
Encoding of Movement in Local Field Potentials from the Wall of
Motor Cortical Lesions in Rats
Ioana Nica
1
, Marjolijn Deprez
2
, Frederik Ceyssens
3
, Kris van Kuyck
2
, Robert Puers
3
, Bart Nuttin
2,4
and Jean-Marie Aerts
1
1
Division Animal and Human Health Engineering, Department of Biosystems, KU Leuven, Leuven, Belgium
2
Research Group Experimental Neurosurgery and Neuroanatomy, KU Leuven, Leuven, Belgium
3
Division Microelectronics and Sensors (MICAS), Department of Electrical Engineering, KU Leuven, Leuven, Belgium
4
Department of Neurosurgery, University Hospitals Leuven, Leuven, Belgium
1 OBJECTIVES
The objective of the current study was to investigate
if electrical oscillatory activity from within the
cavity wall of a motor cortical lesion can be used as
a biomarker in decoding movement. We show
results from 3 rats with unilateral lesion in the
forelimb area of the motor cortex, for which local
field potential (LFP) spectra present significant
modulation within the frequency bands of 6-10 Hz
and 45-90 Hz, corresponding to movement episodes.
2 METHODS
Rats were anaesthetized with a mixture of ketamine
(Nimatek®) and medetomidine hydrochloride
(Narcostart®). A craniotomy over the forelimb area
of the primary motor cortex was made (coordinates:
1.5 mm posterior to 5 mm anterior to bregma, and
0.5 mm to 4.5 mm lateral to bregma), after which the
exposed brain tissue was aspirated to a depth of 1.5
mm. Three weeks later, a polyimide-based thin film
electrode array (Ceyssens et al., 2013) containing 16
platinum electrode contacts, each with a diameter of
350 µm (Fig. 1.a.) was implanted against the cavity
wall. The implant and connector (Omnetics) were
secured in place with stainless steel screws in the
skull and dental cement (Fig. 1.b.).
Open field tests were performed 1 month after
electrode implantation. The rats were placed in
metal cages (dimensions 36x38x35 cm), being free
to move. They were monitored for ~30 min with a
SONY HDR-AS15 camera (sample rate 30 Hz)
placed laterally. Brain electrical activity was
wirelessly recorded at the same time, using the W16
headstage model from Multichannel Systems (MCS
GmbH, Reutlingen, Germany), (see Fig.1.c.). The
LFPs were sampled at 10 kHz and preamplified in
the range of 1Hz to 5 kHz within the headstage.
a). b). c).
Figure 1: a). Electrode array; b). Top view of the
implantation site; c). View of the headstage.
Analyses of both video and LFP recordings were
performed offline in MATLAB (version 2014b; The
MathWorks). An automatic algorithm based on pixel
intensity was implemented to discriminate between
activity and resting intervals. The threshold was set
empirically so as to capture activity such as walking,
self-grooming, standing on hind limbs. Intervals
when no movement of the limbs could be detected
were labelled as part of the ‘resting state’. The
corresponding LFP intervals were then extracted and
divided in 2-sec epochs. On average, 225 (± 55)
epochs per rat were extracted for the active state and
335 (± 80) epochs, respectively, for the resting state.
The LFPs were low-pass filtered and down-
sampled using an equiripple FIR decimator (300 Hz
cutoff frequency, decimation factor of 10) so that
final sampling rate was 1 kHz. We computed the
power spectrum using the ‘periodogram’ Matlab
function (frequency resolution of 0.5 Hz) and we
multiplied power at each frequency bin with squared
frequency to account for the 1/f
2
decay specific to
brain signals (Miller et al., 2009; Buszaki et al.,
2012). A peak in power ~8 Hz and ~50 Hz was
observed on datasets from both behavioural states.
Nica, I., Deprez, M., Ceyssens, F., Kuyck, K., Puers, R., Nuttin, B. and Aerts, J..
Encoding of Movement in Local Field Potentials from the Wall of Motor Cortical Lesions in Rats.
Copyright
c
2015 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
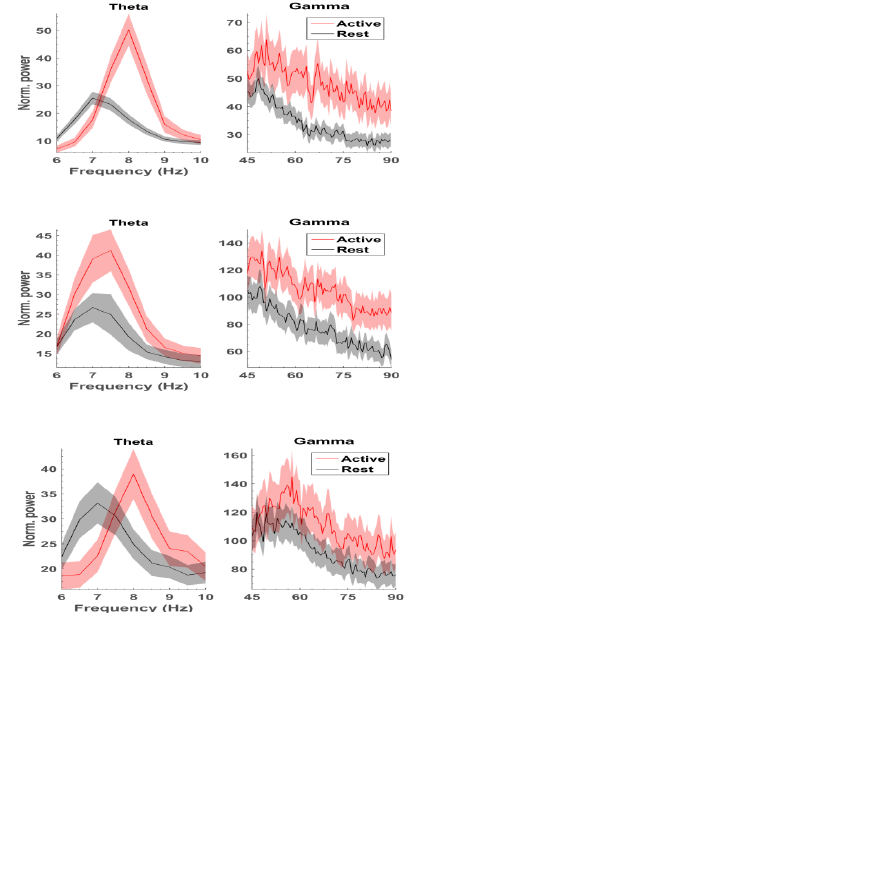
Figure 2: Mean normalized power between significant
channels (± 95% confidence limits) for each animal.
3 RESULTS
We performed two-sample t-tests with unequal
variances to test significant differences between two
sets of features (mean power in theta (6-10 Hz) and
high-gamma (45-90 Hz) band for all 16 electrodes),
to investigate how well each feature discriminates
activity from rest. For each rat, a subset of electrodes
showed significant increases (p<0.05) in theta power
(14 electrodes for rat 1, 9 for rat 2 and 6 for rat 3).
High gamma power was significantly higher
(p<<0.01) in active state on all electrodes.
4 DISCUSSION
We report prominent theta and gamma activity in the
forelimb region of the rat motor cortex, on LFPs
recorded from the wall of a lesion. We show that
these oscillations are strongly linked to motor
behaviour state, in an open-field experimental setup
that required neither learning nor reward. Our results
corroborate previous studies reported in literature on
cortical LFPs from healthy rats, during treadmill
running (von Nicolai et al., 2014), or during reward-
motivated forelimb movement (Igarashi et al. 2013).
Since the results revealed that specific subsets of
electrodes are relevant for each subject, the
robustness of these features could be investigated in
a longitudinal test, while optimizing a decoder able
to detect activity state on a single-trial basis.
Motor cortical lesions can induce various
impairments, therefore it would be of interest to
search for a correlation between theta-gamma
activity and the type of deficit the subject exhibits.
In conclusion, our results suggest that
informative signal features can be extracted from
electrical activity generated in the wall of a motor
cortical lesion. These features should be further
investigated to test the hypothesis that LFPs can help
parametrize state of impairment, on a subject-by-
subject basis.
REFERENCES
Buzsaki, G., Anastassiou, C.A., Koch, C., 2012. The
origin of extracellular fields and currents - EEG,
ECoG, LFP and spikes. Nat. Rev. Neurosci.
13(6):407-420. doi: 10.1038/nrn3241.
Ceyssens, F., van Kuyck, K., Velde, G. V.,
Welkenhuysen, M., Stappers, L., Nuttin, B., Puers, R.,.
2013. Resorbable scaffold based chronic neural
electrode arrays. Biomedical microdevices, 15(3): 481-
493.
Igarashi J., et al., 2013. A θ-γ oscillation code for neuronal
coordination during motor behaviour. J. Neurosci.
33:18515-18530.
Miller, K. J., Sorensen, L. B., Ojemann, J. G. & den Nijs,
M. 2009. Power-law scaling in the brain surface
electric potential. PLoS Comput. Biol. 5, e1000609.
Von Nicolai, C., et al., 2014. Corticostriatal Coordination
through Coherent Phase-Amplitude Coupling. J.
Neurosci. 34(17):5938-5948.
a) Rat 3
b) Rat 2
c) Rat 1
