
RadioBio data: A Moddicom Module to Predict Tumor Control
Probability and Normal Tissue Complication Probability in
Radiotherapy
Nicola Dinapoli
1
, Anna Rita Alitto
1
, Mauro Vallati
2
, Rosa Autorino
1
, Roberto Gatta
1
, Luca Boldrini
1
,
Andrea Damiani
1
, Giovanna Mantini
1
and Vincenzo Valentini
1
1
Department of Radiation Oncology, Universit
`
a Cattolica del Sacro Cuore, Rome, Italy
2
School of Computing and Engineering, University of Huddersfield, Huddersfield, U.K.
Keywords:
Dose Volume Histograms, Radiobiology, Radiotherapy, TCP, NTCP, Vdose, Dvolume.
Abstract:
In this work a system for analysing radiotherapy treatment planning dose-volume data is proposed. The work
starts from the definition of a framework inside a statistical scripting environment (R) used for creating a
software package. The analysis of dose-volume data in radiotherapy of malignant tumours is mandatory for
evaluating the prescribed treatment and for feedback analysis of outcome, both in the direction of tumour
control and in detection of parameters for estimating and predicting toxicity outcome. The statistical analysis
of large amount of clinical data can be slowed by the lack of practice in statistical tools needed, by clinicians,
to perform such kind of analysis. This is the reason that lead our working group in the creation of such a tool.
Finally an example of clinical application of our software is given for the analysis of the outcome of patients
undergoing to radiotherapy for prostate cancer.
1 INTRODUCTION
Radiotherapy is one of the most efficient and most
used treatments in cancer therapy, together with the
chemotherapy and surgery. In the external beam Ra-
diotherapy (EBR) a radiation source is addressed to
send radiation beams (X-ray, gamma-rays or elec-
trons) on the patient, in specific locations, and the
leafs of a collimator are configured in order to repro-
duce in the space a desired 3-dimensional conforma-
tion of radiation doses. Such doses are planned in
order to be able to kill or severely damage the tumour
cells. The 3-dimensional shape of toxic dose is deliv-
ered inside the patient body; therefore, attention must
be payed in order to decrease as much as possible the
delivery of radiations in healthy tissues, so reducing
the likelihood of toxicity.
When planning a radiotherapy treatment, the radi-
ation oncologists have two critical aspects to consider:
(i) providing a total dose on the target that is adequate
for damaging and killing; and (ii) limiting the dose
received by normal tissue, in order to minimise iatro-
genic side effects. Usually, radiation oncologists have
to identify a reasonable trade-off between these two
aspects. Because of that, physicians exploit complex
mathematical models – called radiobiological mod-
els – for estimating the Tumour Control Probability
(TCP) and Normal Tissue Complication Probability
(NTCP). Given such estimations, they can identify
the plan that provides the largest total dose on target,
while limiting the likelihood of normal tissue side ef-
fects.
Despite the fact that numerous models have been
proposed, the identification and exploitation of radio-
biological models is still considered an open and criti-
cal topic. Such models are currently in the aims of the
most important international associations in the field:
for example, the American Society for Radiation
Oncology (ASTRO) in association with the Ameri-
can Association of Physicists in Medicine (AAPM)
supported the creation of Quantitative Analyses of
Normal Tissues Effects in the Clinic (QUANTEC).
QUANTEC project exploits the large amount of data
that modern technologies – like Linear Accelerator
(LINAC) and Treatment Planning Systems (TPSs) –
are able to export, in order to provide more accurate
TCP and NTCP specific for the different involved hu-
man anatomical regions (Bentzen et al., 2010; Marks
et al., 2010b).
One of the most common source of information
exploited by radiobiological models is represented by
Dinapoli, N., Alitto, A., Vallati, M., Autorino, R., Gatta, R., Boldrini, L., Damiani, A., Mantini, G. and Valentini, V.
RadioBio data: A Moddicom Module to Predict Tumor Control Probability and Normal Tissue Complication Probability in Radiotherapy.
DOI: 10.5220/0005693502770281
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 5: HEALTHINF, pages 277-281
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
277

the Dose Volume Histogram (DVH): an histogram
showing on the x-axes the dose and on the y-axes
the volume of the ROI receiving such dose. Each
DVH is a “quick 2D recap”, of the dose absorbed for
each target or normal tissue volume, also called Re-
gion Of Interest (ROI). In literature, the analysis of
DVHs is related to the actual clinical outcome, for ef-
fectively identifying some dose constraints under the
form of clear rules, such as: the minimum dose to the
Planning Target Volume is 95% of the plan reference
point dose; in order to decrease risk of radiation pneu-
monia under 20% it is prudent to limit Mean Lung
Dose within 20 Gy (Marks et al., 2010a) or “to avoid
rectal bleeding or late rectal RTOG grade >= 2 less
than 50% of rectal volume should receive 50 Gy or
less than 35% should receive 60 Gy”(Michalski et al.,
2010).
The classical empirical approach to define such
constrains has been recently overcome by adaptive al-
gorithms, which are able to adaptively find, with the
evidence of the daily clinical practice, also new form
of useful constraints. An important contribute come
from (Naqa et al., 2006) which clearly highlights the
importance of predictors built basing on DVH and
a multi-variate modelling process. Nevertheless all
those experiences are done locally, in different centres
with different tools, and there is no common tools to
build and tune new TCP/NTCP models by a specific
dataset.
Despite collecting and analysing dose information
is considered very important in order to build and im-
prove maths models, there is a poor availability of eas-
ily to access tools for such task. VODCA is the oldest
project and is able to collect DICOM-RT studies and
to analyse them by using R, a well-known statistical
engine. It should be noted that VODCA is not free and
does not provide specific tools, libraries and function
for TCP/NTCP models.
In order to provide a common tool for building
and finely tuning TCP/NTCP models, in this paper
we propose a software library, integrated in a more
complete project called moddicom(Dinapoli et al.,
2015b), that is able to effectively support the process
of generating and improving TCP/NTCP models for
the specific data of a radiotherapy centre. To foster the
exploitation of the tool, and therefore allow a better
use of the knowledge stored in the existing datasets,
the proposed software is freely available as an R pack-
age at https://github.com/kbolab/moddicom.git. In
this paper we provided an overview of the developed
software library, and we show the results of an exper-
imental analysis focused on evaluating the usefulness
of the proposed system.
2 METHODS
The overall structure of the moddicom framework is
shown in Figure 1. Currently, the modules grouped as
R environment are available and ready to be used. The
R environment includes:
• geoLet. This requires the DCMTK Office li-
braries (Eichelberg et al., 2004) and allows to load
DICOM Images, DICOM RTStruct, DICOM RT-
Dose, DICOM RTPlan studies in memory.
• Image Feature Data Analysis. This module al-
low to extract image features for Radiomics (Di-
napoli et al., 2015a) analysis from the previously
loaded DICOM studies.
• RadioBio data models. This module cal-
culates DVHs (starting from the DICOMRT
Struct/Dose/Plan previously loaded by geoLet)
and can tune the parameters of six different radio-
biological maths models for TCP/NTCP mod-
elling.
• Visualisation Module. This module provides
tools to visualise objects and images in 2D/3D in
order to give a practical presentation of the stored
data.
Part of the moddicom framework is still under de-
velopment. The complete moddicom framework will
be able to acquire information directly from modali-
ties and PACS, via DICOM protocol, in order to popu-
late its internal database, which will be subsequently
able to automatically exploit the stored information.
At the moment, the loading is done by searching a
file system and for finding DICOM information.
The geoLet module can search the file system,
from a given path, and load the data stored in
DICOM RT-PLAN/RT-STRUCT and RT-DICOM-
Distribution Doses. The corresponding information
are then used by the RadioBio data module for gen-
erating and fitting a number of mathematical models.
The fitting is also able to analyse the hidden clinical
effects of dose constraint not yet identified as signif-
icant in literature. In the following, the implemented
models are listed and briefly described. The interested
reader is referred to the provided references for details
about the mathematical functions and models.
• Lyman. It is a reformulation of probit model, that
uses different parameters than mean and standard
deviation (Burman et al., 1991).
• Goitein. This model is similar to the Lyman
model, but it is function of logarithm of the Dose
(Shipley et al., 1979; Bentzen and Tucker, 1997).
• Niemierko. It is the translation of a loglogit gen-
eralised linear model as function of T D
50
and γ
50
(Gay and Niemierko, 2007).
HEALTHINF 2016 - 9th International Conference on Health Informatics
278
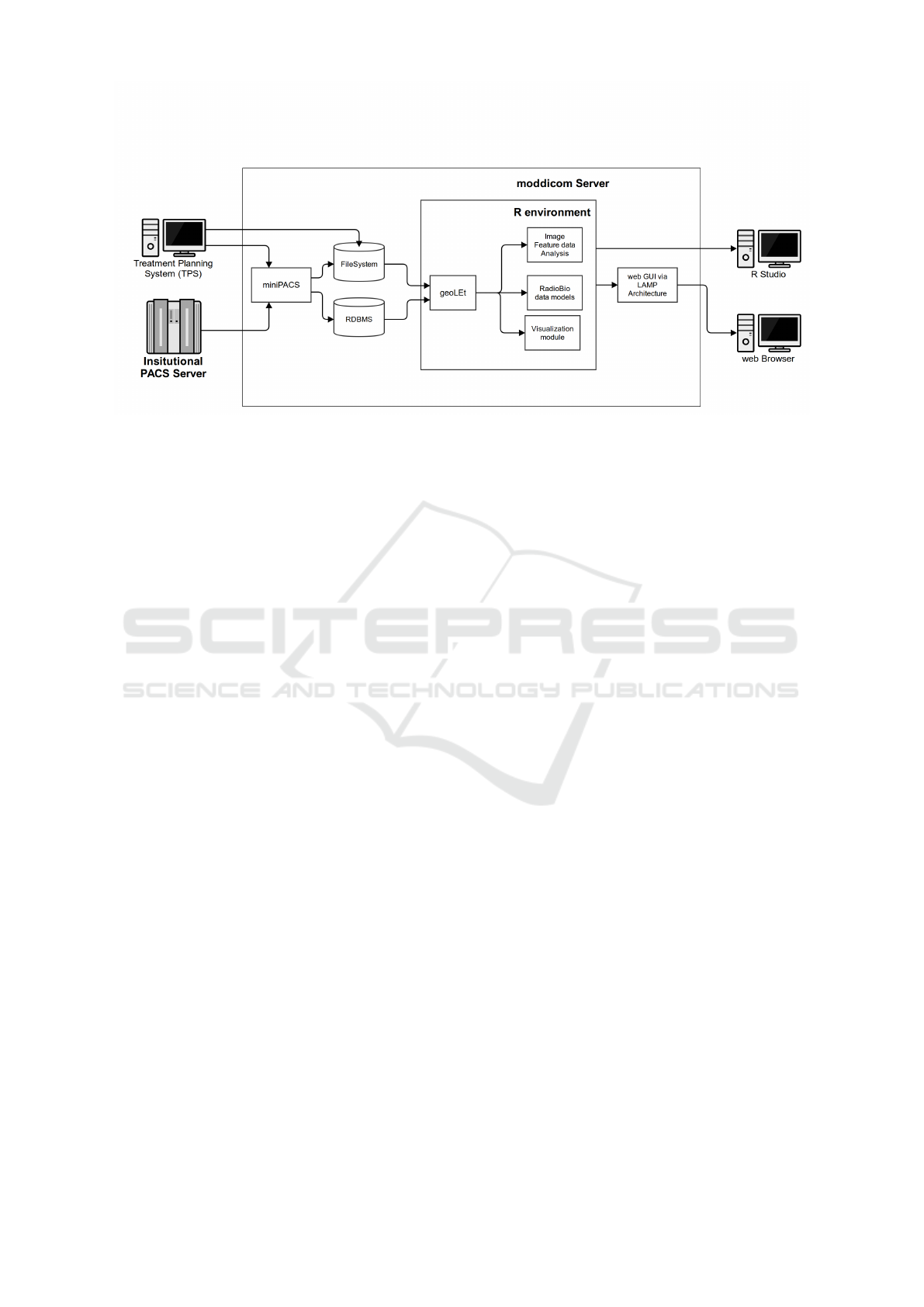
Figure 1: The overall structure of the moddicom architecture. The core is represented in the box titles R environment and is
currently available. The structure includes also the PACS/TPS interface and the web based GUI.
• Munro. This model is an empirical dose/response
curve that fits experimental data. The model is
based on the assumption that this curve is equiv-
alent to a Poisson model (Munro and Gilbert,
1961).
• Okunieff. This model is the equivalent of the lo-
gistic generalised linear model, where the covari-
ates and their coefficients have been reported as
function of T D
50
and γ
50
(Okunieff et al., 1995).
• Warkentin. This model is the equivalent of the
Warkentin Poisson model where the covariates
and their coefficients have been reported as func-
tion of T D
50
and γ
50
(Warkentin et al., 2004).
• Bentzen. Bentzen described dose/response
curves both for tumour and normal tissue re-
sponse. In our implementation, log Poisson model
starts from equation used in Warkentin equation,
but the subsequent evaluation of the relation-
ship of response with the log Dose (Bentzen and
Tucker, 1997).
The previously cited models are considered in
order to automatically find the most significant
Dose/Volume constraint able to fit the model with the
clinical outcomes. A similar approach has been pro-
posed in the past (Naqa et al., 2006), but it is worthy
noting that moddicom represents the first shared and
freely available software tool able to do that.
The RadioBio data module also provide a simula-
tion environment where DVH can be artificially cre-
ated (Van den Heuvel, 2006) and associated to clinical
outcome according with a known statistic. This repre-
sents an useful sandbox to test the implemented math-
ematical models and see their details in action. As
previously cited, RadioBio data module is also linked
with the geoLet module for being able to work with
real clinical data directly taken from DICOM sources.
For providing a complete environment to the re-
searches, a range of tools –including calibration plots
and fitting curve– have been implemented and can be
exploited within the moddicom framework.
3 EXPERIMENTAL RESULTS
In order to test effectively the applicability and the
capability of the proposed RadioBio data moddicom
module, a clinical investigation was performed on
a real dataset of 123 patients treated with Radia-
tion Therapy for prostate cancer: this cohort was
taken from a study performed between September
2010 and May 2014 at the Radiation Oncology Cen-
tre of Gemelli Hospital. Acute and late rectal (GI)
and bladder (GU) toxicity data were collected. Re-
ferring physician clinically examined patients a me-
dian of 5 times during RT treatment, unless oth-
erwise requested by patient himself. RTOG score
(Cox et al., 1995) and CTC-AE v.4.03 (of Health
and Human Services, 2010) were used to stratify
grade of toxicity. Patients’ DVH were collected to
evaluate bladder and rectum dose-volume distribu-
tions. Dose from treatment planning and simulated
delivery was evaluated using the Equivalent Uniform
Dose (EUD) (Niemierko, 1997). For normal tissues,
the EUD represents the uniform dose which leads to
the same probability of injury as the examined inho-
mogeneous dose distribution. The EUD was calcu-
lated from the corresponding dose-volume distribu-
tions (histograms).
To calculate the EUD-based normal tissue com-
plication probability (NTCP), we used parametrisa-
tion of the dose-response characteristics using Lyman
RadioBio data: A Moddicom Module to Predict Tumor Control Probability and Normal Tissue Complication Probability in Radiotherapy
279

Figure 2: Correlation between rectal G1 late toxicity and a
Vdose=59.5 Gy.
Figure 3: Correlation between rectal G1 late toxicity and a
Dvolume=16.44cc
NTCP formula (Burman et al., 1991).
In such comparison DVHs have been clustered ac-
cording with the induced toxicity (Figure 4). The
dose-volume toxicity parametrisation has been con-
ducted by identifying respectively the volume of a
structure receiving a given level of dose (as known as
Vdose) and the dose received by a given volume of the
structure (as known as Dvolume). Such dose-volume
parametrisation is extensively in radiotherapy for cor-
relating outcome and dose distribution data (Marks
et al., 2010b). We observed a correlation between
development of late G1 or superior GI toxicity and
a Vdose equal to 59,5 Gy (see Figure 2) and a Dvol-
ume equal to 16,44 cc (see Figure 3). In particular we
found a significant correlation with D16cc by Mann-
Whitney Test (p = 0.019).
Finally, to elaborate a predictive model for G1 GI
toxicity, we build up a DVH-reduction model by Ly-
man Model Maximum Likelihood Estimation, based
on estimated NTCP under uniform irradiation (EUD)
of the rectum. We found that our patients were dis-
tributed on or close to an S slope showing that 50%
probability of acute toxicity is present with a mean
delivered uniform dose of 66,5 Gy (see FIgure 4).
The results of the analysis, performed using the
Figure 4: The DVH-reduction model based on estimated
complication probability (NTCP) under uniform irradiation
(EUD) of the rectum.
Figure 5: The proposed graphical user interface.
proposed moddicom module, confirmed the evidence
known in literature and also suggested a new possi-
ble significant dose constraint. While the former re-
sult clearly indicates the validity of the approach, the
latter provides some insights into the usefulness of
the RadioBio data module. However, due to the lim-
ited number of recruited patients, further investigation
should be done to confirm the identified constraint.
Remarkably, the analysis was performed by a
physician –although expert user of R– rather than a
technician. Therefore, the use of moddicom can pro-
vide an effective support to physician in their every
day research or clinical activities.
4 CONCLUSIONS
In this paper we presented the RadioBio data module
of moddicom, which is a free and open source library
born to support the adaptive TCP/NTCP modelling.
We tested RadioBio data with 123 patients treated
with Radiation therapy for prostate cancers and we
obtained results in accord with literature. The per-
formed analysis focused on investigating the usability
and usefulness of the proposed module, and it showed
HEALTHINF 2016 - 9th International Conference on Health Informatics
280

that RadioBio data can be an useful tool to mine ef-
fective dose constraints hidden into the shape of Dose
Volume Histograms. Moreover, the considered case-
study can be extended by considering a larger sample
of patients, in order to provide stronger evidences and
better optimise planning procedures in treatment val-
idation and predict possible toxicity. The future chal-
lenge will be the personalisation of treatments and
complications rates reduction.
Future work include the development of a user-
friendly graphical user interface, as proposed in Fig-
ure 5, an experimental analysis considering a larger
number of patients, and the involvement of a larger
number of medical experts.
REFERENCES
Bentzen, S. and Tucker, S. (1997). Quantifying the position
and steepness of radiation dose-response curves. Int J
Radiat Biol., 71(5):531–42.
Bentzen, S. M., Constine, L. S., Deasy, J. O., Eisbruch, A.,
Jackson, A., Marks, L. B., Haken, R. K. T., and Yorke,
E. D. (2010). Quantitative analyses of normal tissue
effects in the clinic (quantec): An introduction to the
scientific issues. Radiation Oncology, 76(3):S3–S9.
Burman, C., Kutcher, G., Emami, B., and Goitein, M.
(1991). Fitting of normal tissue tolerance data to
an analytic function. Int J Radiat Oncol Biol Phys,
21(1):123–35.
Cox, J., Stetz, J., and PajaK, T. (1995). Toxicity criteria
of the radiation therapy oncology group (rtog) and the
european organization for research and treatment of
cancer (eortc). Int. J. Radiation Oncology Biol. Phys,
31(5):1341–1346.
Dinapoli, N., Alitto, A., Vallati, M., Gatta, R., Autorino, R.,
Boldrini, L., Damiani, A., and Valentini, V. (2015a).
Moddicom: A complete and easily accessible library
for prognostic evaluations relying on image features.
In IEEE Engineering in Medicine and Biology Society,
37th Annual International Conference.
Dinapoli, N., Alitto, A. R., Vallati, M., Gatta, R., Au-
torino, R., Boldrini, L., Damiani, A., and Valentini,
V. (2015b). Moddicom: a complete and easily ac-
cessible library for prognostic evaluations relying on
image features. In Proceedings of the 37th Annual
International Conference of the IEEE Engineering in
Medicine and Biology Society (EMBS). IEEE.
Eichelberg, M., Riesmeier, J., Wilkens, T., Hewett, A.,
Barth, A., and Jensch, P. (2004). Ten years of medical
imaging standardization and prototypical implemen-
tation: the dicom standard and the offis dicom toolkit
(dcmtk). In Medical Imaging 2004, pages 57–68.
Gay, H. and Niemierko, A. (2007). A free program for cal-
culating eud-based ntcp and tcp in external beam ra-
diotherapy. Phys Med., 23(3-4):115–25.
Marks, L., Bentzen, S., Deasy, J., Kong, F.-M., Bradley,
J., Vogelius, I., Naqa, I., Hubbs, J., Lebesque, J., Tim-
merman, R., Martel, M., and Jackson, A. (2010a). Ra-
diation dose-volume effects in the lung. Int. J. Radia-
tion Oncology Biol. Phys, 76(3):S70–S76.
Marks, L. B., Yorke, E. D., Jackson, A., Haken, R. K. T.,
Constine, L. S., Eisbruch, A., Bentzen, S. M., Nam, J.,
and Deasy, J. O. (2010b). Use of normal tissue com-
plication probability models in the clinic. Radiation
Oncology, 76(3):S3–S9.
Michalski, J., Gay, H., Jackson, A., Tucker, S., and Deasy,
J. (2010). Radiation dose-volume effects in radiation-
induced rectal injury. Int. J. Radiation Oncology Biol.
Phys, 76(3):S123–S129.
Munro, T. R. and Gilbert, C. W. (1961). The relation be-
tween tumour lethal doses and the radiosensitivity of
tumour cells. Br J Radiol., 34:246–51.
Naqa, I. E., Bradley, J., Blanco, A., Lindsay, P., Vicic, M.,
Hope, A., and Deasy, J. (2006). Multi-variable model-
ing of radiotherapy outcomes including dose-volume
and clinical factors. Int. J. Radiation Oncology Biol.
Phys, 64(4):1275–1286.
Niemierko, A. (1997). Reporting and analyzing dose distri-
butions: a concept of equivalent uniform dose. Medi-
cal physics, 24(1):103–10.
of Health, U. D. and Human Services, National Institutes
of Health, N. C. I. v. (June 14 2010). Common termi-
nology criteria for adverse events (ctcae).
Okunieff, P., Morgan, D., Niemierko, A., and Suit, H.
(1995). Radiation dose-response of human tumors.
Int J Radiat Oncol Biol Phys, 32(4):1227–37.
Shipley, W. U., Tepper, J. E., Prout, G. R., Verhey, L., Men-
diondo, O., Goitein, M., Koehler, A., and Suit, H.
(1979). Proton radiation as boost therapy for local-
ized prostatic carcinoma. JAMA, 241(18):1912–5.
Van den Heuvel, F. (2006). Decomposition analysis of dif-
ferential dose volume histograms. Medical Physics,
33(2):297.
Warkentin, B., Stavrev, P., Stavreva, N., Field, C., and Fal-
lone, B. (2004). A tcp-ntcp estimation module using
dvhs and known radiobiological models and parame-
ter sets. J Appl Clin Med Phys, 5(1):50–63.
RadioBio data: A Moddicom Module to Predict Tumor Control Probability and Normal Tissue Complication Probability in Radiotherapy
281
