Predicting Glaucomatous Progression with Piecewise Regression Model
from Heterogeneous Medical Data
Kyosuke Tomoda
1
, Kai Morino
1
, Hiroshi Murata
2
, Ryo Asaoka
2
and Kenji Yamanishi
1
1
Graduate School of Information Science and Technology, The University of Tokyo, Tokyo 133–8656, Japan
2
Graduate School of Medicine, The University of Tokyo, Tokyo 133–8655, Japan
Keywords:
Glaucoma, Intraocular Pressure, Heterogeneity, Collective Method, Piecewise Linear Regression Model.
Abstract:
This study aims to accurately predict glaucomatous visual-field loss from patient disease data. In general, med-
ical data show two kinds of heterogeneity: 1) internal heterogeneity, in which the phase of disease progression
changes in an individual patient’s time series dataset; and 2) external heterogeneity, in which the trends of
disease progression differ among patients. Although some previous methods have addressed the external
heterogeneity, the internal heterogeneity has never been taken into account in predictions of glaucomatous
progression. Here, we developed a novel framework for dealing with the two kinds of heterogeneity to pre-
dict glaucomatous progression using a piecewise linear regression (PLR) model. We empirically demonstrate
that our method significantly improves the accuracy of predicting visual-field loss compared with existing
methods, and can successfully treat the two kinds of heterogeneity often observed in medical data.
1 INTRODUCTION
1.1 Motivation of our Study
The aim of our study is to construct a novel method
for the treatment of heterogeneous medical data in the
prediction of disease progression. We specifically fo-
cus on data from patients with glaucoma to predict
visual-field loss progression. Medical datasets are
heterogeneous from the following two aspects: exter-
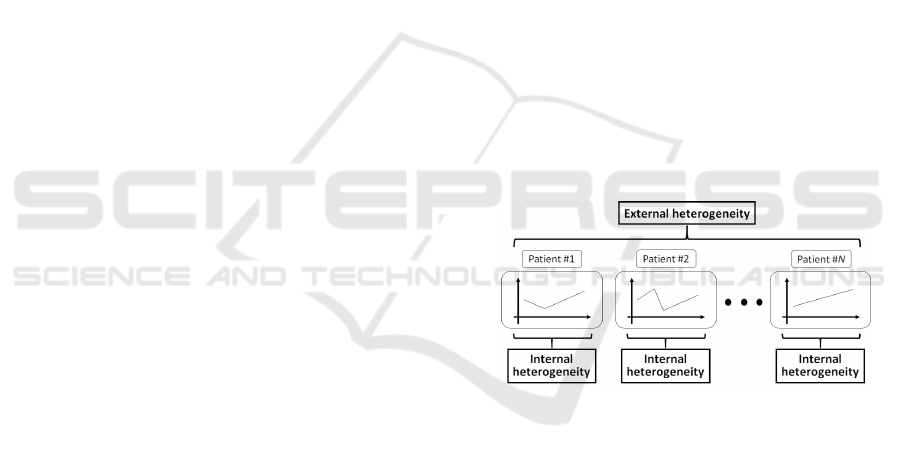
nal heterogeneity and internal heterogeneity (Fig. 1).
External heterogeneity generally refers to the rela-
tionship of measured data among patients; e.g., the
rates of the disease progression differ from patient
to patient. By contrast, internal heterogeneity occurs
within an individual patient; e.g., changes in the phase
of disease progression over time. Therefore, in order
to obtain comprehensive knowledge and trends from
medical data, a method for appropriately dealing with
the characteristic heterogeneity is required.
It is not straightforward to treat heterogeneous
medical data because these two kinds of heterogene-
ity must be resolved in different ways. The main
challenge in this respect is due to the specific struc-
ture of large medical datasets. Although the datasets
are often composed of data from a large number of
patients, data for each patient are usually limited be-
Figure 1: The two kinds of heterogeneity observed in medi-
cal datasets. External heterogeneity is caused by differences
among patients, whereas internal heterogeneity is caused by
changes in each patient’s state over time.
cause of the high costs related to diagnosis, both for
the patients and clinicians. In particular, a detailed
medical examination requires well-trained clinicians,
special medical equipment, and a long period of diag-
nosis. In this paper, we refer to this specific structure
of medical data as medical-data-structure-difficulty.
This difficulty limits the ability to construct a reliable
predictive model for each patient using only the pa-
tient’s information. One way to solve this problem
is to take advantage of hidden relationships among
patients. However, mining for such hidden relation-
ships is often difficult because of the heterogeneous
nature of medical data described above. Therefore,
accurate analysis of medical data requires a method
for overcoming the two heterogeneity problems and
Tomoda, K., Morino, K., Murata, H., Asaoka, R. and Yamanishi, K.
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data.
DOI: 10.5220/0005703900930104
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 5: HEALTHINF, pages 93-104
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
93
the medical-data-structure-difficulty simultaneously.
Various methods have been proposed to over-
come the medical-data-structure-difficulty in a med-
ical dataset, which have mostly involved incorporat-
ing information from other patients to improve the
prediction accuracy (Liang, Z. et al., 2013; Maya,
S. et al., 2014; Murata, H. et al., 2014; Morino, K.
et al., 2015). Therefore, appropriate information is
collected from a patient dataset and a tuned predic-
tor is constructed for the target patient. We here refer
to these types of methods as “collective methods.” In
collective methods, we should carefully analyze the
heterogeneity to collect data appropriately. Most of
the existing collective methods for dealing with data
from glaucoma patients have focused on resolving the
external heterogeneity problem. We here propose a
new collective method that achieves the better pre-
diction accuracy than existing methods, because our
method can cope with both the external and internal
heterogeneity in the medical dataset.
Glaucoma data, the focus of our study, criti-
cally contain both kinds of heterogeneity as well
as the medical-data-structure-difficulty mentioned
above. Glaucoma is an eye disease that causes pro-
gressive damage to a patient’s visual field, which can
ultimately lead to blindness, and is the second-leading
cause of blindness worldwide (Kingman, S., 2004).
Quigley et al. (Quigley, H. A. and Broman, A. T.,
2006) estimated that nearly 80 million people will
suffer from glaucoma by 2020. The glaucomatous
visual-field loss is considered to be irreversible, but
glaucomatous progression can be delayed with appro-
priate treatment. Therefore, suggesting an appropriate
treatment plan at an early stage of disease progres-
sion is a critical factor for improving patients’ quality
of life. Accordingly, the development of methods for
the early prediction of glaucoma progression is par-
ticularly important for effectively treating the disease.
Most of the existing glaucoma prediction meth-
ods involve analyses of visual-field data, which are
associated with the aforementioned medical-data-
structure-difficulty and external heterogeneity prob-
lem. However, in reality, the rate of glaucomatous
progression changes over time for each eye (inter-
nal heterogeneity). Therefore, to improve prediction,
a novel collective method is required to deal with
the internal heterogeneity of data (within-eye level)
in addition to the external heterogeneity (between-
eye level). The internal heterogeneity can be poten-
tially captured with intraocular pressure (IOP) data.
This is based on clinical evidence that the progres-
sion rate of glaucoma increases with an increase in
the IOP value (AGIS Investigators, 2010; Collabora-
tive Normal-Tension Glaucoma Study Group, 1998;
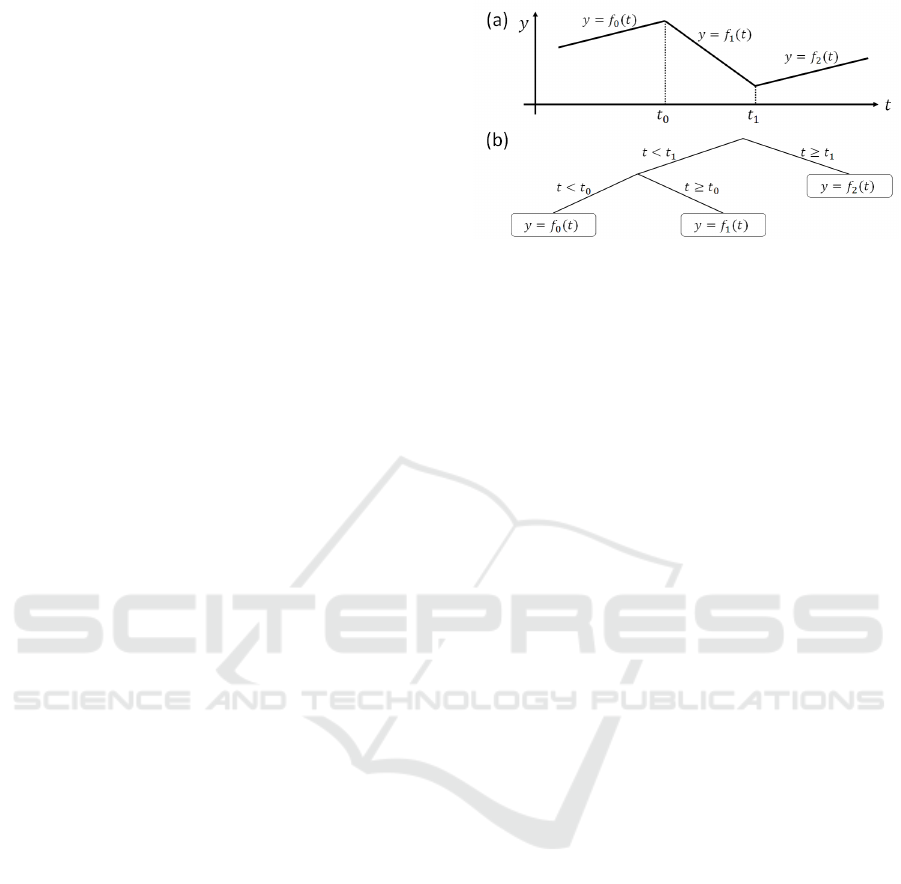
Figure 2: Schematic figure of the piecewise linear regres-
sion model. (a) The piecewise linear regression. (b) The
tree structure expression corresponding to the piecewise lin-
ear regression lines shown in (a).
Satilmis, M. et al., 2003). However, previous col-
lective models for predicting glaucomatous progres-
sion (Liang, Z. et al., 2013; Maya, S. et al., 2014;
Murata, H. et al., 2014) have been trained only with
visual-field data. In this paper, we outline the first ap-
plication of IOP data to the prediction of glaucoma
progression and achieve good prediction accuracy.
1.2 Heterogeneity of Glaucoma Data
Here, we introduce the heterogeneity of glaucoma.
Internal Heterogeneity: The progression rate
of glaucoma essentially changes over several time
points, even when considering the time series of
one eye. Clinical knowledge suggests that the pro-
gression rate of glaucoma varies because of high
IOP values (AGIS Investigators, 2010; Collabora-
tive Normal-Tension Glaucoma Study Group, 1998;
Satilmis, M. et al., 2003). This internal heterogeneity
can be difficult to detect because only limited data can
be obtained from the target eye at certain time points.
Our proposed novel collective method resolves this
internal heterogeneity difficulty by using IOP data.
External Heterogeneity: Glaucoma data are
highly variable among eyes from the following four
perspectives: (1) the disease stage at initial diagnosis;
(2) the progression rate of glaucoma; (3) the average
and fluctuation levels of IOP; (4) the minimum level
of IOP that affects the progress of glaucoma.
1.3 Novelty and Significance
The novelty and significance are summarized below.
1) A Novel Framework for Solving the Het-
erogeneity Problems of Medical Data with a Col-
lective Method. We here propose a novel collec-
tive piecewise linear regression (PLR) model to si-
multaneously deal with the two kinds of heterogene-
ity of medical data and the medical-data-structure-
HEALTHINF 2016 - 9th International Conference on Health Informatics
94
difficulty. In general, a PLR model is suitable for
dealing with the internal heterogeneity problem of
medical data; however, it cannot be easily applied to
the problem of glaucoma progression prediction. A
PLR model (Fig. 2(a)) can be interpreted as a tree-
structured model (Fig. 2(b)); i.e., the edges carry
information about the segmentation, and the nodes
carry information about the regression lines. This
model is powerful because it can reflect the complex
features of medical data by breaking it down into sev-
eral pieces. However, this benefit comes with a disad-
vantage in that piecewise regression requires a large
dataset for good prediction even if the complexity of
the model or the depth of the tree structure is ap-
propriately controlled. Therefore, the medical-data-
structure-difficultyis a barrier to effectivelyanalyzing
the data with existing PLR methods. Our proposed
method overcomes the problems of medical data.
A) Application of a Collective PLR Model with
Medical-data-structure-difficulty. Our proposed
method can be used to train a PLR model with hetero-
geneous medical data. As described above, only lim-
ited data can be obtained for each patient from a large
medical dataset owing to the medical-data-structure-
difficulty. Although effective training algorithms for
a tree-structured model with a very large dataset have
been intensively investigated (Natarajan, R. and Ped-
nault, E., 2002; Vogel, D. S. et al., 2007), there have
been few studies conducted to develop a training al-
gorithm for this type of “big data.” Therefore, our
current study sheds new light on this common prob-
lem and offers a potential solution.
B) A Useful Framework for the Overall Optimiza-
tion of a Piecewise Regression Model Consider-
ing External Heterogeneity. Our novel method opti-
mizes the piecewise model as well as each regression
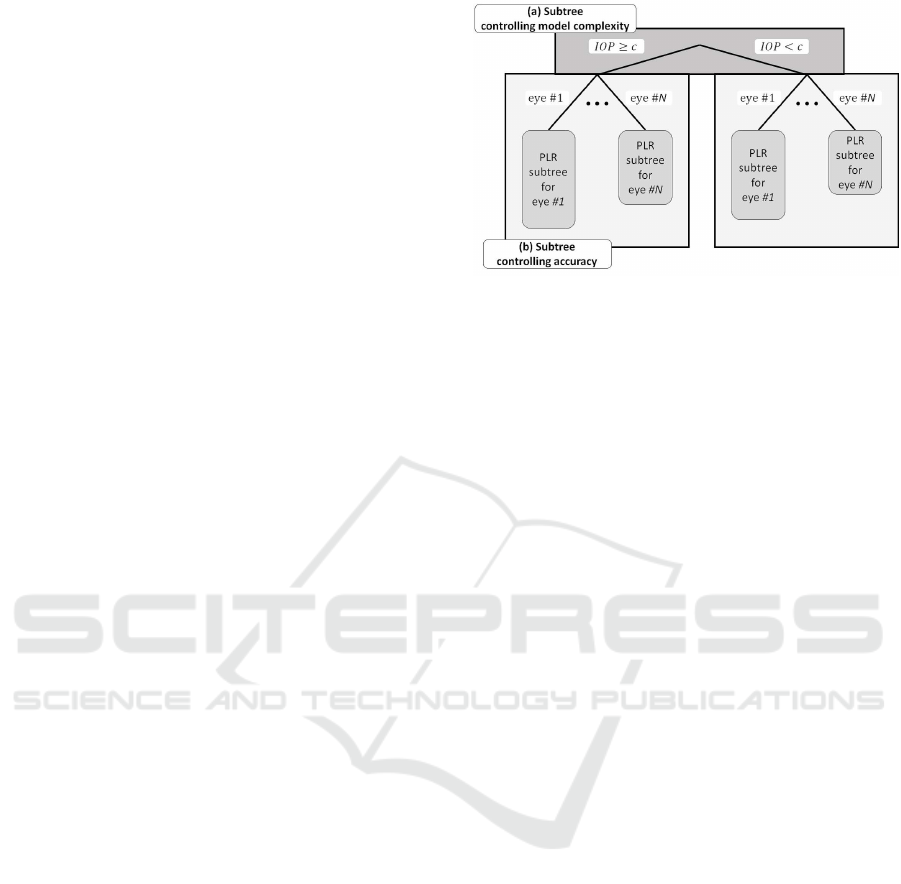
line for each piece simultaneously. Our model (Fig. 3)
consists of two parts: one that controls the model’s
complexity, and the other that controls the model’s
prediction accuracy. This clearly divided model struc-
ture enables the use of other collective regression al-
gorithms besides those employed in this paper (Liang,
Z. et al., 2013). We describe this feature in greater
detail in Sec. 3.2. We optimized the whole model, in-
cluding the segmentation and regression parameters,
using data from similar eyes. We did this optimiza-
tion by applying the statistical model selection crite-
ria. Specifically, we examined a number of existing
information criteria to investigate which gave the best
prediction accuracy.
C) Good Framework of the Collective Piecewise
Regression for Tackling Internal Heterogeneity.
For the collective piecewise regression, our proposed
method provides a good framework that can effec-
Figure 3: Hierarchical tree structure of our IOP-based
piecewise model. The structure of the model can be sep-
arated into two parts (a) controlling the model complexity
and (b) controlling the model accuracy. The PLR model for
each eye also has its tree structure as shown in Fig. 2(b).
tively deal with internal heterogeneity. Our model
was carefully designed to collect data of similar eyes
while coping with the internal heterogeneity related to
disease progression over time. Our method involves
segmentation of time-series data, even within one pa-
tient’s time course, followed by calculation of the re-
gression lines for each piece. We assume that the gra-
dient parameters for each piece in the same state are
common and that the intercept parameters for each
piece are different, so that the internal heterogeneity
can be accurately expressed (see Step 4 in Fig. 5). We
note that our model does not have only common gra-
dient parameters but individualized intercept parame-
ters to reflect each patient’s characteristics. This in-
dividualization cannot be realized by simply sharing
the parameters with all the patients. Hence, our model
can represent each patient’s glaucoma progression ef-
ficiently.
2) Big Impact on the Medical Field of Glau-
coma
A) First Application of IOP to the Prediction of
Glaucoma Progression. To the best of our knowl-
edge, our proposed model represents the first appli-
cation of IOP to the prediction of glaucoma progres-
sion. In existing analyses (Liang, Z. et al., 2013;
Maya, S. et al., 2014; Murata, H. et al., 2014; Maya,
S. et al., 2015; Holmin, C. and Krakau, C. E. T.,
1982; Zhu, H. et al., 2014), only visual-field data
have been used for prediction. Although clinical stud-
ies suggest the importance of IOP to understand dis-
ease progression (AGIS Investigators, 2010; Collabo-
rative Normal-Tension Glaucoma Study Group, 1998;
Satilmis, M. et al., 2003), the high external hetero-
geneity of IOP has limited its application to predictive
models (see Sec. 1.2). We believe that this difficulty
can be overcome with our novel collective method,
which is expected to have a great impact on the med-
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data
95

ical field of glaucoma.
B) Wide Applicability to other Glaucomatous Pre-
diction Models based on Visual-field Data. Any ex-
isting glaucoma prediction method can be converted
into an IOP-based piecewise prediction model using
our framework, which should increase its impact in
the medical field of glaucoma. As mentioned in point
1-B), our model was developedusing a general frame-
work. Therefore, our model is not only applicable to
various existing glaucoma prediction models but also
to models that will be developed in the future. This
wide applicability is practically important for the fu-
ture progress of glaucoma prediction, since any great
prediction model will lose its value after an improved
method is proposed. Owing to its clearly segmentable
structure, our model will be able to embrace other
models, indicating that it has a long life span, and is
flexible and widely applicable.
1.4 Related Works
Conventionally, linear regression analyses of visual-
field time-series data for each eye have been used
to predict the glaucomatous progression (Holmin, C.
and Krakau, C. E. T., 1982). However, simple lin-
ear regression for time series of an individual eye is
not effective when the number of data points is small,
which is often the case for clinical data. Although var-
ious regression models have been applied to the pre-
diction with only data from a target eye, the prediction
accuracy was limited due to the shortage of data (Fu-
jino Y. et al., 2015; Taketani Y. et al., 2015). Thus, we
must overcome this shortage for better prediction.
To make up for this deficiency in clinical data,
some recent studies have proposed collective meth-
ods that exploit visual-field data from other eyes to
predict the glaucomatous progression in a target eye
at an early stage of disease. These studies also pro-
posed some methods for coping with the external het-
erogeneity and medical-data-structure-difficulty from
a data-mining point of view. Liang et al. (Liang, Z.
et al., 2013) proposed a spatio-temporal clustering-
based method. They collected similar eyes in terms
of their spatial and temporal feature of progression.
Then, they used data from the eyes similar to the tar-
get eye for prediction. Zhu et al. (Zhu, H. et al., 2014)
used Bayesian inference to reflect the spatial correla-
tion of progression. Maya et al. (Maya, S. et al., 2014)
used tensor decomposition and a multitask-learning
method to extract multiple features from a heteroge-
neous glaucoma dataset. Murata et al. (Murata, H.
et al., 2014) used Bayesian linear regression to utilize
the measurements of other eyes when making predic-
tions of a target eye. Maya et al. (Maya, S. et al.,
Figure 4: Schematic figure of the glaucomatous pro-
gression. The total deviation (TD) values on a vi-
sual field are schematically shown in shades of gray.
Darker mesh shades indicate a more defective visual
field. The overall aim was to predict TD values at
each mesh at a given target time. 2015) proposed
a hierarchical minimum description length (MDL)-
based clustering method for finding progression pat-
terns, and substituted the clusters used in Liang et
al. (Liang, Z. et al., 2013) with their newly discovered
clusters to more effectivelypredict glaucomatous pro-
gression. However, all of these studies focused on ex-
ternal heterogeneity and did not address the problem
of internal heterogeneity.
To overcome the internal heterogeneity problem,
we employed a PLR model for predicting glaucoma-
tous progression. As we mentioned above, a PLR
model potentially deals with the internal heterogene-
ity problem. However, it is widely known that suffi-
cient data within an appropriate range are required in
order to appropriately estimate each regression line.
Therefore, a large dataset should be used in the learn-
ing phase, which can sometimes cause problems in
applying this model to real situations. Although sev-
eral studies have focused on training models for a tree
structure with a massive dataset (Natarajan, R. and
Pednault, E., 2002; Vogel, D. S. et al., 2007), there is
barely any information on training a PLR model un-
der a situation of medical-data-structure-difficulty.
1.5 Organization
The rest of this paper is organized as follows. In Sec-
tion 2, we introduce some prior knowledge for under-
standing glaucomatous progression. In Section 3, we
present our proposed framework for training a collec-
tive PLR model for tackling the internal and external
heterogeneity. The results of experiments conducted
to evaluate our framework with glaucoma dataset are
described in Section 4. Finally, we provide an overall
conclusion and summary of our method in Section 5.
2 PRELIMINARIES
2.1 Prediction of Glaucoma
Our aim was to predict the visual-field loss precisely
at a given target time, as depicted in Fig. 4. In our
dataset, the visual-field data were measured on 74
meshes of a visual field to obtain a total deviation
(TD) value. This value represents the differences in
the measured light sensitivity on each mesh compared
to age-matched normative data. A negative TD value
HEALTHINF 2016 - 9th International Conference on Health Informatics
96

Figure 4: Schematic figure of the glaucomatous progres-
sion. The total deviation (TD) values on a visual field are
schematically shown in shades of gray. Darker mesh shades
indicate a more defective visual field. The overall aim was
to predict TD values at each mesh at a given target time.
means that the sensitivity is worse than the normative
sensitivity; thus, the TD value decreases as glaucoma
progresses. The rates of progression differ among
the meshes on a visual field; therefore, the TD value
needs to be predicted independently for each mesh.
2.2 Effect of High IOP
Several ophthalmological studies have shown a re-
lationship between IOP and glaucoma progression.
The AGIS Investigators (AGIS Investigators, 2010)
found that a group of patients with glaucomatous
eyes and high IOP lost their visual fields more
quickly than patients with lower IOP. The Collabo-
rative Normal-Tension Glaucoma Study Group (Col-
laborative Normal-Tension Glaucoma Study Group,
1998) observed that the rate of glaucoma progression
in the eyes of patients undergoing treatment, which
involves reduction in IOP, was significantly lower
than that of patients not receiving treatment for IOP
reduction. Satilmis et al. (Satilmis, M. et al., 2003)
demonstrated that the rate of glaucoma progression
was related to the standard deviation in IOP.
Considering these results, the rates of the progres-
sion greatly change at some time points that can be
detected with IOP values. This means that the internal
heterogeneity in the glaucoma dataset can be captured
with IOP. However, the relationship between IOP and
the degree of glaucoma progression has not yet been
fully investigated in ophthalmology. Hence, we do
not employ the raw value of IOP as an explanatory
variable but rather discretize it into “high” and “low”
states to make segmentations of time series according
to whether the undelying IOP is high or low. Further,
the discretization of IOP into the ”two” states makes it
effcient to estimate the parameters of the model. This
is because the more states we separate IOP into, the
smaller the size of each piece becomes.
3 PROPOSED METHOD
3.1 Concept of Proposed Method
Our method makes it possible to decide the appropri-
ate thresholds for separating time series and express
the internally heterogeneous progress of glaucoma.
We optimize the thresholds using an information cri-
terion and estimate gradient and intercept parameters
for each of the separated piece. The outline of our
method is described below. Figure 5 helps to under-
stand its procedure. We note that the following proce-
dure was applied to one prediction-target eye.
Step 1: Normalization for the External Hetero-
geneity of IOP. The mean and standard deviation of
the IOP distribution are highly variable among eyes.
Therefore, the IOP distribution of each eye was nor-
malized to effectively analyze the external hetero-
geneity of IOP.
Step 2: Selection of an IOP Threshold Value c. An
IOP threshold value c is selected to construct the PLR
model from a possible list of IOP threshold values.
We note that a threshold value that has in the previous
iterations is not selected again for model construction.
Step 3: Division of the Visual-field Time Series into
Pieces. The state of an eye (high- or low-IOP state)
is decided using the given IOP threshold c. At each
time point when visual-field data are recorded, if the
standardized IOP score is larger than the given c, the
IOP state at that time point is determined to be in a
high-IOP state (see Sec. 3.4). The time series is then
divided into pieces when the state changes. This pro-
cedure reflects actual clinical knowledge based on the
internal heterogeneity of glaucoma progression.
Step 4: Construction of PLR Models using Our
Collective Method. A PLR model was constructed
using our proposed collective method with the di-
vided pieces. First, a set of similar eyes E was se-
lected to estimate the parameters of the PLR mod-
els. In this case, the training dataset consisted of
|E| eyes that showed similar behavior to the target
eye. In this study, we employed an existing clustering
method (Liang, Z. et al., 2013) to collect data from
similar eyes. However, another collective method
could be used for the same process. Next, we trained
the PLR model only using the eye set E. For each
piece, we fit a linear function of time; i.e., we esti-
mate the gradient and intercept parameters. We ap-
plied the same gradient parameters to pieces in the
same IOP state. On the other hand, the intercept pa-
rameters were individually determined for each piece.
This proper estimation of the gradient and intercept
parameters is the key for effectively constructing a
PLR model (see Sec. 3.5).
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data
97
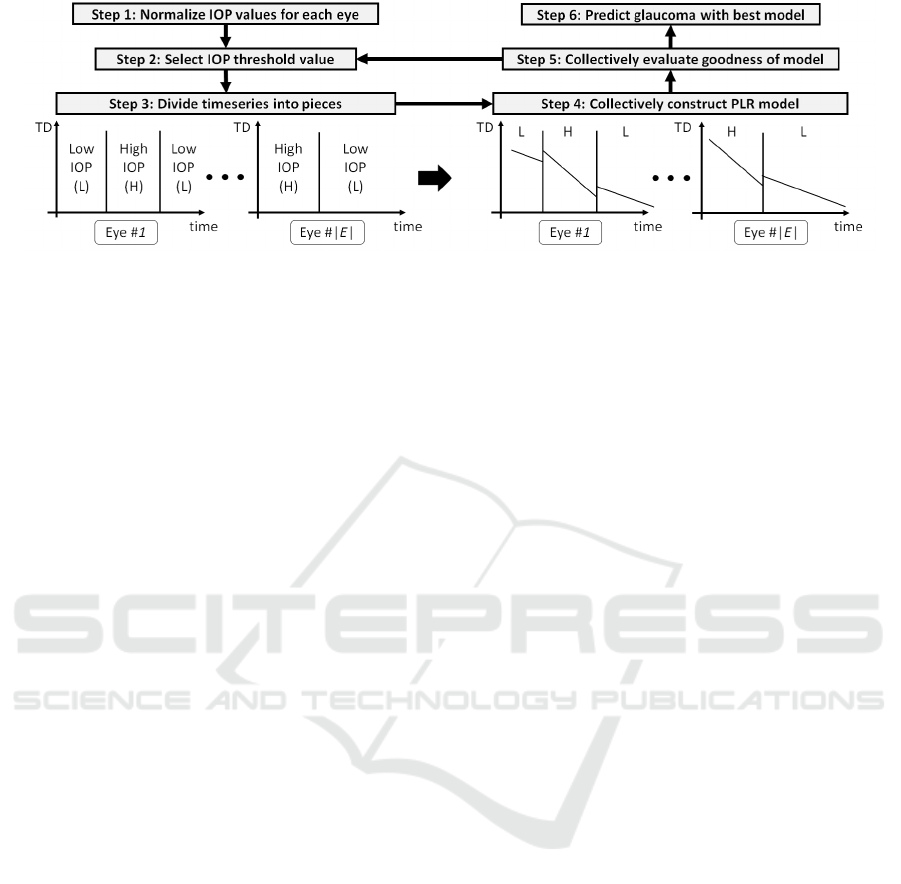
Figure 5: Flow of our algorithm. First, we calculate the standard scores of IOP in Step 1. Then, we iteratively obtain one PLR
model by changing IOP threshold values c in Steps 2-5. Finally, we determine the best PLR model based on an information
criterion and predict the glaucoma progression in Step 6. We note that the gradients corresponding to the same IOP state are
the same for all the eyes belonging to the eye set E composed of eyes similar to the target eye.
Step 5: Evaluation of the Generated PLR Models.
Different PLR models were generated when a differ-
ent threshold c was chosen. Therefore, the best c is
required to obtain the best PLR model. We evaluated
the quality of the generated PLR models on the basis
of statistical model selection criteria. If all possible
thresholds were already selected, the best PLR model
was chosen as the predictor of the target eye, which
was then applied to Step 6. Otherwise, Steps 2-5 were
repeated to construct a new PLR model.
Step 6: Prediction of progression with the best
PLR model. The visual-field loss of the target eye
was predicted with the best PLR model incorporat-
ing both the external and internal heterogeneity of the
glaucoma dataset.
3.2 Shared and Unshared Parts of Our
Collective Method
Our collectivemethod is different from those that sim-
ply use data from other eyes for prediction of a tar-
get eye. The important difference is that we carefully
estimated the model parameters shared among other
eyes.
In the Model Selection Phase (Fig. 3 (a)): The fit-
ness and simplicity of the model are evaluated from
the overall model. The threshold value for the stan-
dardized IOP is common to all similar eyes. This
value is decided by considering the whole model and
each of the predictors for each eye.
In the Estimation Phase (Fig. 3 (b)): The gradient
parameter is shared among each piece in the same
IOP state; therefore, this parameter is estimated using
the data from all similar eyes. Meanwhile, the inter-
cept parameters differ among each piece; therefore,
this parameter is estimated for each eye individually.
This modeling procedure allows for the internal het-
erogeneity of progression to be expressed precisely.
3.3 Problem Settings
Let N denote the number of observed eyes, t
i, j
the
time of the jth measurement of the ith eye, and n
i
the
number of measurements for the ith eye. We repre-
sent the vector of the TD values measured at t
i, j
as
y
i, j
= (y
(1)
i, j
,..., y
(K)
i, j
) ∈ R
K
, where K is the number of
meshes on a visual field, and p
i, j
∈ R is the IOP value
at t
i, j
. We set T
i
:= (t
i,1
,...,t
i,n
i
), Y
i
:= (y
i,1
,..., y
i,n
i
),
and P
i
:= (p
i,1
,..., p
i,n
i
). The whole measured dataset
is represented as D := {(T
1
,Y
1
,P
1
),...,(T
N
,Y
N
,P
N
)}.
We predict the TD value at an arbitrary time point af-
ter the last measurement, given a dataset of N eyes
that includes measurement time, TD and IOP values.
3.4 Judging the IOP State
As mentioned in Sec. 2.2, the higher the IOP, the more
rapidly glaucoma progresses; therefore, the progres-
sion rate changes at certain time points depending on
fluctuations in IOP. To model this internal heterogene-
ity, we propose an IOP-based PLR model. The two
IOP states are defined as the high-IOP state (denoted
as H) and the low-IOP state (denoted as L).
However, IOP also shows external heterogeneity.
To overcome this problem, we focused on the tem-
poral differences within the IOP time series for each
eye. For the ith eye, we used a standardized score
˜p
i, j
= (p
i, j
− ¯p
i
)/σ
i
at t
i, j
calculated with the mean ¯p
i
and standard deviation σ
i
of IOP. Through this nor-
malization of the external heterogeneity of IOP, the
IOP data from other eyes can be treated in the same
manner. Let s
i, j
denote the IOP state of the ith eye at
time t
i, j
, then s
i, j
is defined as
s
i, j
=
(
H, (if ˜p
i, j
≥ c),
L, (if ˜p
i, j
< c),
where c is the threshold constant. Figure 6(a) displays
HEALTHINF 2016 - 9th International Conference on Health Informatics
98
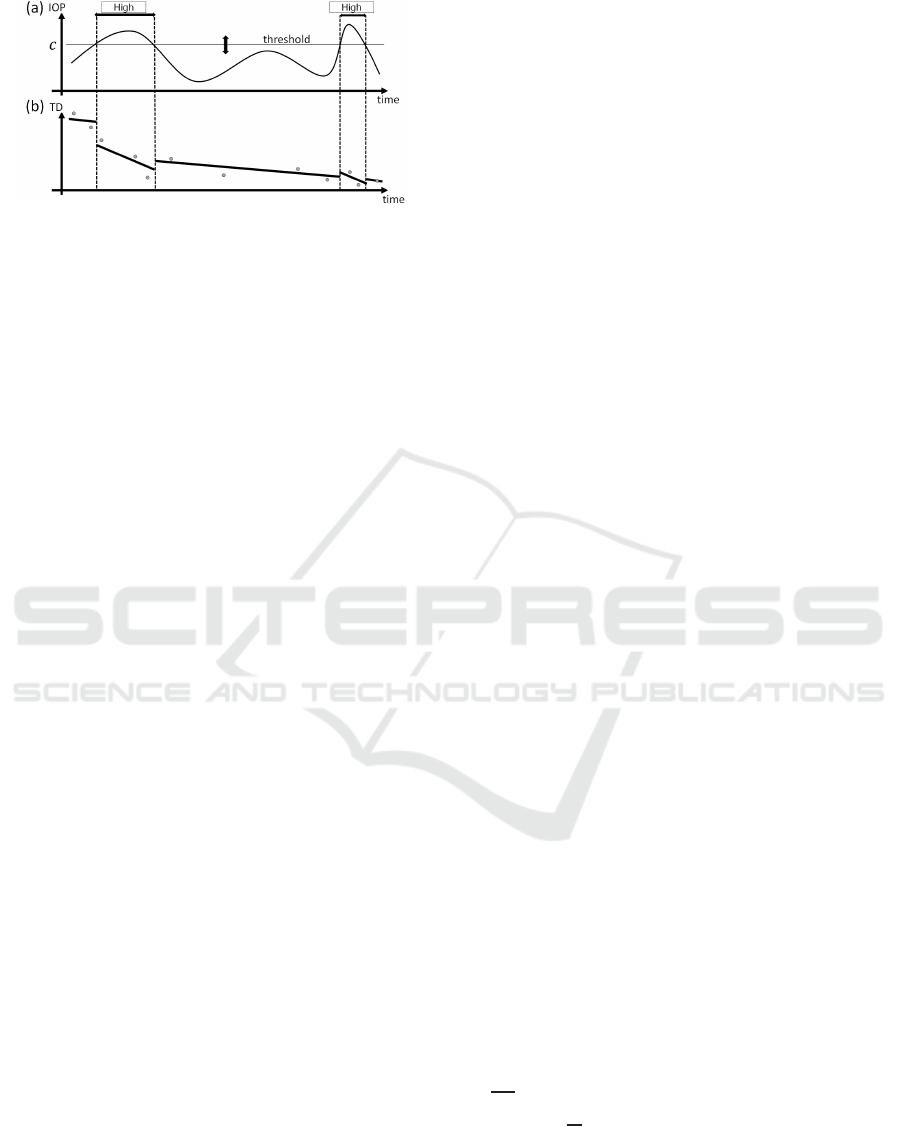
Figure 6: Concept of the proposed method. (a) The time-
series data were divided into intervals with high- or low-IOP
states using the threshold c. (b) The PLR model was trained
based on these intervals. The gradients of the lines within
the same IOP states were the same, and these regression
lines were allowed to be disconnected from the subsequent
lines. The dots on the graph represent the data points.
the classification protocol. Here, we represent I
i,k
(k =
1,... , L
i
) as the set of indices in the interval where
the IOP state remains the same among the series of
IOP states s
i
= {s
i, j
}
n
i
j=1
, calculated as shown above.
We denote L
i
as the number of intervals for the ith
eye. We further assume that this threshold constant is
common between the target eye and similar eyes.
It is worth noting that the training data should not
be partitioned from the target eye data for the follow-
ing two reasons: first, it is difficult to estimate the cor-
rect IOP state, because of the very limited data from
the target eye with a small number of diagnoses; sec-
ond, if the data from the target eye are partitioned,
the number of visual-field data points for the target
eye will be too small to construct the PLR model, be-
cause segmenting the data would further decrease the
amount of training data. Thus, we did not partition
the time series of the target eye, and assumed that it
was in a low-IOP state. This assumption is valid and
realistic, because the interval for the high-IOP state is
usually very short, and therefore the progression after
the final measurement should be in the low-IOP state.
3.5 IOP-based Collective PLR Model
Once the visual-field data for each eye are divided us-
ing the standardized IOP scores, a segmented time-
series dataset is obtained for each eye, or a tree struc-
ture, as shown in Fig. 3. For each high- and low-IOP
state, the proposed model represents a different rate
of progression, or the internal heterogeneity of pro-
gression. Fig. 6 (b) shows our PLR model as applied
to Liang et al.’s method (Liang, Z. et al., 2013).
Liang et al. (Liang, Z. et al., 2013) proposed a
spatio-temporal clustering-based linear-regression
model using data from other eyes. They assumed
the same glaucomatous progression within the
same cluster. Here, we show an application of our
developed PLR model to the temporal-shift linear
regression method (TSLR) using k-NN as a clustering
method (Liang, Z. et al., 2013) . They extracted the
feature vector of ith eye via singular value decompo-
sition of the matrix Y
i
to collect data from the eyes
similar to the target eye. They recognized the first
left-singular vector of Y
i
as the spatial feature vector
of ith eye, and the k-nearest eyes in the spatial feature
vector space are collected as the similar-eyes cluster.
Let E ⊂ {1,...,N} denote the set of indices within a
cluster, and let w
i,k
and b
i,k
denote the gradient and
intercept of the kth piece of the regression line for
the ith eye, respectively. Then, the assumption above
can be formulated as ∀i, j ∈ E, ∀k,h s.t. s
i,k
= s
j,h
=
S, ∃w
S
, w
(l)
i,k
= w
(l)
j,h
= w
(l)
S
, where S ∈ {H,L}. As
stated above, the intercept parameters are not shared
among pieces in the same IOP state within all the eyes
in E, while the gradient parameter is shared. This
gap enables specificity for each piece and can reflect
the internal heterogeneity. Then, the optimal param-
eters
ˆ
φ
(l)
S
= (w
(l)
S
,σ
(l)
S
,{b
(l)
i,S,k
}) are calculated as
ˆ
φ
(l)
S
=
argmin
φ
∑
i∈E
∑
L
i
k=1
∑
j∈I
i,k
n
y
(l)
i, j
−
w
(l)
S
t
i, j
+ b
(l)
i,S,k
o
2
,
where σ
(l)
S
is the standard deviation of the above
errors. Hereafter, we omit the mesh index l for sim-
plicity since each mesh is processed independently.
3.6 Evaluating the Generated Models
As shown above, several tree-structured PLR mod-
els could be obtained with respect to each threshold
value c. The easiest way to evaluate the models is
to select the one with the smallest residual sum of
squares (ERROR). However, this can result in over-
fitting, because the ERROR becomes smaller as the
tree structure of the PLR models deepens and more
detailed data are trained. Therefore, we chose the
best model based on information criteria, consider-
ing a trade-off between simplicity and fitness of a
model. Toward this end, the Akaike Information Cri-
terion (AIC) (Akaike, H., 1973), Bayesian Informa-
tion Criterion (BIC) (Schwarz, G., 1978), and Mini-
mum Description Length Criterion (MDL) (Rissanen,
J., 1986) are well-known information criteria.
The log-likelihood function for the TSLR is
calculated for each IOP state S as logL(φ
S
| D) =
−
1
2σ
2
S
∑
i∈E
∑
L
i
k=1
∑
j∈I
i,k
y
i, j
− (w
S
t
i, j
+ b
i,S,k
)
2
−
n
S
logσ
S
−
n
S
2
log(2π), where φ
S
:= (w
S
,σ
S
,{b
i,S,k
})
and n
S
denote the parameters and the number of data
points for the IOP state S, respectively. Therefore,
AIC, BIC, and MDL are calculated as follows:
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data
99
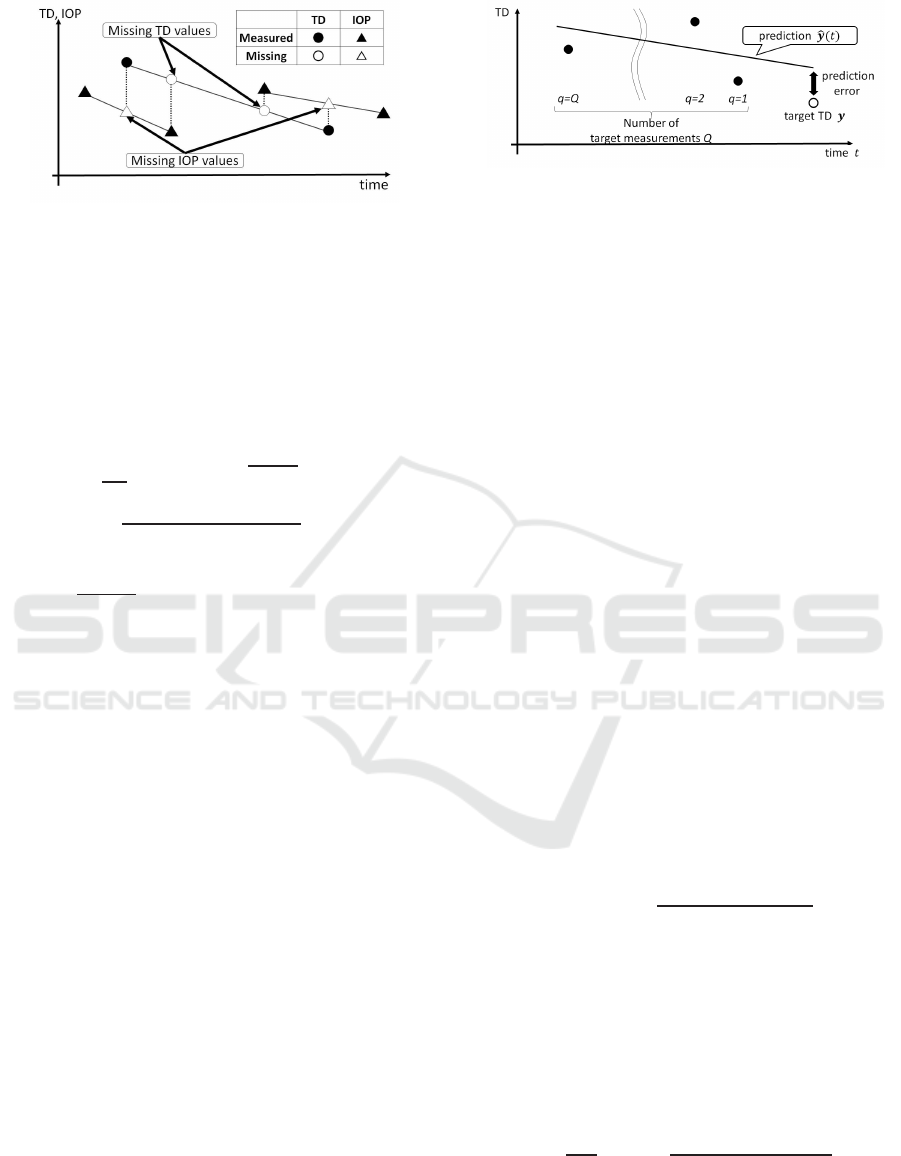
Figure 7: Interpolation of missing TD and IOP values.
AIC = −2logL(φ
S
| D) + 2
∑
i∈T
L
i
+ 2
!
,
BIC = −2logL(φ
S
| D) +
∑
i∈T
L
i
+ 2
!
log
∑
i∈T
n
i
,
MDL = −logL(φ
S
| D) +
∑
i∈T
L
i
+ 2
!
log
∑
i∈T
n
i
+
C
2V
h
arcsinx+ x
p
1− x
2
i
a
w
V/U
b
w
V/U
,
where U =
r
n
p−
∑
P
S
i=1
(q
2
i
/n
i
)
o
/2, V is the vari-
ance of all time stamps in IOP state S, and
C = 2
q
∏
P
S
i=1
n
i
1/(b
P
S
+1
σ
) − 1/(a
P
S
+1
σ
)
∏
P
S
i=1
(a
i
−
b
i
)/(P
S
+1), where (a
w
,b
w
), (a
i
,b
i
), and (a
σ
,b
σ
) rep-
resent the upper and lower bounds of the gradient w
S
,
intercept b
i
, and standard deviation σ
S
, respectively.
We designate p as the squared sum of time stamps,
q
i
as the sum of time stamps in the ith interval, P
S
as
the number of pieces, and n
i
as the quantity of data in
ith interval. The best model and the best threshold are
determined by minimizing the sum of the criteria for
the high- and low-IOP states.
4 EXPERIMENTS
4.1 Data and Parameters
The dataset used in this paper was provided by the De-
partment of Ophthalmology,The University of Tokyo.
This dataset includes visual-field data and IOP data
obtained from N = 939 glaucomatous eyes. The
visual-field data (TD values) were measured with the
Humphrey Field Analyzer (Carl Zeiss Meditec AG,
Dublin, CA, USA), using the SITA-standard 30-2
method (K = 74), controlling for the effects of in-
creasing age on the degree of visual-field loss. There-
fore, the TD value should decrease if glaucoma has
progressed. The TD values were within the range
Figure 8: Predicting the last measurement of a target eye.
We use Q data points from the target eye for training in
addition to data from other eyes.
[−37.0, 4.0] (median: -4, mean: -8.662, standard de-
viation: 10.56). The mean number of measurements
of TD and IOP for each patient was around 11 and 21,
respectively. The standardized-score threshold was
set within [0.1,3.0] (step: 0.1).
4.2 Data Interpolation
Our method requires both visual-field data and IOP
data for each data point. However, most data points
only contain one or the other measure. To facilitate
the use of the dataset, we employed linear interpola-
tion to fill in the missing data using the two neighbor-
ing measurements for the missing measurement, as
shown in Fig. 7. We used these preprocessed data in
all of the experiments, even when IOP values were not
required. The gap between the time stamp from the
last measurement in the training dataset and the target
dataset was within [28.9,889] (median: 227, mean:
258, standard deviation: 117) days.
4.3 Evaluation of Prediction Accuracy
We predict the TD value at the last measurement for
the target eye using the previous Q data points with
data from another N − 1 eyes, as depicted in Fig. 8.
We set Q = 1, . . . ,6. The prediction accuracy was
evaluated with the Root Mean Square Error (RMSE):
RMSE
i
=
s
K
∑
d=1
(y
(d)
i
− ˆy
(d)
i
)
2
/K,
where y
(d)
i
and ˆy
(d)
i
denote the measured and predicted
TD value for the dth mesh of the ith eye, respectively.
Smaller RMSE means better prediction. We also eval-
uated the accuracy of prediction gained in applying
our meta-algorithm to existing methods based on the
Improvement Rate (IR):
IR =
100
N
N
∑
i=1
1−
(RMSE
i
of applied)
(RMSE
i
of original)
.
Greater IR indicates larger enhancement of predic-
tion accuracy among N eyes by applying our meta-
algorithm.
HEALTHINF 2016 - 9th International Conference on Health Informatics
100

In the following sections, we evaluate the effi-
cacy of our proposed method applied to Liang et al.’s
method(Liang, Z. et al., 2013) in terms of RMSE
and IR. We define the optimal selection procedure as
BEST that achieves the smallest RMSE by choosing
the optimal model for each eye; i.e., BEST selects the
best model from all the generated models on the basis
of the TD value to be predicted. We employed leave-
one-out cross-validation in the following analyses.
4.4 Experiment 1: The Best Use of IOP
Experimental Settings: There are several options to
employ IOP information in our method, including the
use of raw values, raw deviations, and standardized
scores. Here, we show the superiority of using the
standardized scores to the other two options in terms
of improvement in prediction accuracy for the BEST
procedure. The thresholds for the raw value and raw
deviation were set within [10, 30] (step: 1) and [0.1,3]
(step: 0.1), respectively. Both units are mmHg.
Results: Table 1 displays the median, mean, and
10%-trimmed mean of the RMSEs using our method
with the three analytical approaches of IOP. As for
the mean and 10%-trimmed mean, the standardized
score significantly improved prediction accuracy for
all Q according to a one-sided Student’s t-test. There
was no significant difference in terms of the median.
Table 2 shows the proportion of cases in which our
method using the standardized IOP scores performed
better in terms of prediction compared to that using
the other two scores. The use of our standardized
scores significantly outperformed the use of the other
two according to a one-sided binomial test.
4.5 Experiment 2: Best Information
Criterion
Experimental Settings: We compared the ERROR,
AIC, BIC, and MDL to determine which criterion is
most suitable for prediction with our method. We also
analyzed the BEST case as reference.
Results: Table 3 presents the median and mean
RMSEs of the predictions obtained with the model for
the ERROR, AIC, BIC, MDL, and BEST. The MDL
performed significantly better than the others in terms
of the mean RMSEs at the 1% significance level with
a one-sided Student’s t-test in most cases, while it
performed better at the 5% significance level when
Q = 3. As for the case of the median RMSEs, the
MDL performed well in most cases but there was no
significant difference. Table 4 shows the proportionof
cases in which our method using the four criteria most
accurately predicted the value. A one-sided binomial
test verified that the MDL significantly outperformed
the other criteria at the 0.1% level in all the cases.
4.6 Experiment 3: Effectiveness of IOP
Experimental Settings: We compared our method
as applied to the original Liang et al.’s method (Liang,
Z. et al., 2013) with the original to investigate the im-
provement in predictive ability. Based on the results
of Experiment 2, we used the MDL. In addition, we
compared our method with the BEST to evaluate the
predictive potential obtained by introducing the IOP.
Results: Table 5 shows the median, mean, and
10%-trimmed mean of the RMSEs for predictions ob-
tained with our proposed method compared to the
original method. As for the mean, the Student’s t-test
verified that our model significantly outperformed the
original at the 0.1% level when the null hypothesis
IR = 0 was set for both trimmed and non-trimmed
cases. For the median, a Mann-Whitney-Wilcoxon
test verified that our model exceeded the predictive
power of the original at the 5% level except when
we set Q = 1. Table 6 shows the proportion of cases
in which our method predicted the value most accu-
rately. A binomial test determined that our method
was significantly better than the original. The results
shown in Tables 5 and 6 revealed that our method ap-
plied with the BEST procedure is highly significantly
superior to the original for prediction accuracy.
4.7 Discussion
Experiment 1: The use of a standardized IOP score
was more effective compared to the use of raw IOP
values or deviations as an indicator of the IOP state.
We believe that this is because the usual state of the
IOP differs from eye to eye; thus a standardized score
can normalize IOP values across eyes, whereas the
raw value and deviation cannot. With the three indi-
cators of IOP, we segmented the IOP states into high
and low states. The fact that the standardized score
was the best indicator of the IOP states indicates a
method for controlling for the heterogeneous differ-
ences among individuals; i.e., solving the external
heterogeneity of IOP. Thus, the scores could correctly
represent the fundamental states of most eyes.
Experiment 2: The results suggested that MDL was
a better choice for improving predictions compared
to others. This result could be due to the following
factors. The framework of MDL, which can take the
structure of a model into account by calculating the
optimal code length, fits well with the property of
our PLR model owing to its clear tree structure (see
Fig. 3). Indeed, several studies have shown that the
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data
101
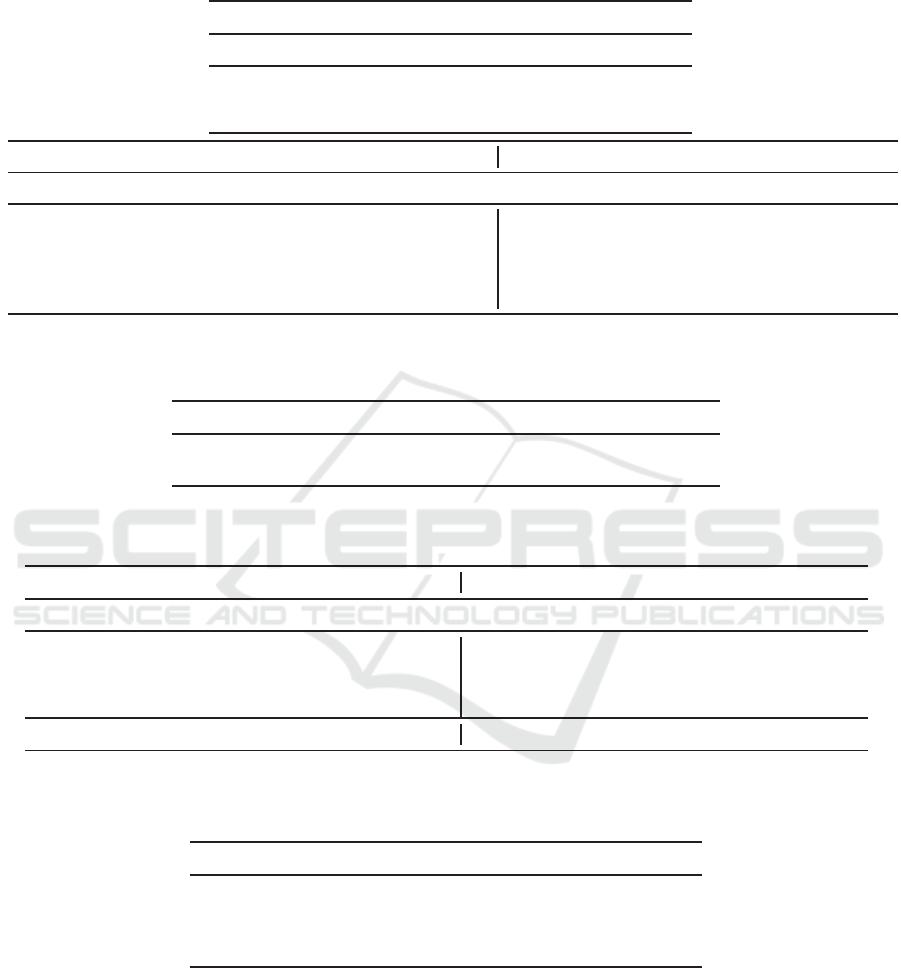
Table 1: Median, mean, and 10%-trimmed mean of RMSEs using the raw values, raw deviations, and standardized scores
of the IOP for each eye. The BEST optimal selection procedure was used for prediction. The symbol ♯ indicates statistical
significance at the 0.1 % level.
Q 1 2 3 4 5 6
(a) Median RMSEs
Std. score 4.026 3.834 3.726 3.673 3.652 3.621
Raw val. 4.112 3.879 3.828 3.759 3.756 3.716
Raw dev. 4.139 3.861 3.770 3.739 3.724 3.666
Q 1 2 3 4 5 6 1 2 3 4 5 6
(b) Mean RMSEs (c) 10%-trimmed Mean RMSEs
Std. score 4.314 4.081 3.991 3.969 3.933 3.875 4.161 3.918 3.809 3.775 3.750 3.708
Raw val. 4.376 4.144 4.054 4.029 3.998 3.939
4.226 3.984 3.872 3.838 3.817 3.771
IR 1.463♯ 1.563♯ 1.576♯ 1.606♯ 1.707♯ 1.834♯
1.262♯ 1.402♯ 1.376♯ 1.391♯ 1.472♯ 1.834♯
Raw dev. 4.456 4.184 4.091 4.100 4.023 3.946
4.241 3.977 3.879 3.851 3.811 3.756
IR 1.550♯ 1.252♯ 1.300♯ 1.501♯ 1.139♯ 1.214♯
0.880♯ 0.803♯ 0.862♯ 0.830♯ 0.777♯ 0.844♯
Table 2: Proportion (%) of cases for which our proposed standardized score of IOP yielded better prediction performance.
Cases for the BEST procedure are shown. The symbol ♯ indicates statistical significance at the 0.1 % level.
Q 1 2 3 4 5 6
v.s. Raw val. 70.54♯ 71.27♯ 69.28♯ 71.41♯ 70.20♯ 73.64♯
v.s. Raw dev. 63.70♯ 61.26♯ 61.44♯ 62.65♯ 62.03♯ 62.74♯
Table 3: Median and mean of RMSEs for predictions with the models selected according to the ERROR/AIC/BIC/MDL, and
the possible best model (BEST). The symbols ∗ and † indicate statistical significance at 5% and 1 %, respectively.
Q 1 2 3 4 5 6 1 2 3 4 5 6
(a) Median RMSEs (b) Mean RMSEs
ERROR 4.461 4.179 4.097 4.083 4.104 4.085 4.738 4.509 4.425 4.425 4.391 4.322
AIC 4.461 4.179 4.096 4.088 4.104 4.087
4.737 4.508 4.425 4.425 4.391 4.323
BIC 4.456 4.173 4.101 4.083 4.099 4.092
4.731 4.505 4.424 4.423 4.387 4.320
MDL 4.422 4.163 4.113 4.087 4.064 4.076
4.682† 4.471† 4.394* 4.388† 4.351† 4.291†
BEST 4.026 3.834 3.726 3.673 3.652 3.621 4.314 4.081 3.991 3.969 3.934 3.875
Table 4: Proportion (%) of cases for which each criterion gave the best performance for prediction. The symbol ♯ indicates
statistical significance at the 0.1 % level.
Q 1 2 3 4 5 6
ERROR 17.26 16.77 17.76 15.80 17.55 15.97
AIC 14.98 13.53 14.75 15.80 13.40 15.26
BIC 19.33 19.81 21.74 19.19 19.63 20.96
MDL 48.43♯ 49.89♯ 45.75♯ 49.20♯ 49.43♯ 47.81♯
MDL works well for tree-structured models (Mehta,
M. et al., 1995; Robnik-
ˇ
Sikonja, M. and Kononenko,
I., 1998). In addition, extremely ineffective models
were not selected when using the MDL, as implied by
the fact that there were significant differences in the
mean, but almost no differences in the median.
Meanwhile, because the MDL is difficult to calcu-
late analytically, it is difficult to apply the MDL to our
proposed method for complex models such as Murata
et al.’s model (Murata, H. et al., 2014). However, suf-
ficiently accurate results were gained with our method
just using ERROR This might be because we assumed
that the gradients of each piece were the same in the
same IOP state, which worked as a kind of regularizer
of the parameters. This result suggests that using ER-
ROR is a feasible solution for more complex models.
HEALTHINF 2016 - 9th International Conference on Health Informatics
102

Table 5: Median, mean, and 10%-trimmed mean of RMSEs. Liang et al.’s method (Liang, Z. et al., 2013) is referred to as the
“original”, and our method as applied to the original method is referred to as the “proposed”. The symbols ∗ and ♯ indicate
statistical significance at the 5% and 0.1 % level, respectively.
Q 1 2 3 4 5 6
(a) Median RMSEs
Original 4.557 4.386 4.246 4.261 4.241 4.205
Proposed (MDL) 4.422 4.163* 4.113* 4.087* 4.064* 4.076*
Proposed (BEST) 4.026♯ 3.834♯ 3.726♯ 3.673♯ 3.652♯ 3.621♯
Q 1 2 3 4 5 6 1 2 3 4 5 6
(b) Mean RMSEs (c) 10%-trimmed Mean RMSEs
Original 369.8 566.6 300.0 262.1 391.4 235.4 4.681 4.487 4.394 4.357 4.317 4.266
Proposed
4.682 4.471 4.394 4.388 4.351 4.291
4.525 4.299 4.208 4.181 4.155 4.116
(MDL)
IR 4.541♯ 5.177♯ 4.985♯ 4.853♯ 4.205♯ 4.182♯
2.670♯ 2.944♯ 2.932♯ 3.108♯ 2.811♯ 2.833♯
Proposed
4.314 4.081 3.991 3.969 3.934 3.875
4.161 3.918 3.809 3.775 3.750 3.708
(BEST)
IR 12.78♯ 14.19♯ 14.54♯ 14.83♯ 14.32♯ 14.50♯
10.92♯ 11.95♯ 12.49♯ 12.76♯ 12.54♯ 12.89♯
Table 6: Proportion (%) of cases in which our method applied to Liang et al.’s method is superior to the original method based
on MDL and BEST. The ♯ indicates that the values are statistically significant at the 0.1 % level.
Q 1 2 3 4 5 6
Proposed (MDL) 71.41♯ 71.38♯ 71.57♯ 71.74♯ 72.74♯ 67.63♯
Proposed (BEST) 99.24♯ 99.35♯ 99.78♯ 99.45♯ 99.23♯ 99.11♯
Experiment 3: Table 5 demonstrates that the appli-
cation of our proposed method to the original Liang
et al.’s method showed much better performance than
the original method. Moreover, our method achieved
better prediction accuracy with small Q. It is well-
known that high IOP exacerbates the progression of
glaucoma (see Sec. 2.2). Therefore, data incorporat-
ing eyes at different IOP states can be anomalous for
long-term prediction of the original method. In our
method, such noise is excluded by segmenting the
data with IOP values, which cannot be realized in the
original method. Therefore, we suppose that this sep-
aration of the data enables our method to produce ac-
curate predictions with less data owing to its stronger
power of expression and purity of the training data.
Table 6 also indicates that our method with MDL
represents a significant improvement over the orig-
inal, and that it can help to improve the outcome
for a large number of eyes. However, about 30 %
of patients would not benefit from our method judg-
ing from the results. This demonstrates that the best
model is not always selected with MDL, and that there
is still room for improvement in considering informa-
tion criteria. Nonetheless, Table 6 shows that insofar
as our framework exploits the IOP and copes with ex-
ternal and internal heterogeneity, it offers more accu-
rate predictions compared to existing methods.
Overall Discussion: We have shown the efficacy of
our collective PLR model in predicting the progres-
sion of glaucoma. Since our method can be applied
to other existing methods, it is expected to serve as an
improvement of current methods by exploiting sup-
plemental data. However, there is still room for im-
provement in the model-selection phase, because the
accuracy of the prediction with the BEST procedure
was much better than that when using other informa-
tion criteria. One possible explanation for this result
is the small number of data entries for the high-IOP
state, making it difficult to comprehensively evaluate
the model for this state. Thus, our future work will fo-
cus on such situations to improvethe proposed model.
5 CONCLUSION
We have proposed a novel collective PLR method
that copes with external and internal heterogeneity as
well as the medical-data-structure-difficulty of medi-
cal datasets. Existing methods cannot cope with the
internal heterogeneity, i.e., a situation where the rate
of progression for individual eyes changes over time.
We have dealt with this internal heterogeneity using
Predicting Glaucomatous Progression with Piecewise Regression Model from Heterogeneous Medical Data
103

a PLR model based on clinical knowledge regarding
the relationship between IOP and the rate of glauco-
matous progression (AGIS Investigators, 2010; Col-
laborative Normal-Tension Glaucoma Study Group,
1998; Satilmis, M. et al., 2003). Our method can
also deal with the external heterogeneity and medical-
data-structure-difficulty by incorporating a collective
method. Therefore, our method is a novel extension
of previous collective methods from both theoretical
and practical aspects, which increases prediction ac-
curacy. Similarly, other methods (Maya, S. et al.,
2014; Murata, H. et al., 2014) are expected to be im-
proved by incorporation of our method.
Medical datasets are commonly plagued by high
levels of heterogeneity, and we have here proposed
a new method that shows good performance in over-
coming this heterogeneity in a glaucoma dataset for
effective predictions of disease progression. We be-
lieve that our method can be extended to tackle simi-
lar difficulties in other medical datasets and we have
provided standardized directions for such analyses.
ACKNOWLEDGEMENTS
We thank Mr. Fujino and Ms. Taketani at the De-
partment of Ophthalmology,The University of Tokyo,
for their useful advice. This work was supported by
CREST, JST.
REFERENCES
AGIS Investigators (2010). The advanced glaucoma in-
tervention study (AGIS): 7. the relationship between
control of intraocular pressure and visual field de-
terioration. American Journal of Ophthalmology,
130:429–440.
Akaike, H. (1973). Information theory and an extension
of the maximum likelihood principle. In Proceedings
of the 2nd International Symposium on Information
Theory, pages 267–281.
Collaborative Normal-Tension Glaucoma Study Group
(1998). Comparison of glaucomatous progression be-
tween untreated patients with normal-tension glau-
coma and patients with therapeutically reduced in-
traocular pressures. American Journal of Opthalmol-
ogy, 126(4):487–497.
Fujino Y., Murata H., Mayama C., and Asaoka R. (2015).
Applying “lasso” regression to predict future visual
field progression in glaucoma patients. Investigative
Ophthalmology & Visual Science, 56(4):2334–2339.
Holmin, C. and Krakau, C. E. T. (1982). Regression anal-
ysis of the central visual field in chronic glaucoma
cases. Acta Ophthalmologica, 60(2):267–274.
Kingman, S. (2004). Glaucoma is second leading cause of
blindness globally. Bulletin of the World Health Or-
ganization, 82(11):887–888.
Liang, Z., Tomioka, R., Murata, H., Asaoka, R., and Ya-
manishi, K. (2013). Quantitative prediction of glau-
comatous visual field loss from few measurements.
In Proceedings of the 2013 IEEE 13th International
Conference on Data Mining 2013, pages 1121–1126.
Maya, S., Morino, K., and Yamanishi, K. (2014). Predicting
glaucoma progression using multi-task learning with
heterogeneous features. In Proceedings of the 2014
IEEE International Conference on Big Data, pages
261–270.
Maya, S., Morino, K., and Yamanishi, K. (2015). Discov-
ery of glaucoma progressive patterns using hierarchi-
cal MDL-based clustering. In Proceedings of the 21st
ACM SIGKDD Conference on Knowledge Discovery
and Data Mining, pages 1979–1988.
Mehta, M., Rissanen, J., and Agrawal, R. (1995). MDL-
based decision tree pruning. In Proceedings of the
1st ACM SIGKDD Conference on Data Mining, pages
216–221.
Morino, K., Hirata, Y., Tomioka, R., Kashima, H., Ya-
manishi, K., Hayashi, N., Egawa, S., and Aihara,
K. (2015). Predicting disease progression from short
biomarker series using expert advice algorithm. Sci-
entific Reports, 5:8953.
Murata, H., Araie, M., and Asaoka, R. (2014). A new ap-
proach to measure visual field progression in glau-
coma patients using variational bayes linear regres-
sion. Investigative Ophthalmology & Visual Science,
55:8386–8392.
Natarajan, R. and Pednault, E. (2002). Segmented regres-
sion estimators for massive data sets. In Proceedings
of the 2nd SIAM International Conference on Data
Mining, pages 566–582.
Quigley, H. A. and Broman, A. T. (2006). The number of
people with glaucoma worldwide in 2010 and 2020.
British Journal of Ophthalmology, 90(3):262–267.
Rissanen, J. (1986). Stochastic complexity and modeling.
Annals of Statistics, 14(3):1080–1100.
Robnik-
ˇ
Sikonja, M. and Kononenko, I. (1998). Pruning re-
gression trees with MDL. In Proceedings of the 13th
European Conference on Artificial Intelligence, pages
455–459.
Satilmis, M., Org¨ul, S., Doubler, B., and Flammer, J.
(2003). Rate of progression of glaucoma correlates
with retrobulbar citation and intraocular pressure.
American Journal of Ophthalmology, 135(5):664–
669.
Schwarz, G. (1978). Estimating the dimension of a model.
Annals of Statistics, 6(2):461–464.
Taketani Y., Murata H., Fujino Y., Mayama C., and Asaoka
R. (2015). How many visual fields are required to pre-
cisely predict future test results in glaucoma patients
when using different trend analyses? Investigative
Ophthalmology & Visual Science, 56(6):4076–4082.
Vogel, D. S., Asparouhov, O., and Scheffer, T. (2007). Scal-
able look-ahead linear regression trees. In Proceed-
ings of the 13th ACM SIGKDD Conference on Knowl-
edge Discovery and Data Mining, pages 757–764.
Zhu, H., Russell, R. A., Saunders, L. J., Ceccon, S.,
Garway-Health, D. F., and Crabb, D. P. (2014). De-
tecting changes in retinal function analysis with non-
stationary weibull error regression and spatial en-
hancement (ANSWERS). PLOS ONE, 9(1):e85654.
HEALTHINF 2016 - 9th International Conference on Health Informatics
104
