
Comparison of GPU-based and CPU-based Algorithms for
Determining the Minimum Distance between a CUSA Scalper and
Blood Vessels
Hiroshi Noborio
1
, Takahiro Kunii
2
and Kiminori Mizushino
3
1
Department of Computer Science, Osaka Electro-Commun. Univ., Kiyotaki 1130-70, 575-0063, Shijo-Nawate, Osaka, Japan
2
Kashina System Co. Hirata-Cho 116-22, 522-0041, Hikone, Shiga, Japan
3
Embedded Wings Co., Ine 5-2-3 562-0015, Minoh, Osaka, Japan
Keywords: Parallel Processing, GPGPU, Z-Buffering, STL, DICOM, CT/MRI, CUSA.
Abstract: In this study, we have designed a GPGPU (General-Purpose Graphics Processing Unit)-based algorithm for
determining the minimum distance from the tip of a CUSA (Cavitron Ultrasonic Surgical Aspirator) scalpel
to the closest point around three types of blood vessel STLs (STereo-Lithographies). The algorithm consists
of the following two functions: First, we use z-buffering (depth buffering) as the classic matured function of
the GPU in order to effectively obtain depths corresponding to image pixels. Second, we use multiple cores
of the GPU for parallel processing so as to calculate the minimum Euclidean distance from the scalpel tip to
the closest z-values of the depths. Therefore, the complexity of the GPU-based algorithm does not depend on
the shape complexity (e.g., patch, edge, and vertex numbers) of the blood vessels.
1 INTRODUCTION
We are currently developing simulators and
navigators for liver surgery. In general, in liver
surgery, the tissue around the affected area (for
example, cancer tissue) is fractured or emulsified
with the CUSA scalpel used for ultrasound surgery
and extracted. Simultaneously, small blood vessels
having a diameter of 0.5 mm or less can be severed
while in hemostasis by cauterizing with an electric
scalpel, but severing larger vessels will cause
significant bleeding and threaten the life of the
patient. To avoid such an issue, it is ideal for the
doctors to perform actual liver surgery as planned
beforehand by regularly confirming the position of
the blood vessels.
In general, DICOM (Digital Imaging and
COmmunication in Medicine) data obtained by CT
(Computed Tomography)/MRI (Magnetic Resonance
Imaging) are used for recording the liver conditions.
In this study, we first classify the cell tissue into the
entire liver, portal veins, arteries, and veins through a
special processing of the DICOM data. This is called
liver segmentation (Zhang, 1994; Foruzan and Chen,
2013). Here, we represent the three types of blood
vessels in an STL-format polyhedron. Further, the
CUSA, which is a device that incises the liver, is
represented in the same STL with a 3D scanner.
This is because a representation in the STL format
accurately maintains the normal vector of the object
surface and the texture and feel of the distance of the
shape can be accurately felt. However, if these are
represented with a polyhedron (such as B-reps), the
basic processing of surgical simulations will be
relatively time consuming, such as the calculation of
the embedded distance between the polyhedra and the
embedded region as well as the calculation of the
shortest distance in all directions including the
operational directions of the polyhedral, as listed in
Table 1.
To begin with, sensory information related to
sight and touch is required for surgery. To obtain such
information, we need to calculate the distance and/or
the intersection between the CUSA and the liver or
the three types of blood vessels. Since the 1980s,
boundary structures (polyhedra such as B-reps [STL
is a type of these structures] and set operations on
primitives such as CSG [Constructive Solid
Geometry]), volume representations such as voxel
arrays and their hierarchical representations (such as
Oct-Tree, OBB [Oriented Bounding Boxes], and
AABB [Axis-Aligned Bounding Box]) have been
128
Noborio, H., Kunii, T. and Mizushino, K.
Comparison of GPU-based and CPU-based Algorithms for Determining the Minimum Distance between a CUSA Scalper and Blood Vessels.
DOI: 10.5220/0005634801280136
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 3: BIOINFORMATICS, pages 128-136
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

Table 1: Advantages and disadvantages of CPU-based models and GPU-based Z-buffer (BES: best, BET: better, NOR:
normal, WOR: worst, *1: Normal vector, history, and shape convexity cannot be used. *2: Normal vector, history, and shape
convexity can be used).
processed. Simultaneously, their distance has been
extracted by many CPU-based algorithms (Canny,
1986; Gilbert et al., 1988; Quinlan, 1994; Noborio et
al., 1989), and/or their intersection has been
determined by other CPU-based algorithms (Noborio
et al., 1989; Gottschalk et al., 1996; Bergen, 1997).
However, irrespective of the method used, the
computational time is proportional to (in the order of
O(n) and O(log(n)) the degree of complexity of the
shape of the polyhedron representing the organ
operated on (with n being the number of surfaces).
As contrasted with the above CPU-based high-
speed algorithms, GPGPU (General-Purpose GPU)
has been recently used for accelerating algorithms of
computer vision, 3D structure modeling, 3D
simulators, sorting, databases, and so on (Hubert,
2007; Miura et al., 2013; Pelletier, 2008; Cederman,
2008; Green et al., 2012; Yasuda, 2008; Taylor et al.,
2008; Lee et al., 2014; Modat et al., 2010). With
respect to the calculation of the distance and the
intersection between a point and an object or multiple
objects, the GPGPU has two advantages. One is fast
digitalization (to digitalize all the objects) by z-
buffering, which is the classic matured function of a
GPU. The other is the fast parallel calculation (to
calculate the minimum Euclidean distance or volume
intersection between a point and an object and/or
multiple objects) by using multicores of a GPU in
parallel.
When an STL is to be processed by z-buffering, a
z-value is calculated for each pixel that lies within the
boundary of the STL. If the z-value at a pixel indicates
that the STL is closer to the viewer than the z-value in
the z-buffer, the z-value recorded in the buffer is
replaced by the STL’s value (Joy, 1996). Further, the
Z value of the fastest patch can be preserved through
the GPU background removal function. Therefore, a
cuboid can be obtained with the width and pixilation
calculated using the Z value of the surface and the
reverse side of the polyhedron, resulting in a cuboid
digital approximation of the polyhedra.
Furthermore, we calculate Euclidean distances
from the tip of the scalpel to rectangular
parallelpipeds in parallel by using multicores of the
GPU in order to select the minimum value as the
shortest distance. Therefore, the computational time
is basically in inverse proportion to the core count.
Note that the GPU core count is still enormous and
increases rapidly. However, the conversion time will
no longer depend on the number of surfaces of the
polyhedron.
Therefore, the superimposition calculation of the
liver and the three types of blood vessel cuboids and
the CUSA scalpel cuboids can be performed instantly
by the GPU, and this enables a rapid calculation, by
the GPU, of the embedded distance and the embedded
regions. From this embedding, for example, an
artificial sense of touch is constructed with the
Kelvin–Voigt model, and this can be experienced
through a tactile feedback device. Further, the
polyhedra can be rapidly transformed in response to
the embedded region, and the concave region
becomes visible (Noborio et al., 2013; Onishi et al.,
2014; Onishi et al., 2015).
On the basis of the abovementioned pre-
processing, in this study, we calculate the shortest
distance from the CUSA tip to the three types of blood
vessels (portal veins, arteries, and veins). Herein, we
design a GPU-based algorithm and, by using a CPU,
compare it with a CPU-based algorithm that
calculates the shortest distance from the CUSA tip to
the three types of blood vessel STLs.
The rest of this paper is organized as follows:
Section 2 describes the classic CPU-based algorithm
and the proposed GPU-based algorithm for
calculating the shortest distance from the CUSA tip
to the three types of blood vessels. With respect to the
advantages and disadvantages of GPU-based and
Comparison of GPU-based and CPU-based Algorithms for Determining the Minimum Distance between a CUSA Scalper and Blood Vessels
129

CPU-based algorithms, Section 3 investigates the
effects of the required bit count for a parallel
processing approach, not using the increasing trends
of the computational time when the surface count
increases, actual time shifts in certain liver incision
simulations, and distance errors. Finally, Section 4
summarizes this study.
Figure 1: (a) Minimum distance between a point T and a
patch P in CPU, and (b) minimum distance between a point
T and a set of rectangular parallelepipeds of P in GPU.
2 ALGORITHM TO CALCULATE
THE SHORTEST DISTANCE
In this section, we discuss the CPU-based and GPU-
based algorithms to calculate the shortest
(Euclidean) distance from the CUSA tip to the three
types of blood vessel STLs.
2.1 CPU-based Algorithm
First, we have the tip of the scalpel and the three types
of blood vessel (portal veins, arteries, and veins)
STLs. Next, patch Ps are sequentially chosen from the
three types of blood vessels, and the Euclidean
distance to these is calculated. Finally, the minimum
distance to all the patches is selected, and this is
considered to be the shortest distance to the three
types of blood vessel STLs.
In general, the minimum values of the Euclidean
distance of T and P are obtained from any of the
distances of an infinite plane including Tip T and
Patch P, an infinite straight line including side E of P
and the distance of T, as well as the top V of both tips
of side E and the distance of T. Accordingly, if the leg
of a perpendicular line from coordinate T to a plane
including Patch P falls within Patch P, the length of
the perpendicular line is the shortest distance (Figure
1(a)). Otherwise, the distance to the side including the
top is the shortest distance (Figure 1(a)). Therefore,
the algorithm to calculate the shortest distance is as
follows:
[Step1] Calculate the normal vector n (size
normalized at 1) from the three top points of the blood
vessel’s STL triangular surface (Patch) Pi.
[Step2] Calculate the vector v from any top point of
the triangular surface to the scalpel point T.
[Step3] Get the inner product of normal vector n and
vector v to find the size of the perpendicular vector
from scalpel tip T to the plane.
[Step4] Only for the distance found in [Step3], find
the point going (on the infinite plane) in the opposite
direction of the normal vector from the scalpel tip.
[Step5] Since the intersection in [Step4] is the
intersection with the infinite plane crossing the three
top points, use an outer product for deciding whether
this intersection is within (including sides and top
points) the triangular surface Pi.
[Step6] If the perpendicular line intersects with the
infinite plane outside of the triangular surface Pi, it is
not the shortest distance from the scalpel tip to the
triangular surface Pi. Therefore, reject the
perpendicular line distance to the plane and proceed
to [Step7]. Otherwise, keep it as the shortest distance
candidate di and proceed to [Step8].
[Step7] Of the distances from the tip of the scalpel to
the three sides, keep the shortest distance as the
shortest distance candidate di.
[Step8] Select the smallest value from all triangular
surfaces (Patch) Pi (i: from 1 to n, n: total surface
count) as di. This is the shortest distance to the three
types of blood vessel STLs from the CUSA tip T.
Here, the distance calculation of “points and
surfaces,” “points and sides,” and “points and tops”
in the existing algorithm is checked in a likely order
(Lin and Canny, 1991).
2.2 GPU-based Algorithm
First, using the z-buffering (depth buffering), we
obtain a set of rectangular parallelepipeds along the Z
(depth) axis (Figures 1(b) and 2) in a two-dimensional
XY array. This is a useful function of the GPU, and
therefore, we can obtain the set of rectangular
parallelepipeds very rapidly. For the three types of
blood vessel STLs, we obtain three sets of rectangular
parallelepipeds around the tip T. A large rectangular
region where a doctor operates on the liver by using
CUSA can be freely selected in the world coordinate
BIOINFORMATICS 2016 - 7th International Conference on Bioinformatics Models, Methods and Algorithms
130
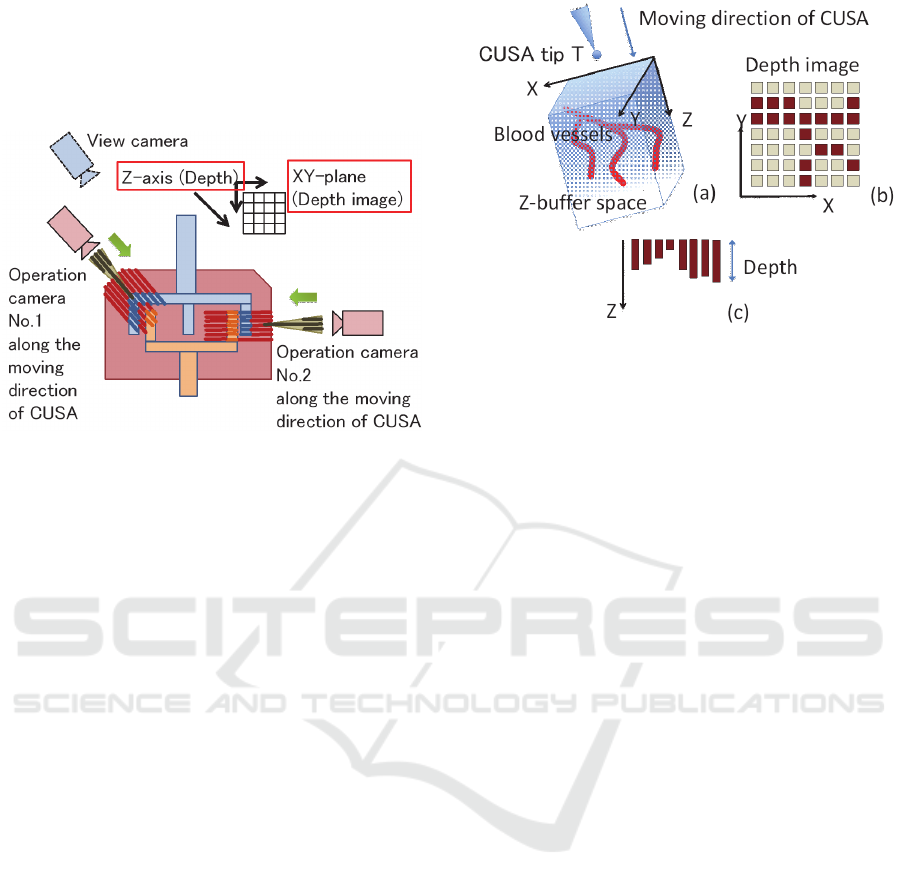
system. In other words, the coordinate system of
depth buffering is defined as a rectangular region,
which implies that rectangular parallelepipeds and the
tip T are transformed into the camera coordinate
system of the depth buffering (Figure 2).
Figure 2: Z-buffer for visible ability and several Z-buffers
for cutting the liver are independently allocated in the world
coordinate (3-D) system.
Next, in the camera coordinate system, we can
calculate the minimum Euclidean distance from the
tip T to the rectangular parallelepipeds in parallel as
the shortest distance by using the many cores of the
GPU (Figure 3). In the assignment to the parallel
processing GPU calculation unit, the thread count is
16 × 16 and the block count is width/16 × height/16.
That is, when the image resolution is 2048 × 2048, a
parallel calculation is performed with a thread count
of 16 × 16 and a block count of 128 × 128.
Finally, in the proposed GPU-based algorithm, we
calculate the distance precision by using two types of
image and depth quantization methods. First, a cuboid
region of X: 106 mm × Y: 106 mm × Z: 213 mm is
established around the scalpel tip to obtain the Z value
of the three types of blood vessel STL surfaces. In
such cases, a 32-bit variable records the Z value, and
the XY image resolution is 2048 × 2048 (Figure 3).
As a result, the quantization error for the Z axis is
about 0.05 nm (=213 mm/2
32
), and the quantization
error for the XY image is about 52 μm (=106
mm/2048). Therefore, the maximum quantization
error of the Euclidean distance used for calculating
the XYZ value by using the three squares theorem is
73.5 μm. From the perspective of a doctor’s
requirements, this error can be sufficiently ignored.
Moreover, the minimum distance to all pixels is the
shortest distance between the scalpel tip T and the
three types of blood vessel STLs (Figure 3).
Figure 3: Many depths (2 values) in the XY image (many
pixels), which are converted from STL blood vessels.
An array z-buffer[x,y] initialized to ∞
begin /* z-buffering in GPU
for each patch P of three types of vessels do {
for each pixel (x,y) that intersects P do {
calculate z-depth of P at (x,y)
if z-depth < z-buffer[x,y] then {
z-buffer[x,y] = z-depth
}
}
}
A vector d-bits[z] initialized to 0
begin /* many cores in GPU
parallel for all pairs of pixels (x,y) and
their z-depth do{
calculate Euclidean distance D between (x,y, z-
buffer[x,y]) and (x
P
,y
P
,z
P
) of tip T of CUSA scalpel
raise a flag at d-bits[z] by D
}
scan d-bits[z] sequentially from left to right in order
to find an initial bit
calculate the distance corresponding to the bit
As shown in Figure 4, even if multiple parallel
processing tasks simultaneously change specific bits,
the existence of their distances is established. In such
cases, if the bit set and the closest XYZ value are
recorded at the same time, multiple proximate vectors
(vectors expressed at the proximity point with the
scalpel tip), which achieve this distance, are
separately recorded. Because all the bits are
initialized, a bit value of 0 implies that a distance
could not be found within the scope corresponding to
any parallel process.
Comparison of GPU-based and CPU-based Algorithms for Determining the Minimum Distance between a CUSA Scalper and Blood Vessels
131

Figure 4: Parallel processing the shortest distance by using
many of the cores in the GPU.
Here, we discuss the relationship between the bit
number and the distance precision in the proposed
algorithm. On the basis of the opinions of doctors, the
ranging scope is set to 0–50 mm, and this is divided
to the extent of 16 × i (variable i is arbitrarily set).
Further, there is a bit for each division, and if there
are any pixels in which the distance from the scalpel
tip to the three types of blood vessels fits within the
bounds of the divisions, this bit is set to 1 (Figure 4).
For example, if i = 1, and the distance error from this
division is 50 mm/16 = about 3 mm, i = 16, then the
distance error from the division is 50 mm/256 = about
0.2 mm. Further, if all arrays are initialized at 0 before
starting the parallel processing for all pixels, when
searching the arrays from 0 after the parallel
processing is finished, in the array in which 1 is first
located, we find the shortest distance by the following
calculation: shortest distance = intermediate value of
the distance range corresponding to the array number
(Figure 4).
Lastly, errors related to this parallel processing are
added to the quantization errors from when the
polyhedra are transformed into Z-buffers. Of course,
in CPU-based algorithms, for parallel processing
errors, there are no quantization errors when the
polyhedra are converted into Z-buffers; there are only
limitations of floating-point numbers (single
precision and double precision). Accordingly,
compared to GPU-based algorithms, there are
relatively few mistakes (Table 2).
Table 2: Comparison of calculation time and distance
precision of CPU- and GPU-based algorithms.
3 EXPERIMENTAL
COMPARISON OF CPU-BASED
ALGORITHMS AND
GPU-BASED ALGORITHM
In this section, we investigate the accuracy of and the
required time for measuring the shortest distance
between the scalpel tip and the three types of blood
vessels. First, we evaluate the computational time for
the two algorithms by monotonically adding the item
count of polyhedral patches that constitute the three
types of blood vessels. Next, we compare the
computational time and the measurement accuracy
when the liver is virtually incised by CUSA.
3.1 PC and Development
Environments
Here, the PC environment and the development
environment related to the CPU and the GPU are
explained in detail.
CPU-related specifications:
(1) PC
OS Windows 8.1 Professional 64-bit
CPU Intel Core i5-2500K CPU 3.30 GHz
Memory 16 GB
(2) Development environment
IDE Microsoft Visual Studio 2012
Renderer Direct 3D 11.0
GPU-related specifications:
(1) PC
VGA GeForce GTX480 CUDA processor core count
480
Graphics clock 700 MHz
Processor clock 1401 MHz
Memory 1536 MB GDDR5
(2) Development environment
Language C++/HLSL
GPGPU Direct Compute
Since the development environments of CPU-based
and GPU-based algorithms are different, the
BIOINFORMATICS 2016 - 7th International Conference on Bioinformatics Models, Methods and Algorithms
132
following comparisons may not be precisely fair.
However, our computer has sufficient memory and
the STL’s data size is not very large. Therefore, the
comparisons are practically useful for designing a
surgical simulator/navigator in the future.
3.2 Evaluation of Computational times
of CPU-based and GPU-based
Algorithms
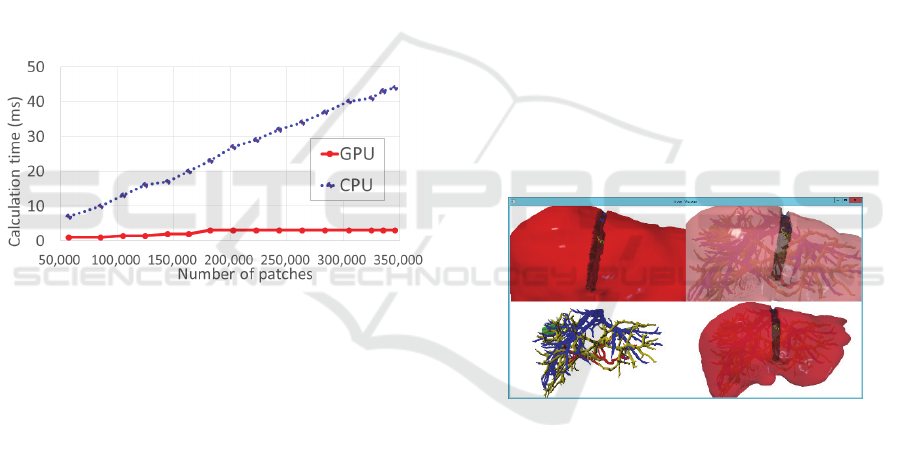
Here, we investigated how the computational time for
each algorithm increases depending on the polyhedra
patch count. First, we place a number of patches for
the three types of blood vessels on the horizontal axis
and monotonically increase the patch count while
increasing the approximate accuracy of the blood
vessel shape to 357992. The computational time for
the CPU- and GPU-based algorithms is shown on the
vertical axis of the graph (unit: milliseconds) (Figure
5).
Figure 5: Computational time of CPU- and GPU-based
algorithms in proportion to the number of patches.
From these results, we found that the computational
time order for algorithms using CPU is O(n) for
surface count n; further, we confirmed that the
computational time increases in proportion to n
(Since this is the shortest distance from the tip of the
scalpel, it is not dependent on the degree of
complexity of the scalpel shape.). This is the same
characteristic as that of conventional algorithms using
CPUs (Lin 1991).
Further, if the accuracy of the polyhedra shape is
increased, the actual computational time will exceed
40 ms. Hence, it will be difficult to obtain not only
the sense of touch but also the reality of sight (The
normal video rate is exceeded.). On the other hand,
the calculation time order of algorithms using the
GPU is O(1), which is a fixed value irrespective of an
increase in n. In the future, since improvement in the
liver segmentation function will allow the recognition
of smaller blood vessels and more accurate shapes,
the polyhedra patch count representing the three types
of blood vessels is expected to increase.
Accordingly, we can state that the computational
time of the GPU-based algorithm, which is
independent of the degree of complexity of the
subject shape, is more desirable than that of the CPU-
based algorithm, which is dependent on the degree of
complexity of the subject shape.
3.3 Changes in Computational Time
for the Shortest Distance between
the Scalpel Tip and the Blood
Vessels in Actual Surgery
Here, we show the computational time required to
derive the shortest distance from the scalpel tip to the
three types of blood vessels when a virtual liver is
actually incised by using a virtual CUSA scalpel.
Figure 6 shows a strobe shot of such an instance, and
Figure 7 shows the time changes exclusively for the
distance calculation of the CPU- and GPU-based
algorithms for each of the motions (the cycle of
moving the scalpel, incising the liver, and measuring
the distance to the three types of blood vessels). Here,
the computational time for the CPU-based algorithm
is more than 40 ms. However, the computational time
for GPU-based algorithms is usually about 3 ms.
Figure 6: Surgery for polyhedral liver and its three types of
blood vessels by using a polyhedron of the CUSA scalpel.
A simulation/navigation of liver surgery needs many
calculation functions such as liver deformation and
real liver sensing and following. The calculation of
the shortest distance is one of them. Therefore, if the
calculation (40 ms) is over the video rate, the surgical
animation of simulation/navigation is sometimes
frozen. Therefore, a faster calculation (2–3 ms) using
a GPU is suitable for real-time simulation/navigation.
A doctor can comfortably follow the instructions for
the surgery when they are provided using smooth
animation. As a result, this real-time animation
reduces the possibility of mistakes made by the
doctor, such as blood vessel injuries in surgery
performed using augmented reality.
Comparison of GPU-based and CPU-based Algorithms for Determining the Minimum Distance between a CUSA Scalper and Blood Vessels
133
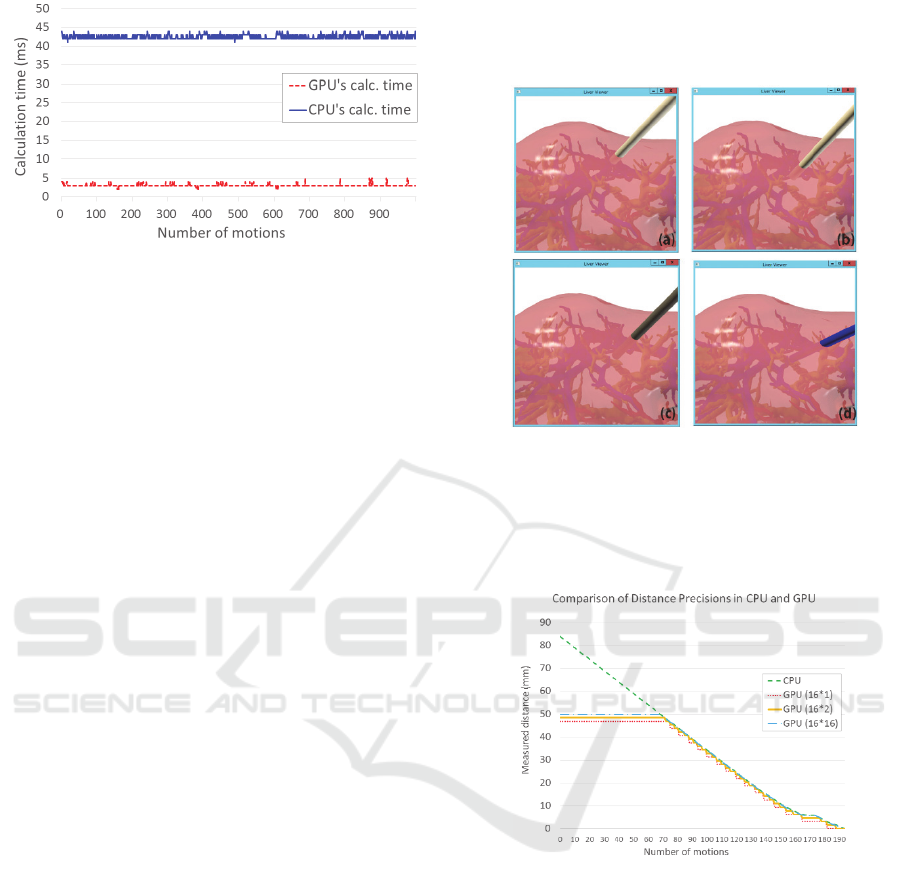
Figure 7: Computational time of CPU- and GPU-based
algorithms in the surgery described in Figure 6.
These results were obtained with the regular-edition
GPU (core count: 480). The current business-use
GPU with thousands of cores will probably be about
five times faster. In contrast, the CPU operating
frequency has hit a plateau since 2007 (around 3
GHz), and Moore’s Law (a three-fold increase in
speed every 2 years) is no longer applicable to the
calculation of computing power. As a
countermeasure, the CPU operating unit counts have
been increased to boost computing power, but the
GPU computing power has improved to a far greater
extent. Therefore, we believe that the GPU-based
algorithm has more of a future than the CPU-based
version.
3.4 Comparison of the Shortest
Distance from the Tip of the Scalpel
to the Three Types of Blood Vessels
Calculated by CPU- and
GPU-based Algorithms in Actual
Surgery
As explained in Section 2 (particularly, the final
paragraph of Section 2.2), GPU-based algorithms
realize parallel processing and maintain its speed. As
a result, certain distance measurement ranges can
only be measured with a certain degree of accuracy
for the distance. On the other hand, in the case of
CPU-based algorithms, no special consideration is
needed for distance measurement ranges or distance
accuracy (naturally, this is still conditioned by the
calculation accuracy of floating-point numbers)
(Table 2).
Here, we actually validated these characteristics.
To begin with, Figure 8 shows the scalpel motion in
a validation experiment, and Figure 9 shows the
trends in distance measurement ranges and distance
accuracy for CPU- and GPU-based algorithms in such
cases. Here, CPU-based algorithms calculated all the
shortest distances without any restrictions, but GPU-
based algorithms calculated the shortest distances for
fixed distance measurement ranges (0–50 mm) with
fixed distance accuracy (50/(16 × i) mm, i: optional).
Figure 8: Motion example in which the CUSA scalpel
accesses three types of blood vessels. In this figure, as long
as the distance decreases monotonously, the color of the
CUSA scalpel changes from white to light gray, dark gray,
and then blue (black). From this color change, a doctor
understands the present state of danger.
Figure 9: Range and precision of distances change when the
CUSA is close to the blood vessels along the sequence of
motions shown in Figure 8.
As compared to the CPU-based algorithm, the GPU-
based algorithm is very fast and its complexity does
not depend on the shape of the blood vessels. On the
other hand, the CPU-based algorithm deals with a
continuous boundary, and therefore, its distance
precision is very good. Unfortunately, the GPU-based
algorithm always digitalizes the shape of blood
vessels by image pixels and their z-buffering.
Consequently, it decreases the distance precision.
BIOINFORMATICS 2016 - 7th International Conference on Bioinformatics Models, Methods and Algorithms
134

4 CONCLUSIONS
In this paper, we proposed an algorithm to calculate
the shortest distance from the scalpel tip to the blood
vessels independent of the degree of complexity of
the shape of the three types of blood vessels. This
satisfies the specifications required by doctors (with
respect to the distance measurement range, distance
accuracy, and computational time).
To begin with, the opinion among liver surgeons
(as opposed to cerebrovascular and cardiovascular
surgeons) is that a positioning accuracy of about 0.5
cm is sufficient for liver surgery (conversely, human
behavioral functions cannot adjust to any higher
degree of accuracy). It is reportedly the case that the
direction of distance measurement, movement of the
scalpel (incision mistakes only occur in this
direction), and distance measurement range are
acceptable at about 10 cm from the tip of the scalpel
(only around the liver cancer to be cut out; the size of
the entire liver is about 20 cm). Moreover, the
surgeons voiced their desire for a real-time nature
(within a range of several milliseconds in all
situations) that calculates the senses of vision and
touch to be emphasized. The algorithm proposed in
this paper meets this request even when the shapes of
the three types of blood vessels inside the liver are
complex.
Currently, the number of cores in a GPU is
increasing every year. The proposed algorithm can
keep pace with this increase. Further, the technology
for blood vessel imaging and its segmentation is
improving every year, with smaller blood vessels as
well as their more detailed shapes being recognized.
Since this results in an enormous surface count for the
STL expressing the three types of blood vessels, the
value of the GPU-based algorithm proposed here that
calculates the shortest distance to the blood vessels is
expected to increase.
Lastly, an issue to consider in the future is the
development of an algorithm with the guidance
control of the scalpel tip to incise about 0.5 cm around
cancer cells in order to remove them. Toward this
end, it is necessary to enable the calculation of the
shortest distance from all directions of the scalpel tip
and calculate the proximity vector from the scalpel tip
to the closest point on the cancer tissue surface in
order to calculate the operational vector of the scalpel.
Further, we developed a CUSA tip for the cutting
region, but if the kidneys or the other organs, and not
the liver, were the target, then CUSA cannot be used;
blades or scissors would have to be employed to make
the incision. In such cases, we would need to
represent the cutting area with a line and not a point.
In the future, we would like to consider expanding the
proposed algorithm to such instances.
ACKNOWLEDGEMENTS
We would like to express our sincere gratitude to
Professor Masanori Kon and Associate Professor
Masaki Kaibori of Kansai Medical University, who
provided us with advice concerning liver surgery, and
Professor Yen-Wei Chen of Ritsumeikan University,
who provided the segmented liver DICOM data.
Further, we would like to note that this research has
been partially supported by the Collaborative
Research Fund for Graduate Schools (A) of the Osaka
Electro-Communication University and a Grant-in-
Aid for Scientific Research of the Ministry of
Education, Culture, Sports, Science and Technology
(Research Project Number: 26289069).
REFERENCES
Zhang Z. Iterative point matching for registration of free-
form curves. Int. J. Comput. Vision 2, pp.119-152,
1994.
Foruzan A.H., Chen Y.W. et al. Segmentation of liver in
low-contrast images using K-means clustering and
geodesic active contour algorithms. IEICE Trans 4,
pp.798-807, 2013.
Canny J.F. Collision detection for moving polyhedral. IEEE
Trans PAMI 2, pp.200-209, 1986.
Gilbert E., Johnson D., Keerthi S. A fast procedure for
computing the distance between complex objects in
three-dimensional space. IEEE J. Robotic. Autom. 2,
pp.193-203, 1988.
Quinlan S. Efficient distance computation between non-
convex objects. IEEE J. Robotic. Autom.’94, pp.3324-
3329, 1994.
Noborio H., Hata H., Arimoto S. Algorithms searching for
the nearest point of 3-D objects using octotree. IPSJ
Trans 3, pp.311-320, 1989 (in Japanese).
Noborio H., Fukuda S., Arimoto S. Fast interference check
method using octree representation. Adv. Robotics 3,
pp.193-212, 1989.
Gottschalk S., Lin M.C., Manocha D. OBBTree: A
hierarchical structure for rapid interference detection.
SIGGRAPH ‘96, New Orleans, pp.171-180, 1996.
Bergen G. Efficient collision detection of complex
deformable models using AABB trees. Journal of
Graphics Tools 4, pp.1-13, 1997.
Hubert N. GPU Gems 3: Programming Techniques for
High-Performance Graphics and General-Purpose
Computation, Addison-Wesley Professional; First
version, 2007.
Miura M., Fudano K., Ito K., Aoki T., Takizawa H.,
Kobayashi H. Performance evaluation of phase-based
Comparison of GPU-based and CPU-based Algorithms for Determining the Minimum Distance between a CUSA Scalper and Blood Vessels
135

correspondence matching on GPUs. Proc. SPIE 8856,
Applications of Digital Image Processing XXXVI,
885614, 26 September 2013.
Pelletier M. G. Parallel algorithm for GPU processing for
use in high speed machine vision sensing of cotton lint
trash. Sensors 8(2), pp.817-829, 2008.
Cederman D. On sorting and load balancing on GPUs.
ACM SIGARCH Computer Architecture News
Archive 36(5), pp.11-18, December 2008.
Green O., McColl R., Bader D. A. GPU merge path - A
GPU merging algorithm. Proc. of the 26th ACM
International Conference on Supercomputing (ICS),
San Servolo Island, Venice, Italy, June 25-29, pp.
331
–
340,
2012.
Yasuda K. Accelerating density-functional calculations
with graphics processing units. Journal of Chemical
Theory and Computation 4(8), pp.1230-1236, August
2008.
Taylor Z. A, Cheng M., Ourselin S. High-speed nonlinear
finite element analysis for surgical simulation using
graphics processing units. IEEE Trans Med Imaging
27(5), pp.650-663, 2008. doi:
10.1109/TMI.2007.913112.
Lee H.-P., Audette M., Joldes G. R. and Enquobahriea A.
neurosurgery simulation using non-linear finite element
modeling and haptic interaction. Proc. SPIE Int. Soc.
Opt. Eng. Author manuscript; available in PMC 2014
Jan 22. Published in final edited form as: Proc. SPIE Int.
Soc. Opt. Eng. 2012 Feb 23; 8316: 83160H. doi:
10.1117/12.911987.
Modat M., Ridgway G. R., Taylor Z. A., Lehmann M.,
Barnes J., Hawkes D. J., Fox N.C., Ourselina S. Fast
free-form deformation using graphics processing units.
Journal of Computer Methods and Programs in
Biomedicine 98(3), pp.278–284, June 2010.
Joy K. The Depth-Buffer Visible Surface Algorithm, On-
Line Computer Graphics Notes,
http://www.idav.ucdavis.edu/education/GraphicsNotes
/Z-Buffer-Algorithm/Z-Buffer-Algorithm.html, 1996.
Lin M., Canny J. A. Fast algorithm for incremental distance
calculation. IEEE Robotics and Automation ’91,
Sacramento, pp.1008-1014, 1991.
Noborio H, Onishi K, Koeda M, Mizushino K, Kunii T,
Kaibori M, Kwon M, Chen YW. A fast surgical
algorithm operating polyhedrons using Z-buffer in
GPU. Proc. of the 9th Asian Conference on Computer
Aided Surgery, Tokyo, pp.110-111, 2013.
Onishi K., Mizushino K., Noborio H., Koeda M. Haptic AR
dental simulator using Z-buffer for object deformation.
Proc. of the HCI International 2014 (16th International
Conference on Human-Computer Interaction), Creta
Maris, Heraklion, Crete Greece, pp.342-348, June 22-
27, 2014.
Onishi K., Noborio H., Koeda M., Watanabe K., Mizushino
K., Kunii T., Kaibori M., Matsui K., Kwon M. Virtual
liver surgical simulator by using Z-buffer for object
deformation. Universal Access in Human-Computer
Interaction (Proc. of HCII 2015), Part III, LNCS 9177,
pp.345-351, Los Angeles, CA, USA, August 2015.
BIOINFORMATICS 2016 - 7th International Conference on Bioinformatics Models, Methods and Algorithms
136
