout the body, including the control of
involuntary activities such as blood circulation,
respiration, digestion, regulation of body
temperature through perspiration, endocrine
function, generative function, and metabolism. One
noteworthy example is the pumping of the heart,
which is caused by periodic excitement of the
sinoatrial node, a rhythm that typically fluctuates.
Fluctuation of heartbeat rhythm is known as heart
rate variability (HRV), and it reflects the health of
the cardiovascular autonomic nervous system.
Research into HRV began in dogs in 1981 using
quantitative spectral analysis with the FFT, and it
has since been studied in humans (Akselrod et al.,
1981, Pomeranz et al., 1985). The prospect that
HRV might be measurable more easily without the
need for large equipment attracted much attention
within the information engineering and biomedical
engineering fields in the 1990s, with advances in
wireless networks and miniaturization of biological
sensors at that time informing attitudes that
considered the prospect feasible. As the autonomic
nervous system innervates many organs, symptoms
of its dysfunction are observed in various diseases,
and recordings of parameters it controls may be
useful for studying the occurrence, pathogenesis,
response to treatment, and prognosis of a multitude
of diseases. While many clinical applications of
HRV analysis have been reported, it has been most
accurately used as a clinical marker of diabetic
neuropathy, as well as a predictive factor for the risk
of sudden cardiac death and the occurrence of
arrhythmias after acute myocardial infarction (Stys
et al., 1998). Moreover, it has been employed to
identify neurological diseases and to confirm the
effects of therapy and medicine targeting them.
Because of the wide range of disease factors
(pathology, prognosis, etc.) that are related to
autonomic nervous system function and dysfunction,
it is a highly regarded health metric in many fields.
However, symptoms of its dysfunction are more
difficult to capture via short-term measurements
compared with other health indices; indeed, in cases
where its function fluctuates based on circadian
changes (e.g., temperature regulation, where body
temperature is cooler during the nighttime), accurate
evaluation becomes difficult because of the need to
211
Akiyama T., Miyazaki T., Ito H., Nogami H. and Sawada R..
Application of a MEMS Blood Flowmeter for Power Spectrum Analysis of Heart Rate Variability.
DOI: 10.5220/0005213302110218
In Proceedings of the International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS-2015), pages 211-218
ISBN: 978-989-758-069-7
Copyright
c
2015 SCITEPRESS (Science and Technology Publications, Lda.)

collect data through continuous monitoring
throughout a subject’s daily activities. Autonomic
nervous system function reacts to situation and
environment in real-time to maintain homeostasis.
Early detection of symptoms of its dysfunction may
be possible by analyzing reactions to return the body
to homeostasis. For example, in the case of diabetes,
the body would be slow to release insulin in
response to high blood sugar; pancreatic
neurological dysfunction could be reflected in HRV.
If these data were accessible by medical institutions
that provide health and medical services, better and
more-effective service could likely be provided.
To this end, we have recently developed a
perhaps more widely applicable solution: a micro-
integrated laser Doppler blood flowmeter (micro-
electromechanical system (MEMS) blood flow
sensor), which is 1/300
th
the volume of a
conventional laser Doppler blood flowmeter
(Higurashi et al., 2003, Kimura et al., 2010). The
weight of this sensor is approximately 1/30
th
of a
conventional instrument, and its power consumption
is only 1/20
th
. The instrument also makes blood flow
measurements possible while subjects are moving by
omitting the optical fiber that is the source of
significant noise in conventional instruments. This
makes MEMS blood flowmetry ideal for the
continuous measurement of blood flow volume over
time. Moreover, since the blood flow data obtained
by MEMS blood flowmetry are a result of analyzing
the skin sympathetic nerves, blood pressure, and
body temperature, MEMS blood flowmetry is
capable of performing multivariate analysis of many
indices, a significant advantage over single-index
methods in the efforts to understand the complex
systems of the living body.
The R-wave of the electrocardiogram is one
candidate parameter often used in HRV analysis to
assess autonomic function. Several commercial
products on the market today allow ECG
measurements during training/movement with an
acceptable accuracy. However, we believe that a
MEMS blood flowmeter is advantageous for
evaluation of the autonomic nervous system for two
reasons. One reason is that a MEMS blood
flowmeter is a small and simple system and, unlike
the ECG, does not require operator skill to affix it in
order to obtain pulse. Another reason is its ready
applicability to multivariable analysis for the
evaluation of autonomic nervous function. If
autonomic nervous system-based changes in pulse
obtained from the pulse waveform of blood flow
volume can be accurately determined, both simple
measurement and further detailed evaluation of
biological state would be possible, which would be a
boon to applications in healthcare and medical
treatments.
At present, R-wave (obtained from
electrocardiography) and pulse (obtained from
acceleration plethysmography) data are typically
used in HRV analysis (Takada et al., 2008). To
confirm whether HRV analysis is possible from the
pulse data obtained from the blood flow volume
waveform of MEMS blood flowmetry, we
performed simultaneous measurement of the R-R
interval (TRR) obtained from an electrocardiogram
and the pulse wave interval (TPP) obtained from a
MEMS blood flowmeter for 100 beats, and then
investigated the relationship between TRR and TPP
by regression analysis (Fig. 1). Furthermore, we
compared the accuracy of the frequency analysis of
the fast Fourier transform (FFT) versus the
maximum entropy method (MEM) for the HRV
signal over 100 beats.
Figure 1: (a) Photograph of the MEMS blood flowmeter;
(b) Measurement principle and schematics of its optical
MEMS chip.
PD: photodiode, DFB-LD: distributed feedback-laser
diode.
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
212

2 MATERIALS AND METHODS
In this section, we provide information on the study
subjects, MEMS blood flowmeter, data analysis, and
experimental method.
2.1 Subjects
The investigation complied with the ethical
standards of the committee responsible for human
experimentation of Kyushu University and with the
Helsinki Declaration of 1964 as revised in 2008. A
total of 6 healthy volunteers (6 males, age range:
21–26 years) participated in this study.
2.2 MEMS Blood Flowmeter
The MEMS blood flowmeter employed in the study
is a blood flowmeter that is integrated with MEMS
technology. Its features of ultra-compactness,
lightweight, low power consumption, and wireless
function make it possible to always be attached to
the body. In addition, since its design omits the
optical fiber that causes significant noise in
conventional blood flowmeters, peripheral blood
flow is measurable while the subject is in motion.
2.2.1 Structure
Figure 1 shows a schematic of the MEMS blood
flowmeter. The MEMS blood flowmeter consists of
the main body and the probe. The main body has a
digital signal processor (DSP), a Bluetooth wireless
link, a display, and a battery. The probe has a sensor
chip and an amplifier circuit. The sensor chip
consists of two crystal silicon substrates, a DFB-LD
(distributed feedback-laser diode) and a PD
(photodiode). One of the silicon substrates contains
two cavities that hold the bonded LD and PD, while
the other contains a microlens and a hole. Gold
etched into the cavity leads the laser beam from the
LD into the microlens. The antireflection-coated
microlens optimizes the laser beam from the LD.
Another silicon substrate is used to seal the cavity.
2.2.2 Principle
The design of the MEMS laser Doppler blood
flowmeter is based on the principle of a laser
Doppler flowmeter (Bonner et al., 1981). The
measurement object is irradiated with a laser beam.
Incident light is scattered multiply by static tissue,
such as a skin and blood vessels, and moving tissue,
such as red blood cells. All light scattered back
Figure 2: Data analysis process.
toward the PD interferes on the PD to form a speckle
pattern. Statistical analysis of the output signal of the
PD with respect to the speckle pattern gives a
relative blood flow.
2.3 Data Analysis
Figure 2 is a flowchart of the data analysis process.
The signal obtained by the MEMS blood flowmeter
is detrended, and smoothing is performed by the
moving average and first-order differential. We
obtain the pulse by detecting the peak of the signal.
The pulse is smoothed by the moving average and
Figure 3: Waveforms of the electrocardiogram and the
MEMS blood flowmeter.
ApplicationofaMEMSBloodFlowmeterforPowerSpectrumAnalysisofHeartRateVariability
213

third-order spline interpolation. Frequency analysis
by the FFT or MEM is performed to obtain the
power spectrum of HRV. Finally, we derive
autonomic indices from the HRV spectrum (details
of index calculations below).
2.3.1 Peak Detection
The signal of the laser Doppler flowmeter is
influenced by peripheral blood flow, which is in turn
influenced by many factors such as blood vessel
conditions, body temperature, vasoconstriction, and
vasodilation by skin sympathetic nerves, vascular
endothelial cell metabolism, respiration, and blood
pressure. In addition, changes in peripheral blood
flow due to heartbeat are smaller than arterial blood
flow. Therefore, we anticipated that the detection of
the signal peak would be difficult compared with
analyses of the clear waveforms in
electrocardiograms and plethysmographs. Methods
of detecting the peak of the pulse waveform may
define the peak as the minimum, maximum, or point
of 10% of the minimum within the waveform. There
are also peak detection methods that instead
emphasize change in the signal in terms of the first
order-differential and second-order differential.
When the latter strategy is applied to peripheral
blood flow, the first-order differential and the
second-order differential may indeed be suitable for
functioning as a high-pass filter for detecting
changes in the signal trend. However, we assumed
that peaks could not be detected more stably with
reference to other, higher-frequency signals in
peripheral blood flow more reliably than simply with
reference to the zero order peak. Therefore, we
consider that the optimum pre-filtering strategy is to
remove only high-frequency fluctuations before the
first-order differential and the second-order
differential. By the above logic, in this study we
decided to perform a first-order differential after
smoothing by the moving average, and detected
peaks as when the signal changed from negative to
positive (Figure 3).
2.3.2 Heart Rate Variability Analysis
Heart rate is controlled by the electrical impulses of
the sinoatrial node, which are in turn controlled by
the central nervous system through the sympathetic
and parasympathetic branches of the autonomic
nervous system. Sympathetic nerves increase heart
rate by accelerating the electrical impulses of the
sinoatrial node, while parasympathetic nerves
decrease heart rate by suppressing them. The central
nervous system also controls various functions based
on the information sent from the sensory organs.
Therefore, fluctuations that are generated by the
body modulate heart rate through the autonomic
nervous system via the central nervous system. Of
particular relevance to our study is that fluctuations
in blood pressure—i.e., the Mayer wave, over a 10-
second period—and respiration cycle are transmitted
by this route to modulate heart rate. An important
fact that can be leveraged for analysis is that the
frequency responses of sympathetic nerves and
parasympathetic nerves are different: about 0.15 and
1 Hz, respectively. As a consequence, although
sympathetic and parasympathetic nerves work
antagonistically in general, heart rate is modulated
by fluctuations in blood pressure (i.e., the 10-s
Mayer wave) when it receives input from the former,
but by fluctuations in both blood pressure and
respiration when it receives input from the latter
(Penaz, 1978). Therefore, it is possible to evaluate
the relative strength of sympathetic and
parasympathetic function on heart rate by frequency
analysis of heart rate fluctuation. These two indices
are defined as follows from each interval:
Low-frequency component (LF) : 0.04-0.15
Hz of power spectral density
High-frequency component (HF) : 0.15-0.40
Hz of power spectral density
Thus, it is possible to evaluate the relative
sympathetic function by calculating LF/HF. In
addition, the HF estimates the parasympathetic
function. However, since the HF is a relative value,
it must be normalized by the total power to be of
analytical use.
2.4 Experimental Method
In order to compare the TPP obtained by MEMS
blood flow sensor and TRR obtained by
electrocardiogram, simultaneous measurement with
conventional electrocardiogram and MEMS blood
flowmeter was conducted on six healthy volunteers
(6 males). The experimental protocol is based on the
method of measuring blood flow as follows: after
attaching the conventional electrocardiogram and
MEMS blood flowmeter to the subject’s body, we
provided a break-time of 10 minutes, after which
heart rate was continuously measured for 100 beats.
We conducted experimental measurements in the
sitting position throughout the experiment. The
MEMS blood flowmeter was attached to the right
index finger. The right hand was fixed at the vertical
level of the heart to prevent it from moving.
Measurements of peripheral blood flow are
deformed by changing the measurement site because
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
214
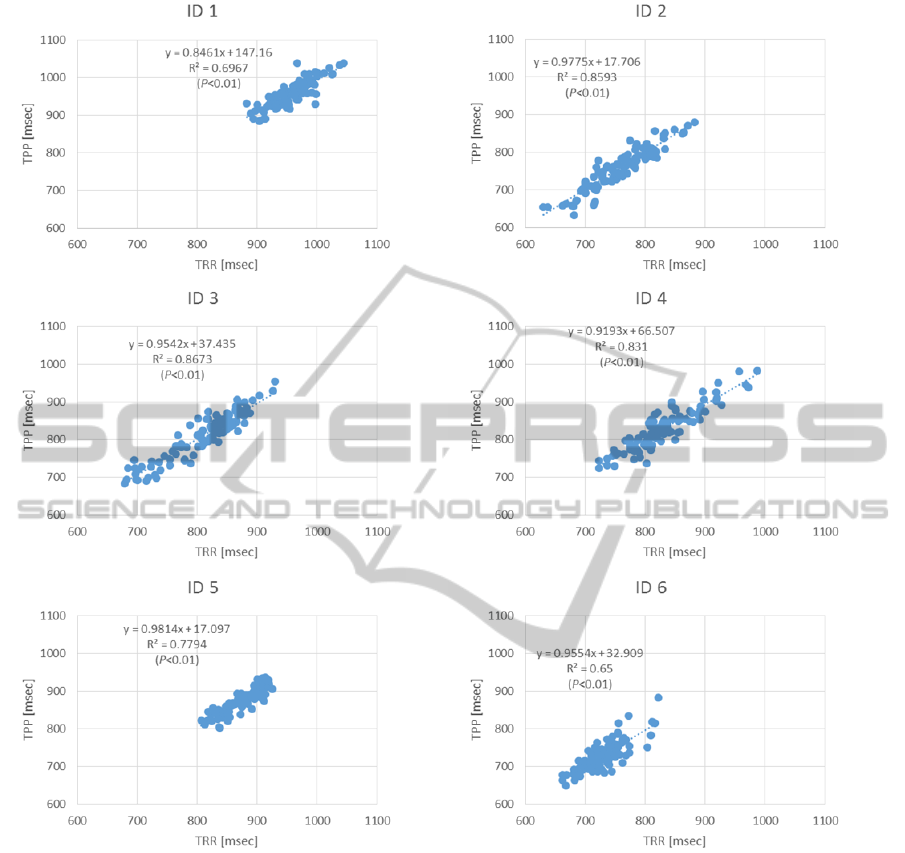
Figure 4: Relationship between the TRR of the electrocardiogram and the TPP of the MEMS blood flowmeter by regression
analysis.
TRR: R-R interval obtained from electrocardiogram, TPP: pulse wave interval obtained from MEMS blood flowmeter.
the density and shape of blood vessels differ across
measurement sites. Therefore, the experiment was
conducted with the flowmeter attached to the same
site throughout. In addition, since contact pressure
by the flowmeter compresses blood vessels and thus
changes peripheral blood flow, the MEMS blood
flowmeter was lightly fixed with double-sided tape.
Furthermore, since peripheral blood flow is altered
by skin temperature, room temperature was held at
constant 25 °C throughout the experiment. The LF
and HF components of the TRR and TPP spectra
were defined as 0.04–0.15 Hz and 0.15–0.40 Hz of
power spectral density, respectively. From the power
spectral density obtained from TRR and TPP, LF%
and HF% (normalized to total power) in addition to
LF/HF were calculated.
3 RESULTS
Figure 4 shows the correlations between TRR and
TPP for each subject. The regression equation and
the coefficient of determination obtained by
regression analysis are also shown. For subjects
ID1–6, the slope was 0.8461, 0.9775, 0.9543,
0.9193, 0.9814, and 0.9554, and the intercept was
ApplicationofaMEMSBloodFlowmeterforPowerSpectrumAnalysisofHeartRateVariability
215

Figure 5: Relationships of LF%, HF%, and LF/HF as calculated by TRR (electrocardiogram) versus by TPP (MEMS blood
flowmeter) by regression analysis. Each dot in the graphs represents a data point from one individual. A total of 6 healthy
male volunteers (age range: 21–26 years) participated in this study. FFT frequency analysis was performed for TRR data
(left side); MEM frequency analysis was performed for TPP data (right side).
LF%: proportion of power spectral density in the low-frequency domain of HRV, HF%: proportion of power spectral
density in the high-frequency domain of HRV, LF/HF: ratio of LF and HF power spectral density, FFT: fast Fourier
transform, MEM: maximum entropy method.
147.16, 17.706, 37.435, 66.507, 17.097, and 32.909,
respectively, with the corresponding coefficients of
determination being 0.6967, 0.8593, 0.8673, 0.831,
0.7794, and 0.65 (p<0.01).
Figure 5 shows the relationships between LF%,
HF%, and LF/HF, as determined by frequency
analysis using FFT versus MEM to analyze TRR
and TPP (respectively). The coefficients of
determination and the regression equation obtained
by regression analysis are also shown.
According to FFT analysis, the slope between
TRR and TPP for LF%, HF%, and LF/HF was
0.7053, 0.7053, and 0.2753, respectively; the
coefficient of intercept was 0.15, 0.1447, and
1.0866. The coefficient of determination was
0.8781, 0.8781, and 0.8946, respectively.
According to MEM analysis, the slope between
TRR and TPP for LF%, HF%, and LF/HF was
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
216

0.8019, 0.7933, and 0.2827, respectively; the
coefficient of intercept was 115975, 172243, and
0.9977. The coefficient of determination was
0.9649, 0.8026, and 0.9181, respectively.
4 DISCUSSION
We conducted a comparison of the RR interval
obtained by an electrocardiogram and the pulse
interval obtained from the pulse signal of our
MEMS blood flowmeter. Coefficients of
determination showed a strong relationship between
the two, and ranged from 0.65–0.87 in the regression
analysis. In addition, autonomic indices obtained via
the former metric showed a strong relationship with
autonomic indices obtained via the latter, with
coefficients of determination ranging from 0.80–
0.96. The MEMS blood flowmeter measures the
pulse signal transmitted from the heart to peripheral
blood vessels. Many factors modulate this pulse
signal. For example, blood vessel structure changes
due to low-frequency fluctuation of skin sympathetic
nerves and vascular endothelial cells. These skin
sympathetic nerves also cause short-term expansion
and contraction of blood vessels. Strong correlation
of blood flow signal modulation with the
electrocardiogram TRR metric was also observed in
this experiment. These results suggest high promise
for evaluating autonomic function and heart rate
variability from the pulse interval of the pulse signal
of MEMS blood flowmeter.
In the future, we will determine whether the
MEMS blood flowmeter can measure relative
changes in autonomic indices induced by
physiological or psychological stressors.
5 CONCLUSIONS
The feasibility of using a MEMS blood flowmeter to
evaluate autonomic function by heart rate variability
was evidenced by comparable findings with
electrocardiogram and pulse waveform data.
Frequency analysis of each signal fluctuation of 100
consecutive heartbeats by the FFT and MEM was
performed, followed by comparison of the resultant
autonomic nervous indices by regression analysis.
We confirmed a strong relationship between
MEMS- and electrocardiogram-derived indices from
a comparison of autonomic function indices and
pulse. We also confirmed the strong relationship
between the electrocardiogram and MEMS blood
flowmeter with power spectrum analysis by both
FFT and MEM. Although MEM has a frequency
resolution higher than the FFT, the FFT also has
comparable ability to analyze data at least to the
extent of the 100 consecutive heartbeats monitored
in this study. The signal obtained by the MEMS
blood flowmeter reflects many indicators such as
skin sympathetic nerve activity (Söderström et al.,
2003), body temperature, and blood pressure, and, to
date, was successfully applied to the evaluation of
hemodynamics during exercise and the detection of
signs of dehydration (Nogami et al., 2011) and
alcohol ingestion (Iwasaki et al., 2012). As
autonomic function evaluation by the MEMS blood
flowmeter becomes more and more feasible in the
future, its applications are expected to grow in a
wide range of fields.
ACKNOWLEDGEMENTS
We would like to express out our special thanks to
the volunteers who participated in the study.
REFERENCES
Akselrod, S., Gordon, D., Ubel, F. A., Shannon, D. C.,
Berger, A. C., & Cohen, R. J. (1981). Power spectrum
analysis of heart rate fluctuation: a quantitative probe
of beat-to-beat cardiovascular control. science,
213(4504), 220-222.
Pomeranz, B., Macaulay, R. J., Caudill, M. A., Kutz, I.,
Adam, D., Gordon, D. A. V. I. D., ... & Cohen, R. J.
(1985). Assessment of autonomic function in humans
by heart rate spectral analysis. American Journal of
Physiology-Heart and Circulatory Physiology, 248(1),
H151-H153.
Stys, A., & Stys, T. (1998). Current clinical applications
of heart rate variability. Clinical cardiology, 21(10),
719-724.
Higurashi, E., Sawada, R., & Ito, T. (2003). An integrated
laser blood flowmeter. Journal of lightwave
technology, 21(3), 591.
Kimura, Y., Goma, M., Onoe, A., Higurashi, E., &
Sawada, R. (2010). Integrated laser Doppler blood
flowmeter designed to enable wafer-level packaging.
Biomedical Engineering, IEEE Transactions on,
57(8), 2026-2033.
Takada, M., Ebara, T., & Sakai, Y. (2008). The
acceleration plethysmography system as a new
physiological technology for evaluating autonomic
modulations. Health evaluation and promotion, 35(4),
373-377.
ApplicationofaMEMSBloodFlowmeterforPowerSpectrumAnalysisofHeartRateVariability
217

Bonner, R., & Nossal, R. (1981). Model for laser Doppler
measurements of blood flow in tissue. Applied optics,
20(12), 2097-2107.
Fredriksson, I., Fors, C., & Johansson, J. (2007). Laser
doppler flowmetry-a theoretical framework.
Department of Biomedical Engineering, Linköping
University.
Rowell, L. B. (1986). Human circulation: regulation
during physical stress (p. 215). Oxford: Oxford
University Press.
Smith, J. J. (1990). Circulatory response to the upright
posture (No. 6). CRC Press.
Yamamoto, Y., & Hughson, R. L. (1991). Coarse-graining
spectral analysis: for studying heart rate variability.
Penaz, J. (1978). Mayer waves: history and methodology.
Automedica, 2(3), 135-142.
Söderström, T., Stefanovska, A., Veber, M., & Svensson,
H. (2003). Involvement of sympathetic nerve activity
in skin blood flow oscillations in humans. American
Journal of Physiology-Heart and Circulatory
Physiology, 284(5), H1638-H1646.
Nogami, H., Iwasaki, W., Abe, T., Kimura, Y., Onoe, A.,
Higurashi, E., ... & Sawada, R. (2011). Use of a simple
arm-raising test with a portable laser Doppler blood
flow meter to detect dehydration. Proceedings of the
Institution of Mechanical Engineers, Part H: Journal
of Engineering in Medicine, 225(4), 411-419.
Iwasaki, W., Nogami, H., Ito, H., Gotanda, T., Peng, Y.,
Takeuchi, S., ... & Sawada, R. (2012). Useful method
to monitor the physiological effects of alcohol
ingestion by combination of micro-integrated laser
Doppler blood flow meter and arm-raising test.
Proceedings of the Institution of Mechanical
Engineers, Part H: Journal of Engineering in
Medicine, 226(10), 759-765.
Ogata, H., Fujimaru, I., Yamada, K., & Kondo, T. (2012).
Suppression of cardiocirculatory responses to
orthostatic stress by passive walking-like leg
movement in healthy young men. J Physiol Anthropol,
31, 24.
Hess, W. R. (1954). Diencephalon, autonomic and
extrapyramidal functions (Vol. 3). Grune & Stratton.
Ohhashi, T., Sakaguchi, M., & Tsuda, T. (1998). Human
perspiration measurement. Physiological
measurement, 19(4), 449.
Homma, S., Nakajima, Y., Toma, S., Ito, T., & Shibata, T.
(1998). Intracerebral source localization of mental
process-related potentials elicited prior to mental
sweating response in humans. Neuroscience letters,
247(1), 25-28.
Tanaka, H., Yamaguchi, H., Matushima, R., & Tamai, H.
(1999). Instantaneous orthostatic hypotension in
children and adolescents: a new entity of orthostatic
intolerance. Pediatric research, 46
(6), 691-691.
Low, P. A., Opfer-Gehrking, T. L., Textor, S. C.,
Benarroch, E. E., Shen, W. K., Schondorf, R., ... &
Rummans, T. A. (1995). Postural tachycardia
syndrome (POTS). Neurology, 45(4 Suppl 5), S19-25.
Wheeler, T., & Watkins, P. J. (1973). Cardiac denervation
in diabetes. British Medical Journal, 4(5892), 584.
Oka, H., Mochio, S., Sato, K., Sato, H., Katayama, K.,
Watanabe, S., ... & Isogai, Y. (1995). Spectral
analyses of RR interval and systolic blood pressure in
diabetic autonomic neuropathy. Journal of the
autonomic nervous system, 52(2), 203-211.
Bonner, R. F., Clem, T. R., Bowen, P. D., & Bowman, R.
L. (1981). Laser-Doppler continuous real-time monitor
of pulsatile and mean blood flow in tissue
microcirculation. In Scattering Techniques Applied to
Supramolecular and Nonequilibrium Systems (pp.
685-701). Springer US.
Kano, T., Shimoda, O., Higashi, K., Sadanaga, M., &
Sakamoto, M. (1993). Fundamental patterns and
characteristics of the laser-Doppler skin blood flow
waves recorded from the finger or toe. Journal of the
autonomic nervous system, 45(3), 191-199.
Abe, M., Ando, Y., Higashi, K., & Kano, T. (1996). Non-
neurogenic periodic fluctuations in heart rate and
vasomotion appearing in familial amyloid
polyneuropathy (FAP) Type I (Met30). Journal of the
autonomic nervous system, 60(1), 71-75.
Ide, J., Yamaga, M., Kitamura, T., & Takagi, K. (1997).
Quantitative evaluation of sympathetic nervous system
dysfunction in patients with reflex sympathetic
dystrophy. The Journal of Hand Surgery: British &
European Volume, 22(1), 102-106.
Ando, Y., Araki, S., Shimoda, O., & Kano, T. (1992).
Role of autonomic nerve functions in patients with
familial amyloidotic polyneuropathy as analyzed by
laser Doppler flowmetry, capsule hydrograph, and
cardiographic R–R interval. Muscle & nerve, 15(4),
507-512.
Mano, T. (1990). Sympathetic nerve mechanisms of
human adaptation to environment—findings obtained
by recent microneurographic studies. Environ. Med,
34, 1-35.
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
218
