
Modeling of an Insect Proprioceptor System based on Different Neuron
Response Times
Daniel Rodrigues de Lima
1
, Michel Bessani
1
, Philip Newland
2
and Carlos Dias Maciel
1
1
Department of Electrical and Computational Engineering, University of S
˜
ao Paulo, S
˜
ao Carlos, S
˜
ao Paulo, Brazil
2
Centre for Biological Science, University of Southampton, Highfield Campus, S017 1BJ Southampton, U.K.
Keywords:
Neuronal Spike Signals, Neuronal Response, Desert Locust, FeCO, Transfer Entropy, Inter-Spike Interval,
Survival Analysis.
Abstract:
This paper analyzes neuronal spiking signals from the Desert Locust Femorotibial Chordotonal Organ (FeCO).
The data comes from records of the insect neuronal response due to external stimulation. We measured the
Inter-Spike Interval (ISI) and calculated Transfer Entropy for investigate different FeCO responses. ISI is a
technique that measures the time between two spikes; and transfer entropy is a theoretical information measure
used to find dependencies and causal relationships. We also use survival functions to assemble FeCO models.
Furthermore, this work uses and compares results of two approaches, one with transfer entropy and other with
ISI measures. The results indicate evidence to support the existence of more than one type of FeCO neuron.
1 INTRODUCTION
The biologically inspired engineering studies biologi-
cal structures intending to find solutions for engineer-
ing problems (Zhang et al., 2015). Examples include
the Particle Swarm Optimization Algorithm (Santos
and Maciel, 2014), the control of exoskeletons using
principles found in biomechanics (Jimnez-Fabin and
Verlinden, 2012) and neuronal circuits (Endo et al.,
2015). Recent works analyzed neural signals (Je-
gadeesan et al., 2015) trying to identify patterns and
connections inside the nervous system (Subramaniam
et al., 2015). They analyzed signals from neural ex-
periments which are records from the neuronal re-
sponses due to some specific stimulus or situations
(Birmingham et al., 2014).
This paper presents the analysis of signals col-
lected on experiments with desert locust neurons. The
experiments were made by stimulating the insect lag
with a forceps that was shaken by a Gaussian White
Noise (GWN). Figure 1 shows the signal analysis
schematic and presents a sample of the recorded sig-
nals. The data analyzed come from a large and multi-
variate neurobiological data set obtained from a neu-
ral insect network (Newland and Kondoh, 1997). This
network produces and controls the movements of the
desert locust hind leg (Angarita-Jaimes et al., 2012).
Specifically, the data analyzed here were collected on
the Femorotibial Chordotonal Organ (FeCO). FeCO
is a proprioceptor that detects movements from the
tibia relative to the femur of a desert locust hind leg
(Burrows et al., 1988), and its behavior is equivalent
to others found in more complex structures (Vidal-
Gadea et al., 2010).
Previous studies investigated neuronal structure
models in the FeCO data (Maciel et al., 2012).
They also investigated characteristics of the signal
transmission channel (Endo et al., 2015). While
doing these experiments, transfer entropy measure-
ments (Schreiber, 2000) suggested the existence of
different kinds of FeCO. Transfer entropy is an
information-theoretic measurement, which allows to
detect non-linearity (Nichols et al., 2005), establish
causal relationships (Barnett et al., 2009), and also
find interaction delays between signals (Pampu et al.,
2013). Some curves obtained from these measure-
ments showed different FeCO responses and pointed
the possible existence of different groups of neurons.
Those curves motivated this investigation about how
many kinds of FeCO neurons exist.
In order to investigate different FeCO responses
we combined two analyzes: transfer entropy and the
Inter-Spike Interval (ISI) (Schwalger et al., 2015).
Classically, ISI measures the amount of time spent be-
tween spikes, trying to find a probability distribution
for them (Chen et al., 2009). However, this study uses
ISI measurements to determine parameters to classify
the FeCO in different groups of neurons. The pa-
Lima, D., Bessani, M., Newland, P. and Maciel, C.
Modeling of an Insect Proprioceptor System based on Different Neuron Response Times.
DOI: 10.5220/0005706202190226
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 4: BIOSIGNALS, pages 219-226
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
219

GWN
27HZ
GWN
200HZ
FeCO
Figure 1: Schematic of the signal analysis presenting the collected signals (bottom) and their interaction (top). In the ex-
periment, a Gaussian Withe Noise (GWN) with a bandwidth of 200Hz passed trough a low-pass filter with cutoff frequency
manually adjusted to 27Hz or 58Hz. The filtered signal shook a forceps that moved the locust leg stimulating its nervous
system. The FeCO signal is the neuronal response to the excitation, and it is also a spiking signal. This diagram is the primary
assumption, which considers only one kind of FeCO.
rameters found are used in a statistical representation,
which uses survival functions (Lawless, 2011) to as-
semble FeCO models.
Survival Analysis aims to explore the behavior of
sets of individuals according to the time interval nec-
essary for an event to happen (Collett, 2003). Ex-
amples include a system failure (Achcar and Moala,
2015) or a patient longevity (MacKenzie et al., 2014).
The survival technique also analyzes data from remis-
sion time (Cabrero et al., 2015), which is the time
spent between two occurrences of an event (Bewick
et al., 2004). ISI is the time spent between two spikes
(two occurrences), and it is possible to use survival
functions to create FeCO models.
The study goal is to investigate whether the FeCO
response rates would change under the same stimula-
tion. For that, we analyzed FeCO signals recorded
during two different experiments: one applying a
GWN stimulation limited to 27Hz; and another lim-
ited to 58Hz. To perform the analysis, we used ISI
technique and statistical treatments as a criterion to
divide and classify the signals into different groups.
We also used transfer entropy to reinforce our as-
sumption about the existence of different kinds of
FeCO and investigate the differences between the
groups found. Finding different types of FeCO, the
diagram in Figure 1 will need to be changed. This
study will be useful then to check if the diagram from
Figure 1 is sufficient to represent the FeCO neuronal
structure or if a more complex model is required.
Furthermore, using the two analyzes, ISI and
transfer entropy, it is possible to identify different
kinds of FeCO neurons. Additionally, next section
will present the survival analysis and transfer entropy
theories. They are followed by the methodology sec-
tion that describes the experiment. Also, the results
section will present the FeCO analysis, followed by
the conclusion and next steps.
2 THEORY
This section presents the theories used to analyze the
FeCO data, and assemble the models created, respec-
tively Transfer Entropy, and Survival Analysis.
2.1 Transfer Entropy
We used the property of transfer entropy detect time
delays (Pampu et al., 2013) as a reference measure
to check the algorithms consistency by calculating
the propagation time between the signals 200Hz and
27Hz. We also used it to look for dependencies be-
tween FeCO and 27Hz signals. Additionally, it will
be used to reinforce our assumption of different kinds
of FeCO.
Transfer entropy has significant properties. The
first one is that it indicates the shared information be-
tween two random variables, making possible to de-
termine connections and dependencies (Runge et al.,
2012). A second one is that transfer entropy uses
a time lag to introduce directional sense (Schreiber,
2000), causal sense (Wibral et al., 2012) and find
time-delays (Ito et al., 2011). These properties make
it similar to time-delayed mutual information (Jin
et al., 2010). However, time-delayed mutual infor-
mation does not distinguish information exchanged
from shared information, while transfer entropy does
(Schreiber, 2000).
Transfer entropy (Vicente et al., 2011) is defined
as follows by
T E(X;Y ) =
∑
p(y
t+u
, y
d
y
t
, x
d
x
t
)log
p(y
t+u
|y
d
y
t
,x
d
x
t
)
p(y
t+u
|y
d
y
t
)
, (1)
where t represents the discrete time index, u the pre-
diction time, and x
d
x
t
and y
d
y
t
are the delay vectors.
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
220
2.2 Survival Analysis
A survival time variable (T) is a non-negative random
variable representing the time until an event occurs
(Klein and Moeschberger, 2003). If f(t) represents
T probability density function, and F(t) represents
its cumulative distribution function, then the survival
function (S(t)) is defined as follows:
S(t) = Pr(T ≥ t) = 1 −F(t) =
Z
∞
t
f (x)dx, (2)
which represents the probability of an event do not
occur during time t.
Another concept related to the survival variable T
is the hazard function h(t) defined as:
h(t) = lim
∆t→0
P(t ≤ T < t + ∆t|T ≥ t)
∆t
. (3)
The function h(t) represents the instantaneous
rate of a given event occurs at time t (Klein and
Moeschberger, 2003). For ∆t → 0,
P(t ≤ T < t + ∆t) → f (t) (4)
and using the conditional probability rule (Walpole
et al., 2014) we can rewrite (3),
P(A|B) =
P(A ∩ B)
P(B)
⇒ h(t) =
f (t)
S(t)
. (5)
In the experiments, the excitation signals had a
constant bandwidth, hence we considered that the
spike rate is also constant. Consequently, the hazard
function is also constant, and in this case, it is possible
to use an exponential distribution,
f (t) = λe
−λt
= µe
−
t
µ
, (6)
with survival function
S(t) = e
−λt
= e
−
t
µ
, (7)
where λ is the spike rate (spikes/ms), and µ is the dis-
tribution mean (ms/spike) (Harrell, 2013).
To obtain the exponential models for the data sets,
we sampled the time between spikes (ISI), and calcu-
lated a mean time between spikes (MTBS - µ) on each
one of the signals.
3 MATERIALS AND METHODS
The experiments used thirty five adult desert locusts,
Schistocerca gregaria (Forsk
˚
al), male and female, ar-
ranged ventral-side-uppermost in modeling clay. By
cutting a piece of cuticle in their anterior distal fe-
mur, the FeCO apodeme was exposed and gripped by
a forceps attached to a shaker (Ling Altec 101). A
small window opened in their ventral thorax exposed
the metathoracic ganglion that was immobilized on a
wax covered with a silver platform. The sheath was
treated with protease (Sigma type XIV) for 1 min be-
fore recording. After that, microelectrodes filled with
potassium acetate and with DC resistances of 50-80
MΩ were driven through the sheath. They entered
into the neuropilar processes of the nonspiking lo-
cal interneurons, the axons of sensory neurons, or
the somata of spiking local interneurons. Intracellular
recordings were made using an Axoclamp 2A ampli-
fier (Axon Instruments, USA).
A GWN signal shook the forceps. The GWN
was produced by filtering a pseudo random binary
sequence (CG-742, NF Circuit Design Block) band-
limited to 27Hz or 58Hz with low-pass filters (SR-
4BL, NF Circuit Design Block) with a decay of 24
dB/octave. This generated a signal with a Gaus-
sian probability density function in the bandwidth
of interest. The signal vibrated the forceps holding
the apodeme, stimulating and evoking the interneu-
rons responses, as shown in Figure 2. The signals
were stored on magnetic tape using a PCM-DAT data
recorder (RD-101 T, TEAC, Japan) and then sampled
at a rate of 10 kHz offline to a computer.
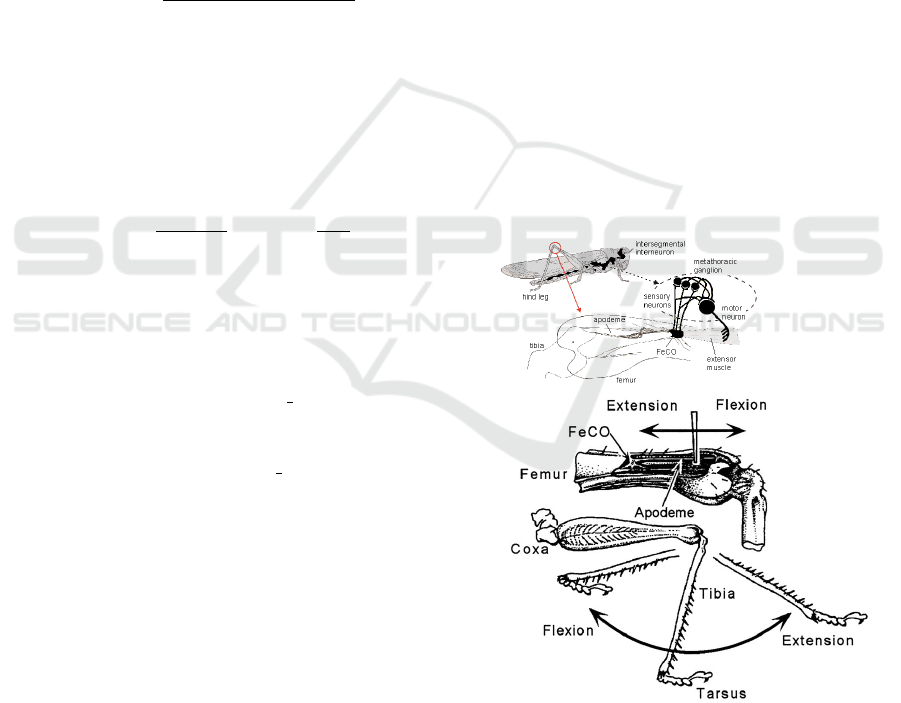
Figure 2: Desert Locust neuronal system (top) with the
neurons where the signals were collected, highlighting
the FeCO. Correspondent leg movement for the exten-
sion/flexing caused by the forceps excitation (bottom).
Modeling of an Insect Proprioceptor System based on Different Neuron Response Times
221
4 RESULTS
This section presents the ISI Analysis on 27Hz, and
58Hz signals, and transfer entropy measurements in
the 27Hz signals.
4.1 ISI Analysis
Experiment with 27Hz Signals
The MTBS on each one of the 27Hz signals were
measured, and their confidence intervals were calcu-
lated. Table 1 shows the results after processing the
27Hz data set, calculating the MTBS and confidence
intervals considering an α = 5% for all 25 samples.
Table 1: MTBS and confidence intervals for 27Hz signals.
Sample
CI Low MTBS CI Up
(ms/spike) (ms/spike) (ms/spike)
1 15.79 16.92 18.05
2 19.52 20.32 21.12
3 21.69 22.50 23.30
4 21.75 22.60 23.45
5 21.72 22.67 23.61
6 22.01 23.01 24.02
7 21.95 23.18 24.40
8 22.46 23.49 24.52
9 23.73 24.66 25.58
10 24.06 25.35 26.65
11 24.45 25.56 26.68
12 25.68 26.91 28.15
13 26.14 28.71 31.27
14 28.42 29.89 31.35
15 30.75 32.63 34.50
16 31.42 33.05 34.69
17 33.25 35.84 38.43
18 34.95 36.82 38.69
19 45.31 49.10 52.89
20 46.83 49.79 52.76
21 46.85 49.83 52.80
22 50.00 53.26 56.53
23 51.90 54.12 56.34
24 58.15 62.16 66.18
25 58.14 62.28 66.41
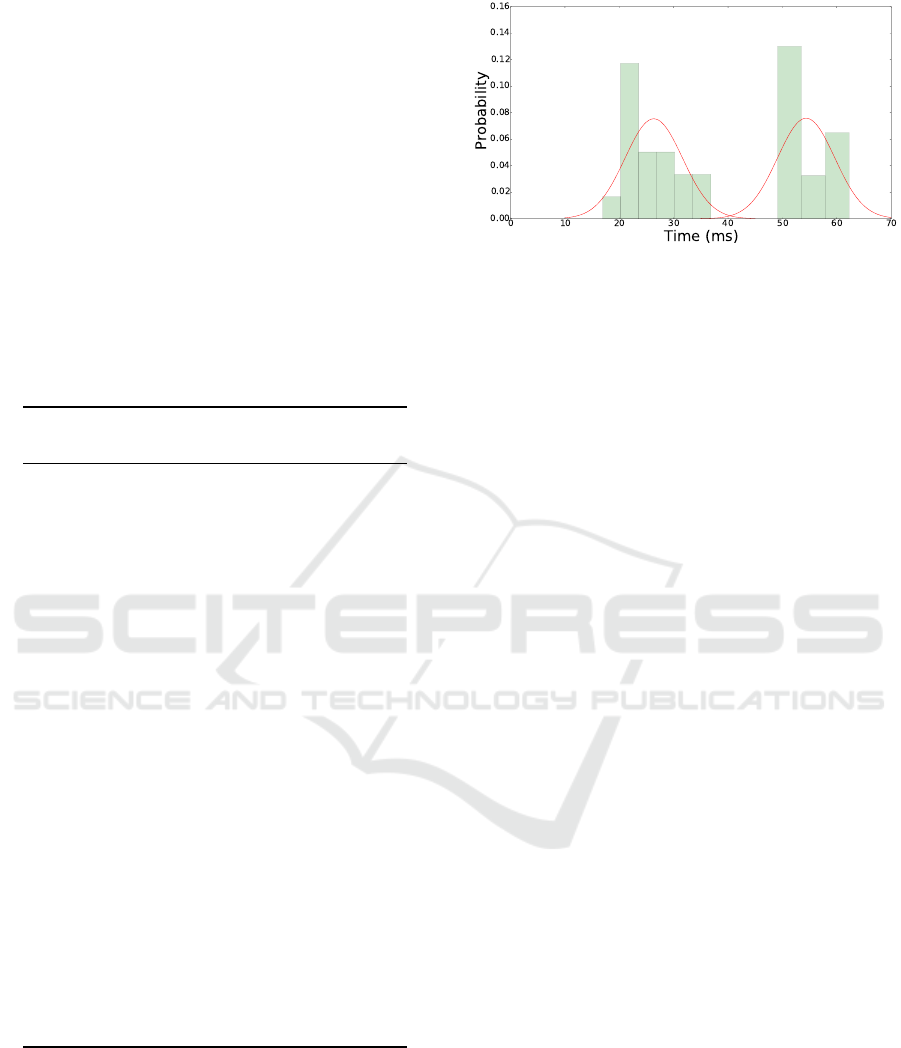
Table 1, presents a gap between samples 18 and
19 confidence intervals. The same gap appears in the
MTBS histogram presented in Figure 3. These gaps
point to the existence of two groups, i.e., two different
neuronal responses rates.
Since samples 1 and 2 confidence intervals are dis-
connected, we performed a hypothesis test comparing
the MTBS, testing if there are two or three groups of
Figure 3: Histogram of the MTBS (Table 1) calculated for
the 25 signals in the 27Hz data set. It is possible to note a
gap indicating the presence of two different groups.
neurons (Walpole et al., 2014). The tests assumed a t
statistic with an α = 5%, and followed the hypothesis
H
0
: µ = µ
i
H
1
: µ 6= µ
i
.
(8)
The hypothesis H
0
indicates that the MTBS mea-
sured (µ
i
) is equal to the group mean (µ), while the
hypothesis H
1
indicates the opposite. The hypothesis
H
0
was accepted with a significance level α = 5% for
all MTBS inside each group.
Experiment with 58Hz Signals
The time between each spike on the 58Hz signals
was measured in order to calculate the MTBS and
their confidence intervals. Table 2 shows the results
after processing all the 58Hz data set, calculating
the MTBS and confidence intervals considering an
α = 5%.
From Table 2, it is possible to see that the sam-
ples confidence intervals cannot be separated. This
suggests only one group of neurons in the 58Hz data.
This is reinforced by Figure 4 that shows the his-
togram of MTBS. To assure this assumption, a hy-
pothesis test (Equation 8) for the MTBS was made
with an α = 5%. As a result, the hypothesis H
0
was
accepted for all MTBS (µ
i
).
A consideration must be made for the 58Hz exper-
iment, since its results opposed to the ones presented
in Figure 3. The number of samples in the 58Hz ex-
periment is the same of the first group found in the
27Hz experiment; thence it is possible that the 58Hz
experiment only recorded signals from the first group.
However, it is known that there are nonlinear compo-
nents in the desert locust neuronal system (Dewhirst
et al., 2013). This implies that the output signal could
present an entirely different response once that the in-
put excitation is changed.
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
222
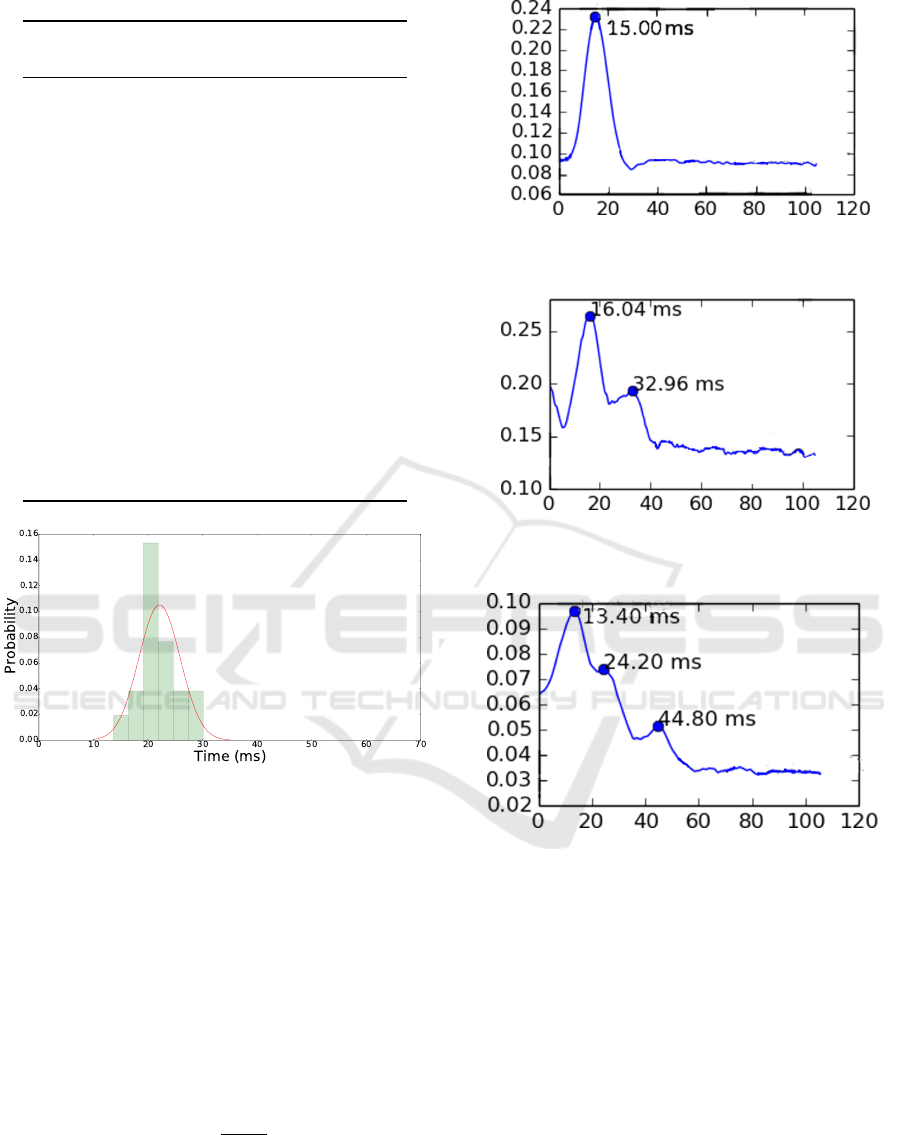
Table 2: MTBS and confidence intervals for 58Hz signals.
Sample
CI Low MTBS CI Up
(ms/spike) (ms/spike) (ms/spike)
1 17.87 18.57 19.27
2 18.33 19.03 19.73
3 19.23 19.92 20.60
4 19.38 20.12 20.87
5 19.40 20.15 20.90
6 19.53 20.35 21.18
7 19.58 20.37 21.16
8 19.67 20.43 21.19
9 19.88 20.69 21.50
10 20.51 21.32 22.13
11 21.82 22.84 23.86
12 22.43 23.46 24.48
13 22.56 23.46 24.36
14 22.96 24.12 25.28
15 24.84 25.79 26.74
16 25.45 26.53 27.60
17 27.36 29.17 30.98
18 28.86 30.17 31.47
Figure 4: Histogram of MTBS (Table 2) calculated for the
18 signals in the 58Hz data set. It is possible to note only
one group of neurons.
4.2 Transfer Entropy
After making ISI analyzes and find evidences of two
possible kinds of neuronal response in the 27Hz data
set, we made transfer entropy measurements for the
signals inside the 27Hz experiment.
The filter used adds a fixed and well known time
delay for a given cutoff frequency f
c
(Manal and
Rose, 2007). The equation for the time delay is given
by
t
delay
=
0.416
f
c
, (9)
and since f
c
was 27Hz, it is expected that transfer en-
tropy presents a 15.41ms delay between 200Hz and
27Hz signals. Figure 5 shows that the time-delay
found is close to the expected.
Figure 5: Transfer entropy calculated between 200Hz and
27Hz signals for references purposes. The time delay found
is close to the expected with an error of 2.6%.
Figure 6: Transfer entropy calculated between 27Hz and
FeCO signals in the first group of neurons, showing two
maximum points.
Figure 7: Transfer entropy calculated between 27Hz and
FeCO signals in the second group of neurons, showing three
maximum points.
We calculated transfer entropy between 27Hz and
FeCO signals in the first group, which is presented in
Figure 6. It is important to note that the graph shows
two maximum points.
We also calculated transfer entropy between 27Hz
and FeCO signals in the second group. Figure 7
presents the results and points three time-delays.
Figure 6 differs from Figure 7 in the number of
time-delays found. The first figure presents two max-
imum points: one at 16.04 ms, and another at 32.96
ms. The second presents three maximums: one at
13.40 ms, other at 24.20 ms, and another at 44.80 ms.
Modeling of an Insect Proprioceptor System based on Different Neuron Response Times
223
4.3 FeCO Survival Models
We found two groups of neurons in the 27Hz exper-
iment and one group in the 58Hz. Using MTBS it
is possible to construct models representing their re-
sponse due to a fixed excitation. Thence, it is consid-
ered the Survival models for that construction.
Considering that the distribution mean is the mean
of the group, we calculated confidence intervals with
an α = 5% for each group mean, as shown in Table 3.
Table 3: Parameters calculated for the 27Hz experiment.
Group
λ µ CI 95%
(spikes/ms) (ms/spike) Low Up
1st 0.04 26.34 23.64 29.04
2nd 0.02 54.36 51.55 57.12
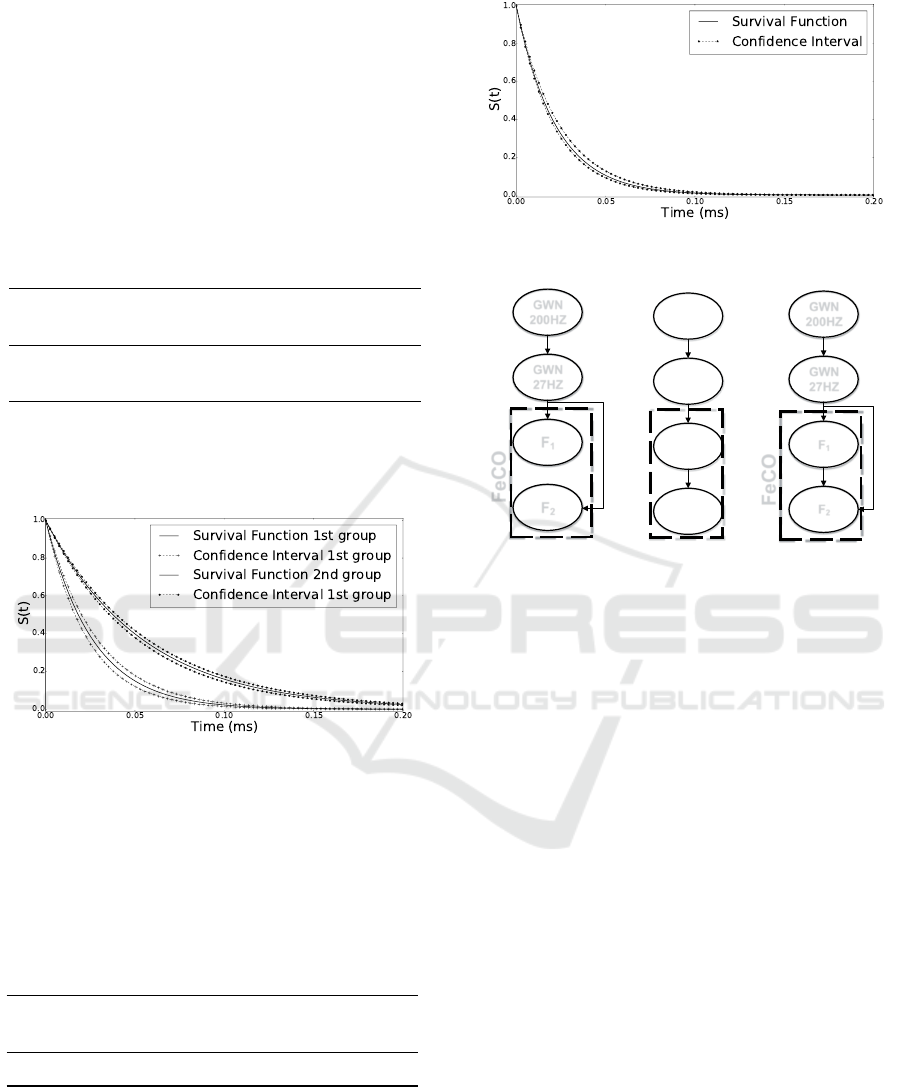
Figure 8 presents a graphic visualization, and it
is possible to notice that there is no common region
either for the functions or their confidence intervals.
Figure 8: Survival probability functions for the two groups
of neurons. The confidence intervals of those groups do not
touch, evidencing the existence of two groups of neurons.
In the 58Hz experiment we found only one neu-
ronal response rate. Considering that the distribution
mean is the group mean, we calculated a confidence
interval with an α = 5% for the group, as shown in
Table 4.
Table 4: Parameters calculated for the 58Hz experiment.
Group
λ µ CI 95%
(spikes/ms) (ms/spike) Low Up
1st 0.05 22.11 20.89 24.27
4.4 FeCO Model
Results pointed two different neuronal responses,
which brings two possibilities. The first one is that it
happens because there are two different FeCO struc-
tures. The second is that it happens because of the
Figure 9: Survival probability function for the group of neu-
rons found in the 58Hz data set with its confidence intervals.
GWN
27HZ
GWN
200HZ
F
1
F
2
FeCO
GWN
27HZ
GWN
200HZ
F
1
F
2
FeCO
GWN
27HZ
GWN
200HZ
F
1
F
2
FeCO
Figure 10: Models suggested for the FeCO. A model with
two different neurons that are independent (left). Other
model where only the fist kind of FeCO affects the second
type (middle). A model showing an influence of the first
kind of FeCO, but it also presenting a direct influence of the
27Hz signal on the second kind of FeCO (right).
neuronal configuration. Figure 10 presents three hy-
pothetical neuronal configurations.
The structures from Figure 10 are only sugges-
tions for the neuronal circuit configuration, which
need deeper investigations to confirm. The first con-
figuration assumes two different kinds of FeCO, and
they receive direct influence from the excitation sig-
nal. The second and the third configurations can as-
sume two distinct kinds of FeCO or only just two dif-
ferent response rates given by the same kind of FeCO
with different configurations. Both, third and second
configurations, suggest a model organized in layers
where there is a preprocessing process.
5 CONCLUSION
In this paper, we used two analyzes to investigate dif-
ferent neuronal responses due to the same excitation.
We analyzed signals from the desert locust metatho-
racic ganglion. Specifically we looked at the data
from the Desert Locust Femorotibial Chordotonal Or-
gan, which is a spiking neuron. The diagram shown in
Figure 1 was the simplest representation possible for
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
224

the FeCo experiment, ignoring the existence of any
other kind of FeCO neurons.
Figure 1 presented a diagram connecting the
200Hz signals to the 27Hz signals, and then 27Hz
to FeCO, and does not consider any other influences,
such as other neurons. However, here we pointed ev-
idence that suggest the existence of different kinds of
FeCO neurons. These findings show that it is possible
to obtain different response rates for FeCO neurons
even when they receive the same stimulation. There-
fore, it implies that there is a different connection than
the one presented in Figure 1.
Our assumption of a different connection than the
one showed in Figure 1 is supported by three differ-
ent analyses. The histograms of MTBS from Figure
3 pointed the existence of different neurons using the
ISI technique, and statistical hypothesis tests supports
these results. The survival models also showed two
different curves with non-overlapping confidence in-
tervals. Additionally, Figure 6 and Figure 7 shows
two different responses for each group using the trans-
fer entropy technique.
All these measurements and tests point the exis-
tence of more than one kind of FeCO neuron. A con-
sideration must be made for the 58Hz signals since
they presented only one group, a result different than
the one showed for 27Hz signals. It is known that
there are nonlinear components inside the neuronal
system (Dewhirst et al., 2013), and this implies that
the output response could be entirely different once
the input excitation is changed.
We know that the algorithms for transfer entropy
are correct because of the reference measurement pre-
sented in Figure 5. A closer look at Figure 7 indicates
that it presents one more maximum point than Figure
6. By comparing the responses of Figure 6 and Figure
7 we made new assumptions about the connections in-
side the FeCO, they are presented in Figure 10.
The assumptions presented in Figure 10 suggest
two particular cases. The first one with a structure
where there are no influence or iterations between the
two kinds of FeCO. The second case is that there is an
interaction between them. This case is supported by
Figure 7 that presents one more maximum point than
Figure 6, indicating a model organized in layers.
Those three possible structures shown in Figure
10 are not the only possible configuration for the neu-
ronal system; the locust may have a complex struc-
ture. Future works are required to determine the best
connections and the best structure. Therefore, this is
our next steps when we intend to investigate the best
structure model using Dynamical Bayesian Networks
(Meyer-Baese and Schmid, 2014).
Moreover, this analysis indicates that the FeCO
neuronal connection structure is more complex than
the one presented in Figure 1. Additionally, we pre-
sented models based on survival functions for the
FeCO response. Those models can also be used to
perform simulations in future works.
ACKNOWLEDGEMENT
The authors would like to thank the Brazilian Federal
Agency for Support and Evaluation of Graduate Edu-
cation - CAPES; and also thank the National Counsel
of Technological and Scientific Development CNPq
for the Project number 475064/2013-5.
REFERENCES
Achcar, J. A. and Moala, F. A. (2015). Use of copula func-
tions for the reliability of series systems. Interna-
tional Journal of Quality & Reliability Management,
32(6):617–634.
Angarita-Jaimes, N., Dewhirst, O. P., Simpson, D. M., Kon-
doh, Y., Allen, R., and Newland, P. L. (2012). The dy-
namics of analogue signalling in local networks con-
trolling limb movement. European Journal of Neuro-
science, 36(9):3269–3282.
Barnett, L., Barrett, A. B., and Seth, A. K. (2009). Granger
causality and transfer entropy are equivalent for gaus-
sian variables. Phys. Rev. Lett., 103:238701.
Bewick, V., Cheek, L., and Ball, J. (2004). Statistics review
12: survival analysis. CRITICAL CARE-LONDON-,
8:389–394.
Birmingham, K., Gradinaru, V., Anikeeva, P., Grill, W. M.,
Pikov, V., McLaughlin, B., Pasricha, P., Weber, D.,
Ludwig, K., and Famm, K. (2014). Bioelectronic
medicines: a research roadmap. Nature Reviews Drug
Discovery, 13(6):399–400.
Burrows, M., Laurent, G., and Field, L. (1988). Propriocep-
tive inputs to nonspiking local interneurons contribute
to local reflexes of a locust hindleg. The Journal of
neuroscience, 8(8):3085–3093.
Cabrero, M., Jabbour, E., Ravandi, F., Bohannan, Z., Pierce,
S., Kantarjian, H. M., and Garcia-Manero, G. (2015).
Discontinuation of hypomethylating agent therapy in
patients with myelodysplastic syndromes or acute
myelogenous leukemia in complete remission or par-
tial response: Retrospective analysis of survival after
long-term follow-up. Leukemia Research, 39(5):520
– 524.
Chen, L., Deng, Y., Luo, W., Wang, Z., and Zeng, S. (2009).
Detection of bursts in neuronal spike trains by the
mean inter-spike interval method. Progress in Natural
Science, 19(2):229 – 235.
Collett, D. (2003). Modelling survival data in medical re-
search. Texts in statistical science. Chapman & Hall,
London, New York.
Modeling of an Insect Proprioceptor System based on Different Neuron Response Times
225

Dewhirst, O., Angarita-Jaimes, N., Simpson, D., Allen, R.,
and Newland, P. (2013). A system identification anal-
ysis of neural adaptation dynamics and nonlinear re-
sponses in the local reflex control of locust hind limbs.
Journal of Computational Neuroscience, 34(1):39–58.
Endo, W., Santos, F., Simpson, D., Maciel, C., and New-
land, P. (2015). Delayed mutual information infers
patterns of synaptic connectivity in a proprioceptive
neural network. Journal of Computational Neuro-
science, 38(2):427–438.
Harrell, F. E. (2013). Regression modeling strategies: with
applications to linear models, logistic regression, and
survival analysis. Springer Science & Business Me-
dia.
Ito, S., Hansen, M. E., Heiland, R., Lumsdaine, A., Litke,
A. M., and Beggs, J. M. (2011). Extending transfer
entropy improves identification of effective connectiv-
ity in a spiking cortical network model. PLoS ONE,
6(11):e27431.
Jegadeesan, R., Thakor, N., and Yen, S. C. (2015). Wire-
less for peripheral nerve prosthesis and safety. In
Neural Engineering (NER), 2015 7th International
IEEE/EMBS Conference on, pages 648–651.
Jimnez-Fabin, R. and Verlinden, O. (2012). Review of
control algorithms for robotic ankle systems in lower-
limb orthoses, prostheses, and exoskeletons. Medical
Engineering & Physics, 34(4):397 – 408.
Jin, S.-H., Lin, P., and Hallett, M. (2010). Linear and non-
linear information flow based on time-delayed mu-
tual information method and its application to cor-
ticomuscular interaction. Clinical Neurophysiology,
121(3):392–401.
Klein, J. P. and Moeschberger, M. L. (2003). Survival
analysis: techniques for censored and truncated data.
Springer Science & Business Media.
Lawless, J. F. (2011). Statistical models and methods for
lifetime data, volume 362. John Wiley & Sons.
Maciel, C. D., Simpson, D. M., and Newland, P. L. (2012).
Inference about multiple pathways in motor control
limb in locust. In BIOSIGNALS, pages 69–75.
MacKenzie, T., Gifford, A. H., Sabadosa, K. A., Quinton,
H. B., Knapp, E. A., Goss, C. H., and Marshall, B. C.
(2014). Longevity of patients with cystic fibrosis in
2000 to 2010 and beyond: survival analysis of the
cystic fibrosis foundation patient registry. Annals of
internal medicine, 161(4):233–241.
Manal, K. and Rose, W. (2007). A general solution for the
time delay introduced by a low-pass butterworth digi-
tal filter: An application to musculoskeletal modeling.
Journal of Biomechanics, 40(3):678 – 681.
Meyer-Baese, A. and Schmid, V. J. (2014). Pattern Recog-
nition and Signal Analysis in Medical Imaging. Else-
vier.
Newland, P. L. and Kondoh, Y. (1997). Dynamics of neu-
rons controlling movements of a locust hind leg ii.
flexor tibiae motor neurons. Journal of Neurophysi-
ology, 77(4):1731–1746.
Nichols, J., Seaver, M., Trickey, S., Todd, M., Olson, C.,
and Overbey, L. (2005). Detecting nonlinearity in
structural systems using the transfer entropy. Physi-
cal Review E, 72(4):046217.
Pampu, N., Vicente, R., Muresan, R., Priesemann, V.,
Siebenhuhner, F., and Wibral, M. (2013). Transfer
entropy as a tool for reconstructing interaction delays
in neural signals. In Signals, Circuits and Systems
(ISSCS), 2013 International Symposium on, pages 1–
4.
Runge, J., Heitzig, J., Marwan, N., and Kurths, J. (2012).
Quantifying causal coupling strength: A lag-specific
measure for multivariate time series related to transfer
entropy. Phys. Rev. E, 86:061121.
Santos, F. P. and Maciel, C. D. (2014). A pso approach for
learning transition structures of higher-order dynamic
bayesian networks. In Biosignals and Biorobotics
Conference (2014): Biosignals and Robotics for Bet-
ter and Safer Living (BRC), 5th ISSNIP-IEEE, pages
1–6.
Schreiber, T. (2000). Measuring information transfer. Phys-
ical review letters, 85(2):461.
Schwalger, T., Droste, F., and Lindner, B. (2015). Statis-
tical structure of neural spiking under non-poissonian
or other non-white stimulation. Journal of Computa-
tional Neuroscience, 39(1):29–51.
Subramaniam, K., Hooker, C. I., Biagianti, B., Fisher,
M., Nagarajan, S., and Vinogradov, S. (2015). Neu-
ral signal during immediate reward anticipation in
schizophrenia: Relationship to real-world motivation
and function. NeuroImage: Clinical, 9:153 – 163.
Vicente, R., Wibral, M., Lindner, M., and Pipa, G. (2011).
Transfer entropya model-free measure of effective
connectivity for the neurosciences. Journal of Com-
putational Neuroscience, 30(1):45–67.
Vidal-Gadea, A. G., Jing, X., Simpson, D., Dewhirst, O. P.,
Kondoh, Y., Allen, R., and Newland, P. L. (2010).
Coding characteristics of spiking local interneurons
during imposed limb movements in the locust. Jour-
nal of neurophysiology, 103(2):603–615.
Walpole, R., Myers, R., Myers, S., and Ye, K. (2014). Prob-
ability and statistics for engineers and scientists.
Wibral, M., Wollstadt, P., Meyer, U., Pampu, N., Priese-
mann, V., and Vicente, R. (2012). Revisiting wiener’s
principle of causality #x2014; interaction-delay re-
construction using transfer entropy and multivariate
analysis on delay-weighted graphs. In Engineering in
Medicine and Biology Society (EMBC), 2012 Annual
International Conference of the IEEE, pages 3676–
3679.
Zhang, Q., Yang, X., Li, P., Huang, G., Feng, S., Shen, C.,
Han, B., Zhang, X., Jin, F., Xu, F., and Lu, T. J. (2015).
Bioinspired engineering of honeycomb structure us-
ing nature to inspire human innovation. Progress in
Materials Science, 74:332 – 400.
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
226
