
Tackling the Problem of Data Imbalancing for Melanoma Classification
Mojdeh Rastgoo
1,2
, Guillaume Lemaitre
1,2
, Joan Massich
1
, Olivier Morel
1
, Franck Marzani
1
,
Rafael Garcia
2
and Fabrice Meriaudeau
1
1
LE2I UMR6306, CNRS, Arts et M
´
etiers, Universit
´
e de Bourgogne Franche-Comt
´
e,
12 rue de la Fonderie, 71200 Le Creusot, France
2
ViCOROB, Universitat de Girona, Campus Montilivi, Edifici P4, 17071 Girona, Spain
Keywords:
Imbalanced, Classification, Melanoma, Dermoscopy.
Abstract:
Malignant melanoma is the most dangerous type of skin cancer, yet melanoma is the most treatable kind of
cancer when diagnosed at an early stage. In this regard, Computer-Aided Diagnosis systems based on machine
learning have been developed to discern melanoma lesions from benign and dysplastic nevi in dermoscopic
images. Similar to a large range of real world applications encountered in machine learning, melanoma clas-
sification faces the challenge of imbalanced data, where the percentage of melanoma cases in comparison
with benign and dysplastic cases is far less. This article analyzes the impact of data balancing strategies at
the training step. Subsequently, Over-Sampling (OS) and Under-Sampling (US) are extensively compared in
both feature and data space, revealing that NearMiss-2 (NM2) outperform other methods achieving Sensitivity
(SE) and Specificity (SP) of 91.2% and 81.7%, respectively. More generally, the reported results highlight that
methods based on US or combination of OS and US in feature space outperform the others.
1 INTRODUCTION
Malignant melanoma is the deadliest type of skin can-
cer, accounting for the vast majority of skin cancer
deaths (American-Cancer-Society, 2014). According
to latest reports, melanoma causes over 20,000 deaths
annually in Europe (Forsea et al., 2012). In 2014,
the American Cancer Society also reported that the
number of new diagnosed cases is 76,100 with 9710
estimated deaths (American-Cancer-Society, 2014).
Nevertheless, melanoma is the most treatable kind of
cancer if diagnosed early.
Melanoma is clinically diagnosed through vi-
sual inspection and deep analysis of the lesion,
using clinical imaging techniques such as dermo-
scopic imaging. The clinical diagnosis of early
stage melanoma is commonly based on the “ABCDE”
rule (Abbasi et al., 2004), defined as Asymmetry, ir-
regular Borders, variegated Colors, Diameters greater
than 6 mm and its Evolution over time. These inspec-
tions and analysis are challenging since, first different
lesions such as melanoma and dysplastic nevi share
similar characteristics in terms of “ABCD” and sec-
ond the necessity to perform patient follow-up over
the years. Therefore, the research communities have
dedicated their efforts to develop computerized lesion
analysis algorithms for classification of melanoma le-
sions.
When studying skin lesions, akin to other med-
ical applications, the percentage of malignant cases
is far less when compared with benign cases. This
problem is frequently referred as “class imbalance”
problem (Prati et al., 2009) and has been encountered
in multiple areas such as telecommunication man-
agements, bioinformatics, fraud detection, and med-
ical diagnosis. Imbalanced data substantially com-
promises the learning process since most of the
standard machine learning algorithms expect bal-
anced class distribution or an equal misclassification
cost (He et al., 2009).
Medical data are prone to such drawbacks due to
the fact that the portion of diseased samples or pa-
tients is far lower than healthy cases. Furthermore,
the detection and classification of minority malignant
cases are highly essential so that the Sensitivity (SE)
of developed algorithms need to be maximized. Con-
sequently, the problem of imbalanced data is usually
addressed by employing different techniques which
do not impair the topology of the data. Despite the
fact that classification of malignant melanoma has
been extensively studied (Rastgoo et al., 2015a), up
to our knowledge, only few works tackled the issue
32
Rastgoo, M., Lemaitre, G., Massich, J., Morel, O., Marzani, F., Garcia, R. and Meriaudeau, F.
Tackling the Problem of Data Imbalancing for Melanoma Classification.
DOI: 10.5220/0005703400320039
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 2: BIOIMAGING, pages 32-39
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
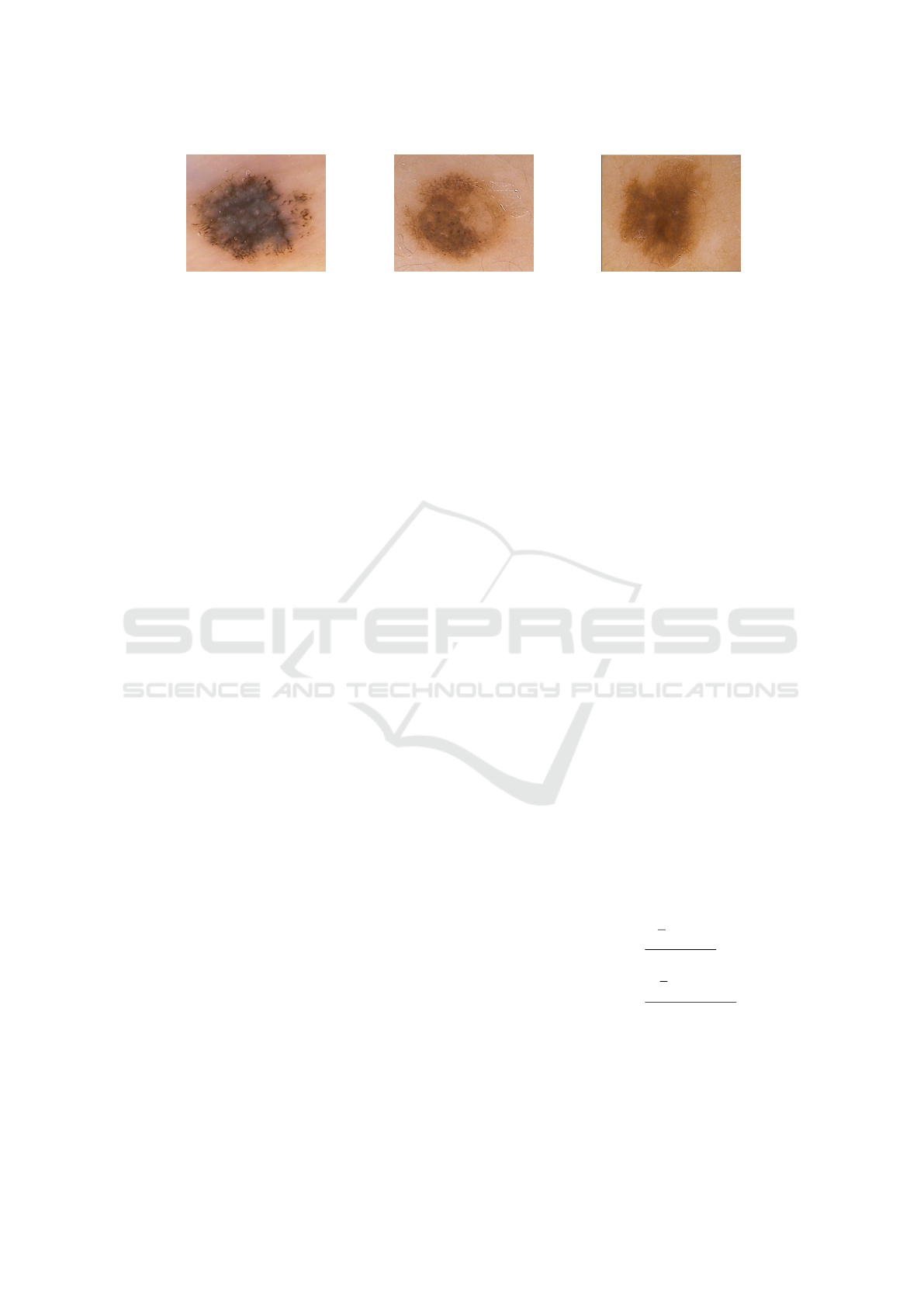
(a) Melanoma lesion (b) Dysplastic lesion (c) Benign lesion
Figure 1: Samples of PH
2
dataset, representing melanoma, dysplastic and benign lesions, respectively.
implied by imbalanced dataset (Barata et al., 2014,
Celebi et al., 2007). Barata et al. generate new
synthetic samples by adding a Gaussian noise
with fixed parameters to the samples belonging to
the minority class (Barata et al., 2014). Celebi et
al. and Capdehourat et al. over-sampled their
dataset using Synthetic Minority Over-sampling
TEchnique (SMOTE) (Chawla et al., 2002) to im-
prove the SE of their algorithm (Celebi et al., 2007,
Capdehourat et al., 2009).
This paper provides an insight to the specific
problem of classification of imbalanced dataset for
malenoma. To proceed, we review different tech-
niques proposed by the machine learning commu-
nity and compile a comprehensive quantitative eval-
uation. The rest of this paper is organized as follows:
an overview of the classification framework designed
to investigate data balancing techniques is presented
in Sect. 2. The balancing strategies are explained in
depth in Sect. 3 and the validation and classification
are discussed in Sect. 4. A quantitative evaluation is
discussed in Sect. 5 followed by a concluding section.
2 MATERIAL AND METHODS
Figure 4 illustrates and summarizes the experiment
designed to explore the data imbalance problem dur-
ing the classification of dermoscopic images. The
experimentation is based on the works presented
in (Rastgoo et al., 2015a, Rastgoo et al., 2015b) and
follows a cross-validated classification evaluation
framework. Details of the dataset used for the exper-
iments are given in Sect. 2.1. The extracted features
correspond to the highest performing subset of fea-
tures according to the latter mentioned studies and are
presented in Sect. 2.2.
2.1 Dataset
In order to allow future comparisons, we choose
to work with the only public dermoscopic dataset
PH
2
(Barata et al., 2014). This dataset is acquired
at Dermatology Service of Hospital Pedro Hispano,
Matosinhos, Portugal (Barata et al., 2014) with Tue-
binger Mole Analyzer system with a magnification of
20×. The 8-bits RGB color dermoscopic images were
obtained under the same conditions with a resolution
of 768 px ×560 px. This dataset contains 200 dermo-
scopic images divided into 160 benign and dysplastic
and 40 melanoma lesions. In terms of Ground Truth
(GT), histological diagnosis and segmentation of the
lesions are provided.
Due to an imbalance limitation of one of the tech-
niques here studied, the experimentation is conducted
on a data subset with an imbalance ratio of 1:3. Thus,
the subset is composed of 39 melanoma and 117 be-
nign and dysplastic lesions, randomly selected. Fig-
ure 1 shows three samples of this dataset, represent-
ing melanoma, dysplastic, and benign lesion, respec-
tively.
2.2 Feature Extraction
The Color Variance and Histogram (C
1
):
descriptor contains the mean and variance of
the color channels {R, G, B, H, S, V, L, A, B}
and a 42 bins histogram for each channel of the
set {R, G, B}. Thus, the final descriptor is made
of 144 features.
The Opponent Color Space Angle and Hue Histo-
gram: textbf(C
2
) is a robust and rotation in-
variant feature descriptor derived from the RGB
channels (Van De Weijer and Schmid, 2006),
such that:
H = arctan
√
3(R −G)
R + G −2B
!
,
θ
O
d
= arctan
√
3
R
0
d
−G
0
d
R
0
d
+ G
0
d
−2B
0
d
!
, (1)
where d denotes the spatial coordinates of (x, y)
and R
0
d
, G
0
d
, B
0
d
denote the first order derivatives
of RGB channels with respect to the coordinates.
This color descriptor is built by taking a 42 bins
histogram for the opponent angle θ
O
d
and the hue
Tackling the Problem of Data Imbalancing for Melanoma Classification
33

9
12
34
10
25
28
99
64 56
Original Image
-16 -13
9
-15 0 3
74
39
31
Local Difference
-1 -1 1
-1 1
1 1 1
Sign Component
16 13
9
15 3
74
39
31
Magnitude Component
(a)
CLBP
s
CLBP
m
CLBP
c
Grey level
of center
Local
Differences
Binary Patterns
CLBP Map
CLBP
Histogram
Original
Image
(b)
Figure 2: CLBP descriptor process, (a) represents an example on how local distances, sign and magnitude components are
calculated and (b) shows an overall view of CLBP process.
channel (H), for a final descriptor size of 84 di-
mensions.
Completed Local Binary Pattern (CLBP) (T
1
):
is a completed modeling of Local Binary
Pattern, especially designed for texture classifi-
cation (Guo and Zhang, 2010). This descriptor
encodes the magnitude and sign differences of
the central pixel with its neighbors and the grey
level of the central points in the local patterns
rather than only the sign differences (see Fig 2).
The sign CLBP
S
, magnitude CLBP
M
, and central
grey level CLBP
C
binary pattern are created by
encoding the local distance components and the
central grey levels to binary patterns. The CLBP
are calculated for each pixel in a given image and
the final descriptor is defined as their histogram.
The rotation invariant, uniform, and normalized
CLBP features is calculated considering a radius
of 24 px. The descriptor is composed of 26
dimensions.
Gabor Filter (T
2
): is a linear filter which is defined
as a modulation of a Gaussian kernel with a
sinusoidal wave. This filter is formulated in
Eq. (2) as two Gaussians with standard deviations
of σ
x
and σ
y
that vary along x and y axes and
it is modulated by a complex sinusoidal with a
wavelength of λ. Here θ represents the orientation
of the Gabor filter, ψ is the phase offset and s is
the scale factor. The filter bank is created using
six different orientations equally spaced in the
interval [0, π], along 4 scales with a downsizing
factor of 2:
g(x, y) = exp
−
x
02
2σ
2
x
+
y
02
2σ
2
y
cos
2π
x
0
λ
+ ψ
, (2)
where
x
0
= s(x cosθ + y sin θ),
y
0
= s(−x sinθ + y cos θ).
The final descriptor is composed 48 feature di-
mensions.
3 BALANCING STRATEGIES
Considering a binary classification problem, the class
with the smallest number of samples is defined as the
minority class and its counterpart is defined as the
majority class. The problem of data balancing cor-
responds to equalize the number of samples of both
the minority and majority classes. This task can be
achieved in either data or feature space.
3.1 Data Space Sampling
Data space sampling is related with the generation
of new synthetic samples by modifying the original
data ahead of any feature extraction processes. Over-
Sampling (OS) is performed on the original dataset by
generating synthetic melanoma images based on two
types of deformation (Rastgoo et al., 2015b). Further-
more, cubic b-spline interpolation is used with both
methods to approximate non-integer points in the im-
age. These deformations are considered since they
are more likely to occur, due to non-planar surface of
some body parts, skin wrinkles, camera rotation, and
position.
Random Deformation using Gaussian Motion:
achieved by deforming the original im-
age by adding a random Gaussian motion
N (µ, σ) = (0, 5) at each pixel compounded with
a global rotation of 80
◦
.
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
34

(a) (b) (c)
Figure 3: Data space transformation: (a) original synthetic data, (b) RDGM deformation, (c) BD deformation.
Barrel Deformation: corresponds to a deformation
of the original image using barrel distortion com-
pounded with a global rotation of 145
◦
.
A synthetic example illustrating the results of these
deformation is presented in Fig. 3.
3.2 Feature Space Sampling
Three strategies can be employed to overcome the
problem of imbalanced dataset: (i) Under-Sampling
(US), (ii) OS, and (iii) a combination of both. The
following sections give an overview of the techniques
used to tackle this issue.
3.2.1 Under-Sampling
Considering the problem of imbalanced, US is per-
formed such that the number of samples of the major-
ity class is reduced to be equal to the number of sam-
ples of the minority class. The following methods are
considered to perform such balancing.
Random Under-Sampling (RUS): is performed by
randomly selecting without replacement a subset
of samples from the majority class such that the
number of samples is then equal in both minority
and majority classes.
Tomek Link (TL): can be used to under-
sample the majority class of the original
dataset (Tomek, 1976). Let define a pair of
Nearest Neighbour (NN) samples (x
i
, x
j
) such
that their associated class label y
i
6= y
j
. The pair
(x
i
, x
j
) is defined as a Tomek Link (TL) if, by
relaxing the class label differentiation constraint,
there is no other sample x
k
defined as the NN
of either x
i
or x
j
. US is performed by removing
the samples belonging to the majority class and
forming a TL. It can be noted that this US strategy
does not enforce a strict balancing between the
majority and the minority classes.
Clustering Under-Sampling (CUS): refers to the
use of a k-means to cluster the feature space such
that k is set to be equal to the number of samples
composing the minority class. Hence, the cen-
troids of these clusters define the new samples of
the majority class.
NearMiss: offers three different meth-
ods to under-sample the majority
class (Mani and Zhang, 2003). In NearMiss-
1 (NM1), samples from the majority class are
selected such that for each sample, the average
distance to the k NN samples from the mi-
nority class is minimum. NearMiss-2 (NM2)
diverges from NM1 by considering the k farthest
neighbours samples from the minority class.
In NearMiss-3 (NM3), a subset M containing
samples from the majority class is generated
by finding the m NN from each sample of the
minority class. Then, samples from the subset
M are selected such that for each sample, the
average distance to the k NN samples from the
minority class is maximum. In our experiment, k
and m are fixed to 3.
Neighborhood Cleaning Rule (NCR): consists of
applying two rules depending on the class of
each sample (Laurikkala, 2001). Let define x
i
as a sample of the dataset with its associated
class label y
i
. Let define y
m
as the class of the
majority vote of the k NN of the sample x
i
. If y
i
corresponds to the majority class and y
i
6= y
m
, x
i
is rejected from the final subset. If y
i
corresponds
to the minority class and and y
i
6= y
m
, then the
k NN are rejected from the final subset. In our
experiment k is fixed to 3.
3.2.2 Over-Sampling
In the contrary, the data balancing can be performed
by OS in which the new samples belonging to the mi-
nority class are generated aiming at equalizing the
number of samples in both classes. Two different
methods are considered.
Random Over-Sampling: is performed by ran-
domly replicating the samples of the minority
class such that the number of samples is equal in
both minority and majority classes.
SMOTE: is a method to generate synthetic samples
in the feature space (Chawla et al., 2002). Let
Tackling the Problem of Data Imbalancing for Melanoma Classification
35

Figure 4: Framework outline.
define x
i
as a sample belonging to the minority
class. Let define x
nn
as a randomly selected sam-
ple from the k NN of x
i
, with k set to 3. There-
fore, a new sample x
j
is generated such that x
j
=
x
i
+ σ(x
nn
−x
i
), where σ is a random number in
the interval [0, 1].
3.2.3 Combination of OS and US
Subsequently, OS methods can be combined with US
methods to clean the subset created. In that regard,
two different combinations are tested.
SMOTE + TL: are combined to clean the samples
created using SMOTE (Batista et al., 2003).
SMOTE over-sampling can lead to over-
fitting which can be avoided by removing
the TL from both majority and minority
classes (Prati et al., 2009).
SMOTE + Edited Nearest Neighbour: are
combined for the same aforementioned rea-
son (Batista et al., 2004).
4 CLASSIFICATION
The classification is performed using a Random
Forests (RF) classifier. RF is an ensemble of decision
trees (Breiman, 2001) which generalizes the classifi-
cation process by applying two types of randomiza-
tion: at the tree level, each tree is fed by a bootstrap
made of S
0
samples built from the original data of size
S such that S = S
0
, and at the node level, a subset of
feature dimensions m is randomly selected from the
original dimension M such that m =
√
M. The trees
in RF are grown to their maximum length without any
pruning. Each tree in the ensemble casts a unit vote
in the final prediction and the final prediction is based
on combination of all the votes. RF is used with 100
un-pruned trees and the original feature dimension of
size M = {144, 84, 228, 26, 48, 74, 254, 276, 302}
4.1 Validation
We used a 10-fold cross-validation scheme to vali-
date our classifier with stratified sampling. However,
differently from the usual 10-fold cross-validation, 8
folds were kept for training and 2 folds for testing at
each iteration. The training set is balanced using pre-
viously described imbalanced techniques. The clas-
sification performance are reported in terms of aver-
age SE (TPR) and Specificity (SP) (TNR) over 10
runs of cross-validation. The visual and analytic in-
terpretation of these evaluation measures are depicted
in Fig. 5.
To select the best performance, similarly to
(Barata et al., 2013) we consider to evaluate the re-
sults based on a cost function, which defines the trade
off between SE and SP. This function is formulated
as:
C =
c
10
(1 −SE) + c
01
(1 −SP)
c
10
+ c
01
, (3)
where, c
10
and c
01
are the costs of incorrectly classi-
fying a melanoma and non-melanoma lesions, respec-
tively. In cancer classification such as melanoma, cor-
rectly identifying the cancer lesions has high impor-
tance (i.e., high SE). Thus incorrect classification of
melanoma is not desired and c
10
is a greater error and
evidently more costly and should be penalized more.
In order to achieve a high SE without significantly re-
ducing the value of SP, Barata et al. proposed to set
c
10
= 1.5 ×c
01
and c
01
= 1 (Barata et al., 2013). We
considered the same configuration for our cost func-
tion.
5 EXPERIMENTAL RESULTS
The classification results are reported in Table 1 using
the aforementioned features, the RF classifier, and the
different imbalancing techniques presented in Sect. 3
and Sect. 4.
Table 1 can be divided into three main parts rep-
resenting the results using imbalance data (IB), the
balancing in the data space OS and the balancing in
the feature space. These strategies are separated by
a double horizontal line. The strategies performed in
the feature space are subdivided into either OS or US
or a combination of OS follow by US (see horizontal
dashed line in Table 1).
In this table, based on the previously defined cost
function, the best performance for each feature set are
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
36
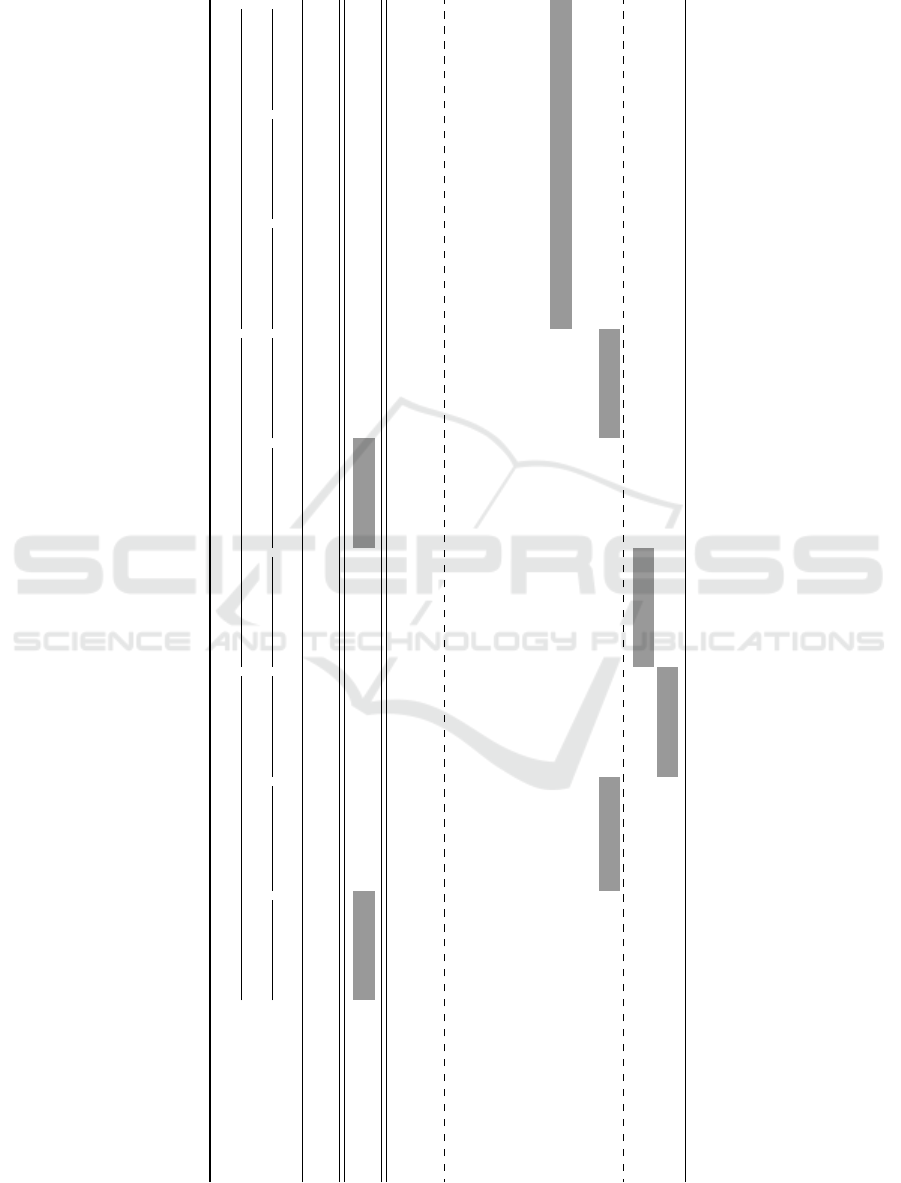
Table 1: The obtained results with different balancing techniques for color and texture features using a RF classifier. The first
and second highest results for each feature set are highlighted in dark and lighter gray colors, respectively.
Features Color Texture Combined
C
1
C
2
C
1,2
T
1
T
2
T
1,2
T
1
, C
1,2
T
2
, C
1,2
T
1,2
, C
1,2
Balancing techniques SE SP SE SP SE SP SE SP SE SP SE SP SE SP SE SP SE SP
IB 52.5 89.6 75.0 88.7 71.2 87.5 38.7 91.7 60.0 96.2 66.2 93.7 73.7 89.6 71.2 89.6 71.2 92.5
OS 93.7 66.7 80.0 86.2 82.5 87.1 43.7 83.7 72.5 90.0 70.0 91.7 77.5 87.1 81.2 88.3 78.7 88.3
ROS 55.0 80.8 80.0 84.2 72.5 85.4 42.5 82.1 60.0 89.2 66.2 87.9 75.0 85.4 73.7 86.2 73.7 85.8
SMOTE 60.0 82.5 78.7 84.6 75.0 70.0 56.2 74.2 61.2 87.5 84.2 87.1 78.7 85.0 73.7 84.6 73.7 85.0
RUS 72.5 72.9 86.2 80.0 78.7 80.0 67.5 53.3 76.2 76.2 85.0 78.7 91.2 75.0 85.0 78.7 92.5 78.3
TL 51.2 86.2 76.2 87.9 67.5 88.3 37.5 87.9 65.0 90.4 68.7 91.7 73.7 88.7 63.7 90.0 72.5 91.2
CUS 81.2 67.9 80.0 84.6 86.2 80.4 56.2 65.8 70.0 77.5 85.0 77.1 83.7 81.2 80.0 84.2 83.7 82.9
NM1 67.5 72.1 86.2 79.2 85.0 82.5 72.5 43.7 80.0 62.5 87.5 66.7 85.0 82.1 86.2 80.4 87.5 80.8
NM2 70.0 72.9 86.2 81.2 85.0 82.9 76.2 48.7 86.2 40.8 86.2 51.2 87.5 82.1 92.5 77.5 91.2 81.7
NM3 82.5 75.0 87.5 80.8 85.0 80.4 73.7 55.8 72.5 82.5 82.5 80.4 83.7 81.2 85.0 80.0 86.2 80.4
NCR 66.2 76.7 87.5 81.2 85.0 82.1 67.5 67.9 75.0 85.8 82.5 83.3 86.2 81.7 82.5 85.0 83.7 85.4
SMOTE + ENN 76.2 73.3 85.0 81.2 85.0 82.1 81.2 56.2 76.2 82.1 80.0 79.6 86.2 81.2 83.7 82.5 78.7 82.9
SMOTE + TL 75.0 73.7 83.7 82.5 87.5 80.8 72.5 59.2 77.5 82.1 78.7 78.7 85.0 82.1 77.5 82.9 88.7 82.5
Tackling the Problem of Data Imbalancing for Melanoma Classification
37
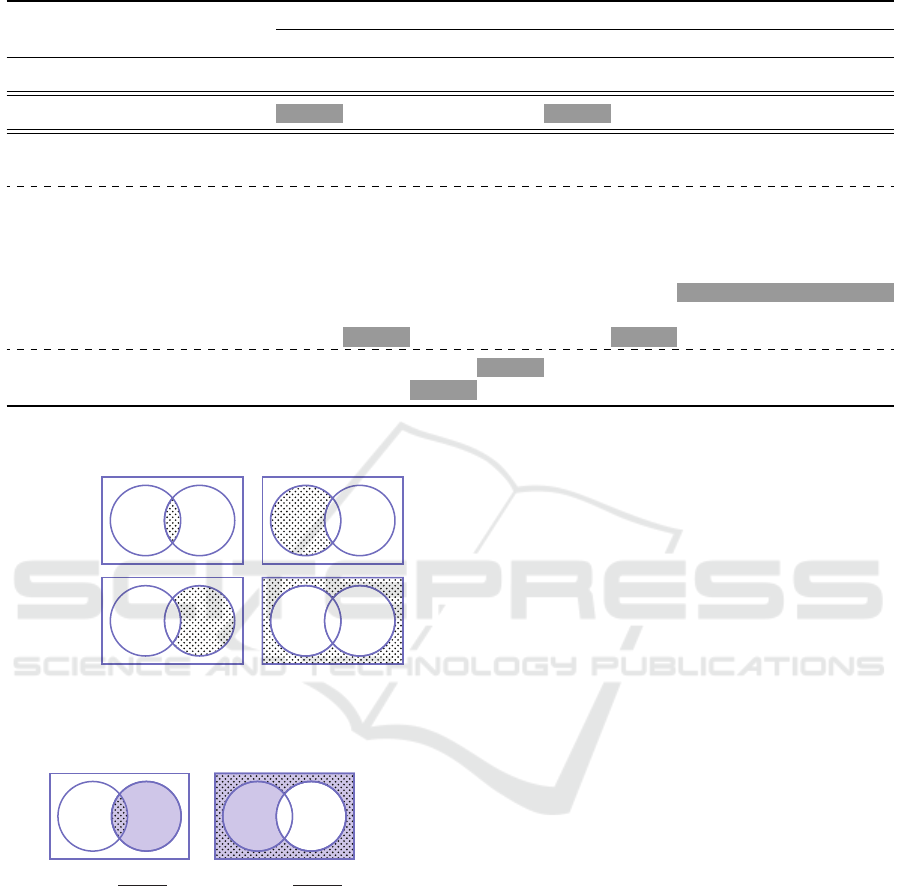
Table 2: The classification costs, C for different balancing techniques and feature sets.
Balancing techniques Classification cost, C
C
1
C
2
C
1,2
T
1
T
2
T
1,2
T
1
, C
1,2
T
2
, C
1,2
T
1,2
, C
1,2
IB 0.3267 0.1950 0.2225 0.4008 0.2550 0.2275 0.1992 0.2142 0.2025
OS 0.1708 0.1750 0.1567 0.4025 0.2050 0.2133 0.1867 0.1592 0.1742
OS 0.3467 0.1833 0.2233 0.4167 0.2833 0.2508 0.2083 0.2125 0.2142
SMOTE 0.3100 0.1892 0.2133 0.3658 0.2825 0.2317 0.1875 0.2192 0.2175
Random Under-Sampling (RUS) 0.2733 0.1625 0.2075 0.3817 0.2375 0.1750 0.1525 0.1750 0.1317
TL 0.3475 0.1908 0.2417 0.4233 0.2483 0.2208 0.2025 0.2575 0.2000
Clustering Under-Sampling 0.2408 0.1817 0.1608 0.3992 0.2700 0.1817 0.1725 0.1833 0.1658
NM1 0.3067 0.1658 0.1600 0.3900 0.2700 0.2083 0.1617 0.1608 0.1517
NM2 0.2883 0.1575 0.1583 0.3475 0.3192 0.2775 0.1467 0.1350 0.1258
NM3 0.2050 0.1517 0.1683 0.3342 0.2350 0.1833 0.1725 0.1700 0.1608
Neighborhood Cleaning Rule (NCR) 0.2958 0.1500 0.1617 0.3233 0.2067 0.1717 0.1558 0.1650 0.1558
SMOTE+ENN 0.2492 0.1650 0.1617 0.2875 0.2142 0.2017 0.1575 0.1675 0.1958
SMOTE+TL 0.2550 0.1675 0.1517 0.3283 0.2067 0.2125 0.1617 0.2033 0.1375
Actual Class
A+ A-
Predicted Class
P+
P
+
−
A
−
+
P
+
−
A
−
+
P-
P
+
−
A
−
+
P
+
−
A
−
+
(a) Confusion matrix with truly and falsely positive
samples detected (TP, FP) in the first row, from left to
right and the falsely and truly negative samples detected
(FN, TN) in the second row, from left to right.
P
+
−
A
−
+
SE =
T P
T P+F N
P
+
−
A
−
+
SP =
T N
T N+FP
(b) Sensitivity and Specificity evaluation, cor-
responding to the ratio of the doted area over
the blue area.
Figure 5: Evaluation metrics: (a) confusion matrix, (b) Sen-
sitivity - Specificity.
highlighted in the shaded cells. Table 2 shows the
obtained cost value for each configuration. Strategies
with low cost function are synonymous with a better
SE and SP trade-off.
The obtained results indicate that balancing tech-
niques are essential and improve the classification
performance. For this case study the US techniques
and their combination with OS techniques outper-
form the OS techniques. Due to the characteristics
similarities of melanoma and dysplastic lesions, it
is expected to have correlated feature space among
melanoma and dysplastic lesions. Subsequently, the
miss-leading samples could be removed using US and
lead to better performance. Specifically to our pur-
pose, NM2 algorithm with the combination of all the
features (T
1,2
C
1,2
) with the lowest cost value, achieve
the highest SE and SP of 91.2% and 81.7%. Using the
same feature combination, RUS achieve the second
lowest cost with SE and SP of 92.5% and 78.3%, re-
spectively. The NM2 algorithm also achieve the third
lowest cost with combination of Gabor and color fea-
tures (T
2
, C
1,2
) with SE and SP of 92.5% and 77.5%,
respectively. Focusing only on OS techniques, OS in
data space outperforms the techniques performing in
feature space.
Comparing the color features, opponent color an-
gle and hue histogram feature descriptor, C
2
, has a
better performance than well-used color statistics, C
1
.
In texture domain, Gabor descriptor, T
2
, outperforms
CLBP features, T
1
. Finally, the combination of color
and texture features outperforms any other feature
combination.
6 CONCLUSION
In this paper, we analyzed the impact of data bal-
ancing techniques for the classification of malignant
melanoma. Therefore, we presented an extensive
comparison of twelve OS and US techniques in both
feature and data space. These techniques were evalu-
ated on the only public dermoscopic dataset, the PH
2
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
38

dataset, in order to provide a chance for future com-
parison. The obtained results particularly highlight
the advantage of balancing the training set over using
the original data, particularly for the methods based
on US (NM2, NCR) and combination of OS and US in
feature space. Furthermore, OS in data space outper-
forms the techniques performing in the feature space.
This study also showed that combining color and tex-
ture features will lead to better performance.
REFERENCES
Abbasi, N. R., Shaw, H. M., et al. (2004). Early diagnosis
of cutaneous melanoma: revisiting the abcd criteria.
Jama, 292(22):2771–2776.
American-Cancer-Society (2014). Cancer facts & figures
2014.
Barata, C., Marques, J. S., and Emre Celebi, M. (2013).
Towards an automatic bag-of-features model for the
classification of dermoscopy images: The influence of
segmentation. In Image and Signal Processing and
Analysis (ISPA), 2013 8th International Symposium
on, pages 274–279. IEEE.
Barata, C., Ruela, M., Francisco, M., Mendonc¸a, T., and
Marques, J. (2014). Two systems for the detection of
melanomas in dermoscopy images using texture and
color features. IEEE Systems Journal,, 8(3):965–979.
Batista, G. E., Bazzan, A. L., and Monard, M. C. (2003).
Balancing training data for automated annotation of
keywords: a case study. In WOB, pages 10–18.
Batista, G. E., Prati, R. C., and Monard, M. C. (2004). A
study of the behavior of several methods for balancing
machine learning training data. ACM Sigkdd Explo-
rations Newsletter, 6(1):20–29.
Breiman, L. (2001). Random forests. Machine learning,
45(1):5–32.
Capdehourat, G., Corez, A., Bazzano, A., and Mus
´
e, P.
(2009). Pigmented skin lesions classification using
dermatoscopic images. In Progress in Pattern Recog-
nition, Image Analysis, Computer Vision, and Appli-
cations, pages 537–544. Springer.
Celebi, M. E., Kingravi, H. A., et al. (2007). A method-
ological approach to the classification of dermoscopy
images. Computerized Medical Imaging and Graph-
ics, 31(6):362–373.
Chawla, N. V., Bowyer, K. W., Hall, L. O., and Kegelmeyer,
W. P. (2002). Smote: synthetic minority over-
sampling technique. Journal of artificial intelligence
research, pages 321–357.
Forsea, A., Del Marmol, V., de Vries, E., Bailey, E., and
Geller, A. (2012). Melanoma incidence and mortal-
ity in europe: new estimates, persistent disparities.
British Journal of Dermatology, 167(5):1124–1130.
Guo, Z. and Zhang, D. (2010). A completed modeling of
local binary pattern operator for texture classification.
IEEE Transactions on Image Processing, 19(6):1657–
1663.
He, H., Garcia, E., et al. (2009). Learning from imbalanced
data. Knowledge and Data Engineering, IEEE Trans-
actions on, 21(9):1263–1284.
Laurikkala, J. (2001). Improving identification of diffi-
cult small classes by balancing class distribution.
Springer.
Mani, I. and Zhang, I. (2003). knn approach to unbalanced
data distributions: a case study involving information
extraction. In Proceedings of Workshop on Learning
from Imbalanced Datasets.
Prati, R. C., Batista, G. E., and Monard, M. C. (2009). Data
mining with imbalanced class distributions: concepts
and methods. In IICAI, pages 359–376.
Rastgoo, M., Garcia, R., Morel, O., and Marzani, F.
(2015a). Automatic differentiation of melanoma from
dysplastic nevi. Computerized Medical Imaging and
Graphics, 43:44–52.
Rastgoo, M., Morel, O., Marzani, F., and Garcia, R.
(2015b). Ensemble approach for differentiation of ma-
lignant melanoma. In The International Conference
on Quality Control by Artificial Vision 2015, pages
953415–953415. International Society for Optics and
Photonics.
Tomek, I. (1976). Two modifications of cnn. IEEE Trans.
Syst. Man Cybern., 6:769–772.
Van De Weijer, J. and Schmid, C. (2006). Coloring local
feature extraction. In Computer Vision–ECCV 2006,
pages 334–348. Springer.
Tackling the Problem of Data Imbalancing for Melanoma Classification
39
