
Discontinuous Dielectrophoresis
A Technique for Investigating the Response of Loosely Adherent Cells to High
Shear Stress
Rebecca Soffe
1
, Sara Baratchi
2
, Shi-yang Tang
1
, Peter McIntyre
2
,
Arnan Mitchell
1
and Khashayar Khoshmanesh
1
1
School of Electrical and Computer Engineering, RMIT University, 124 La Trobe St., Melbourne, Australia
2
Health Innovations Research Institute and School of Medical Sciences, RMIT University, Plenty Rd., Melbourne, Australia
Keywords: Discontinuous Dielectrophoresis, Dielectrophoresis, Microfluidics, Shear-Induced Stress, Intracellular
Calcium Signalling, HEK-293.
Abstract: The functioning of cells under mechanical stress influences several cellular processes, for example
proliferation, organogenesis, and transcription. Current techniques used to examine mechanical stress on
loosely adherent cells, are however, primarily focused on single individual cells being stimulated, or require
time-consuming surface coating techniques; and are limited in the level of shear stress that can be supplied
to immobilised cells. Here we report the process of the technique, discontinuous dielectrophoresis; which
enables high shear stress analysis of clusters of immobilised loosely adherent cells, we have analysed the
performance of the system using Saccharomyces cerevisiae yeast cells, up to a shear stress of 42 dyn/cm
2
.
Additionally, we provide application experimental results from investigating shear induced calcium
signalling of HEK-293-TRPV4 cells at flow rates of 2.5, and 120 µl/min, corresponding to shear stress
levels of 0.875 and 42 dyn/cm
2
, respectively. In summary, discontinuous dielectrophoresis will enable the
investigation of the mechanotransduction behaviour of loosely adherent cells under physiologically relevant
shear stresses. Additionally, discontinuous dielectrophoresis provides the capability for parallelism, and
dynamic control over the microenvironment, as previously explored by different microfluidic platforms
without the capacity for high shear stress analysis of loosely adherent cells.
1 INTRODUCTION
Our technique differs from existing permanent
immobilisation techniques, as it enables loosely
adherent cells to be investigated under shear stress
magnitudes higher than previously reported
(Mendoza et al., 2010, Yamamoto and Ando, 2013).
Various conventional techniques either on-chip
or off-chip are used to immobilise both adherent and
non-adherent cells, to carry out various biological
assays and microscopy techniques, in the absence of
shear stress. Furthermore, predominantly
conventional methods are focused on surface
modification techniques, which include for example,
ligands, biometric peptides, and cell adhesive
peptides. Such techniques do not facilitate the robust
immobilisation required for applying high shear
stress, to loosely adherent cells; potentially as some
systems are not intentionally designed for high shear
stress analysis (Mutreja et al., 2015, El-Ali et al.,
2006, Nahavandi et al., 2014, Berthier et al., 2012,
Yamamoto and Ando, 2013, Baratchi et al., 2014,
Voldman, 2006). However, Baratchi et al. utilised
surface modification techniques on a microfluidic
platform to apply shear stress, however this was
limited to the lower end of the physiological shear
stress range (Baratchi et al., 2014).
Moreover, microfluidic-based immobilisation
techniques such as dielectrophoresis,
magnetophoresis, or acoustophoresis, require the
electric, magnetic, or acoustic field, be active
throughout the duration of the experimentation to
keep cells immobilised, and often limited to low
shear stress levels to avoid dislodgement of cells
(Soffe et al., 2015b, Ding et al., 2012, Voldman,
2006). In contrast, in our technique the electric field
is activated for just 120 seconds, which in turn
minimizes the negative impact on cells and
simplifies the experimental procedure (Soffe et al.,
2015a).
Soffe, R., Baratchi, S., Tang, S-y., McIntyre, P., Mitchell, A. and Khoshmanesh, K.
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress.
DOI: 10.5220/0005654700230033
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 1: BIODEVICES, pages 23-33
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
23

Although permanent immobilisation of proteins by
means of dielectrophoresis has been previously
demonstrated by T. Yamamoto, et al. (Yamamoto
and Fujii, 2007), our technique differs from their
work, as it enables the permanent immobilisation of
multiple cells rather than small clusters of proteins;
additionally, and more importantly our technique
enables the patterned cells to remain attached to the
surface even at high shear stress levels.
2 DISCONTINUOUS
DIELECTROPHORESIS
Discontinuous dielectrophoresis is a technique based
on dielectrophoresis; however, the application of the
electric field is minimised, and high flow rates
producing a shear stress over a cluster of cells can be
achieved (Soffe et al., 2015a). Discontinuous
dielectrophoresis overcomes limitations of
dielectrophoresis, as previously mentioned,
regarding the reduced activation period of the
electric field and the ability to conduct experiments
in biologically relevant suspension media, due to the
deactivation of the electric field. Furthermore,
immobilised cells are able to withstand high levels
of shear stress; here we report experiments using
shear stress levels up to 42 dyn/cm
2
.
In this section we give a brief overview of the
theory of dielectrophoresis, the design of the
microfluidic platform used to develop discontinuous
dielectrophoresis, the procedure of discontinuous
dielectrophoresis, and analysis of the discontinuous
dielectrophoresis, in terms of trapping efficiency of
immobilised cells. Taking note, that Saccharomyces
cerevisiae yeast cells are used as our model cell,
which is commonly used to show proof of concept
technologies and known to be non-adherent.
2.1 Dielectrophoresis Overview
Dielectrophoresis is a phenomenon, in which a non-
uniform electric field is used to induce motion into
polarisable particles; consequently, label-free
manipulation can be achieved. Currently
dielectrophoresis systems have been demonstrated
for the manipulation, sorting, immobilisation, and
characterisation of a variety of bio-particles.
The response of a particle in an electric field is
governed by the dielectric properties, such as
structural, morphological, and chemical
characteristics. Furthermore, the time average
dielectrophoretic force (<F
DEP
>) experienced on a
spherical particle developed by Morgan and Green,
is governed by the following equation (Morgan and
Green, 2003, Chapter 4):
〈
〉
=2
∇
|
|
(1)
Where r is the radius of the cell, ɛ
o
and ɛ
med
, are
the permittivity of free space (8.854x10
-12
F/m) and
the suspension medium, respectively; in addition
Re{f
CM
}, is the real component of the Clausius-
Mossotti factor, and E
rms
, is the electric field root-
mean-squared. A more extensive analysis is
presented in the succeeding subsection, for the
electric field, and dielectrophoretic force induced by
the interdigital microelectrodes.
The Clausius-Mossotti factor (f
CM
) for a
homogenous spherical structure is given by (Morgan
and Green, 2003, Chapter 3):
=
∗
−
∗
∗
+2
∗
,
(2)
where, complex permittivity, ɛ
*
, is given by:
∗
=−
,=
√
−1,
(3)
where, ɛ
*
cell
and ɛ
*
med
, are the complex
permittivities of the cell and suspension medium,
respectively; in addition, σ, is the electrical
conductivity, and ω, is the angular frequency of the
applied signal. The real part of the complex variable
Clausius-Mossotti factor, provides an indication of
the behaviour of a particle within the electric field at
various medium.
Furthermore, this behaviour is presented in
Figure 1a for three different medium conductivities
(200, 500, 1000 µS/cm) over a frequency range of
10
4
to 10
8
Hz, calculated with the geometric and
dielectric properties for the Saccharomyces
cerevisiae yeast cell as given in Table 1. The cells
were suspended in an isotonic low electrical
conductivity (LEC, 8.5% w/v sucrose, 0.3%
dextrose, ~ 100 µS/cm) buffer, with the conductivity
adjusted with the addition of phosphate-buffered
saline (PBS). Such that when the real component of
the Clausius-Mossotti factor is positive, the cells are
attracted to the microelectrodes, a phenomenon that
is commonly known as positive dielectrophoresis
(Figure 1b). On the other hand, when the real
component of Clausius-Mossotti factor is negative,
the cells are repelled from the microelectrodes; a
phenomenon more commonly known as negative
dielectrophoresis (Figure 1b).
The Clausius-Mossotti factor response (Figure
1) is heavily influenced by the complex permittivity
of the cell (ɛ
*
cell
). In our case we are using a yeast
cell, which has a cell wall, thus, a two shell model
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
24

Table 1: Geometric and dielectric properties of viable
yeast cells (Urdaneta and Smela, 2007).
Parameter Value
Cell diameter 8 µm
Membrane thickness 8 nm
Wall thickness 220 nm
Cytoplasm conductivity 0.2 S/m
Cytoplasm permittivity 50ɛ
o
F/m
Membrane conductivity 25e-8 S/m
Membrane permittivity 6ɛ
o
F/m
Wall conductivity 14e-3 S/m
Wall permittivity 60ɛ
o
F/m
Figure 1: (a) The real part of Clausius-Mossotti factor
response to frequency for yeast cells at medium
conductivities of (i) 200, (ii) 500, and (iii) 1000 µS/cm, for
cells suspended in LEC, with conductivities adjusted with
PBS. Schematic representation of: (b) positive, and (c)
negative dielectrophoresis.
encompassing the geometric and dielectric
properties of the yeast cells given in Table 1, which
is given by (Huang et al., 1992):
∗
=
∗
+2
∗
−
∗
∗
+2
∗
−
∗
−
∗
∗
+2
∗
,
(4)
∗
=
∗
+2
∗
−
∗
∗
+2
∗
−
∗
−
∗
∗
+2
∗
.
(5)
Where, ɛ
*
cyto
, ɛ
*
mem
, ɛ
*
wall
, and ɛ
*
cyto-mem
are the
complex permittivities of the cell cytoplasm,
membrane, wall, and the equivalent combined
homogenous cell cytoplasm and membrane,
respectively.
2.2 Dielectrophoresis Platform Design
The microfluidic platform consists of two main
components, the microelectrodes and the
microchannel; both are designed to facilitate the
maximum immobilised cell population visible when
using microscopy techniques (Figure 2). In the
following subsections the design and fabrication
procedure will be outlined; including, the analysis of
the resulting electric field and shear stress contours
produced through the microfluidic channel, when
applying a flow rate between 0 and 120 µl/min,
corresponding to a shear stress range from
0 to 42 dyn/cm
2
.
Figure 2: (a) Overview of the device design:
(i) Photograph of the microfluidic platform; schematic
with corresponding relevant dimensions of features of:
(ii) Plan view of the setup overview; (iii) Microchannel;
and (iv) Microelectrodes.
2.2.1 Microelectrode Design and Fabrication
To maximise the uniformity of the patterning of
immobilised cells an interdigital microelectrode
design was invoked, as presented in Figure 2.
Consequently, this maximised the cell population
immobilised within microscopic imagining range,
under a 10× objective (with a 1.5 × multiplier) on a
Nikon Eclipse (TE 2000). The microelectrodes were
designed to have a gap and width of 40 µm, which is
the smallest possible feature resolution available to
us to fabrication limitations. Furthermore, the active
region was designed to be 300 µm, such that
spanning the entire imaging range.
The microelectrodes were fabricated in two
stages. Initially the thin films where fabricated using
evaporation on a glass microscope slide, using a
gold on chrome process, at thickness of 1500 and
500 Å, respectively. The microelectrodes where then
patterned using standard microfabrication
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress
25

techniques, including photolithography and wet
etching (Nasabi et al., 2013).
2.2.2 Electric Field Analysis
To determine the influence of the electric field on
the time averaged dielectrophoretic force
experienced on a spherical cell, the electric field
contours are determined (Figure 3). Laplace
equations are solved within the microfluidic channel
(design presented in succeeding subsection), through
applying electric potentials accordingly, to
determine the electric field contours.
Figure 3: (a) Contours of the gradient of the square of the
electric field (E, V
2
/m
3
) and corresponding
dielectrophoretic force (<F
DEP
>, N).
Using the general rule of thumb that there is zero
electric flux along the surfaces of the microchannel,
other than the glass substrate (microscope slide)
where the microelectrodes are fabricated; thus, flux
is zero along the sidewalls and top of the
microchannel. Thus, resulting in a flux relationship
given as:
∇
=0.
(6)
Additionally, the electric field strength (E) is
determined by taking the gradient of the electric
potential (ϕ), resulting in the following relationship:
=−∇
.
(7)
Furthermore, as seen in Equation 1 the time average
dielectrophoretic force is proportional to the electric
field strength as given by (Figure 3):
〈
〉
∝∇
,
(8)
Simulations indicated the maximum force
experienced on the cells was 1.40e
-10
N (Figure 3).
2.2.3 Microchannel Design and Fabrication
Microchannel was designed to encompass the width
of the active electric field, being 300 µm
(Figure 2). Consequently, the width of the
microchannel was designed to have a width of
500 µm, to ensure that the strongest section of the
electric field (tip region of the microelectrodes) was
not within the imaging region. Furthermore, the
channel was designed to have an arbitrary height of
80 µm, which allowed cells to immobilise along the
microelectrodes, and cells and suspension to wash
over the immobilised cells without dislodging them.
The polydimethylsiloxane (PDMS) microchannel
(500 x 80 µm) was fabricated using standard soft
lithography and replica molding techniques
(Whitesides et al., 2001). The PDMS was cured
using a standard ratio of Sylgard 184, with the base
to curing agent ratio 10:1 (Dow Corning
Corporation, MI).
2.2.4 Velocity and Shear Stress Profiles
In order to determine the shear stress being applied
to the immobilised cells on the microelectrodes,
computational fluidic dynamic simulations were
carried out (Figure 4).
Figure 4: Contours of the shear stress across the substrate.
Given that the flow is laminar and the liquid is
assumed to be Newtonian the following equations
apply. The continuity equation is given by:
∇∙=0.
(8)
Furthermore the momentum of the liquid is given by:
∙∇
=−∇+
∇
.
(9)
Where, U, P, ρ
liquid
, and µ
liquid
, are velocity, pressure,
density, and dynamic viscosity of the liquid,
respectively. Note that the assumed boundary
conditions of ambient pressure at the inlet, desired
flow rate through entire microchannel, and no-slip at
the sidewalls is used to evaluate these equations. To
determine the shear stress over the glass substrate
(microscope slide), one assumes that the
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
26

immobilised cells do not influence the
hydrodynamic properties of the cells; thus, the
resulting shear stress () is given by:
τ
|
_
=
.
(10)
Furthermore, the resulting shear stress profiles can
be determined along the glass substrate; such as
presented in Figure 4 for a flow rate of 120 µl/min,
corresponding to a shear stress of 42 dyn/cm
2
. The
relationship of flow rate (Q) and shear stress for our
microfluidic platform is given by:
=
67.5
,
(11)
where, W and H, are the width and height of the
microchannel. Additionally, the relationship drag
force exerted on a cell is given by is given by:
Drag = 4
.
(12)
The maximum reported flow rate of 120 µl/min,
corresponds to a drag force of
8.40e
-10
N.
2.3 Discontinuous Dielectrophoresis
Procedure
The procedure required for discontinuous
dielectrophoresis, is systematic; however, in some
cases slightly different tactics need to be used, such
as when using stains that are sensitive to shear
stress. The fundamental discontinuous
dielectrophoresis procedure is presented in Figure 5
and each stage will be discussed in detail in the
subsections.
2.3.1 Sample Preparation and Application
Samples need to be suspended in an isotonic low
electrical conductivity (LEC) buffer, composed of
8.5% w/v sucrose, and 0.3% dextrose in deionised
water; ensuring that the final suspension
conductivity is 200 µS/cm. In necessary, the solution
conductivity can be increased through the addition
of phosphate-buffered saline, or any other relevant
biological buffer. The cell suspension is then
transpired to the inlet reservoir of the microfluidic
platform at a flow rate of 2.5 µl/min (Figure 2). A
low flow rate is used to ensure that cells can be
immobilised once the electric field is activated.
Additionally, this is advantageous when doing high
shear stress analysis, as this minimises the pre-
exposure to shear stress. In the case for yeast cells
for a 100 ml volume, we added 20 mg of dried S.
cerevisiae yeast cells (Sigma-Aldrich).
Figure 5: Schematic representation of procedure used for
the discontinuous dielectrophoresis strategy:
(a) Activation of the electric field for 120 s, excited by a
10 MHz 5 V
pk-pk
sinusoid, to immobilise the cells along the
microelectrodes; with the cell suspension flown over the
microelectrodes at a flow rate of 2.5 µl/min;
(b) Deactivation of the electric field after 120 s of
activation, with the flow rate remaining consistent;
(c) Once cells have stabilised on the surface and reached
equilibrium the flow rate can be increased to the desired
flow rate, such as 60 µl/min.
2.3.2 Activation of the Electric Field
Once cells are consistently flowing over the
microelectrodes, the microelectrodes are activated,
through the application of a 10 MHz sinusoid
operating at 5 V
pk-pk
. The microelectrodes are kept
active for a period of 120 s (Figure 5a). This was to
minimise the duration of the electric field, thus
minimising any harmful effects on the cells due to
being within an electric field. Furthermore, the cell
population can be controlled by an increase in the
activation duration of the electric field; however, we
recommend increasing the initial cell population.
The dielectrophoretic and shear forces on the cell
influence the capacity of the cell to remain
immobilised to the substrate. Such that the cells are
immobilised using a flow rate of 2.5 µl/min, which
corresponds to a shear stress of 0.875 dyn/cm
2
,
resulting in a drag force of 1.77e
-11
N being exerted
on the cells. However, the force dielectrophoretic
force exerted on the cells is determined to be
1.40e
-10
N (Figure 3). Consequently, as the
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress
27

dielectrophoretic force is greater than the drag force
generated, the cells are immobilised on the substrate;
rather than being washing directly over the electrode
in the event of a higher flow rate, which produces a
greater shear stress and subsequent force.
2.3.3 Deactivation of the Electric Field
Once the field has been active for 120 s, the electric
field is turned off. Deactivation of the electric field,
resulted in the additional layers of cells and cells not
correctly immobilised in the first layer being
dislodged and washed away. Consequently, a single
layer of immobilised cells remained, in the
imagining range (Figure 5b). In general, it was
observed that non-viable cells would not initially
immobilise or was dislodged with increasing flow
rate. Once the electric field is deactivated, the cell
suspension can be exchanged to a suitable biological
imaging media, in our case we exchanged for
HEPES. The cells were then left for five minutes to
stabilise in their immobilised location and reach
equilibrium, especially in the occurrence that the
media was exchanged to a biologically relevant
media. Once these criteria were met, the flow rate
was increased to the desired flow rate and resulting
shear stress, as presented in Figure 5c for 60 µl/min.
Furthermore once the electric field is inactivated,
the dielectrophoretic force no longer influences the
forces experienced on the cell. In the event the flow
rate is increased to 120 µl/min, corresponding to a
drag force of 8.40e
-10
N. This force is considerably
larger than the maximum dielectrophoretic force,
demonstrating the adhesive attraction between the
cell surface and the glass substrate produced during
the discontinuous dielectrophoresis procedure.
2.3.4 Yeast Trapping Efficiency
To analyse the effectiveness of discontinuous
dielectrophoresis, we investigated the trapping
efficiency. Initially the cells are immobilised using
the aforementioned procedure, then the flow rate of
the system is increased sequentially in three minute
intervals, at various flow rates between 2.5 and
100 µl/min. Trapping efficiency was evaluated as:
ɳ
=
_
.100%,
(13)
where, n
remaining_cells
, and n
intial
are the number of
remaining and initial cell counts of immobilised
viable cells.
Analysis of the trapping efficiency of yeast cells
was carried out using three different exciting
waveform voltages, when supplying the electric field
for the 120 s duration. The three selected voltages
selected where 2.5, 5, and 10 V
pk-pk
, as presented in
Figure 6a for cells suspended in LEC. Additional
analysis was carried out using the optimal voltage of
5 V
pk-pk
, in which the suspension media was
exchanged for HEPES after deactivation of the
electric field. For the scenario that the cells were
kept in LEC, the change in voltage provided no
significant change in the trapping efficiency which
was determined to be 27% when using a 5 V
pk-pk
sinusoid. However, when the suspension media was
exchanged for HEPES when immobilisation was
achieved using a signal operating at 5 V
pk-pk
the
trapping efficiency increased to 82%. This was
conjectured to be attributed to the additional ions
present in HEPES compared to LEC.
Figure 6: Yeast trapping efficiency analysis (a) Influence
of the exciting waveform voltage used during electric field
activation, at (i) 2.5, (ii) 5, and, (iii)10 V
pk-pk
. (a) Influence
of the medium used after deactivation of the electric field
both conducted at an exciting waveform voltage of
5 V
pk-pk
, in (i) LEC, and (ii) HEPES.
3 APPLICATION
To highlight the functionality of our system, we
investigated the response of intracellular calcium
signalling of HEK-293-TRPV4 cells to shear
induced stress. Shear stress is one of many stimuli
that cells respond to, others include, thermal and
capsicum, for example. These stimuli regulate
various biological processes, such as proliferation,
apoptosis, transcription, and proliferation (Ahern,
2013, Jaalouk and Lammerding, 2009, Polacheck et
al., 2013, Lin and Corey, 2005).
HEK-293 cells are transfected to express the
TRPV4 (transient receptor potential vaniloid)
mechanosensitive ion channel, as HEK-293 cells are
limited in the ion channels it expresses. TRPV4 ion
channels are of interest as they play a role in
controlling vascular homeostasis and tone (Mendoza
et al., 2010, Nilius et al., 2004, Baratchi et al., 2014).
Furthermore, studies into the influence of shear
stress has been restricted as using current methods
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
28

for used for HEK-293 cells limits the maximum
level of shear induced stress without extensive cell
dislodgement.
3.1 HEK-293-TRPV4 Cell Preparation
HEK-293 T-REx (Life Sciences) cell lines where
prepared off-chip, will full experimentation
including viability assays carried out on the
microfluidic platform. Firstly, the cells were grown
in tetracycline-free DMEM media supplemented
10% FBS, blasticidin (5 µg/ml) and hygromycin
(50 µg/ml). Secondly, 12 hours before
experimentation, the TRP channel expression was
induced in the HEK-293 cells using 0.1 µg/ml of
tetracycline (Poole et al., 2013). Taking note, that
HEK-293 expresses a limited number of TRP
channels; thus, making HEK-293 a good candidate
to analysis calcium influx through the TRPV4
channel. Additionally, non-transfected HEK-293 T-
REx cells were used as a negative control; results
not presented here, trapping efficiency was not
affected by being non-transfected, additionally no
response was observed in the occurrence of shear
induced stress.
In the case for investigating shear induced
intracellular calcium signalling, the cells required
further preparation at the time of experimentation;
such that the cells were loaded with Fluo-4AM, and
suspended in HEPES-buffered saline solution
(140 mM NaCl, 5 mM KCl, 10 mM HEPES, 11 mM
D-glucose, 1 mM MgCl
2
, 2 mM CaCl
2
, and 2 mM
probenecid, adjusted to pH 7.4). After bring
incubated at 37
o
C
for 30 minutes, 25 µl of the HEK-
293-TRPV4 cell suspension is suspended in 1000 µl
of LEC, making sure not to gently mix the solution,
to minimise pre-exposure to shear induced stress.
Additionally, the viability of the cells was
examined on-chip with propidium iodide (PI)
(10 µg/ml) staining, at the completion of each
experiment.
3.2 Experimental Procedure
Considerations
Initially cells needed to be characterised on the
designed dielectrophoresis platform, such that the
Clausius-Mossotti factor response to frequency was
investigated. The effect of the technique of
discontinuous dielectrophoresis was compared to
conventional dielectrophoresis, to highlight the
significant improvement on mortality rates of cells.
Furthermore, the trapping efficiency was
investigated, to ensure that discontinuous
dielectrophoresis was going to be suitable for
investigating high shear induced stress. Finally, once
the response of HEK-293 cells to discontinuous
dielectrophoresis was characterised, shear induced
intracellular calcium signalling was investigated, at
shear stress levels of 0.875 and 42 dyn/cm
2
.
3.2.1 HEK-293 Clausius-Mossotti Factor
The crossover frequency for HEK-293 cells was
examined on chip for three different suspension
medium conductivities. Consequently, the measured
crossover frequencies were determined to be 55 ± 7,
145 ± 17, and 285 ± 45 MHz, corresponding to
conductivities of 200, 500, and 1000 µS/cm,
respectively. Conductivities were achieved by
adjusting the initial LEC buffer with the addition of
PBS. Once the crossover frequencies were obtained,
the Clausius-Mossotti factor can be determined for
the three conductivities over the frequency range of
10
4
to 10
8
Hz, as presented in Figure 7, taking note
the geometric and dielectric properties of HEK-293
cells presented in Table 2. Furthermore, the
equivalent single shell model is used, as expressed in
Equation 4, unlike yeast, as HEK-293 cells do not
have a cell wall. Such that dielectrophoresis
experiments involving HEK-293 cells, were
conducted using a conductivity of
200 µS/cm, using an operating frequency of
10 MHz, which is well within the positive
dielectrophoresis range (Figure 7ai). Additionally,
the combined use of this medium conductivity and
operating frequency minimised the manifestation of
electrothermal effects, such as vortices and
electrolysis.
Table 2: Geometric and dielectric properties of viable
HEK-293 cells used to determine dielectrophoresis
behaviour (Soffe et al., 2015a).
Parameter Value
Cell diameter 12.5 µm
Membrane thickness 7 nm
Cytoplasm conductivity 0.5 S/m
Cytoplasm permittivity 60ɛ
o
F/m
Membrane conductivity 7e-14 S/m
Membrane permittivity 9.5ɛ
o
F/m
3.2.2 HEK-293 Cell Viability
Viability assays for HEK-293 were conducted under
three different environmental scenarios, for a period
of 60 minutes, with the microelectrodes excited with
a 10 MHz sinusoid operating at 5 V
pk-pk
(Figure 8).
Viability assays were limited to 60 minutes, as the
longest duration for a shear stress experiment was 30
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress
29
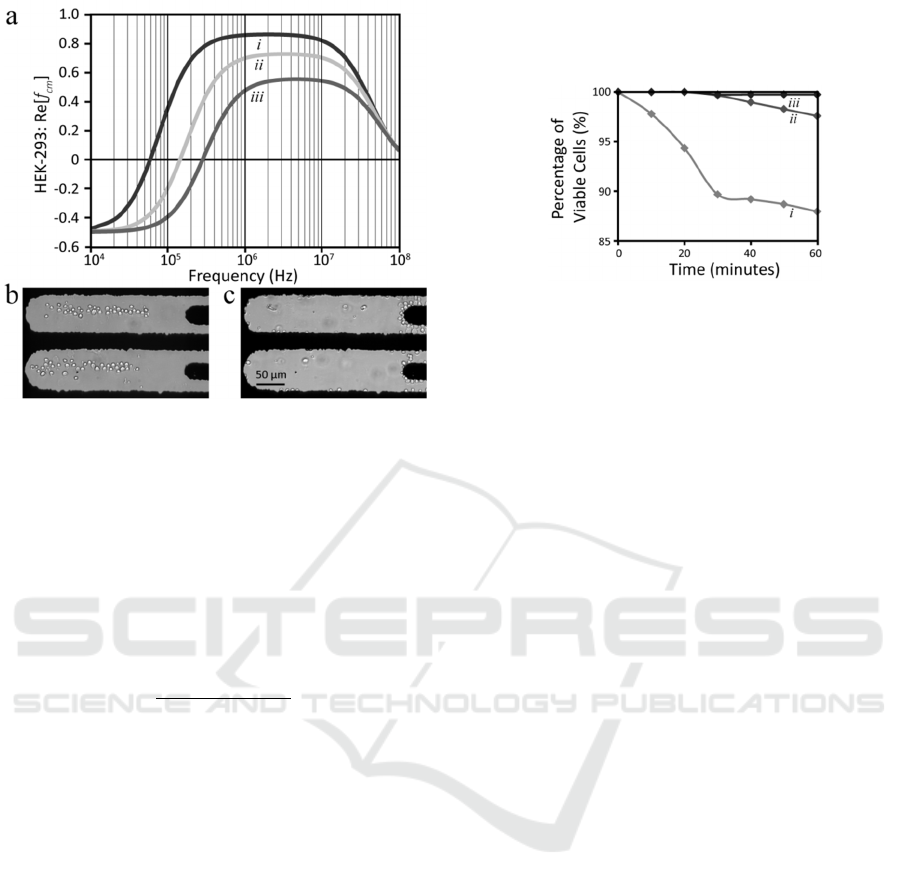
Figure 7: (a) The real part of Clausius-Mossotti factor
response to frequency for HEK-293 cells at medium
conductivities of (i) 200, (ii) 500, and (iii) 1000 µS/cm.
Conductivities were achieved by adjusting the initial LEC
buffer with the addition of PBS Experimental images of
(b) positive; and (c) negative dielectrophoresis; taken
when determining the crossover frequency for HEK-293
cells.
minutes, so 60 minutes should suffice. Cell viability
was determined by evaluating, the following
equation:
ɳ
_
=
_
_
.100%,
(14)
where, n
remaining_viable
, and n
intial_viable
are the number
of remaining and initial cell counts of viable cells.
Non-viable cells were determined through PI
staining, and excluded from the viable cell count.
Scenario one (Figure 8i), conventional
dielectrophoresis, such that the cells are suspended
in LEC and the electric field is activated for the
entire 60 minutes and two additional minutes to be
comparable to the other two scenarios. In scenario
two (Figure 8ii), the electric field was activated for
a period of 120 s, and the cells are suspended in
LEC. In scenario three (Figure 8iii), the electric
field was activated for a period of 120 s, and the
cells are suspended in HEPES once the field is
deactivated.
The viability assays indicated that the continued
presence of the electric field (Scenario one)
significantly affects the viability of cells, such that
88% of cells are viable. However, the viability
percentage is significantly increased when
discontinuous dielectrophoresis is invoked; such that
approximately 98% of the cells are viable regardless
of cell suspension. Although cells suspended in
HEPES have a slightly higher viability rate
(Scenario Three).
Figure 8: HEK-293-TRPV4 cell viability analysis using
PI; conducted using three different scenarios:
(i) Conventional dielectrophoresis with cells suspended in
LEC (constant electric field activation); (ii) Discontinuous
dielectrophoresis with cells suspended in LEC; and
(iii) Discontinuous dielectrophoresis with cells suspended
in HEPES.
3.2.3 HEK-293 Trapping Efficiency
A more extensive trapping efficiency analysis was
carried out for HEK-293, due to the cell being more
susceptible to environmental influences, such as: the
supplied waveform parameters during electric field
application; and the suspension media, which assists
in regulating cellular behaviour. The influence of the
exciting waveform was examined, using three
different voltages, being 2.5, 5, and
10 V
pk-pk
for three different scenarios
(Figure 9a-c), with the trapping efficiency
determined using Equation 13. In scenario one
(Figure 9a), conventional dielectrophoresis, a
supply voltage of 5 V
pk-pk
produced the most
effective trapping efficiency of 94%. However, in
scenario two (Figure 9b), discontinuous
dielectrophoresis was invoked, with the cells
suspended in LEC, all initially immobilised cells
were dislodged at a flow rate of 100 µl/min
(35 dyn/cm
2
). Furthermore, in scenario three
(Figure 9c), discontinuous dielectrophoresis was
invoked, with the cells suspended in HEPES. In this
scenario, the percentage of immobilised cells
significantly increased, regardless of the supplied
voltage, to a trapping efficiency between 84% and
90%. In comparison to scenario two, this indicated
that the presence of a biologically relevant media, is
crucial in ensuring cells remain immobilised.
Optimal conditions presented in Figure 9d, the
three aforementioned scenarios, are displayed for a
supply voltage of 5 V
pk-pk
(Figure 9di-iv).
Additionally, a control comparison was conducted
(Figure 9di), achieved by allowing cells suspended
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
30
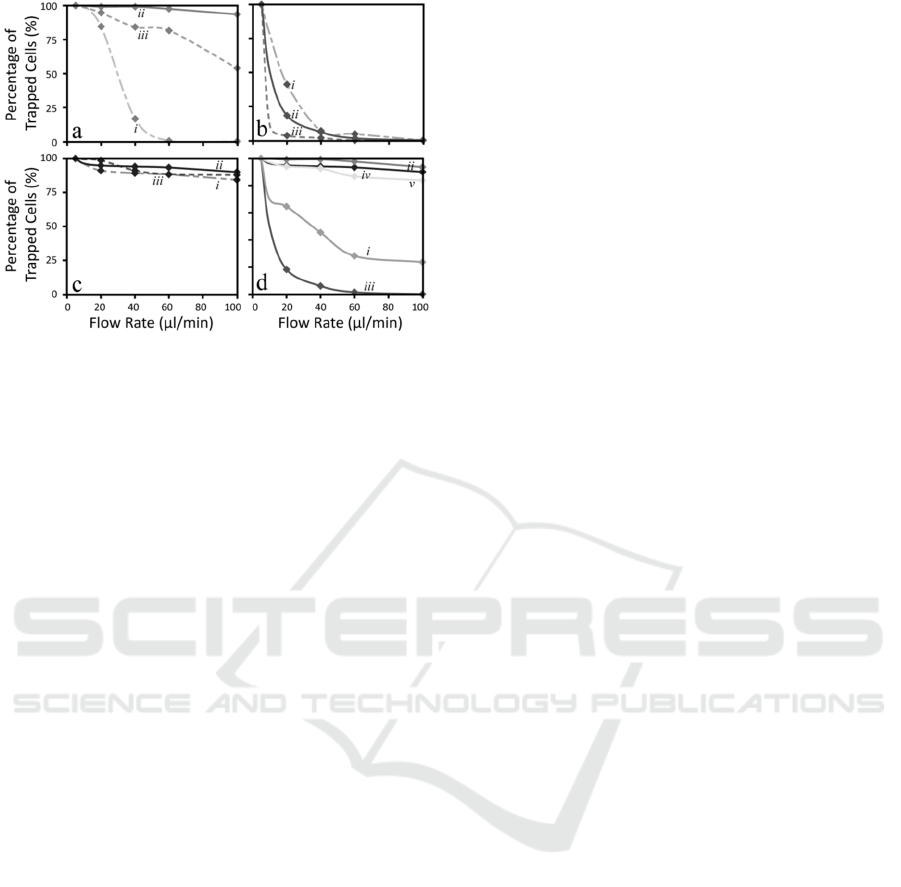
Figure 9: Trapping efficiency of HEK-293-TRPV4 under
different environmental scenarios. For (a) Conventional
dielectrophoresis with cells suspended in LEC (constant
electric field activation); (b) Discontinuous
dielectrophoresis with cells suspended in LEC; and
(c) Discontinuous dielectrophoresis with cells suspended
in HEPES. Furthermore, for each scenario the exciting
waveform voltage used during electric field activation was
examined at (i) 2.5, (ii) 5, and (iii) 10 V
pk-pk
.
(d) Conducted under different environmental scenarios in
optimal conditions (supplying sinusoid waveform
operating at 10 MHz 5 V
pk-pk
): (i) Control, with no
dielectrophoresis with cells suspended in HEPES;
(ii) Conventional dielectrophoresis with cells suspended in
LEC; (iii) Discontinuous dielectrophoresis with cells
suspended in LEC; (iv) Discontinuous dielectrophoresis
with cells suspended in HEPES; and (v) Discontinuous
dielectrophoresis with cells suspended in HEPES using a
PDMS substrate instead of a glass substrate.
in HEPES to rest on the non-treated glass substrate
for 30 minutes; thus, simulating cells being
immobilised by dielectrophoresis. At a flow rate of
100 µl/min, equivalent to 35 dyn/cm
2
, the control
experiments led to a trapping efficiency of 24%,
which was a significant improvement of cells
suspended in LEC using discontinuous
dielectrophoresis (scenario two), in which case all
the cells were dislodged. However, cells either
trapped using conventional dielectrophoresis or
discontinuous dielectrophoresis with cells suspended
in HEPES, resulted in the optimal trapping
efficiencies.
An additional comparison was made to examine
the importance of the substrate, such that
discontinuous dielectrophoresis was invoked, using
a platform with microelectrodes fabricated on
PDMS on glass substrate (Figure 9dv) (Nasabi et
al., 2013). These results indicated that,
discontinuous dielectrophoresis when using a
biological relevant media is produces the highest
trapping efficiency, when applying high shear
stresses, such as 35 dyn/cm
2
(100 µl/min).
Although, the application of conventional
dielectrophoresis using a supply voltage of 5 V
pk-pk
,
resulted in the highest trapping efficiency (93%), the
continued electric field application affected cell
viability as presented in Figure 8. Thus,
conventional dielectrophoresis was determined not
suitable for shear stress analysis of HEK-293 cells.
On the other hand, discontinuous dielectrophoresis,
with a slightly lower trapping efficiency of 90%,
minimised effects on cell viability (Figure 8), and
enabled experimentation to be carried out in a
biologically relevant media (HEPES).
3.3 Intracellular Calcium Signalling
Analysis of HEK-293-TRPV4
Intracellular calcium signalling is important as it
facilitates in the regulation of several biological
processes. The calcium ion (Ca
2+
) is of importance,
as it regulates a variety of spatial and temporal
signals. The movement of calcium ions is facilitated
through the stimulation of permeable ion channels,
such as the TRPV4 ion channel. The influx of
calcium ions through calcium permeable ion channel
into the plasma membrane, occurs due an induced
stimulation of their selective stimulus, such as shear
stress a form of mechanical stimulation (Mendoza et
al., 2010, Baratchi et al., 2014). The level of
intracellular calcium level ([Ca
2+
]
i
), due to calcium
influx is measured through the use of calcium
sensitive dyes.
A comparison was undertaken of the influence of
shear stress on the behaviour of intracellular calcium
influx, using HEK-293 cells expressing TRPV4.
Cells were prepared off-chip with Fluo-4AM, a
calcium sensitive dye, as outlined previously.
Intensity measurements were then acquired using an
inverted microscope, equipped with a
photomultiplier tube, a near infrared camera
(QuantEM:512SC, Photometrics), and a 10×
objective (CFI Plan Apo Lambda 10×). With the
assistance of NIS Elements, microscope imaging
software (Basic Research, Nikon Instruments), the
intensity measurements were able to be processed.
Intracellular calcium signalling analysis was then
carried out using discontinuous dielectrophoresis
with cells suspended in HEPES, with the
microelectrodes excited with a 10 MHz sinusoid
operating at 5 V
pk-pk
. The influence of shear stress on
intracellular calcium signalling through the TRPV4
ion channel, was conducted by subjecting
immobilised cells to shear stress for a period of
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress
31

Figure 10: HEK-293-TRPV4 cell response to shear stress
achieved using flow rates of (i) 2.5, and (ii) 120 µl/min.
With the retrospective images of the immobilised cells in:
(a) Bright field; (b) Fluorescent images of cells loaded
with Fluo-4AM obtained at 60, 300, and 720 s; and (c) PI
fluorescent images taken at 1020 s; 300 s after the addition
of PI to the microfluidic platform. (d) Corresponding
normalised intensity profile over period of 720 s.
720 s, and measuring the intensity of the calcium
dye (Fluo-4AM) (Figure 10). A shear stress of
42 dyn/cm
2
(120 µl/mi), was selected, as this shear
stress is in the upper region of the physiological
shear stress range (Figure 10ii); consequently,
highlighting the capability of discontinuous
dielectrophoresis of cells remaining immobilised
under high levels of shear stress. The resulting
intensity profiles where compared against a
negligible shear stress of 0.875 dyn/cm
2
(2.5 µl/min), to maintain the cells with a fresh
supply of HEPES (Figure 10i).
A shear stress level of 42 dyn/cm
2
resulted in a
percentage of activated cells of 73.1 ± 12.5%, and
maximum fold increase in [Ca
2+
]
i
of 2.27 ± 0.07. In
comparison to negligible shear stress
(0.875 dyn/cm
2
), which resulted in a percentage of
activated cells of 3.25 ± 1.2%, and a maximum fold
increase in [Ca
2+
]
i
of 1.17 ± 0.09. Furthermore, a
decrease in the cellular and peak response times was
observed with the higher shear stress level. Such that
the cellular response time decreased from 130 ± 40 s
to 77 ± 6 s, and the peak response time decreased
from 426 ± 36 s to 392 ± 18 s, for shear stress levels
of 0.875, and 42 dyn/cm
2
, respectively.
Consequently, indicating that the calcium influx
intensifies with higher shear stress levels, and the
importance of being able to investigate high shear
stress on a cellular physiological level.
4 CONCLUSIONS
Discontinuous dielectrophoresis provides a strategy
for analysing the response of loosely adherent cells
to high levels of shear stress. We have demonstrated
procedure of discontinuous dielectrophoresis using
S. cerevisiae yeast cells, and the capacity of the
immobilised cells to withstand high levels of shear
stress. We then in turn, investigated the capability of
the system using HEK-293 cells, a commonly used
cell line for biological assays. The experimental
considerations were investigated for using HEK-293
cells for discontinuous dielectrophoresis, such as:
Clausius-Mossotti factor response to frequency;
viability and trapping efficiency comparison of
conventional and discontinuous dielectrophoresis for
cells suspended in LEC or HEPEs. For cells
immobilised using discontinuous dielectrophoresis,
using HEPES as the suspension media after electric
field deactivation, resulted in a trapping efficiency
of 90% when experiencing a shear stress level of
42 dyn/cm
2
. Additionally, discontinuous
dielectrophoresis minimises cell mortality rates,
such that after a period of 60 minutes, approximately
98% of cells were deemed viable, through
propidium iodide staining.
The capacity of the system for biological
analysis under high shear stress was then
demonstrated, for investigating the influence of
shear stress on intracellular calcium signalling of
HEK-293-TRPV4 cells; which indicated the shear
stress intensifies the calcium influx. This technique
has the ability for investigating various cell
responses to high levels of shear stress, as presented
here and demonstrated for HEK-293-TRPV4 cells.
Furthermore, the platform offers the potential of
parallelism, and dynamic analysis through changing
the microenvironment within the microchannel, such
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
32

as thermal stimuli, in the presence of high shear
stress.
ACKNOWLEDGEMENTS
Khashayar Khoshmanesh acknowledges the
Australian Research Council for funding, under the
Discovery Early Career Researcher Award
(DECRA) scheme, (project DE120101402).
REFERENCES
Ahern, G. P. 2013. Transient receptor potential channels
and energy homeostasis. Trends in Endocrinology &
Metabolism, 24, 554-560.
Baratchi, S., Tovar-Lopez, F. J., Khoshmanesh, K., Grace,
M. S., Darby, W., Almazi, J., Mitchell, A. & Mcintyre,
P. 2014. Examination of the role of transient receptor
potential vanilloid type 4 in endothelial responses to
shear forces. Biomicrofluidics, 8, 044117(1-13).
Berthier, E., Young, E. W. K. & Beebe, D. 2012.
Engineers are from PDMS-land, Biologists are from
Polystyrenia. Lab on a Chip, 12, 1224-1237.
Ding, X., Lin, S. C., Kiraly, B., Yue, H., Li, S., Chiang, I.
K., Shi, J., Benkovic, S. J. & Huang, T. J. 2012. On-
chip manipulation of single microparticles, cells, and
organisms using surface acoustic waves. Proceedings
of the National Academy of Sciences, 109, 11105-9.
El-Ali, J., Sorger, P. K. & Jensen, K. F. 2006. Cells on
chips. Nature, 442, 403-411.
Huang, Y., Holzel, R., Pethig, R. & Xiao, B. W. 1992.
Differences in the AC electrodynamics of viable and
non-viable yeast cells determined through combined
dielectrophoresis and electrorotation studies. Physics
in Medicine and Biology, 37, 1499.
Jaalouk, D. E. & Lammerding, J. 2009.
Mechanotransduction gone awry. Nature reviews
Molecular cell biology, 10, 63-73.
Lin, S.-Y. & Corey, D. P. 2005. TRP channels in
mechanosensation. Current opinion in neurobiology,
15, 350-357.
Mendoza, S. A., Fang, J., Gutterman, D. D., Wilcox, D.
A., Bubolz, A. H., Li, R., Suzuki, M. & Zhang, D. X.
2010. TRPV4-mediated endothelial Ca2+ influx and
vasodilation in response to shear stress. American
Journal of Physiology-Heart and Circulatory
Physiology, 298, H466-H476.
Morgan, H. & Green, N. 2003. AC electrokinetics:
colloids and nanoparticles. Baldock, England:
Research Studies Press Ltd.
Mutreja, I., Woodfield, T. B. F., Sperling, S., Nock, V.,
Evans, J. J. & Alkaisi, M. M. 2015. Positive and
negative bioimprinted polymeric substrates: new
platforms for cell culture. Biofabrication, 7, 025002.
Nahavandi, S., Tang, S.-Y., Baratchi, S., Soffe, R.,
Nahavandi, S., Kalantar-Zadeh, K., Mitchell, A. &
Khoshmanesh, K. 2014. Microfluidic Platforms for the
Investigation of Intercellular Signalling Mechanisms.
Small, 10, 4810-4826.
Nasabi, M., Khoshmanesh, K., Tovar-Lopez, F. J.,
Kalantar-Zadeh, K. & Mitchell, A. 2013.
Dielectrophoresis with 3D microelectrodes fabricated
by surface tension assisted lithography.
ELECTROPHORESIS, 34, 3150-3154.
Nilius, B., Vriens, J., Prenen, J., Droogmans, G. & Voets,
T. 2004. TRPV4 calcium entry channel: a paradigm
for gating diversity. American Journal of Physiology-
Cell Physiology, 286, C195-C205.
Polacheck, W. J., Li, R., Uzel, S. G. & Kamm, R. D. 2013.
Microfluidic platforms for mechanobiology. Lab on a
Chip, 13, 2252-2267.
Poole, D. P., Amadesi, S., Veldhuis, N. A., Abogadie, F.
C., Lieu, T., Darby, W., Liedtke, W., Lew, M. J.,
Mcintyre, P. & Bunnett, N. W. 2013. Protease-
activated Receptor 2 (PAR(2)) Protein and Transient
Receptor Potential Vanilloid 4 (TRPV4) Protein
Coupling Is Required for Sustained Inflammatory
Signaling. Journal of Biological Chemistry, 288,
5790-5802.
Soffe, R., Baratchi, S., Tang, S.-Y., Nasabi, M., Mcintyre,
P., Mitchell, A. & Khoshmanesh, K. 2015a. Analysing
the calcium signalling of cells under high shear flows
using discontinuous dielectrophoresis Scientific
Reports, 5, 11973.
Soffe, R., Tang, S.-Y., Baratchi, S., Nahavandi, S., Nasabi,
M., Cooper, J. M., Mitchell, A. & Khoshmanesh, K.
2015b. Controlled Rotation and Vibration of Patterned
Cell Clusters Using Dielectrophoresis. Analytical
Chemistry, 87, 2389-2395.
Urdaneta, M. & Smela, E. 2007. Multiple frequency
dielectrophoresis. ELECTROPHORESIS, 28, 3145-
3155.
Voldman, J. 2006. Engineered systems for the physical
manipulation of single cells. Current opinion in
biotechnology, 17, 532-537.
Whitesides, G. M., Ostuni, E., Takayama, S., Jiang, X. &
Ingber, D. E. 2001. SOFT LITHOGRAPHY IN
BIOLOGY AND BIOCHEMISTRY. Annual Review
of Biomedical Engineering, 3, 335-373.
Yamamoto, K. & Ando, J. 2013. Endothelial cell and
model membranes respond to shear stress by rapidly
decreasing the order of their lipid phases. Journal of
cell science, 126, 1227-1234.
Yamamoto, T. & Fujii, T. 2007. Active immobilization of
biomolecules on a hybrid three-dimensional
nanoelectrode by dielectrophoresis for single-
biomolecule study. Nanotechnology, 18, 495503.
Discontinuous Dielectrophoresis - A Technique for Investigating the Response of Loosely Adherent Cells to High Shear Stress
33
