
Respiratory Effort Belts in Postoperative Respiratory Monitoring:
Pilot Study with Different Patients
Tiina M Seppänen
1, 2
, Olli-Pekka Alho
2, 3, 4
, Merja Vakkala
2, 5
, Seppo Alahuhta
2, 5
and Tapio Seppänen
1, 2
1
Center for Machine Vision and Signal Analysis, University of Oulu, P.O Box 4500, 90014 Oulu, Finland
2
Medical Research Center Oulu, Oulu University Hospital and University of Oulu, Oulu, Finland
3
Department of Otorhinolaryngology, Oulu University Hospital, Oulu, Finland
4
Research Unit of Otorhinolaryngology and Ophthalmology, University of Oulu, Oulu, Finland
5
Department of Anesthesiology, Oulu University Hospital, Oulu, Finland
Keywords: Airflow Waveform, Respiratory Airflow, Respiratory Rate, Respiratory Volume.
Abstract: Respiratory complications are common in patients after the general anaesthesia. Respiratory depression often
occurs in association with postoperative opioid analgesia. Currently, there is a need for a continuous non-
invasive respiratory monitoring of spontaneously breathing postoperative patients. We used calibrated
respiratory effort belts for the respiratory monitoring pre- and postoperatively. Used calibration method
enables accurate estimates of the respiratory airflow waveforms. Five different patients were measured with
the spirometer and respiratory effort belts at the same time. Preoperative measurements were done in the
operating room just before the operation, whereas postoperative measurements were done in the recovery
room after the operation. We compared three calibration models pre- and postoperatively. Postoperative
calibration produced more accurate respiratory airflows. Results show that not only the tidal volume, minute
volume and respiratory rate can be computed precisely from the estimated respiratory airflow, but also the
respiratory airflow waveforms are very accurate. The method produced accurate estimates even from the
following challenging respiratory signals: low airflows, COPD, hypopneic events and thoracoabdominal
asynchrony. The presented method is able to produce estimates of postoperative respiratory airflow
waveforms to enable accurate, continuous, non-invasive respiratory monitoring postoperatively.
1 INTRODUCTION
Respiratory complications are common in patients
after the general anaesthesia. Respiratory depression
often occurs in association with postoperative opioid
analgesia (Etches, 1994; Gamil and Fanning, 1991;
Taylor et al., 2005). Adequate respiration monitoring
postoperatively is important, so that respiratory
depression can be identified as early as possible
(George et al., 2010; Lynn and Curry, 2011).
During general anaesthesia, mechanical
ventilation is used and, consequently, monitoring of
gas exchange and respiration can be done accurately.
During postoperative care, respiratory status can be
assessed with oxygen saturation (SpO
2
)
measurements, blood gas measurements, subjective
clinical assessment and intermittent, manual
measurements of respiratory rate (Lynn and Curry,
2011; Ramsay et al., 2013). The problems with these
methods are that they are slow and, in addition,
subjective methods are also unreliable and give
inconsistent results (Lovett, 2005). There is a need for
a continuous non-invasive respiratory monitoring of
spontaneously breathing postoperative patients.
Respiratory depression and subsequent adverse
outcomes can arise from pain, residual operating
room anaesthetics and administration of opioids for
pain management (Cepeda et al., 2003). Inadequate
respiration can result in respiratory complications,
morbidity, mortality and excessive costs.
A few studies have been recently published on
monitoring postoperative respiration continuously
and non-invasively. Drummond et al., (2013) have
studied respiratory rate and breathing patterns of
postoperative subjects using encapsulated tri-axial
accelometer taped to a subject’s body. They found
that abnormal breathing patterns are extremely
common. Voscopoulos et al., (2015; 2014a; 2014b)
76
Seppänen, T., Alho, O-P., Vakkala, M., Alahuhta, S. and Seppänen, T.
Respiratory Effort Belts in Postoperative Respiratory Monitoring: Pilot Study with Different Patients.
DOI: 10.5220/0005695000760082
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 5: HEALTHINF, pages 76-82
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

have studied minute ventilation, tidal volume and
respiratory rate of postoperative subjects using
impedance-based electrodes placed to a subject’s
body.
Recently, we published a novel calibration
method to produce accurate estimates of respiratory
airflow signals from respiratory effort belt signals
(Seppänen, 2013). Here, the method is used in order
to produce estimates of postoperative respiratory
airflow waveforms to enable accurate, continuous,
non-invasive respiratory monitoring postoperatively.
Pre- and postoperative measurement data of different
patients are used to demonstrate the performance of
the method.
2 MATERIALS AND METHODS
2.1 Materials
The study protocol was approved by the Regional
Ethics Committee of the Northern Ostrobothnia
Hospital District. Five patients who had lumbar back
surgery and were expected to need opioid analgesia
postoperatively were recruited to the study. Exclusion
criteria were BMI (Body Mass Index) over 40 and the
planned surgical wound being in the area where
respiratory effort belts were placed. The
characteristics of the volunteers are given in Table 1.
Table 1: Characteristics of volunteers.
Patient Gender
Age
[years]
BMI
[kg/m
2
]
Disease
1 M 68 21.8 None
2 M 41 30.3 None
3 F 77 22.4 None
4 M 64 28.1 COPD
5 M 67 27.4 Sleep apnea
Respiratory effort belt signals were recorded with
the polygraphic recorder (Embletta Gold, Denver,
Colorado, USA). The recorder had inductive
respiratory effort belts for rib cage and abdomen with
the sampling rate of 50 Hz. For calibrating the
respiratory effort belt signals, simultaneous
respiratory airflow signal was recorded with a
spirometer (Medikro Pro M915, Medikro Oy,
Kuopio, Finland), which had a sampling rate of 100
Hz. Mask (Cortex Personal-Use-Mask, Leipzig,
Germany) was attached to the mouthpiece of the
spirometer. The spirometer could record at most 1
min long signals.
2.2 Measurement Protocol
The measurements for each patient were done in two
parts: 1) short measurement session (5 min)
preoperatively; and 2) longer measurement session (3
h) postoperatively.
The first measurements were done in the
operating room just before the operation without any
sedative premedication. The rib cage respiratory
effort belt was placed on the xyphoid process and the
abdominal belt was placed above the umbilicus. The
mask of the spirometer was put to the volunteer’s face
and its airtightness was secured. The signals were
recorded until two successful recordings of the 1 min
were obtained. The places of respiratory effort belts
were marked with drawing ink on the skin, so that it
was possible to place the belts to the same places
postoperatively. The mask and respiratory effort belts
were removed.
As soon as it was possible, measurements were
continued postoperatively in the recovery room. The
rib cage respiratory effort belt and the abdominal
respiratory effort belt were placed to the previously
marked places. They recorded the signals during the
whole 3 hour measurement period. Every 10 min, the
mask of the spirometer was put to the volunteer’s
face, its airtightness was secured and 1 min
measurement with the spirometer was recorded.
Participation to the study did not affect the routine
management of the patients.
2.3 Calculation of Respiratory Airflow
Estimates
In this study, we applied our recently published
respiratory effort belt calibration method (Seppänen,
2013). The method was therein tested against various
breathing style changes and body position changes,
and compared with the state-of-the-art methods. Our
method outperformed the other methods showing
high robustness to the breathing style changes and
body position changes.
Our method is an extension to conventional
multiple linear regression method so that 1) it uses
number N of consecutive input signal samples and
linear filtering for estimation of each output signal
sample and 2) it is based on polynomial regression to
model different transfer functions between the input
and output. The method is based on optimally trained
FIR (Finite Impulse Response) filter bank constructed
as a MISO (Multiple-Input Single-Output) system
between the respiratory effort belt signals and the
spirometer signal. Three polynomial transfer
functions were tested: linear terms only (M1), linear
Respiratory Effort Belts in Postoperative Respiratory Monitoring: Pilot Study with Different Patients
77

terms and cross-product term (M2), and linear terms
with second order terms (M3).
Figure 1 and equation 1 show the realization of the
filter bank for model M2. Similar realizations can be
derived also for models M1 and M3.
,
(1)
where
,
and
denote the N tap coefficients of
filters FIR
1
, FIR
2
and FIR
3
, respectively:
1
,
2
,…,
, i = 1,2,3. Superscript T
denotes matrix transpose. Parameter denotes
respiratory airflow from spirometer and is zero-
mean Gaussian error. Vectors
and
include N
consecutive signal samples from the rib cage
respiratory effort belt signal and abdominal
respiratory effort belt signal, respectively:
,
1
,…,
1
, j = 1,2 and k
= N,…,n. Variable n is the number of observations
used in the calibration.
Figure 1: Extended calibration method of respiratory effort
belt signals.
is an (n-N+1) × (3 × N) matrix formed from the
vectors
and
:
…
…
…
.
(2)
During the calibration, tap coefficients
,
and
are estimated with the method of least-squares. The
least-squares estimator of the parameter vector
,
,
is given by
.
(3)
The length of the vector
is 3 × N. Finally, the
respiratory airflow signal estimated from the rib cage
and abdominal respiratory effort belt signals through
the FIR filter bank is
.
(4)
In Figure 1, there is the delay element z
-D
included at
the output. There is always a small delay between the
spirometer signal and respiratory effort belt signals
due to the physiological reasons and internal delays
of measuring devices. Thus, the signals have to be
time-synchronized by searching for a proper value for
D (Seppänen, 2013).
We made two different test setups: 1) the data of
the second preoperative measurement minute were
used to train the estimation model and the data of all
the postoperative measurement minutes were used to
test the estimation model (PRE setup); and 2) the data
of the first postoperative measurement minute were
used to train the estimation model and data of the rest
postoperative measurement minutes were used to test
the estimation model (POST setup).
The similarity of spirometer signals and estimated
respiratory airflow signals were assessed by
computing R
2
(coefficient of determination) values.
Tidal volumes, minute volumes and BPM (Breaths
per Minute, respiratory rate) were calculated from the
spirometer signals and estimated respiratory airflow
signals. Relative errors were calculated.
3 RESULTS AND DISCUSSION
Signals were recorded according to the protocol
described in Section 2.2. There were altogether 93
simultaneous measurement minutes with spirometer
and respiratory effort belts. Five of these had to be
discarded due to a malfunction of the spirometer. One
of them had to be discarded due to a malfunction of
the polygraphic recorder. In addition to that, five
postoperative measurement minutes of patient 5 had
to be discarded, because he had serious difficulties to
wake up and stay awake in the recovery room. During
the measurements patients received opioid analgesia
as many times as they needed: 3, 2, 1, 7 and 4 times
for patients 1-5, respectively.
During the measurements a number of problems
related to PRE setup were observed. Firstly, places of
respiratory effort belts can interchange before POST
setup by mistake. Secondly, there may be a need to
tighten or loosen the respiratory effort belts after the
operation, because fluids can accumulate in the body
or can leave from the body during the operation. This
leads to a situation where the estimation model
trained with a preoperative data is not valid anymore.
Thirdly, if there are complications during the
operation or if the operation is prolonged otherwise,
the estimation model trained with a preoperative data
can be erroneous for the postoperative data. With the
POST setup, no problems were observed.
HEALTHINF 2016 - 9th International Conference on Health Informatics
78
3.1 Accuracy of Airflow Estimates
The selection of the best model and FIR filter length
(N value) depended on whether waveform accuracy
(R
2
), tidal volume (V
T
) error, minute volume (V
minute
)
error or BPM error values were studied. Table 2
summarizes the results when PRE setup was used for
training and testing all estimation models. It is seen in
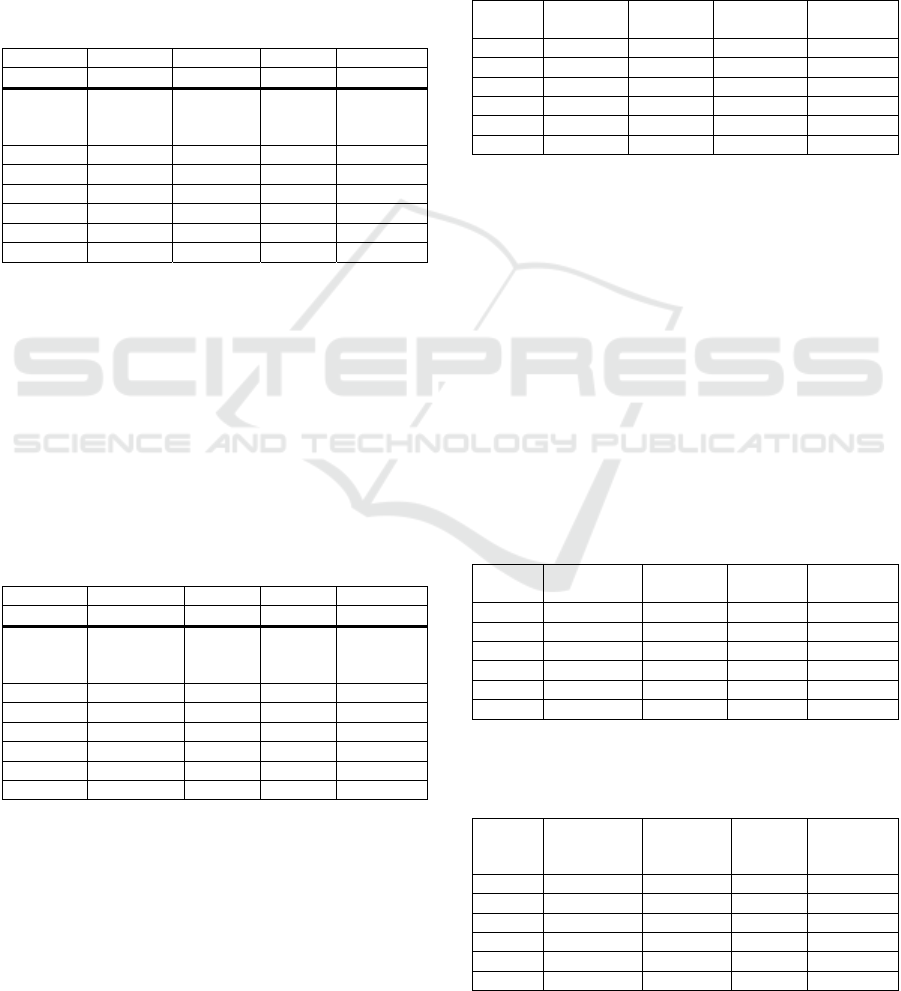
Table 2 that model M1 produced the best results.
Table 2: Results (average value ± SD) with the best models
when PRE setup was used for training and testing all
estimation models.
Best model M1 M1 M1 M1
FIR size N = 8 N = 8 N = 8 N = 16
Patient R
2
Abs (V
T
error) [%]
Abs
(V
minute
error) [%]
BPM error
1 0.88 ± 0.05 12.8 ± 10.7 12.5 ± 9.5 0.03 ± 0.07
2 0.87 ± 0.06 26.7 ± 7.7 25.1 ± 8.2 0.01 ± 0.01
3 0.94 ± 0.02 10.9 ± 2.9 14.2 ± 5.0 0.09 ± 0.30
4 0.87 ± 0.06 15.9 ± 6.0 21.7 ± 8.6 0.09 ± 1.14
5 0.43 ± 0.17 70.2 ± 14.1 72.4 ± 12.3 0.48 ± 0.79
Average 0.82 ± 0.18 24.1 ± 21.4 26.0 ± 21.3 0.29 ± 0.70
Table 3 summarizes the results when POST setup
was used for training and testing all estimation
models. In this case, it is seen that model M2
produced the best results with this data. As an
important difference to the preceding results in Table
2, more accurate waveforms were received since R
2
values were higher and BPM error values lower.
Also, the volume error values decreased to the
fractions.
Table 3: Results (average value ± SD) with the best model
when POST setup was used for training and testing all
estimation models.
Best model M2 M2 M2 M2
FIR size N = 8 N = 16 N = 16 N = 8
Patient R
2
Abs (V
T
error) [%]
Abs
(V
minute
error) [%]
BPM error
1 0.90 ± 0.04 11.4 ± 7.9 9.9 ± 6.8 0.01 ± 0.01
2 0.95 ± 0.01 5.9 ± 4.5 5.8 ± 5.0 0.01 ± 0.01
3 0.94 ± 0.04 5.4 ± 4.7 6.3 ± 4.2 0.10 ± 0.30
4 0.88 ± 0.09 8.7 ± 6.0 11.9 ± 10.2 0.83 ± 1.13
5 0.91 ± 0.02 10.5 ± 9.3 8.2 ± 6.3 0.01 ± 0.00
Average 0.91 ± 0.06 5.8 ± 6.3 8.5 ± 7.1 0.21 ± 0.63
It can be seen from Table 2 and Table 3, that if the
smallest error results are sought, then there is a need
to use several models. However, if there is, for
example, a need to get accurate respiratory rate only,
then one should choose POST setup and model M2
with N value 8. On the other hand, one may wish to
use only one model with good overall performance.
The results for that are presented next. Model M3
(including linear and 2
nd
order terms) produced
clearly worse results than the other models, thus only
the results of using models M1 and M2 are presented
here. Table 4 and Table 5 present the results when the
estimation model M1 with N value 8 was used with
PRE and POST setups, respectively.
Table 4: Results (average value ± SD) of the calibration
when estimation model M1 (N = 8) was used with PRE
setup.
Patient R
2
Abs (V
T
error) [%]
Abs (V
minute
error) [%]
BPM error
1 0.88 ± 0.05 12.8 ± 10.7 12.5 ± 9.5 0.09 ± 0.27
2 0.87 ± 0.06 26.7 ± 7.7 25.1 ± 8.2 0.01 ± 0.01
3 0.94 ± 0.02 10.9 ± 2.9 14.2 ± 5.0 0.09 ± 0.29
4 0.87 ± 0.06 15.9 ± 6.0 21.7 ± 8.6 1.00 ± 1.06
5 0.43 ± 0.17 70.2 ± 14.1 72.4 ± 12.3 0.37 ± 0.55
Average 0.78 ± 0.20 24.1 ± 21.4 26.0 ± 21.3 0.32 ± 0.68
Table 6 and Table 7 present the results when the
estimation model M2 with N value 8 was used with
PRE and POST setups, respectively.
It is clearly seen from Tables 4-7 that POST setup
produced superior results. Respiratory airflow
waveforms are much more accurate, average R
2
increased from 0.78 to 0.91 with both models M1 and
M2. In addition to that, tidal volume errors, minute
volume errors and BPM errors are smaller. However,
when the average results of Table 5 and Table 7 are
compared, it can be seen that models M1 and M2 with
N value 8 produced both very good results and that
there are little differences between the results.
Table 5: Results (average value ± SD) of the calibration
when estimation model M1 (N = 8) was used with POST
setup.
Patient R
2
Abs (V
T
error) [%]
Abs (V
minute
error) [%]
BPM error
1 0.91 ± 0.04 11.6 ± 7.8 8.9 ± 7.1 0.01 ± 0.01
2 0.94 ± 0.02 6.9 ± 7.0 5.7 ± 5.6 0.01 ± 0.01
3 0.94 ± 0.04 5.6 ± 4.5 6.0 ± 3.8 0.10 ± 0.30
4 0.87 ± 0.09 8.4 ± 4.9 11.9 ± 8.9 0.82 ± 1.13
5 0.90 ± 0.02 9.6 ± 5.3 10.9 ± 4.9 0.12 ± 0.35
Average 0.91 ± 0.06 8.4 ± 6.1 8.6 ± 6.7 0.23 ± 0.64
Table 6: Results (average value ± SD) of the calibration
when estimation model M2 (N = 8) was used with PRE
setup.
Patient R
2
Abs (V
T
error) [%]
Abs
(V
minute
error) [%]
BPM error
1 0.82 ± 0.08 17.8 ± 9.8 21.3 ± 8.3 0.10 ± 0.27
2 0.76 ± 0.09 42.7 ± 8.8 51.7 ± 9.6 0.04 ± 0.12
3 0.93 ± 0.03 13.3 ± 5.1 15.3 ± 6.4 0.09 ± 0.30
4 0.87 ± 0.06 15.4 ± 6.7 19.6 ± 8.6 0.91 ± 1.11
5 0.39 ± 0.21 55.1 ± 9.5 65.5 ± 15.1 1.67 ± 1.47
Average 0.78 ± 0.20 26.9 ± 17.8 32.4 ± 21.1 0.49 ± 0.96
Respiratory Effort Belts in Postoperative Respiratory Monitoring: Pilot Study with Different Patients
79

3.2 Example Cases
Figure 2 depicts short segment of example signals
with low airflow. Estimation model M2 (N = 16) was
used with the POST setup for the measurement
signals of patient 3. In this case, R
2
was 0.94, tidal
volume error 1.5 %, minute volume error 10.9 % and
BPM error 0.01. The spirometer signal is estimated
with excellent accuracy.
Figure 3 depicts an example of apneic event. In
this case, estimation model M1 (N = 8) was used with
the POST setup for the measurement signals of
patient 5. During obstruction, the rib cage ceases to
move, but abdomen is moving. There is no air
exchange, so there is no airflow signal either. It can
be seen from the figure, that during the obstruction
airflow was zero, but because there was movement in
respiratory effort belts, the estimated respiratory
airflow signal also showed activity. The same
phenomenon was also encountered by Drummond et
al., (2013) and Voscopoulos et al., (2013). However,
apneic events can be detected from the changed
pattern of movements from respiratory effort belt
signals. Both of them have lower amplitudes than
before or after the apneic event. Especially the rib
cage respiratory effort belt is almost motionless
during the apneic event. Detected parts of the
respiratory airflow estimate could then be replaced
with zero airflow.
Table 7: Results (average value ± SD) of the calibration
when estimation model M2 (N = 8 was used with POST
setup.
Patient R
2
Abs (V
T
error) [%]
Abs (V
minute
error) [%]
BPM error
1 0.90 ± 0.04 11.4 ± 7.6 10.0 ± 7.0 0.01 ± 0.01
2 0.95 ± 0.01 6.1 ± 4.8 6.2 ± 5.3 0.01 ± 0.01
3 0.94 ± 0.04 5.3 ± 4.5 6.2 ± 4.0 0.10 ± 0.30
4 0.88 ± 0.09 8.8 ± 3.9 11.4 ± 10.1 0.83 ± 1.13
5 0.91 ± 0.02 13.1 ± 8.9 9.6 ± 6.4 0.01 ± 0.00
Average 0.91 ± 0.06 8.8 ± 6.6 8.7 ± 7.1 0.21 ± 0.63
Figure 2: Short segment of example signals with low
airflow: spirometer signal (thin line) and the estimated
respiratory airflow signal (bold line).
Figure 4 depicts hypopneic event of patient 4 with
COPD. Here, estimation model M2 (N = 8) was used
with the POST setup. In this case, R
2
was 0.81, tidal
volume error was 0.8 %, minute volume error was
3.24 % and BMP error was 1.08. It can be seen, that
the method was able to estimate respiratory airflow
very well even in this kind of complicated situations.
Figure 3: Short segment of example signals during apneic
event. Upper subfigure: spirometer signal (solid line) and
the estimated respiratory airflow signal (dotted line). Lower
subfigure: rib cage respiratory effort belt signal (solid line)
and abdominal respiratory effort belt signal (dotted line).
Figure 4: Example signals of COPD patient. Upper
subfigure: spirometer signal (solid line) and the estimated
respiratory airflow signal (dotted line). Lower subfigure: rib
cage respiratory effort belt signal (solid line) and abdominal
respiratory effort belt signal (dotted line).
Measurement data of patient 4 included
thoracoabdominal asynchrony more or less during the
whole measurement session. Figure 5 depicts one
example of this. Estimation model M2 (N = 8) was
used with the POST setup and the results were the
following: R
2
was 0.94, tidal volume error was 4.7 %,
minute volume error was 2.6 % and BPM error was
0.02. These results are consistent with our earlier
findings indicating that our method produces very
good results with thoracoabdominal asynchrony
signals too (Seppänen, 2013).
HEALTHINF 2016 - 9th International Conference on Health Informatics
80

Figure 6 demonstrates the performance difference
between PRE and POST setups. In this case,
estimation model M1 (N = 16) was used firstly with
the PRE setup and secondly with the POST setup. The
measurement data was from patient 1. It can be seen
from Figure 6 that there were clear differences with
the estimated respiratory airflows (PRE setup with
dotted line and POST setup with bold line). Following
numerical results demonstrate these differences
further. The results for the PRE setup were: R
2
was
0.82, tidal volume error was 34.8 %, minute volume
error was 35.8 % and BPM error was 0.00.
Equivalently results for the POST setup were: R
2
was
0.93, tidal volume error was 14.6 %, minute volume
error was 13.9 % and BPM error was 0.00. Although,
the PRE setup was otherwise remarkably worse than
POST setup in this case, BPM was estimated very
accurately.
Figure 5: Example signals of thoracoabdominal
asynchrony. Upper subfigure: spirometer signal (solid line)
and the estimated respiratory airflow signal (dotted line).
Lower subfigure: rib cage respiratory effort belt signal
(solid line) and abdominal respiratory effort belt signal
(dotted line).
Figure 6: Short segment of example signals depicting the
difference of PRE and POST setups: spirometer signal (thin
line), the first estimated respiratory airflow signal (PRE
setup, dotted line) and the second estimated respiratory
airflow (POST setup, bold line).
3.3 Limitations of Study
The study included a number of limitations. Firstly,
the study included only five patients. The study
should be repeated with a larger data set in order to
draw more general conclusions. Secondly, respiratory
effort belts cannot be used if the surgical wound is in
the area where the belts are placed. However, the
proposed method could be applied to the
measurement data acquired with other sensors
without this kind of restriction, such as acceleration
sensors. Thirdly, as was pointed out in Section 3.2
during the apneic event there is no respiratory airflow
but still estimated respiratory airflow shows
otherwise. This could be resolved by detecting the
changed pattern of movements from respiratory effort
belts and replacing these parts of the respiratory
airflow estimate with zero airflow. This remains
future work.
4 CONCLUSIONS
Here, a method was proposed to estimate accurate
continuous respiratory airflow postoperatively. The
data from respiratory effort belts were calibrated with
a spirometer using an extended multiple linear
regression method. The results showed that training
the estimation model with the postoperative data
produced much more accurate results than training
the estimation model with the preoperative data.
It was demonstrated with data from five different
patients in postoperative situation that estimated
respiratory airflow signals have very accurate
waveforms. In addition, tidal volume, minute volume
and respiratory rate can be calculated remarkably
accurately from these signals. The method produced
very good estimates even from challenging
respiration signals: low airflows, COPD, hypopneic
events and thoracoabdominal asynchrony.
In summary, the presented method is able to
produce estimates of postoperative respiratory
airflow waveforms to enable accurate, continuous,
non-invasive respiratory monitoring postoperatively.
ACKNOWLEDGEMENTS
Finnish Cultural Foundation, North Ostrobothnia
Regional Fund and International Doctoral
Programme in Biomedical Engineering and Medical
Physics (iBioMEP) are gratefully acknowledged for
financial support.
Respiratory Effort Belts in Postoperative Respiratory Monitoring: Pilot Study with Different Patients
81

REFERENCES
Cepeda M.S., Farrar J.T., Baumgarten M., Boston R., Carr
D.B., Strom B.L., 2003. Side effects of opioids during
short-term administration: Effect of age, gender, and
race. Clinical Pharmacology & Therapeutics, 74(2):
102-112.
Drummond G.B., Bates A., Mann J., Arvind D.K., 2013.
Characterization of breathing patterns during patient-
controlled opioid analgesia. British Journal of
Anaesthesia, 111(6): 971-978.
Etches R., 1994. Respiratory depression associated with
patient-controlled analgesia: a review of eight cases.
Canadian Journal of Anaesthesia, 41(2): 125-132.
Gamil M., Fanning A., 1991. The first 24 hours after
surgery. A study of complications after 2153
consecutive operations. Anaesthesia, 46: 712-715.
George J.A., Lin E.E., Hanna M.N., Murphy J.D., Kumar
K., Ko P.S., Wu C.L., 2010. The effect of intravenous
opioid patient-controlled analgesia with and without
background infusion on respiratory depression: a meta-
analysis. Journal of Opioid Management, 6(1): 47-54.
Lovett P.B., Buchwald J.M., Sturmann K., Bijur P., 2005.
The vexatious vital: neither clinical measurements by
nurses nor an electronic monitor provides accurate
measurements of respiratory rate in triage. Annals of
Emergency Medicine, 45(1): 68-76.
Lynn L.A., Curry J.P., 2011. Patterns of unexpected in-
hospital deaths: a root cause analysis. Patient Safety in
Surgery, 5:3.
Ramsay M.A.E., Usman M., Lagow E., Mendoza M.,
Untalan E., De Vol E., 2013. The accuracy, precision
and reliability of measuring ventilatory rate and
detecting ventilatory pause by rainbow acoustic
monitoring and capnometry. Anesthesia & Analgesia,
117(1): 69-75.
Seppänen, T.M., Alho, O.-P., Seppänen T., 2013. Reducing
the airflow waveform distortions from breathing style
and body position with improved calibration of
respiratory effort belts. Biomedical Engineering
Online, 12:97.
Taylor S., Kirton O.C., Staff I, Kozol R.A., 2005.
Postroperative day one: a high risk period for
respiratory events. The American Journal of Surgery,
190(5): 752-756.
Voscopoulos C.J., MacNabb C.M., Brayanov J., Qin L.,
Freeman J., Mullen G.J., Ladd D., George E., 2015. The
evaluation of a non-invasive respiratory volume
monitor in surgical patients undergoing elective surgery
with general anesthesia. Journal of Clinical Monitoring
and Computing, 29(2): 223-230.
Voscopoulos C., Ladd D., Campana L., George E., 2014.
Non-invasive respiratory volume monitoring to detect
apnea in post-operative patients: case series. Journal of
Clinical Medicine Research, 6(3): 209-214.
Voscopoulos C.J., MacNabb C.M., Freeman J., Galvagno
S.M., Ladd D., George E., 2014. Continuous
noninvasive respiratory volume monitoring for the
identification of patients at risk for opioid-induced
respiratory depression and obstructive breathing
patterns. Journal of Trauma and Acute Care Surgery,
77(3): S208-S215.
Voscopoulos C, Brayanov J., Ladd D., Lalli M., Panasyuk
A., Freeman J., 2013. Evaluation of a novel noninvasive
respiration monitor providing continuous measurement
of minute ventilation in ambulatory subjects in a variety
of clinical scenarios. Anesthesia & Analgesia, 117(1):
91-100.
HEALTHINF 2016 - 9th International Conference on Health Informatics
82
