
A Semiautomatic Image Processing Tool to Measure Small Structures
in Magnetic Resonance Images of the Brain at 7 Tesla
Application to Hippocampus Subfields of Patients with Mild Cognitive Impairment
Alessandra Retico
1
, Graziella Donatelli
2
, Mauro Costagli
3,4
, Laura Biagi
3
, Maria Evelina Fantacci
1,5
,
Daniela Frosini
6
, Gloria Tognoni
6
, Mirco Cosottini
2,4
and Michela Tosetti
3,4
1
Istituto Nazionale di Fisica Nucleare, Sezione di Pisa, Pisa, Italy
2
Dipartimento di Ricerca Traslazionale e delle Nuove Tecnologie in Medicina e Chirurgia, Università di Pisa, Pisa, Italy
3
IRCCS Fondazione Stella Maris, Pisa, Italy
4
Fondazione Imago7, Pisa, Italy
5
Dipartimento di Fisica, Università di Pisa, Pisa, Italy
6
Dipartimento di Medicina Clinica e Sperimentale, Università di Pisa, Pisa, Italy
Keywords: Image Processing Methods, Magnetic Resonance Imaging, Hippocampus, Mild Cognitive Impairment,
Alzheimer Disease.
Abstract: The current availability of Magnetic Resonance (MR) systems that operate at ultra high magnetic field (≥ 7
Tesla) allows the representation of anatomical structures at sub-millimeter resolution. Interestingly, small
structures of the brain, such as the subfields of the hippocampus, the inner structures of the basal ganglia
and of the brainstem become visible. Suitable software packages that allow analyzing and measuring such
small structures are not currently readily available. We developed a semi-automated procedure to measure
the thickness of the stratum radiatum and lacunosum-moleculare (SRLM) of the hippocampus. The change
in the thickness of this subfield of the hippocampal formation is supposed to have a role in the pathological
cognitive decline. Once we developed and validated the semiautomatic procedure on the 7T high-resolution
T2*-weighted images of a healthy volunteer, we carried out a preliminary study on a population affected by
Mild Cognitive Impairment to investigate the correlations of the SRLM thickness with the clinical scores of
the patients, e.g. the Mini-Mental State Examination score and the Free and Cued Selective Reminding Test.
1 INTRODUCTION
The use of ultra high-field (UHF) Magnetic
Resonance Imaging (MRI) systems, operating at
magnetic field strength of 7 Tesla and above, has
opened new perspectives in clinical research studies
(Kraff, 2015; van der Zwaag, 2015). Among the
established advantages of UHF is the high-resolution
structural imaging. Interestingly, small structures of
the brain, such as subfields of the hippocampus
(Thomas, 2008), become visible (Figure 1), but the
understanding of their potential role in the onset of
the Alzheimer’s Disease is still an open research
issue (Kerchner, 2012; Brown, 2014). Several
groups of researchers are working to make available
software packages to identify and measure the
hippocampal substructures with manual,
semiautomatic and automatic procedures (Van
Leemput, 2009; Wisse, 2012; Pipitone, 2014). Their
reliability and usefulness is still under evaluation by
the international scientific community. In particular,
none of the segmentation algorithms that have been
presented so far is able to measure an extremely thin
structure such as the stratum radiatum and
lacunosum-moleculare (SRLM) of the hippocampus.
Recent studies highlighted the relevance of this thin
hippocampal subfield in the early stages of
pathological cognitive decline, especially the mild
Alzheimer’s Disease. Cognitive decline can
represent a symptom of normal aging but also a
forerunner of dementia; within this spectrum of
disease, the Mild Cognitive Impairment (MCI),
defined as a cognitive decline greater than expected
for age and education that can evolve to dementia, is
an interesting transitional stage to investigate.
124
Retico, A., Donatelli, G., Costagli, M., Biagi, L., Fantacci, M., Frosini, D., Tognoni, G., Cosottini, M. and Tosetti, M.
A Semiautomatic Image Processing Tool to Measure Small Structures in Magnetic Resonance Images of the Brain at 7 Tesla - Application to Hippocampus Subfields of Patients with Mild
Cognitive Impairment.
DOI: 10.5220/0005818001240128
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 2: BIOIMAGING, pages 124-128
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

Figure 1: High-resolution coronal image of the
hippocampus in vivo acquired at 7 T with 2D T2*-
weighted GRE sequence (TE=22 ms, TR=240 ms, in-
plane resolution 0.3×0.3 mm
2
, slice thickness = 2 mm). A
slice through the hippocampus body is visible, showing
detailed structures including cornu ammonis (CA), dentate
gyrus (DG), stratum radiatum and lacunosum-moleculare
(SRLM), subiculum (SUB) and parahippocampal gyrus
(PHG).The overall thickness of the SRLM was shown to
be lower in patients with mild Alzheimer’s Disease
compared to healthy subjects (Kerchner, 2010) and to
correlate with memory performance (Kerchner, 2012).
The aim of the present work is to develop a
semiautomatic procedure to measure the SRLM
thickness by using 7T high-resolution T2*-weighted
MR images. Once the algorithm is set up on a
sample image of a healthy volunteer, the
semiautomatic procedure is used to investigate the
relationship between SRLM thickness and clinical
scores in MCI patients.
2 METHODS
We propose a semiautomatic procedure to measure
the thickness of the SRLM, which imports some
basic ideas of the algorithm presented by Kerchner
et al (2012) with the same purpose, and extend it to
make the measure more reliable and reproducible.
2.1 The Semiautomatic Algorithm to
Measure the SRLM Width
First of all an oblique coronal slice prescribed
perpendicularly to the axis of the hippocampus,
where the SRLM is visible, is presented to the user,
who is asked to select the centre of the Region of
Interest (ROI) for the following steps (see Figure 2).
The ROI is zoomed to allow the user to modify the
image contrast and the brightness and to draw a line
by mouse clicking along the curved shape of the
SRLM. Then, the algorithm that estimates the
SRLM width automatically starts running. It
prompts the user intermediate graphs and figures,
which are useful to check in real time the quality and
the reliability of the estimated SRLM width obtained
at the end of the procedure. Going into the details of
the procedure, it consists in the following steps
(Figure 2):
1) the user defined line following the curve
shape of the SRLM is interpolated with a
spline;
2) the normal directions to the spline are
computed and the normal vectors are overlaid
to the original ROI and shown to the user;
3) the image intensity profiles along the normal
directions are computed and mounted in 2D
image, where the hippocampus appears as
unrolled along the SRLM;
4) the SRLM appears as a dark straight band in
this 2D image, which is finally squeezed
along the SRLM direction to obtain the
average of the image intensity across all
normal profiles;
5) as this averaged intensity profile shows a
Gaussian shape, a fit with a Gaussian
function is carried out, and its width (4 σ) is
retained as a measure of the SRLM thickness.
As shown in Figure 2, a first check on the quality
and reliability of the SRLM measure obtained
through this semiautomatic procedure can be done
directly by the user while running the software. For
the procedure to be correctly executed, the manually
delineated line should be placed in the middle of the
SRLM or close to either its upper or lower
boundary. Only in this case the hippocampus will be
correctly unrolled and the picture in Figure 2(d) will
contain straight bands. This correct alignment of the
profiles prevents their average to blur and the SRLM
width to be overestimated.
2.2 Reproducibility of the Measure
The reproducibility of the measure of the SRLM
provided by the semiautomatic procedure has to be
checked. Variability in repeated measures are indeed
induced by the choice of the points along the SRLM
profile operated by the end user, which is scarcely
reproducible from run to run. To this purpose one of
the user of the proposed semiautomatic tool was
asked to repeat ten times the same measurement for
both the left and right hippocampi of one subject.
By means of these measurements the variability
of the measurements has been estimated.
A Semiautomatic Image Processing Tool to Measure Small Structures in Magnetic Resonance Images of the Brain at 7 Tesla - Application
to Hippocampus Subfields of Patients with Mild Cognitive Impairment
125
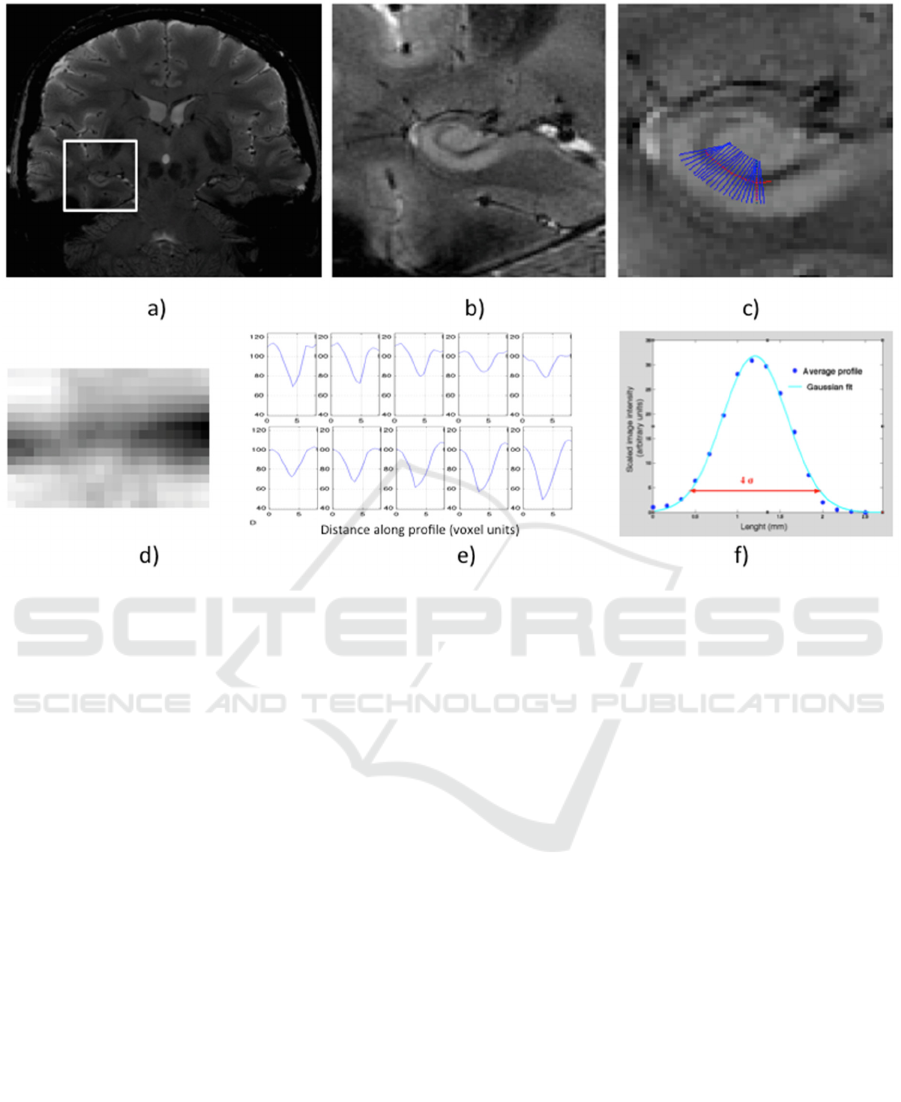
Figure 2: Main steps of the semiautomatic procedure to estimate the thickness of the stratum radiatum and lacunosum-
moleculare (SRLM): a) the user selects a point in the region of interest (ROI); b) the ROI is enlarged to allow the user to
draw the SRLM profile by mouse clicking; c) these point are interpolated with a spline and the normal vectors to the
profiles are computed and shown; d) the image intensity profiles along the normal segments are combined in a linear image
(the “unrolled” SRLM); e) a subsample of the profiles are shown to the user for a visual check; f) the linearized SRLM
image (d) is squeezed along the SRLM direction, i.e. the profiles (e) are averaged and the SRLM thickness is derived from
the width of the Gaussian fit (4 σ).
3 SET UP AND VALIDATION OF
THE ALGORITHM
3.1 Practical Implementation of the
Algorithm on MRI Data
The algorithm has been implemented in Matlab
(R2009b, The MathWorks, Inc.), and its execution is
managed by a very basic GUI, which only aims to
allow the software usage by researchers with a
limited expertise in the Matlab environment.
To set up the algorithm and to fix all the free
parameters, the brain 7T-MR examination of a
healthy volunteer was considered. MR images were
acquired with a 7T MR950 scanner (GE Healthcare
Medical Systems, Milwaukee, WI, USA) equipped
with a 2ch-Tx/32ch-Rx head coil (Nova Medical,
Wilmington, MA USA).
The acquisition protocol included a high-
resolution 2D T2*-weighted sequence prescribed
perpendicular to the longitudinal axis of the
hippocampus and covering the hippocampal body
(gradient-recalled echo –GRE–, TE=22 ms, TR=240
ms, in-plane resolution 0.3×0.3 mm
2
, slice thickness
= 2 mm).
Data are converted from the DICOM to the
NIFTI format as a prerequisite to run our algorithm.
As we are only interested in the thickness on the
SRLM, regardless its contrast with respect to the
surrounding brain structures, we do not need to
normalize the MR image intensity.
The user selects points along the SRLM, which
are connected through spline interpolation. The
obtained user-defined profile is sampled so to
accommodate at least 15 normal segments, whose
length is set to 2.5 mm. This length has been
optimized on data to properly cover the SRLM
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
126
thickness plus an additional margin, which is
necessary to obtain a stable Gaussian fitting.
3.2 Test of the Reproducibility of the
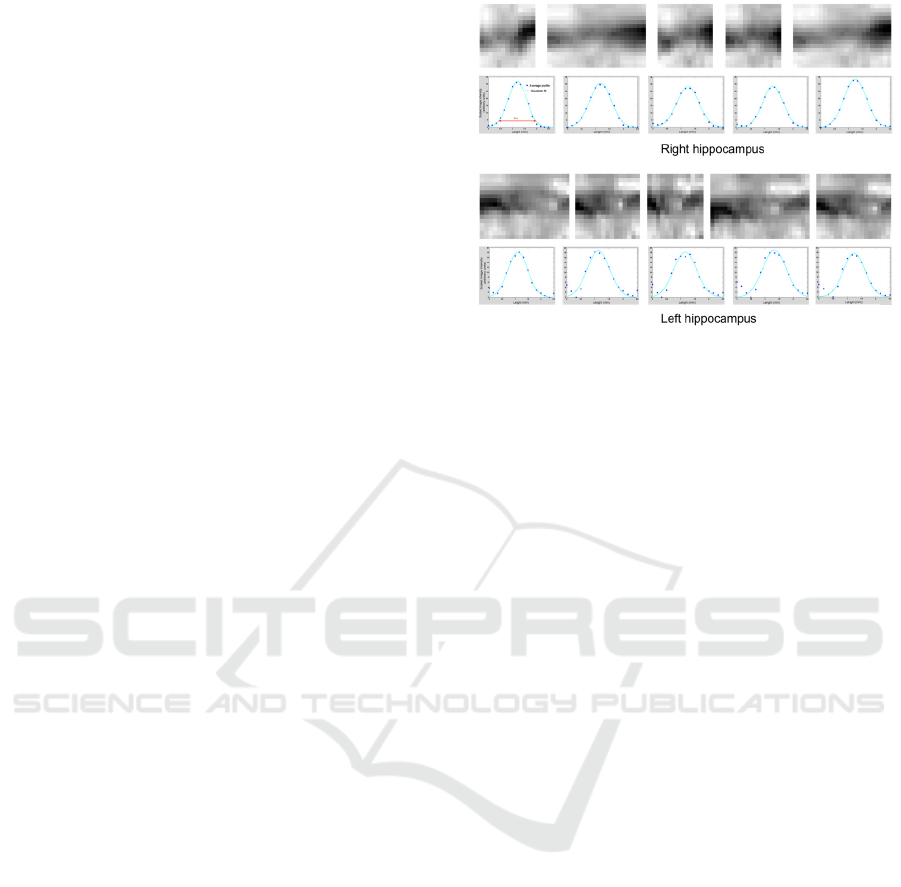
Measure of the SRLM Thickness
The thickness of the SRLM of the left and the right
hippocampi of the healthy volunteer has been
measured ten times. The variability in the
appearance of the linearized SRLM images obtained
at each repetition of the experiment is visible in
Figure 3, where only five out of the ten measures per
side are shown for demonstration purpose. Also the
Gaussian fits on the average profile are visible in the
figure.
We obtained the following measures of the
SRLM (average ± standard deviation [range of
values]): (1.61±0.10) mm [1.51–1.83 mm] for the
left hippocampus and (1.53±0.04) mm [1.47–1.61
mm] for the right hippocampus. The measure is
considered as highly reproducible, as the estimated
error is limited to few per cents of the measured
thickness, i.e. the 6% and the 3% for the left and
right hippocampus, respectively.
4 APPLICATION TO MCI
SUBJECTS
Once the semiautomatic algorithm has been
developed and validated on the 7T MR image of a
healthy volunteer, it has been used in a clinical study
involving subjects affected by MCI.
4.1 Data Sample
Ten MCI patients underwent a brain 7T-MR
examination including a high-resolution 2D T2*-
weighted sequence targeting hippocampus. All
patients underwent a neuropsychological battery
including the Mini-Mental State Examination
(MMSE) and the Free and Cued Selective
Reminding Test (giving great attention to the free
recall FCSRT-FR).
4.2 SRLM Measures on MCI Patients
and Correlation with Clinical Tests
An experienced neuroradiologist used the
semiautomatic image processing tool we developed
to delineate the SRLM profile on a coronal oblique
slice of the 7T T2*-weighted MRI of each MCI
patient. The thickness of the SRLM was estimated.
Figure 3: Test of the reproducibility of the SRLM
measure. Five repetitions of the algorithms for the left and
right hippocampi are shown (out of the 10 repetitions used
for the test): the user-defined SRLM profile slightly
changes from run to run leading to different “unrolled”
SRLM images, and slightly different averaged profile and
Gaussian fit.
This estimation has been done for both the right and
the left hippocampi.
The correlation between the measured SRLM
thickness and the numerical scores provided by the
neuropsychological tests were analysed according to
the Spearman’s rank test. Our preliminary results
showed that the average SRLM thickness (average
value between the left and right hippocampi)
correlated with MMSE score (r=0.60; p<0.1; n=10)
while the average SRLM thickness of the right
hippocampus correlated with the FCSRT-FR
(r=0.97; p<0.05; n=5).
5 CONCLUSIONS
We present in this paper a semiautomatic image
processing tool to measure the thickness of small
structures in anatomical images. The high-resolution
structural imaging that can be acquired with MRI at
UHF (7T) allows the visualization of very thin
anatomical structures. Suitable tools to measure their
size in a reproducible way might have a fundamental
role in clinical research studies.
As a case study we focused on the measure of
one of the hippocampal subfields, the SRLM, which
is supposed to be involved in early degenerative
changes related to pathological cognitive decline.
We developed the procedure on a representative 7T
T2*-weighted MR image of a healthy volunteer, and
tested it on a sample of subjects affected by MCI.
We found that the SRLM thickness correlated with
A Semiautomatic Image Processing Tool to Measure Small Structures in Magnetic Resonance Images of the Brain at 7 Tesla - Application
to Hippocampus Subfields of Patients with Mild Cognitive Impairment
127

the MMSE and the FCSRT-FR neuropsychological
scores.
The choice of a semiautomatic approach instead
of a fully automated one was dictated by two main
reasons: 1) the definition of the structures of interest
varies according to international protocols still under
study by the community of neurologists
(Yushkevich, 2015); 2) a semiautomatic tool can at
this stage help defining eloquent structures of
interest for specific pathologies in exploratory
studies by the community of neuroradiologists and
neurologists. Then, once the target anatomy of
interest is fully delineated, software developers can
further improve the semiautomatic tools to make
them fully automatic.
The possibility to extract from 7T brain MRI
quantitative features related to the underlying
pathological condition of the MCI subjects can open
the way to the development and application of more
robust predictive models of early diagnosis of
Alzheimer's disease based on machine learning
techniques (Chincarini, 2011; Retico, 2015).
Among the limitations of the present work are:
the need to implement other segmentation strategies
to obtain the global extent of the anatomical
structure under evaluation (e.g. the entire
hippocampus) to account for its effect in the
statistical analysis; the 2D nature of the procedure,
which can be extended to 3D data, where available;
the need to substantiate the reproducibility test
results with more than one subject’s data and to
validate the algorithm reliability in an inter-rater
reliability test.
Finally, the algorithm proposed in this paper,
despite tailored to measure the SRLM thickness, can
be used to measure the thickness of other thin
anathomical structures represented in 7T MR
images. Moreover, as the proposed approach is only
based on the assumption of a Gaussian shape of the
image intensity profile, it can be extended with few
modifications to measure the thickness of different
anathomical thin structures appearing in images
acquired with other imaging modality.
ACKNOWLEDGEMENTS
This work has been partially founded by the Italian
Ministry of Health and the Tuscany Government
(RF-2009-1546281 Clinical impact of ultra high-
field MRI in neurodegenerative diseases diagnosis
PI: M. Cosottini) and by the National Institute for
Nuclear Physics (nextMR project).
REFERENCES
Brown, TI, Hasselmo, ME et al., 2014. ‘A high-resolution
study of hippocampal and medial temporal lobe
correlates of spatial context and prospective
overlapping route memory’, Hippocampus, vol. 24,
no. 7, pp. 819-839.
Chincarini, A, Bosco, Pet al., 2011. ‘Local MRI analysis
approach in the diagnosis of early and prodromal
Alzheimer's disease’, Neuroimage, vol. 58, no. 2, pp.
469-480.
Kerchner, GA, Hess, CP et al., 2010. ‘Hippocampal CA1
apical neuropil atrophy in mild Alzheimer disease
visualized with 7-T MRI’, Neurology, vol. 75, pp.
1381-1387.
Kerchner, GA, Deutsch et al., 2012. ‘Hippocampal CA1
apical neuropil atrophy and memory performance in
Alzheimer's disease’, Neuroimage, vol. 63, pp. 194-
202.
Kraff, O, Fischer, A et al., 2015. ‘MRI at 7 Tesla and
Above: Demonstrated and Potential Capabilities’, J
MRI, vol. 41, pp. 13-33.
Pipitone, J, the Alzheimer's Disease Neuroimaging
Initiative et al., 2014. ‘Multi-atlas segmentation of the
whole hippocampus and subfields using multiple
automatically generated templates’, Neuroimage, vol
101, pp. 494-512.
Retico, A, Bosco P et al., 2015. ‘Predictive Models Based
on Support Vector Machines: Whole-Brain versus
Regional Analysis of Structural MRI in the
Alzheimer's Disease’, J Neuroimaging, vol. 25, no. 4,
pp. 552-563.
Thomas, BP, Welch, EB et al., 2008. ‘High-Resolution 7T
MRI of the Human Hippocampus In Vivo’, J MRI,
vol. 28, pp. 1266-1272.
van der Zwaag, W, 2015. ‘Recent applications of UHF-
MRI in the study of human brain function and
structure: a review’, NMR in Biomedicine, DOI:
10.1002/nbm.3275.
van Leemput, K, Bakkour, et al., 2009. ‘Automated
Segmentation of Hippocampal Subfields From Ultra-
High Resolution In Vivo MRI’, Hippocampus, vol. 19,
pp. 549-557.
Wisse, LEM, Gerritsen, L et al., 2012. ‘Subfields of the
hippocampal formation at 7 T MRI: In vivo
volumetric assessment’, Neuroimage, vol. 61, pp.
1043-1049.
Yushkevich, PA & Hippocampal Subfields Group (HSG).
2015. ‘Quantitative comparison of 21 protocols for
labeling hippocampal subfields and parahippocampal
subregions in in vivo MRI: Towards a harmonized
segmentation protocol’, Neuroimage, vol. 111, pp.
526-241.
BIOIMAGING 2016 - 3rd International Conference on Bioimaging
128
