
A Framework for Creating Realistic Synthetic Fluorescence
Microscopy Image Sequences
Matsilele Mabaso
1
, Daniel Withey
1
and Bhekisipho Twala
2
1
MDS(MIAS), Council for Scientific and Industrial Research, Pretoria, South Africa
2
Department of Electrical and Electronic Engineering, University of Johannesburg,
Auckland Park, Johannesburg, South Africa
Keywords: Synthetic Image Sequences, Microscopy Bioimaging, Spot Detection.
Abstract: Fluorescence microscopy imaging is an important tool in modern biological research, allowing insights into
the processes of biological systems. Automated image analysis algorithms help in extracting information
from these images. Validation of the automated algorithms can be done with ground truth data based on
manual annotations, or using synthetic data with known ground truth. Synthetic data avoids the need to
annotate manually large datasets but may lack important characteristics of the real data. In this paper, we
present a framework for the generation of realistic synthetic fluorescence microscopy image sequences of
cells, based on the simulation of spots with realistic motion models, noise models, and with the use of real
background from microscopy images. Our framework aims to close the gap between real and synthetic
image sequences. To study the effect of real backgrounds, we compared three spot detection methods using
our synthetic image sequences. The results show that the real background influences spot detection,
reducing the effectiveness of the spot detection algorithms, indicating the value of synthetic images with a
realistic background in system validation.
1 INTRODUCTION
Advances in bioimaging based on fluorescence
microscopy have become fundamental in biomedical
and medical research. The use of fluorescence
microscopy and specific staining methods makes the
biological molecules to appear as bright particles
called spots. These bright particles are local intensity
maxima whose intensity level is significantly
different from their neighbourhood. This technique
generates a huge amount of data which is degraded
by factors such as noise and non-uniformity in the
background. Automated image analysis algorithms
are used to study and analyse these images.
Evaluation of these algorithms in real image datasets
requires manual annotation to estimate the ground
truth. However, the process of manual annotation
requires an expert to follow hundreds of spots
moving in an image sequence. This process can be
tedious, susceptible to errors and the ground truth
varies when repeated.
To avoid the problem of manual annotation,
several studies (Genovesio et al., 2006; Sbalzarini
and Koumoutsakos, 2005; Smal et al., 2010; Yoon et
al., 2008; Ruusuvuori et al., 2008; Ruusuvuori et al.,
2010; Rezatifighi et al., 2013) introduced the use of
synthetic image sequences to simulate real
microscopy images. The use of synthetic images
became popular because they contain the ground
truth data and give the opportunity to compare and
validate the results of automated methods. Most
existing frameworks for the creation of synthetic
image sequences (Feng et al., 2011; Smal et al.,
2010; Smal et al., 2008) make certain assumptions,
such as: no background structures, fixed shape for
spots and fixed signal to noise ratio.
(a) (b)
Figure 1: A selection of images with multiple spots. (a)
real fluorescence microscopy image, and (b) synthetic
image with a real background. The background in (b) was
obtained from a different study.
Mabaso, M., Withey, D. and Twala, B.
A Framework for Creating Realistic Synthetic Fluorescence Microscopy Image Sequences.
DOI: 10.5220/0005699200850092
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 2: BIOIMAGING, pages 85-92
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
85
These assumptions make creations of synthetic
images simpler; however, they do not fully reflect
the complexity of real images.
In this work, we describe a powerful framework
for creating realistic synthetic image sequences. The
approach presented in this study is based on the use
of real microscopy image sequences, unlike other
frameworks that simulate the entire image sequence.
Instead of learning the background and trying to
simulate it, our framework makes use of real
microscopy images with synthetic spots. To simulate
our spots we place a Gaussian profile directly into
the real image. These will result in partially-
synthetic image sequences. The proposed framework
is described for 2D images but can also be extended
to 3D.
This paper is organized as follows: Section 2
gives some related work, followed by Section 3
which explains the framework strategy, then Section
4 and 5 present the experimental set-up and results,
and, finally, Section 6 concludes the study.
2 RELATED WORK
There exist several studies for the creation of
synthetic sequences of microscopy images.
Smal (Smal, 2015) proposed a framework that
can mimic images acquired using fluorescence
microscopy. The procedure can simulate background
structures and spots, however, it does not fully
mimic the background of real images, and spot
motion is not considered. Another study (Genovesio
et al., 2006) generated synthetic images using a
mixture of Gaussians to form the background. Their
study modelled some image properties however, it
lacked the properties of a real background structures.
Similar to (Genovesio et al., 2006), (Yoon et al.,
2008) proposed a framework which can model the
movements of spots in microscopy images.
However, (Yoon et al., 2008) did not take into
account the background in microscopy images.
There exist few methods which can model the
effects of image noise, spot motion, and realistic
background in synthetic microscopy images. The
work by (Smal, 2015) can model noise and
background but the motion of spots was not
considered. Another work by (Rezatifighi et al.,
2013) uses HDome transformation (Vincent, 1993)
to estimate the background in real microscopy image
sequences. Although, their study can model spot
motion and noise, it still lacks important
characteristics of real data.
A recent study by (Chenouard et al., 2014)
compared the performance of different tracking
methods using synthetic image sequences. Their
sequences contained spots moving in random walk
with varying velocities, and Gaussian noise was
used to simulate the kind of noise found in
microscopy images. However, the disadvantage of
their sequences is lack of background structures.
One of the major conclusions in their study is the
need for synthetic image sequences with realistic
background.
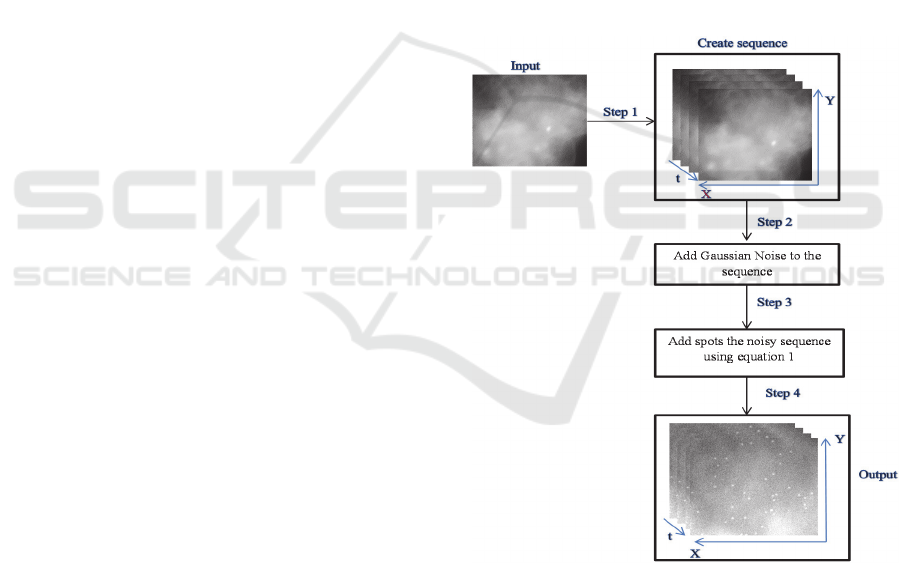
3 OUR FRAMEWORK
To generate our realistic synthetic image sequences,
we propose an improved framework as shown in,
Figure 2, which is able to create realistic synthetic
image sequences of fluorescence microscopy.
Figure 2: A diagram showing the steps involved in our
framework for the creation of synthetic image sequences.
3.1 Reference Data
An example of a real microscopy image with mRNA
spots is shown in Figure 1(a).
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
86

3.2 Background Modelling
Existing work on creating synthetic image sequences
are based on estimating the background either by
using HDome (Vincent, 1993) or Gaussian mixture
model (Genovesio et al., 2006). The disadvantage of
estimating the background is that it will still be
different from the real background. In our
framework, instead of simulating the background,
we make use of real microscopy images (without
spots) and add the spots. The real images were
obtained from our collaborator, the Synthetic
Biology Research Group at the CSIR.
3.3 Spot Model
Fluorescence microscopy images contain a number
of bright particles (spots) superimposed on an
uneven background, as shown in Figure(a). The
most common approach to model these spots is to fit
a Gaussian intensity profile (Cheezum et al., 2001;
Zhang et al., 2007; Carter et al., 2005). In this work,
we considered a 2D Gaussian function with four
parameters, the position, and , standard
deviation, and peak intensity. The model for a single
spot is given by:
,
(1)
The parameters,
,
describes the width of the
spots, and, , the spot amplitude. In order to model
an isotropic spot, the parameters,
and
were set
to be equal.
3.4 Spot Motion Models
The movements of spots in microscopy imaging can
be described using some statistical models of
motion. A number of studies (Genovesio et al.,
2006; Feng et al., 2011; Rezatifighi et al., 2013)
suggested the use of three kinds of models to
describe the kinds of spot movements in microscopy
images (Genovesio et al., 2006). The models
include random walk, first order linear extrapolation,
and second order linear extrapolation, modelling
Brownian motion, constant speed, and constant
acceleration movements, respectively, which are
motions representative of biological motion
(Lakadamyali et al., 2003). To model the movement
of spots using the above mentioned motion models,
we used a plugin developed by (Chenouard, 2015)
3.5 Noise Generation
There exists many noise sources in microscopy
imaging which affects the image quality. To
simulate the kind of noise found in microscopy
imaging, we used additive Gaussian noise with mean
of zero and varying standard deviation, ~μ
0,
. Gaussian models are commonly used
models in microscopy imaging.
3.6 Signal to Noise Ratio
The quality of images can be expressed in terms of
signal to noise ratio (SNR). The SNR measures the
amount of noise in image and is widely used in
image processing. The signal to noise ratio was
defined as the ratio of spot intensity,
, divided
by the noise standard deviation,
;
(2)
4 EXPERIMENTAL SET-UP
4.1 Synthetic Sequences
The framework presented in this study is capable of
simulating different kinds of microscopy image
sequences. In order to study the effect of real
background on synthetic image sequences, we
created two types of synthetic image scenarios. The
first scenario consisted of image sequences with no
background structures (named NOBGND) and the
second scenario consisted of synthetic sequences
with real fluorescence background structures.
BGND refers to background. For the second
scenario four realistic synthetic image sequences
(named, BGND0, BGND1, BGND2 and BGND3)
were created by varying the background as shown
in, Figure 3, All the scenarios were corrupted by
Gaussian noise, with the mean of zero and varying
standard deviation {2.86, 5, 10, 20}. The following
signal to noise ratios (SNR) levels were explored {7,
4, 2, 1} where the spot intensity was 20 gray levels.
Each synthetic image sequence created was of 100
time steps with image dimension of 512 by 512
pixels. The density of spots in each image of a
sequence was on the order of {50-100} and the spots
motion models were governed by Brownian motion.
The spot numbers, dynamics, start and end were
randomized in order to mimic the kinds of properties
in real microscopy images. MATLAB was used to
A Framework for Creating Realistic Synthetic Fluorescence Microscopy Image Sequences
87

add spots, and the OMERO.matlab-4.3.3 toolbox
was used to read and save images.
4.2 Detection Methods
In order to study the effect of real background on
synthetic image sequence, we compared results from
three spot detection methods applied to our synthetic
image sequences. These methods were chosen based
on their implementation availability and they were
also being used in different comparison studies
(Ruusuvuori et al., 2010; Smal et al., 2010). The
detection algorithms compared are, Isotropic
Undecimated Wavelet Transform (IUWT) (Olivo-
Marin, 2008), Feature Point Detection (FPD)
(Sbalzarini and Koumoutsakos, 2005) and HDome
Transformation (Smal et al., 2010; Vincent, 1993).
A detailed description of each method is found in
Appendix A.
4.3 Performance Measure
In order to test the performance of the three
detection methods, we computed several measures:
true positives (TP), false positives (FP) and false
negatives (FN). True positives are detected spots
that correspond to the ground-truth spots. If the
detected spot does not correspond to the ground
truth it is considered as a false positive. A missed
ground truth spot is considered as a false negative.
Two performance measures are considered in this
study, Recall and Precision (Allalou et al., 2010).
Recall measures the ratio of correctly detected spots
overall ground-truth spots, and is defined as:
(a)
(b)
(c)
(d)
(e)
Figure 3: Examples of synthetic image sequences created
using our framework. (a) NOBGND, and (b) BGND0, (c)
BGND1, (d) BGND2, and (e) BGND3.
(3)
Precision measures the ratio of correctly detected
spots among all detected spots and defined as:
(4)
Where
the number of true positives is,
is
the number of false negatives and
the number of
false positives.
Then, the
measure is computed as a
weighted average of the two measures, precision and
recall:
2
(5)
A good detection method should have the value of
approaching one.
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
88
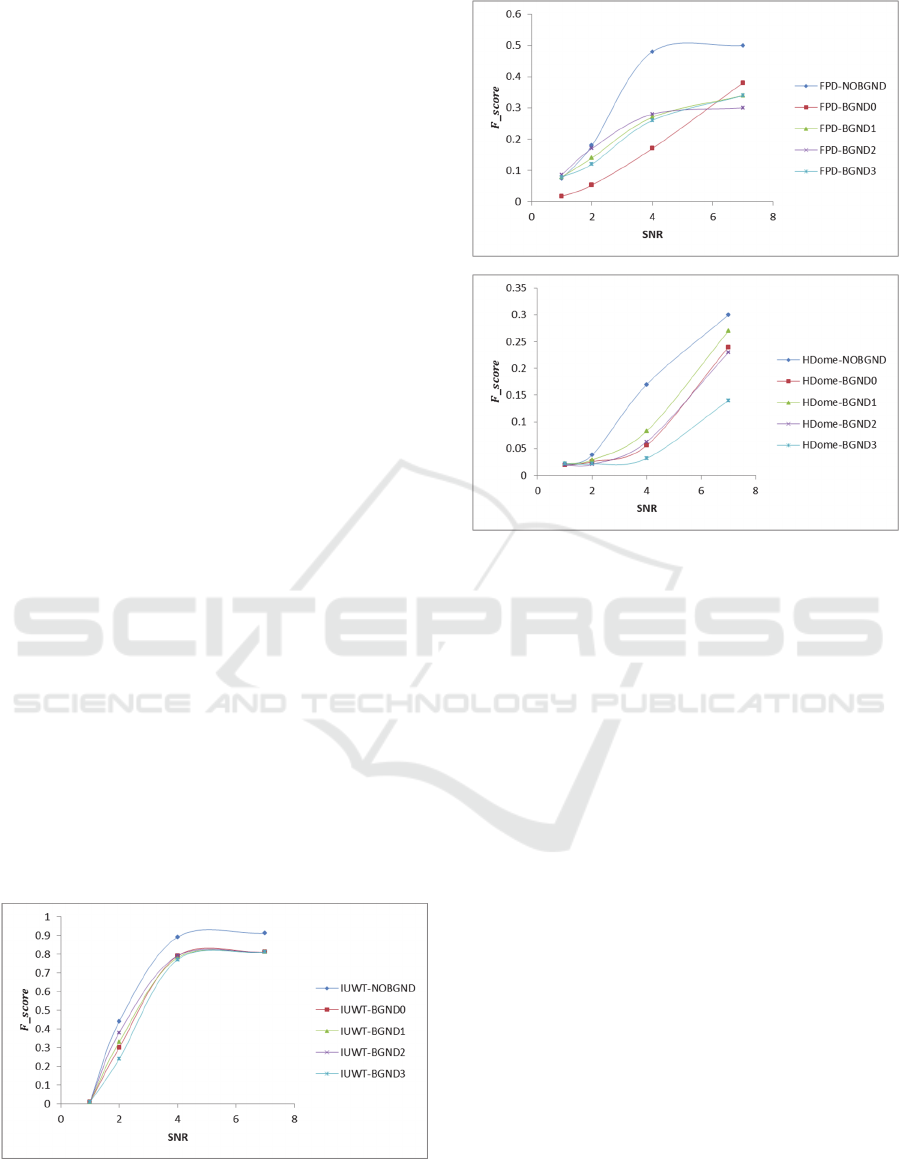
5 EXPERIMENTAL RESULTS
We evaluate the performance of three detection
methods using synthetic image sequences consisting
of five experimental scenarios. The first scenario
consisted of image sequences with no background
structures, NOBGND. This will help with the
evaluation of the performance of the algorithms as a
function of image noise (SNR). The second to fifth
scenario consisted of image sequences with a real
background (BGND0, BGND1, BGND2 and
BGND3). The second to fifth experimental scenarios
were used to evaluate the performance of the
methods as a function of real background and image
noise. In all the scenarios, the spot motion was
governed by Brownian motion. For each method, the
performance measures, Recall, Precision and
,
were computed. It’s important to mention that the
only difference between image scenarios was the
background, and all other properties were the same.
Figure 4 shows the results of all detection
methods in terms of
. The results show that all
methods performed well on NOBGND sequences
compared to sequences with a background. It is
noted that the HDome and FPD methods fail to
reach
nearly one on NOBGND test case at
SNR=7; because the challenges of handling
overlapping spots. It turns out that the performance
of the algorithms decreases when the real
background is introduced. The decrease in
performance of the algorithms could be explained by
the increase in the number of false positives (FP)
and false negatives (FN) detected by the algorithms
when the background is introduced, and thus
affecting the
. In all experiments, the IUWT
method performed better compared to other
methods. However at SNR=2 or below all methods
drop in performance for all experiments.
(a)
(b)
(c)
Figure 4: The curves of
versus SNR for the detection
methods applied to two synthetic image scenarios as
described in Section 4.1. (a) IUWT, (b) FPD and, (c)
HDome. All methods perform less well with realistic
background.
6 CONCLUSIONS
In this work we presented a framework for the
simulation of fluorescence microscopy images
sequences and also study the effect of real
background on synthetic image sequences. The
framework improves the modelling of real
microscopy image sequences by including realistic
spots, realistic noise, and realistic motion with real
image background. The synthetic image sequences
created using this framework offer a better way to
evaluate different detection and tracking algorithms
since the ground truth is available. Our evaluation
results showed that the performance of three
detection methods is reduced when tested with
synthetic image sequences exhibiting realistic
background, compared to the sequences which had
no background. This showed that the real
background has an effect on spot detection algorithm
performance. The performance of the detection
methods is reduced in the presence of background
structures.
A Framework for Creating Realistic Synthetic Fluorescence Microscopy Image Sequences
89

ACKNOWLEDGEMENTS
This work was carried out with the financial support
of the Council for Scientific and Industrial Research
(CSIR) and the Electrical and Electronic
Engineering Department at the University of
Johannesburg, South Africa. We would also like to
thank the Synthetic Biology research group at the
CSIR for providing us with real microscopy images.
REFERENCES
Allalou, A., Pinidiyaarachchi, A. and Wählby, C. (2010)
'Robust signal detection in 3D fluorescence
microscopy ', Cytometry, vol. 77A, no. 1, pp. 86-96.
Carter, B.C., Shubeita, G.T. and Gross, S.P. (2005)
'Tracking single particles: a user-friendly quantitative
evaluation', Physical Biology, vol. 2, no. 1, pp. 60-72.
Cheezum, M.K., Walker, W.F. and Guilford, W.H. (2001)
'Quantitative comparison of algorithms for tracking
single fluorescent particle', Biophysical Journal, vol.
81, pp. 2378-2388.
Chenouard, N. (2015) Particle tracking benchmark
generator, [Online], Available:
http://icy.bioimageanalysis.org/plugin/Particle_trackin
g_benchmark_generator [1 September 2015].
Chenouard, N., Ihor, S., de Chaumont, F., Maska, M.,
Sbalzarini, I.F., Gong, Y., Cardinale, J., Carthel, C.,
Coraluppi, S., Winter, M., Cohen, A.R., Godinez,
W.J., Rohr, K., Kalaidzidis, Y., Liang, L., Duncan, J.,
Shen, H., Xu, Y., Magnusson, K.E., Jalden, J. et al.
(2014) 'Objective comparison of particle tracking
methods', Nature Methods, vol. 11, no. 3, pp. 281-290.
Crocker, J.C. and Grier, D.G. (1996) 'Methods of Digital
Video Microscopy for Colloidal Studies', Journal of
Colloid and Interface Science 179, pp. 298-310.
Feng, L., Xu, Y., Yang, Y. and Zheng, X. (2011) 'Multiple
dense particle tracking in fluorescence microscopy
images based on multidimensional assignment',
Journal of Structural Biology, vol. 173, pp. 219-228.
Genovesio, A., Liendl, T., Emiliana, V., Parak, W.J.,
Coppey-Moisan, M. and Olivo-Marin, J.-C. (2006)
'Multiple particle tracking in 3d+t microscopy:
Method and application to the tracking of endocytosed
quantum dots', IEEE Transactions on Image
Processing, vol. 15, no. 5, pp. 1062-1070.
Lakadamyali, M., Rust, M.R., Babcock, H.P. and Zhuang,
X. (2003) 'Visualizing infection of individual
influenza viruses', Proceedings of the National
Academy of Science of the United States of America,
9280-9285.
Olivo-Marin, J.-C. (2008) 'Extraction of spots in biological
images using multiscale products', Pattern
Recognition, vol. 35, no. 9, pp. 1989-1996.
Rezatifighi, S.H., Pitkeathly, W.T., Goud, S., Hartley, R.,
Mele, K., Hughes, W.E. and Burchfield, J.G. (2013) 'A
framework for generating realistic synthetic sequences
of total internal reflection fluorescence microscopy
images', 10th IEEE International Symposium on
Biomedical Imaging, 157-160.
Ruusuvuori, P., Äijö, T., Chowdhury, S., Garmaendia-
Torres, C., Selinummi, J., Birbaumer, M., Dudley,
A.M., Pelkmans, L. and Yli-Harja, O. (2010)
'Evaluation of methods for detection of fluorescence
labeled subcellular objects in microscope images',
BMC Bioinformatics, vol. 11, pp. 1-17.
Ruusuvuori, P., Lehmussola, A., Selinummi, J., Rajala, T.,
Huttunen, H. and Yli-Harja, O. (2008) 'Benchmark set
of synthetic images for validating cell image analysis
algorithms', Proceedings of the 16th European Signal
Processing Conference, EUSIPCO.
Sbalzarini, I.F. and Koumoutsakos, P. (2005) 'Feature
Point Tracking and Trajectory Analysis for Video
Imaging in Cell Biology', Journal of Structural
Biology, vol. 151, no. 2, pp. 182-195.
Smal, I. (2015) Synthetic data generator
, September,
[Online], Available:
http://smal.ws/wp/software/synthetic-data-generator/.
Smal, I., Loog, M., Niessen, W. and Meijering, E. (2010)
'Quantitative comparison of spot detection methods in
fluorescence microscopy', IEEE Trans on Medical
Imaging, vol. 29, no. 2, pp. 282-301.
Smal, I., Meijering, E., Draegenstein, K., Galjart, N.,
Grigoriev, I., Akhmanova, A., van Royen, M.,
Houtsmuller, A.B. and Niessen, W. (2008) 'Multiple
object tracking in molecular bioimaging by Rao-
Blackwellized marginal particle filtering', Medical
Image Analysis , vol. 12, no. 6, pp. 764-777.
Vincent, L. (1993) 'Morphological grayscale
reconstruction in image analysis: Applications and
efficient algorithms ', IEEE Trans Image Process, vol.
2, pp. 176-201.
Yoon, J.W., Bruckbauer, A., Fitzgerald, W.J. and
Klenerman, D. (2008) 'Bayesian inference for
improved single molecule fluorescence tracking',
Biophysical Journal, vol. 94, pp. 4932-4947.
Zhang, B., Zerubia, J. and Olivo-Marin, J.-C. (2007)
'Gaussian approximations of fluorescence microscope
PSF models', Applied Optics, vol. 46, no. 10, pp. 1-34.
APPENDIX-A
Spot Detection Methods
Isotropic Undecimated Wavelet Transform
The method of IUWT was proposed in (Olivo-
Marin, 2008) for the detection of spots in biological
images. The algorithm is based on the assumption
that spots will be present at each scale of wavelet
decomposition and thus will appear in the multiscale
product. The ́ trous wavelet transform step is based
on the convolution of the image , row by row
and column by column with a symmetric low pass
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
90

filter
1,4,6,4,1
16
⁄
, resulting in a smoothed
image
,. This process is repeated for scale
levels, augmenting the filter with 2
1 zeros
between taps in each case. The corresponding
wavelet coefficients,
,, are given as:
,
,
,
0
.
(6)
Then a hard thresholding step is applied to reduce
the effect of noisy wavelet coefficients.
,
,,
,
0,
,
(7)
With
, where
is the standard deviation of
noisy wavelet coefficients at scale and 3.
Thus, after hard thresholding, a multiscale
product of each wavelet coefficient is computed to
get a correlation image,
,,
,
,
.
(8)
This correlation image
,
, is binarized with
equation (9) and the resulting connected components
yield the final particles detected.
,
255,
,
.
0,
(9)
Where,
, is the predetermined detection level. A
spot is accepted only at positions where the
correlation is above
,
Feature Point Detection
The method of feature point detection was proposed
in (Crocker and Grier, 1996) and used for the
detection of bright particles in (Sbalzarini and
Koumoutsakos, 2005). The algorithm consists of
four steps:
1) Image Restoration: this step corrects the
imperfection in the image by using a box-car
average estimation and simultaneously enhances
spot-like structures by convolving with a
Gaussian kernel. The convolution kernel is given
by:
1
1
4
1
21
,
(10)
where
and are normalization factors,
defines
the kernel width and is a user-tunable constant,
thus the final image after restoration is given by:
,
,
,
(11)
where , and , are pixel coordinates in the
image and kernel, respectively.
2) Estimating the Particle Location: this is done
by locating local intensity maxima in the filtered
image,
,. A local maximum is considered
to be a spot if it has the highest intensity within a
local window and the intensity is in the
highest percentile. These local maximum are
identified using a gray scale dilation with a disc
as the structural element. Then pixels of the
filtered image with the same value as the dilation
transformed image are taken as candidate
locations.
3) Refining the Particle Location: this step
reduces the standard deviation of the position
measurement. It is based on the assumption that
the local intensity maximum of the point at
,
is near the geometric center
,
of
the spot. The offset is approximated by the
distance to the gray-level centroid in the filtered
image,
,:
1
,
.
(12)
Factor
, is the sum of all pixels values over
feature point given as:
,
.
(13)
Then the refined location estimate is determined as:
,
,
.
(14)
4) Non-particle Discrimination: this step rejects
false identifications from sources such as auto
fluorescence and dust. This step is based on the
intensity moments of order 0 and 2, and
identifies true particles as those within a cluster
in the
,
plane.
HDOME
The method of HDome transformation was proposed
in (Vincent, 1993) and used in biological application
in (Smal et al., 2010). The method is based on the
mathematical morphology:
,
,
,
,
where
,
denotes the results of
subtracting a constant, h, from a gray-scale image
A Framework for Creating Realistic Synthetic Fluorescence Microscopy Image Sequences
91

,, and
,
is the morphological
reconstruction of the gray-scale image, , from
,
. The gray-level reconstruction is
obtained by geodesic dilation of
,
under
,. The algorithm starts by reducing
background noise by convolving the original gray
scale image with a LoG filter and simultaneously
enhancing particles. Then HDome method is applied
to the filtered image to keep spots of height superior
to the threshold .
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
92
