
Spectral and Lifetime Measurements of the Endogenous Fluorescence
Variation of Freshly Resected Human Samples over Time
Measuring Endogenous Fluorescence Changes at Different Moment after
Tumor or Epileptic Cortex Excision
M. Zanello
1,2
, A. Ibrahim
1
, F. Poulon
1
, P. Varlet
3
, B. Devaux
2
and D. Abi Haidar
1,4
1
IMNC Laboratory, UMR 8165-CNRS, Orsay, France
2
Department of Neurosurgery, Sainte Anne Hospital, Paris, France
3
Department of Neuropathology, Sainte Anne Hospital, Paris, France
4
University of Paris Diderot-Paris7, Paris, France
Keywords: Lifetime Measurement, Spectroscopy, Autofluorescence, Metastasis and Cortical Human Samples.
Abstract: Analysis of human tissue endogenous fluorescence is becoming a new modality of medical imaging. Its
capacities represent the missing link between macroscopic radiological tools such as MRI and CT-scan and
the surgeon view during surgical procedures. However, numerous aspects of this signal are not well known.
Time dependence is one of these aspects. The aim of this work is to investigate the autofluorescence
changes with time. Five ex vivo human samples were studied. Spectral and lifetime measurements were
acquired each hour. Fluorescence intensity decreased slightly with time. This decrease existed for healthy
and tumoral samples and did not affect the differences between them: higher fluorescence intensity for
control samples compared to tumor samples. Lifetime values showed a slight decrease too for both type of
tissue. This work is the first report of fresh human brain samples multimodal autofluorescence analysis with
time.
1 INTRODUCTION
Progress in medical imaging change the everyday
life of doctors. Since the 1950’s, doctors had to
deliver care based only on clinical examination. If
they developed an expertise that tends to disappear
nowadays, this approach had some major
drawbacks: for instance, appendicitis diagnosis
based on clinical symptoms resulted in more than
40% of false positive diagnosis (Raman, 2008).
Neurosurgery has benefited greatly over the last
years from technologies such as computed
tomography scan, magnetic resonance imaging or
ultrasonography (Fontana, 2014). However, it is still
impossible for a neurosurgeon to know in real time
and with certitude if he performs a gross total tumor
resection. This limit can be overcome by modern
optical imaging basing on fluorescence contrast.
Analysis of the human tissue endogenous
fluorescence emission gives information on the
tissue microenvironment in real time without any
exogenous dye. If promising results were reported
(Liu, 2014), some aspects of the autofluorescence
signal are not precisely known. For instance
variation of this signal in function of time has yet to
be investigated. Two photon-imaging microscopes
are only present in laboratories nowadays.
Consequently, there is a significant time lapse
between tumor resection and optical analysis due to
the selection and transport of the sample between
operating room and microscopy laboratory. Even if
we used confocal endomicroscope during the
neurosurgical procedure, this time delay still exists:
neurooncology interventions last often several hours
and boundaries appear at the end of the intervention.
The aim of this preliminary work is to record
spectral and lifetime components of the endogenous
fluorescence on human samples and to monitor its
variation with time after excision.
2 MATERIALS AND METHODS
2.1 Optical Setup
Description of the set-up has been previously
Zanello, M., Ibrahim, A., Poulon, F., Varlet, P., Devaux, B. and Haidar, D.
Spectral and Lifetime Measurements of the Endogenous Fluorescence Variation of Freshly Resected Human Samples over Time - Measuring Endogenous Fluorescence Changes at Different
Moment after Tumor or Epileptic Cortex Excision.
DOI: 10.5220/0005654900130017
In Proceedings of the 4th International Conference on Photonics, Optics and Laser Technology (PHOTOPTICS 2016), pages 15-19
ISBN: 978-989-758-174-8
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
15

published (Abi Haidar, 2015), and it is represented
in figure 1. Briefly, The excitation source is
composed by two separated laser diodes from
PicoQuant coupled with a shutter and emitting
pulses of 70 ps centered at 405 nm (LDH-P-C-405B)
and 375 nm (LDH-P-C-375B) with a repetition rate
of 40 MHz. The laser beam is coupled into an
optical fiber from SEDI ATI Fibres optiques (HCG
M0200T) specifically dedicated to the excitation
source. It is a multimode fiber with 200 µm of core
diameter and a numerical aperture (NA) equal to
0.22, preceded by an injector coupled with a band
pass filter centered at 375 or 407 nm. The average
power is less than 1 mW at the fiber output. The
spatial resolution was of 500 µm (Leh, 2012). The
fluorescence is collected by a second multimode
fiber (HCG M0365T) with a core diameter of 365
µm and a NA equal to 0.22, separated from the first
fiber and with a collimator at its proximal output,
coupled with a high pass filter. A beam splitter
separates the collected fluorescence into two
detectors. For spectral measurements a cooled
spectrometer (QP600-1-UV-VIS, Ocean Optics) was
used. For time resolved measurements, the collected
fluorescence was guided to a Photomultiplier Tube
(PMT) (PMA-182 NM, PicoQuant GmbH, Berlin,
Germany). Temporal resolution of the PMT was 220
ps. The synchronization output signal from the diode
driver and the start signal from the PMT were
connected to their respective channels on the data
acquisition board Time-Correlated Single Photon
Counting (TCSPC) (TimeHarp 200, PicoQuant
GmbH, Berlin, Germany). Motorized filter wheel
(FW102C, Thorlabs, Newton, USA) was placed in
front of the PMT allowing the selection of spectral
emission band. With the 405 nm excitation
wavelength, we used five filters (Semrock, New
York, USA): 450 ± 10 nm, 520 ± 10 nm, 550 ± 30
nm, 620± 10 nm and 680 ± 10 nm corresponding to
five endogenous fluorophores: reduced
Nicotinamide adenine dinucleotide (NADH), flavin
(FAD), lipopigments (Lip), porphyrin (Porph) and
chlorin, respectively. With the 375nm excitation
wavelength, we worked only with two of the filters,
the 450 ± 10 nm and 520 ± 10 nm filters. Lifetime
and spectroscopic measurements were acquired on
the same set up and two seconds are required to
measure each fluorophore lifetime.
The spectral measurements were processed using
homemade Matlab software and fluorescence
lifetime data were collected and analysed via the
Symphotime software (PicoQuant, GmbH, Berlin,
Germany). A specific mechanical support was
mounted on a motorized micro translator stage
(Thorlabs, Newton, USA) for XY scanning. The X-
dimension scanning velocity was 100 µm/s and the
acquisition time during X-line scanning was 3 sec
per fluorescence spectrum.
To be as close as possible to the in vivo
conditions, this optical set-up was placed in the
Neuropathology Department of Sainte Anne
Hospital (Paris, France).
Figure 1: The optical setup for spectral and lifetime
fluorescence measurements.
2.2 Samples
Samples were provided from adult patients operated
in the Sainte Anne Hospital Neurosurgery
Department (Paris, France). The protocol was
approved by the Institutional Review Board of
Sainte Anne Hospital (Ref CPP S.C.3227). Five
samples had been analyzed: three metastasis samples
(tumor samples) and two epilepsy surgery samples
(control sample). A senior pathologist selected each
sample on a fresh resected specimen. A sample was
analyzed if: 1) there was enough material for gold-
standard histopathology; 2) resected specimen was
representative of a tumor or a healthy tissue.
2.3 Protocol and Data Acquisition
Autofluorescence measurements have been made at
different times starting from T
0
: time of the
reception of the sample in the Neuropathology
Department. The same region on the sample has
been measured every 60 minutes, between T
0
and T
0
plus five hours. The first four samples (Metastasis 1
and 2; Cortex 1 and 2) were studied during three
hours: at T
0
; and every 60 minutes, respectively T
1
,
T
2
and T
3
. The fifth sample (Metastasis 3) was
studied at T
0
, T
1
, T
2
, T
3
and also after four and five
hours, respectively T
4
and T
5
.
PHOTOPTICS 2016 - 4th International Conference on Photonics, Optics and Laser Technology
16

Table 1: Mean maximum fluorescence intensity of the five
endogenous fluorophores at different times with 405 nm
excitation wavelength: after excision (T0), one hour after
excision (T1), two hours after excision (T2) and three
hours after excision (T3).
Excitation
wavelength (nm)
405
Spectra fitting:
Fluorophore
NAD
H
FAD Lip Porph Clorin
Sample
Time after
selection
Fluorescence intensity (a.u.)
Metastasis 1
T0 2.29 8.00 7.81 7.95 16.50
T1 0.38 9.89 8.58 9.47 16.65
T2 0.59 7.70 8.30 7.69 15.47
T3 0.77 6.90 6.13 5.71 11.39
Metastasis 2
T0 0.08 12.2 9.61 7.82 8.01
T1 0.41 11.6 7.66 8.00 8.92
T2 3.07 13.3 7.13 12.56 9.48
T3 0.14 7.85 9.30 6.34 8.95
Metastasis 3
T0 6.25 26.1 23.1 39.5 46.18
T1 1.36 19.1 18.8 35.2 41.7
T2 1.68 17.9 15.9 25.5 32.9
T3 5.51 20.2 12.9 22 32.7
Cortex 1
T0 5.47 20.4 13.1 29.7 30.06
T1 5.08 11.6 10.5 15.3 16.34
T2 4.21 11.5 9.02 14.4 14.85
T3 4.53 13.3 9.61 18.7 16.67
Cortex 2
T0 3.99 21.1 14.0 9.92 16.80
T1 7.31 16.3 13.4 14.3 16.93
T2 1.44 7.96 4.24 8.50 6.87
T3 3.19 17.2 12.4 10.6 16.64
Fluorescence lifetime acquisitions were made on a
selected Region of Interest (ROI) of the sample.
Spectral acquisitions were made on a line for the
first four samples. This longitudinal acquisition
allowed us to investigate a large part of the sample.
For the fifth sample (Metastasis 3), spectral
acquisitions were made on a single ROI during five
hours.
Samples were conserved in physiological
solution during the procedure to be as close as
possible to the brain during neurosurgery.
2.4 Results
2.4.1 Spectral Emission
Fluorescence intensity showed a slight decrease
within time. A strong decrease (>50%) of the
fluorescence intensity was observed for the longest
time intervals (T4 and T5). For the majority of fitted
spectra, maximum fluorescence intensity of the five
investigated endogenous fluorophores was lower
after three hours (T3) than at the initial measurement
(T0). Note that this decrease was not always
observed and a large variability existed on the data
set. The mean fluorescence intensity of each
explored fluorophore is detailed on Table 1 at at 405
nm excitation wavelength and for every sample.
Figure 2 summarizes the results at 375 nm excitation
wavelength.
At 405 nm excitation wavelength, cortex
samples showed stronger fluorescence intensity
values than metastasis and this remained true during
all the protocol. At 375 nm excitation wavelength,
the opposite situation seemed to exist (figure 2) even
if partial results did not allow any conclusion.
To go further and to explore change in the
spectra shape, we recorded the last metastasis
sample until the fifth hour and on the same ROI.
Results are presented in figure 3 and figure 4.
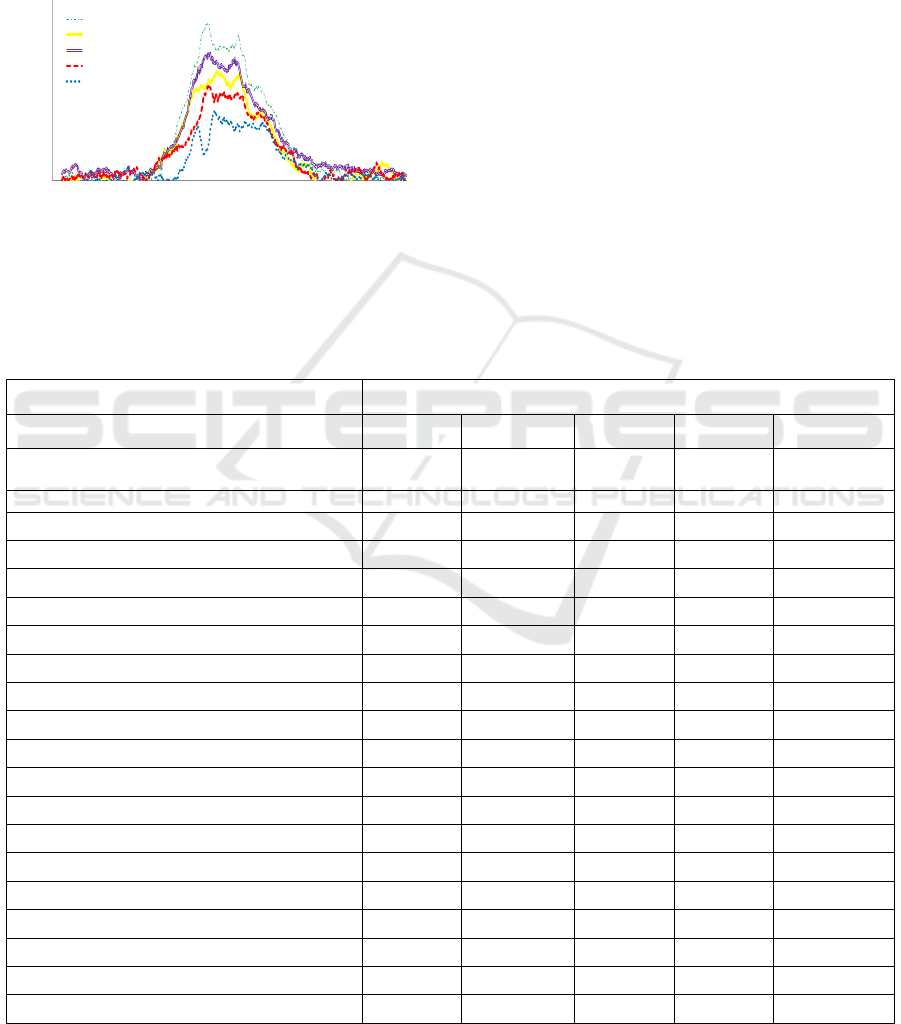
Figure 2: Time variation of the mean maximum
fluorescence intensity emission of NADH and FAD for
different samples: Metastasis 1 (M1), Metastasis 2 (M2)
and Cortex 1 (cortex) at 375 nm excitation wavelength.
Figure 3: The spectral shape of the metastasis 3 sample
fluorescence emission until the fifth hour and on the same
ROI using 405 nm excitation wavelength.
0
5
10
15
20
25
30
35
40
45
5
0
200 300 400 600 700 800 900
F
l
u
o
r
e
s
c
e
n
c
e
i
n
t
e
n
s
i
t
y
(
a
.
u
.
)
W
a
v
e
l
e
n
g
t
h
(
n
m
)
T0
T1
T2
T3
T5
Spectral and Lifetime Measurements of the Endogenous Fluorescence Variation of Freshly Resected Human Samples over Time -
Measuring Endogenous Fluorescence Changes at Different Moment after Tumor or Epileptic Cortex Excision
17
Spectral shape was also affected with time: at
T0, three peaks existed around 600 nm, 680 and 700
nm with 405 nm excitation wavelength and the first
two first peaks (600 nm and 680 nm) were also
present at 375 nm, whereas after five hours (T5), no
peak was visible with both 375 nm and 405 nm
excitation wavelengths. These changes could be
related to tissue oxygenation.
Figure 4: the spectral shape of the metastasis 3 sample
fluorescence emission until the fifth hour and on the same
ROI using 375 nm excitation wavelength.
2.4.2 Lifetime Measurements
Table 2 summarizes the lifetime fluorescence
measurements overtime for the three-metastasis
samples and the two samples of healthy human
cortex using 405 nm excitation wavelength.
The three-metastasis samples showed the same
trend: lifetime values shortened slightly with time at
405 excitation wavelengths. Longer lifetime values
were observed at 375 nm excitation wavelength for
the first two filters (450 ±10 nm and 520 ±10 nm)
with regard to 405 nm excitation wavelength values.
Lifetime values ranged from 1.69 ns to 5.63 ns.
Interestingly, at 405 nm, the values for the last two
filters (620 ±10 nm and 680 ±10 nm) seemed longer
for metastasis samples than for cortex samples. The
variation of the fluorescence lifetime could be
related to the viability of the tissue. Indeed after
three hours the cellular structure changed as we can
easily notice on histological analysis done right after
the surgery or after T3. Consequently the
microenvironement of the tissue changes too, which
Table 2: Lifetime measurements of the five endogenous fluorophores at different times with 405 nm excitation wavelength:
after excision (T0), one hour after excision (T1), two hours after excision (T2) and three hours after excision (T3).
Excitation wavelength (nm) 405
Filter wavelength (nm) 450 ± 10 520 ± 10 550 ± 30 620 ± 10 680 ± 10
Sample
Time after selection
Lifetime
value (ns)
Lifetime
value (ns)
Lifetime
value (ns)
Lifetime
value (ns)
Lifetime
value (ns)
Metastasis 1
T0 2.98 3.28 3.99 2.66 1.93
T1 2.40 2.27 3.48 2.19 1.80
T2 2.60 2.46 3.73 2.09 1.81
T3 3.04 3.01 3.94 2.47 2.15
Metastasis 2
T0 2.70 2.94 3.70 2.69 1.86
T1 2.35 2.64 3.52 2.62 2.01
T2 2.33 2.97 3.40 2.63 1.85
T3 2.17 2.52 3.47 2.33 1.69
Metastasis 3
T0 2.20 2.07 3.74 5.63 4.32
T1 2.64 2.93 3.93 3.96 3.74
T2 2.81 2.81 3.83 4.21 3.48
T3 2.59 3.05 3.81 3.62 3.19
Cortex 1
T0 2.24 2.37 3.45 1.88 1.76
Cortex 2
T0 3.14 2.94 4.13 2.03 1.75
0
5
10
15
20
25
200
300
400
600
700
800
900
F
l
u
o
r
e
s
c
e
n
c
e
i
n
t
e
n
s
i
t
y
(
a
.
u
.
)
Wavelength
(nm)
T0
T1
T2
T3
T5
PHOTOPTICS 2016 - 4th International Conference on Photonics, Optics and Laser Technology
18

could affect lifetime measurements. In general, three
hour after tissue excision, no (remarkable) clear
difference was observed on fluorescence lifetime
measurements between all studied fluorophores on
these metastasis samples.
Healthy cortex samples seemed to present shorter
lifetimes values than metastasis samples especially
for the last two filters (620 ±10 nm and 680 ±10 nm)
corresponding to porphyrin and chlorin, at 405 nm
excitation wavelength.
3 DISCUSSIONS AND
CONCLUSION
Our work, the first study of fresh human brain
samples autofluorescence over time, led to four main
conclusions 1) fluorescence intensity decreased
slightly with time, 2) spectral shape was
considerably modified over time, 3) fluorescence
lifetime measurements decreased marginally with
time too, and 4) concerning ex vivo studies,
autofluorescence measurements should be acquired
in less than three hours after excision.
This investigation is crucial to validate the use of
endogenous fluorescence as a new imaging tool.
More than 70% of intracranial surgery last longer
than two hours and duration of surgery is a risk
factor for extracranial complications ( Golebiowski,
2015). Prior to develop a device able to help
neurosurgeon during his interventions, it is
necessary to take into account the potential variation
of autofluorescence over time.
Our work revealed that autofluorescence
decreased with time after extraction but that this
decrease was highly variable for fluorescence
intensity and not strong for both fluorescence
intensity and lifetime measurements. This is in
accordance with the only reference on
autofluorescence variation with time (Groce, 2003).
Metastasis and control samples (cortex providing
from epilepsy surgery) showed the same trend to a
slight decrease in fluorescence intensity and lifetime
values with time. Higher fluorescence intensity
values at 405 nm excitation wavelength for the
control samples compared to metastasis samples
were found during all the protocol. If multimodality
is the clue to overcome previous limits of
autofluorescence per operative use (Marcu, 2012)
(Groce, 2014), it is not possible to distinguish
healthy boundaries and tumor with the spectra
shape: spectra did not show any recognizable peak
five hours after extraction.
These results are preliminary and need to be
confirmed and specified. Larger cohort with more
ROI for lifetime measurements is required.
Our work underlines the necessity to take into
account clinical issue to develop and calibrate an
adequate and precise tool to help neurosurgeon
performing gross total resection. Close collaboration
between clinical and scientific teams is required to
investigate brain autofluorescence.
ACKNOWLEDGEMENTS
This Work as a part of the MEVO project was
supported by “Plan Cancer” program founded by
INSERM (France), by CNRS with “Défi
instrumental” grant, and the Institut National de
Physique Nucléaire et de Physique des Particules
(IN2P3).
REFERENCES
Raman SS, Osuagwu FC, Kadell B, Cryer H, Sayre J, Lu
DSK.2008 Effect of CT on false positive diagnosis of
appendicitis and perforation. N Engl J Med.
Fontana EJ, Benzinger T, Cobbs C, Henson J, Fouke
SJ.,2014 Sep;119(3):491–502, The evolving role of
neurological imaging in neuro-oncology. J
Neurooncol. 2014 Sep;119(3):491–502.
Liu W, Bakker NA, Groen RJ., 2014;121(3):665–
73.Chronic subdural hematoma: a systematic review
and meta-analysis of surgical procedures: A systematic
review. J Neurosurg.
Abi Haidar D, Leh B, Zanello M, Siebert R, 2015 Spectral
and lifetime domain measurements of rat brain
tumours. J Biomed. optics express
Leh B, Siebert R, Hamzeh H, Menard L, Duval M-A,
Charon Y, et al., 2012 Oct;17(10):108001, Optical
phantoms with variable properties and geometries for
diffuse and fluorescence optical spectroscopy. J
Biomed Opt.
Golebiowski A, Drewes C, Gulati S, Jakola AS, Solheim
O. 2015, Is duration of surgery a risk factor for
extracranial complications and surgical site infections
after intracranial tumor operations? Acta Neurochir
Croce AC, Fiorani S, Locatelli D, Nano R, Ceroni M,
Tancioni F, et al., 2003 Mar;77(3):309–18, Diagnostic
potential of autofluorescence for an assisted
intraoperative delineation of glioblastoma resection
margins. Photochem Photobiol.
Marcu L., 2012 Feb;40(2):304–31, Fluorescence Lifetime
Techniques in Medical Applications. Ann Biomed
Eng.
Croce AC, Bottiroli G., 2014, Autofluorescence
spectroscopy and imaging: a tool for biomedical
research and diagnosis. Eur J Histochem
Spectral and Lifetime Measurements of the Endogenous Fluorescence Variation of Freshly Resected Human Samples over Time -
Measuring Endogenous Fluorescence Changes at Different Moment after Tumor or Epileptic Cortex Excision
19
