
Neuronal Patterns in the Cavity Wall of Lesions during Gait Cycle in
a Rat Model of Brain Lesion Cavities
Ioana Nica
1
, Marjolijn Deprez
2
, Frederik Ceyssens
3
, Kris van Kuyck
2
, Robert Puers
3
, Bart Nuttin
2,4
and Jean-Marie Aerts
1
1
Division Measure, Model & Manage Bioresponse (M3-BIORES), Department of Biosystems, KU Leuven, Leuven, Belgium
2
Research Group Experimental Neurosurgery and Neuroanatomy, KU Leuven, Leuven, Belgium
3
Division Microelectronics and Sensors(MICAS), Department of Electrical Engineering, KU Leuven, Leuven, Belgium
4
Department of Neurosurgery, University Hospitals Leuven, Leuven, Belgium
Keywords: Beta Oscillations, Frequency Modulations, Local Field Potential, Motor Cortex, Rehabilitation, Brain
Lesion, Gait.
Abstract: Oscillatory neural activity was reported to have various physiological roles in information processing of
brain functions. It is now established that extracellular activity in the motor cortex encodes aspects of
movement, involving planning and motor control. Oscillatory patterns have also been hypothesized to play a
role in brain recovery and functional remapping. In this study, we measured neural activity from within the
cavity wall of a motor cortex lesion, in a rat model, while the animals performed a skilled walking task. We
aim at providing a possible framework of analysis, focused on revealing oscillatory patterns in the cavity
wall and their correlation with motor deficits, by using a combination of spectral features, involving power
spectra and coherence estimates in the beta and gamma frequency bands.
1 INTRODUCTION
Synchronization phenomena have been shown to
play a significant role in processing information in
the nervous system, in the case of normal or
pathological brain activity. Strongly synchronized
oscillations occurring at the neuronal level can be
identified in measurements performed at the
population level, like local field potentials (LFPs).
Oscillations are reported to be related to the
planning and generation of movements in healthy
subjects. In a number of invasive and non-invasive
studies, in humans and animal models, prominent
10-30 Hz beta oscillations have been observed in
motor cortical and subcortical structures in idle
subjects (Jasper et al., 1949; Toma et al. 2000;
Neuper and Pfurtscheller, 2001; Alegre et al., 2002);
beta oscillations tend to get suppressed when the
subject begins planning a movement, while the 40-
90 Hz gamma band oscillations emerge (Bouyer et
al., 1981; Donogue et al., 1998; Hamada et al., 1999;
Aoki F. et al., 2001). On the other hand, oscillations
are also an expression of motor deficits. In
Parkinson's disease, increased synchronization in
beta oscillations in the basal ganglia has been
reported as probable underlying cause in
manifestation of resting tremor, bradykinesia, and
rigidity (Raz et al., 1996, 2000; Ruskin et al., 1999,
2003; Goldberg et al., 2004).
More recently, it has been hypothesized that
synchrony might play a key role in triggering self-
repair processes after brain injury. It is thought that
spontaneous modulation of extracellular fields and
emergence of synchronous activity act as a signal for
axonal sprouting (Carmichael et al., 2002) and may
influence Hebbian plasticity mechanisms, that favor
rewiring of neural pathways (Murphy and Corbett,
2009).
In this study, we aimed at developing a
methodology to quantify patterns of oscillatory
neural activity within the wall of cortical lesions.
The approach allows investigation of motor deficits
in relation with continuous neural input in the region
of lesions induced on the motor cortex of rats.
289
Nica I., Deprez M., Ceyssens F., van Kuyck K., Puers R., Nuttin B. and Aerts J..
Neuronal Patterns in the Cavity Wall of Lesions during Gait Cycle in a Rat Model of Brain Lesion Cavities.
DOI: 10.5220/0005275802890294
In Proceedings of the International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS-2015), pages 289-294
ISBN: 978-989-758-069-7
Copyright
c
2015 SCITEPRESS (Science and Technology Publications, Lda.)

2 METHODS
We lesioned the contralateral forelimb area of the
primary motor cortex (M1) in nine rats that have
been trained to walk along a horizontal ladder with
unequally spaced rungs. We examined gait cycle
kinematics visible from video recordings of the rat
performing the task, and spectral features of the
simultaneously recorded LFPs.
The lesion caused a decrease in movement
precision, grasping strength and crossing speed in
the affected limb. However, the rats were still
capable of movement planning and control.
Oscillatory activity in electrophysiological signals
can be captured with a wide range of time-frequency
transforms. Since we recorded LFPs via a 4x4 multi-
electrode array that spans over the wall of the lesion,
we propose a space-time-frequency method of
analysis. We focus on power and inter-electrode
coherences in the beta and gamma bands, to
investigate dynamical relationships between electric
activity captured at the 16 electrode locations.
2.1 Animal Preparation and Surgical
Techniques
Before training started, the animals (nine male
Sprague-Dawley rats) were housed individually and
put on a restricted feeding schedule until the body
weight reduced with ± 10%. They were trained for
four consecutive days to walk over the horizontal
ladder in one direction. After training, a lesion was
created by aspiration of the forelimb area in the
primary motor cortex. The lesion was made in the
contralateral hemisphere to the preferred limb
(which was determined using the pasta matrix
reaching task). Lesions were made at the following
coordinates: 5 mm anterior to 1 mm posterior to
bregma, and 0.5 to 4.5 mm lateral to bregma. The
rats were allowed to recover for 3 weeks after which
the electrode array (Fig.1.a) was implanted against
the lesion cavity wall. After implantation, the lesion
cavity was filled with medical silicone (Kwik-Sil) to
keep the electrode array in place. Arrays were
implanted against the center and posterior cavity
wall, since the array was not large enough to cover
the entire lesion cavity wall (this placement was
determined based on the first rat, where it was not
possible to implant the electrode against the anterior
wall). The implant was secured with screws and
dental cement (Fig.1.b). Electrode arrays were
produced in house by the Electrotechnical
Engineering department (MICAS-ESAT) of the KU
Leuven. They consisted of 16 Platinum contacts
each with a diameter of 350 µm. Electrode contacts
were spaced ± 1.3 mm apart from each other.
a. b. c.
Figure 1: a. electrode array; b. view of the lesion site after
electrode and connector implantation; c. top view of the
rat head with headstage and connector.
2.2 Experimental Setup and
Recordings
Two weeks after electrode implantation, the animals
motor ability was tested by performing the
horizontal ladder walking they were trained for. We
performed three trials per rat, during which LFP and
video recordings were performed simultaneously.
Video recordings were taken at a sampling rate
of 40Hz with an iDS GigE camera (iDS Imaging
Development Systems GmbH, Obersulm, Germany).
To check for lost video frames, a LED flashing
protocol was implemented, so that a bursting flash
could be visible on the upper part of the frame at a
predetermined rate. Based on this, we implemented a
custom automatic video tracking algorithm in
Matlab to detect the flashes and synchronize camera
frames with LFP timestamps.
LFP recordings were performed at a sampling
rate of 10 kHz, wirelessly, using the W16 model
from Multichannel Systems (MCS GmbH,
Reutlingen, Germany), placed on top of the rat’s
head (Fig.2). The electrodes were connected to the
headstage via an Omnetics connector (npd-18-VV-
GS). The LFPs were preamplified in the range of
1Hz to 5 kHz and digitized within the headstage,
before the transfer to the receiver.
a. b.
Figure 2: a. side view during the ladder test; b. view of
the headstage location.
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
290

2.3 Data Processing
The data processing was performed in Matlab,
version 2013b, using the available toolboxes and
custom algorithms.
2.3.1 Behaviour Labelling
The video recordings were analysed frame by frame,
to identify and describe quantitatively components
of the rat gait cycle according to a scoring system
presented elsewhere (Metz and Whishaw, 2009).
Extracted features are summarized in Table 1.
The initial group consisted of a number of 20
rats, 6 of which died before the experiment was
concluded. Based on the video analysis, 5 more rats
were excluded, since the high number of stops and
long delays while crossing the ladder would not
allow for a significant number of consecutive
completed gait cycles to be analysed. Three trials per
rat were analysed, the results presented in Table 1
represent the mean over all movements identified in
the three trials.
We analysed general features like latency (time
needed to cross the ladder) and number of times the
rat stops while being on the ladder, but also
quantitative features related to the accuracy of foot
placement and forepaw dexterity (according to
(Metz and Whishaw, 2009)).
Limb placement was scored on a scale from 0 to
6, and the individual scores were defined as follows:
midportion of the palm of the limb is placed on the
rung with full weight support (6 points); the limb is
partially placed on the rung with either wrist or
digits of the forelimb or heel or toes of a hind limb
(5 points); the limb aims for one rung but it is placed
on another rung without touching the first one or,
alternatively, the limb is placed on a rung but
quickly repositioned (4 points); the limb is placed on
a rung, but before it bears the weight, it is
repositioned on another rung (3 points); the limb is
placed on a rung, slips off slightly during weight
bearing without disrupting the gait cycle (2 points);
the limb is placed on the rung and slips off when
weight bearing which leads to a fall (1 point); the
limb completely misses the rung and falls (0
points). We analysed the movement of all four
limbs: left and right forelimbs (LFl, RFl) and left
and right hind-limbs (LHl, RHl).
Out of these, only the slips (score of 0-3) were
considered errors of foot placement.
Accuracy of forepaw placement and ability to
grasp were also considered, using a four-category
scale, as follows: digits completely flexed around
the rung (3 points), digits flexed in a 45˚ angle (2
points), digits placed in a 90˚ angle on the rung (1
point); forepaw partially placed on the rung, with
either wrist or tips of digits resulting in no grasp (0
points).
These scores provide an indication regarding the
position of the lesion and the functional area
affected; for instance, in some rats, the lesion
equally affects the contralateral fore- and the hind-
limbs.
2.3.2 LFP Analysis
We are interested in capturing oscillatory events and
synchrony, therefore, we focused on spectral
features of the local field potentials.
LFPs were down-pass filtered with a cut-off
frequency of 300 Hz (elliptic filter with passband
ripple of 0.5 dB and stopband attenuation of 60dB).
Since the recordings were performed wirelessly, no
powerline interference was visible and the data was
artefact-free.
Based on the video analysis, intervals of gait
cycles were identified and included in the analysis,
Table 1. Features used in scoring rat kinematics.
Rat (impaired
limb)
Latency
(sec)
Total
number
of stops
Limb fault scoring
(0 - 6)
Number of
slips
(Fl/Hl)
Forepaw
digit score
(0 - 3)
LFl RFl LHl RHl Left Right LFl RFl
2 (RFl) 9.57 6 5.1 5 5.8 4.7 0/1 2/4 1.3 1
9 (RFl) 11.9 5 5.3 5.7 5.2 5.5 2/2 0/1 0.9 1.8
11 (RFl) 8.93 5 5.6 5.4 5.2 3.9 0/3 3/6 1.6 1.6
4 (LFl) 8.07 2 5.2 5.8 4.7 5.6 1/2 0/0 0.8 1.4
6 (LFl) 9.6 0 4.6 5.3 4.6 4.8 4/3 0/3 0.9 0.9
8 (LFl) 9.64 4 5 5.2 5 4.6 2/2 0/3 0.5 0.7
10 (LFl) 9.54 6 4.9 5.8 5.8 5.4 1/3 0/0 0.8 1.3
13 (LFl) 10.2 4 4.8 4.7 4.7 5 4/3 2/2 1.4 1.5
14 (LFl) 9.17 6 5.3 5.7 5.6 5.1 0/0 0/2 1.6 1.5
NeuronalPatternsintheCavityWallofLesionsduringGaitCycleinaRatModelofBrainLesionCavities
291
(only intervals comprising of consecutive steps were
taken into consideration, moments when the rat
stopped were excluded).
We estimated coherence between the 16 channel
recordings. Coherence - or, equivalently, magnitude-
squared coherence (C
x,y
) - provides a statistical
measure of the similarity between signals at a given
frequency and is defined in terms of the cross-
spectral density of two signals x(t) and y(t) (G
xy
),
and normalized by their autospectra (G
xx
, G
yy
) as
follows:
,
,
(1)
Magnitude-squared coherence estimates were
performed in Matlab, on epochs of approximately
250 msec (LFPs and video recordings were
synchronized and the interval between 10
consecutive video frames was included in the
coherence calculations) by taking a window of 50
msec, with 80% overlap.
a.
b.
c.
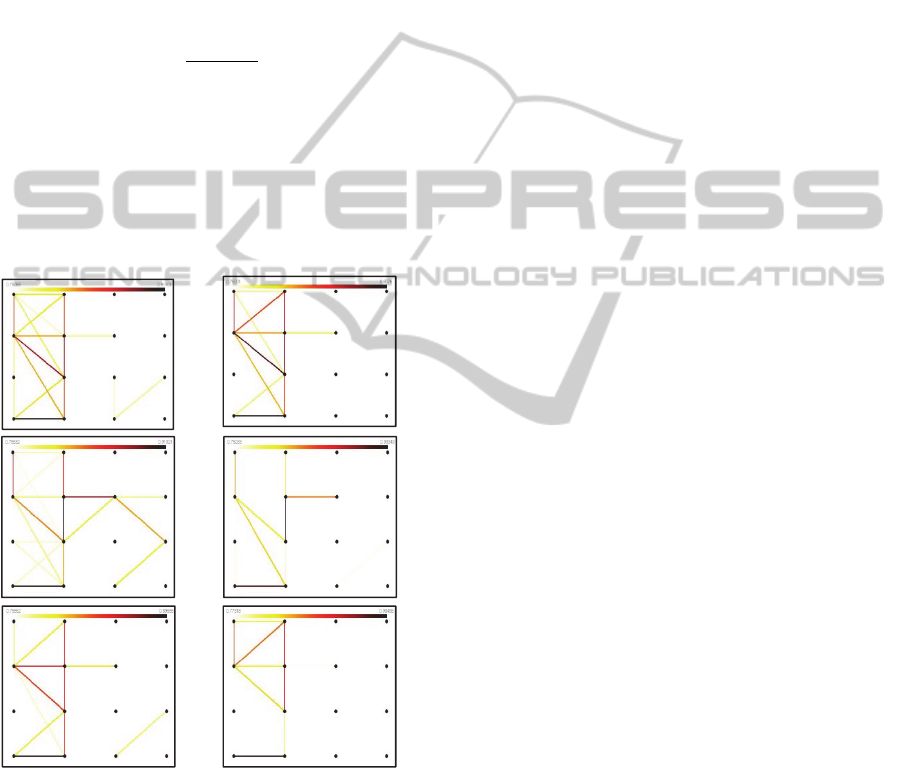
Figure 3: Example of patterns of coherences in the beta
band on 100 msec epoch prior to (left column) and
immediately after (right column) movement initiation. The
coherence patterns were generated based on averages of at
least 8 steps from the gait cycle of rat 2 such that in Figure
3.a.: epochs were selected for good steps; in Figure 3.b.:
epochs were selected for under-reached steps; in Figure
3.c.: epochs were selected for over-reached steps. The
orientation of the maps with respect to global coordinates
is North - anterior, South - posterior.
Coherence values were calculated for 120
possible pairs between the 16 electrodes, for
frequencies below 250 Hz. The resulting values
were averaged for the intervals of 15-30 Hz (beta
band), 30-45 Hz (low gamma) and 45-90 Hz (high
gamma).
Only coherence values beyond a threshold,
consisting of 125% of the mean over the 250 msec
epoch were taken into consideration and further
organized in a 16x16 adjacency matrix, used to
generate an undirected graph, that can be interpreted
as a map of significant coherences at a certain
frequency band (see Fig. 3).
1,
0,
(2)
The vertices represent electrode locations and the
edges connecting them indicate the strength of the
coherence, according to the colormaps. This way of
estimating coherences over short time-windows can
be used to interpret data on-line, and it was
implemented so as to match the sampling rate of the
video, as a way of visualizing behavior and neural
activity simultaneously. It incorporates information
about frequency, time localization and spatial
location of most active connected sites within the
lesion in a bi-dimensional form.
3 RESULTS AND DISCUSSION
Based on preliminary analysis, our hypotheses are as
follows: (i) coherence maps could be used to better
understand the nature of an individual’s deficit, by
using as feature the location of the ‘maximally
connected’ electrode locations (these are vertices
with maximum number of significant coherence
pairs); (ii) the quality of a gait cycle could be
quantified and predicted, by determining the
structure of a specific coherence map per individual
rat, on a preliminary training set of steps (with
features such as the location of the significant
coherence pairs and their strength); (iii) a possible
feature in detecting movement onset could be the
number of significant coherence pairs (out of a
maximum of 120) between the 16 electrode contacts.
In Figure 4, a representative example for the
power content of the signals during one ladder run is
provided: as observed, the lower, 10-Hz frequency
power is consistently high and non-discriminative,
while there is a continuous power shift between beta
and the higher gamma-band, which was not
observed to be directly correlated to the movement
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
292
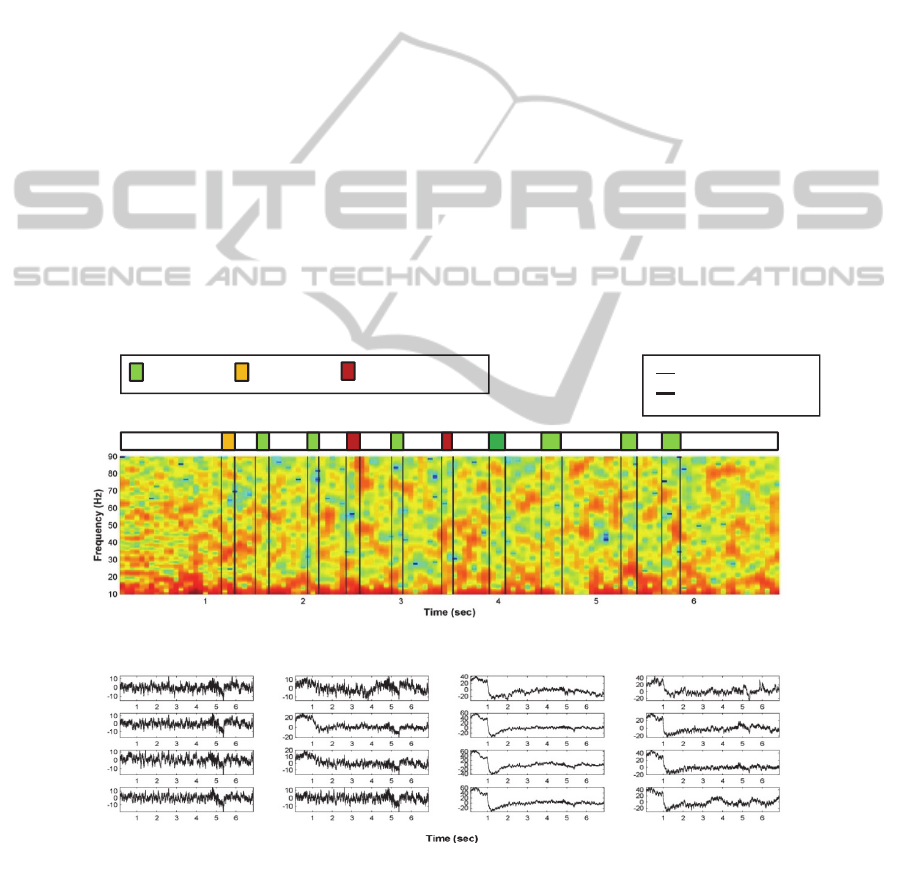
onset, gait pattern or the specific type of errors in
movement coordination. However, it is this power
shift in power content in the higher frequency bands
that motivated the choice for beta and gamma bands
in the coherence investigation.
The behavior analysis revealed the deficit of
some rats to be more extensive than expected. For
instance, in Rat 2, the higher deficit is apparent in
the hind-limb. Therefore, in the corresponding
coherence maps illustrated in Figure 3, we expect
the region in the lower half to be also active, to
account for the involvement of the hind limb
functional area of the motor cortex and as shown,
the pattern is indeed concentrated in the left side of
the map. However, the main challenge in
interpreting these results is related to the question of
how observable the system is from our recordings,
since there is a strict dependence on the placement
of the electrode array within the lesion.
Regarding the second hypothesis, a recurrent
observation is that on correct steps,
there is a
focalized center of coherence, whereas on the onset
of an erroneous step, the connections are more
dispersed.
However, we investigated a very limited number
of examples from individual rats, a validation would
require an extensive longitudinal study.
The third hypothesis is sustained by the
observation that, on movement onset, the number of
significant beta coherences decreases, while the
number of gamma coherences increases, where a
significant level was defined as the value at least
equal to 125% of the mean coherence over all
coherence maps calculated within one ladder run.
An example is provided in Figure 5, where the
average number of significant coherence pairs is
approximated on a window equivalent to the interval
spanned by 10 video frames. This feature is then
plotted against time, for the course of a ladder run.
The vertical lines indicate the intervals of swing on
the affected limb (left fore-limb, in the example
provided, for rat 6). To be noted is that all the other
limbs are moving as part of a normal gait cycle
sequence. An interesting question would be whether
or not there exists a feature discriminating initiation
of swing on each limb, or if there is a rather more
global biomarker, specific to the initiation of gait. In
order to test this idea, the resting state condition
should be included in the experiment.
Figure 4: Example of power content and signal waveform for the 4x4 electrode array during one ladder run. Figure 4.a.:
mean power spectra content of the 16 LFP channels in the 10-90 Hz band during one ladder run (from rat 4, swing intervals
identified for the left forelimb; power spectrum calculated on windows of 20 msec, with 80% overlap); the intervals of gait
swing and the labels for each step are provided according to the color code. Figure 4.b.: Corresponding LFP traces from the
16 channels.
b
.
a.
Step starts
Grasping
Correct step Under-reach Over-reach;
NeuronalPatternsintheCavityWallofLesionsduringGaitCycleinaRatModelofBrainLesionCavities
293
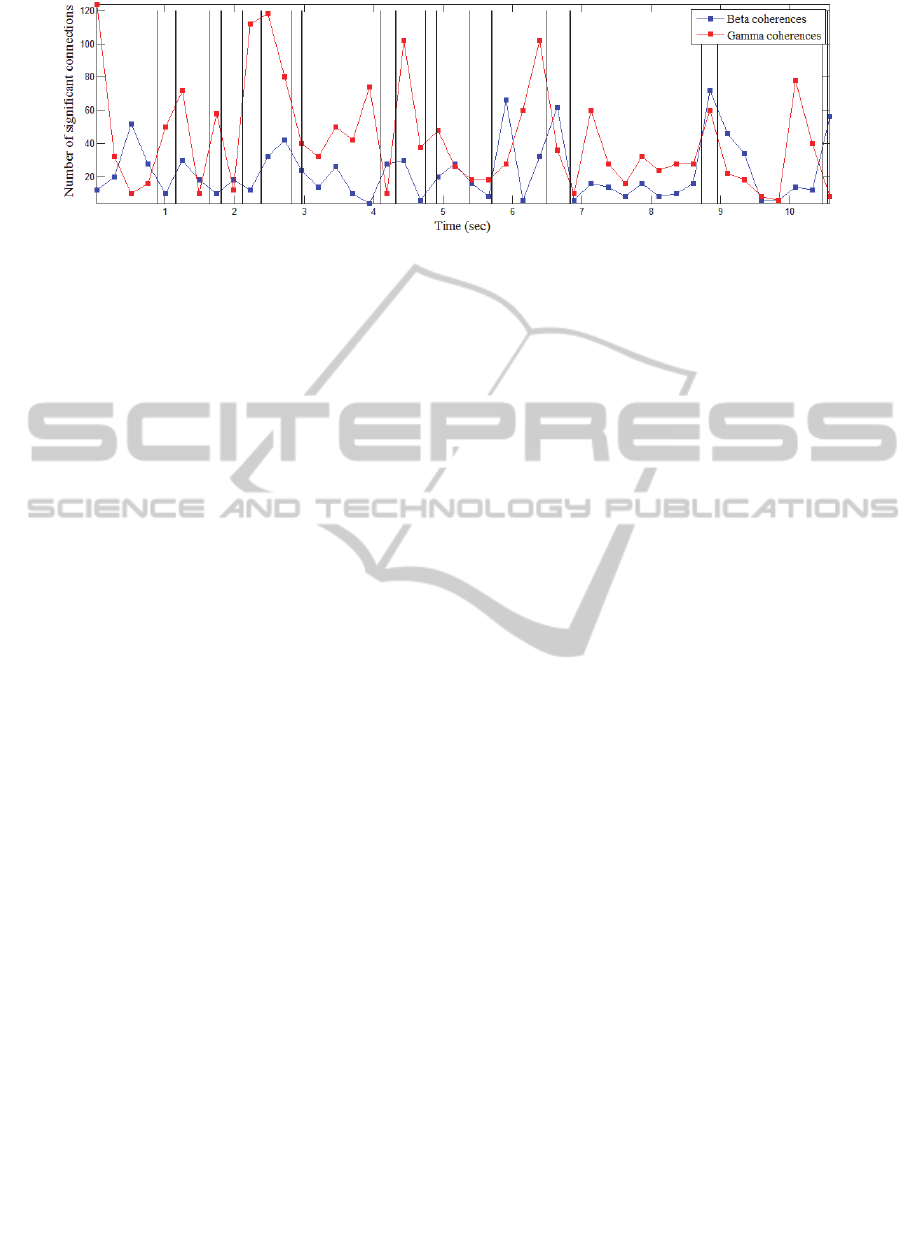
Figure 5: Variation of number of connections during a ladder run test.
4 CONCLUSIONS AND FUTURE
WORK
In order to characterise motor deficits in a rat model
of brain lesion cavities, we developed a
methodology involving behaviour analysis along
with investigation of local field potentials
originating from the cavity wall of the cortical
lesion.
Based on preliminary results, coherence
measures of the neural activity within the cavity wall
may prove valid in discribing the nature of the
functional deficit, but also, the configuration of
coherence maps may constitute signatures for
movement initiation and coordination. An
immediate objective would be to design a more
extensive cross- and longitudinal study, to test the
two hypotheses.
The method we propose for generating coherence
maps cand be easily implemented in an on-line
configuration, to characterise signal similarity in
three dimensions: frequency, space and time.
REFERENCES
Aoki, F., et al., 2001. Changes in power and coherence of
brain activity in human sensorimotor cortex during
performance of visuomotor tasks.
Alegre, M., et al., 2002. Beta electroencephalograph
changes during passive movements: sensory
afferences contribute to beta event-related
desynchronization in humans. Neurosci. Lett., 331, 29.
Donogue J.P. et al., 1998.Neural discharge and local field
potential oscillations in primate motor cortex during
voluntary movements, J. Neurophysiol., 79, 159.
Goldberg JA, Rokni U, Boraud T, Vaadia E, Bergman H
(2004) Spike synchronization in the cortex-basal
ganglia networks of parkinsonian primates reflects
global dynamics of the local field potentials. J
Neurosci 24: 6003-6010.
Hamada, Y., Miyashita, E., and Tanaka, H., 1999.
Gamma-band oscillations in the ‘barel cortex’ preced
rat’s exploratory whisking. Neuroscience, 88, 667.
Jasper, H., and Penfield, W., 1949. Electrocorticograms in
man: effect of voluntary movement upon the electrical
activity of the precentral gyrus, Arch. Psychiatrie und
Z. Neurologie, 182, 163.
Metz, G.A., Whishaw, I.Q., 2009. The ladder rung
walking task: a scoring system and its practical
application. J. Vis. Exp. (28), e1204,
doi:10.3791/1204.
Murphy, T. H., and Corbett, D., 2009. Plasticity during
stroke recovery: from synapse to behaviour. Nat. Rev.
Neurosci. 10, 861–872.
Neuper, C., and Pfurtscheller, G., 2001. Evidence for
distinct beta resonance frequencies in human EEG
related to specific sensorimotor cortical areas. Clin.
Neurophysiol., 112, 2084.
Raz, A., Feingold A., Zelanskaya V, Vaadia E, Bergman
H (1996) Neuronal synchronization of tonically active
neurons in the striatum of normal and parkinsonian
primates. J Neurophysiol 76: 2083-2088.
Raz A, Vaadia E, Bergman H (2000) Firing patterns and
correlations of spontaneous discharge of pallidal
neurons in the normal and the tremulous 1-methyl-4-
phenyl-1,2,3,6-tetrahydropyridine vervet model of
parkinsonism. J Neurosci 20: 8559-8571.
Ruskin DN, Bergstrom DA, Kaneoke Y, Patel BN, Twery
MJ, Walters JR (1999a) Multisecond oscillations in
firing rate in the basal ganglia: robust modulation by
dopamine receptor activation and anesthesia. J
Neurophysiol 81:2046-2055.
Toma, K., et al., 2000. Desynchronization and
synchronization of central 20-Hz rhythms associated
with voluntary muscle relaxation: a
magnetoencephalographic study. Exp. Brain Res., 134,
417.
BIOSIGNALS2015-InternationalConferenceonBio-inspiredSystemsandSignalProcessing
294
