
Use of Electrical Stimulation of the Posterior Tibial Nerve in Patients
with Bladder Hyperactivity as a Substitute for Pharmacological Therapy
based on Solifenacin Succinate and Oxybutynin Hyloridrate
Fabiana Bianchi Perez
1
, Jos
´
e Natal de Souza
2
, Nathalia Cristina Rosa
3
,
Cristiano Jacques Miosso
4
and Adson Ferreira da Rocha
4
1
Department of Medicine, University of Brasilia, Brasilia, DF, Brazil
2
Department of Pharmacology, University Alfredo Nasser, Aparecida de Goi
ˆ
ania, GO, Brazil
3
Department of Physiology, University Alfredo Nasser, Aparecida de Goi
ˆ
ania, GO, Brazil
4
University of Bras
´
ılia at Gama, Gama, DF, Brazil
Keywords:
Electrical Stimulation, Neurogenic Bladder Incontinence, Posterior Tibial Nerve, Parasympathetic System
Inhibitor, Anticholinergic Syndrome.
Abstract:
In a previous paper, we proposed a technique for the therapeutic use of Transcutaneous Electrical Nerve Stim-
ulation (TENS) of the posterior tibial nerve. The goal was to treat patients with signs of urinary incontinence
(UI) due to detrusor overactivity (DO). We now conduct a comparison between our proposed TENS technique
and the most commonly used treatment against DO, namely the pharmacological therapy based on solifenacin
succinate and oxybutynin hydrochloride. Our goal is to show that the TENS of the posterior tibial nerve treats
UI associated with DO, as well or better than the pharmacological treatment, but without the adverse side ef-
fects of the drugs (the so-called cholinergic syndrome). Method: we applied the proposed posterior tibial nerve
TENS in 21 patients, including the TENS Burst anaesthetic current. We treated each patient in 10 sessions, 2
times per week, for 20 minutes each session. Each patient could control the current intensity, in order to keep
it at comfortable levels, and in order to still be able to move the halux, thus simulating the Babinski reflex.
Results: we observed that with the proposed TENS approach and with the removal of the pharmacological
treatment, the cholinergic syndrome symptoms disappeared after 3 days without drugs, whereas we attained
the modulation of the non-inhibited detrusor contractions. Conclusion: this research provides evidence of the
proposed TENS technique’s efficiency in eliminating the effects of the hyperactive bladder, while avoiding the
symptoms of cholinergic syndrome associated to the more common pharmacological treatment. The statistical
tests showed that the reductions of DO symptoms and of the cholinergic syndrome were significantly improved
over the pharmacological treatment (p < 0.01). This improves patients’ life quality by reducing or eliminating
the social discomfort and hygiene problems associated to UI while avoiding the cholinergic syndrome.
1 INTRODUCTION
Detrusor hyperactivity is a type of urinary inconti-
nence, associated to severe social discomfort and hy-
giene problems. It also leads to important changes in
urinary frequency (including an increased number of
enuresis cases) and to the need for using urinary-loss
protection devices (Coyne et al., 2003; O’Conor et al.,
1998).
Among the possible treatment modalities, phar-
macological therapy is a common approach (Diokno
and Ingber, 2006), based for example on solifenacin
succinate and oxybutinin hydrochloride. However,
this approach usually leads to strong side effects, such
as dry mouth, blurred vision, and intestinal consti-
pation, which are jointly called the cholinergic syn-
drome.
In this context, we propose and evaluate a differ-
ent type of treatment based on electrical stimulation
of the posterior tibial nerve, with the purpose of re-
ducing the detrusor hyperactivity while avoiding the
cholinergic syndrome. We applied the proposed pro-
tocol in 10 sessions and with 21 patients, and our re-
sults suggest that the treatment reduces the effects of
detrusor hyperactivity without the side effects of the
pharmacological treatment.
Perez, F., Souza, J., Rosa, N., Miosso, C. and Rocha, A.
Use of Electrical Stimulation of the Posterior Tibial Nerve in Patients with Bladder Hyperactivity as a Substitute for Pharmacological Therapy based on Solifenacin Succinate and Oxybutynin
Hyloridrate.
DOI: 10.5220/0005826602750281
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 1: BIODEVICES, pages 275-281
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
275

2 BACKGROUND
In pharmacological therapy applied to disorders of the
detrusor and external urhetral sphincter, it is impor-
tant to analyze the neurophysiology of urination, and
its physio-pathological disorders. The injury or ill-
ness of the nervous system is an important compli-
cation of the urinary tract (UT). Specially, the neu-
rogenic bladder has several possible causes such as
spinal cord injury, stroke, brain stem injury, lesions in
the cerebral cortex, peripheral nervous system injury,
multiple sclerosis, quadriplegia and paraplegia.
2.1 Urination Control
The urination control is a neural-physiological pro-
cess which is controlled by the sympathetic and the
parasympathetic systems. The continence phase is
controlled by the sympathetic system with the alpha
1 and beta 2 adrenergics receptors. These are re-
sponsible for the contraction of the external sphincter
ureter and the perineum and detrusor muscles. The
parasympathetic system, on the other hand, controls
the urination itself based on a different mechanism,
using the perineum muscle and urethral sphincter to
relax and the detrusor muscle contract. Figure 1 illus-
trates this process.
2.2 Bladder Hyperactivity
Involuntary loss of urine is a problem of social order
and hygiene, causing embarrassment and changes in
behavior such as social isolation, low self-esteem and
psychosocial disorders (Oliveira et al., 2007). The
most common etiology of urinary incontinence is neu-
rogenic (Monteiro et al., 2009; van der Pal et al.,
2006; Coyne et al., 2003; Fischer-Sgrott et al., 2009).
The four handles or neurological pathways in the
control of urination, and which are related to each
other, are: the core trunk detrusor cerebral cortex
(loop I), the core detrusor muscle spinal / sacral brain-
stem (loop II)-sacral urethral sphincter of the bladder
(loop III), and the sacral-brain (loop IV). Pathways I
and IV are responsible for voluntary control of urina-
tion. Pathways II and III, on the other hand, regulate
the contractions of the detrusor bladder emptying to
promote and coordinate efforts between the detrusor
and urethra (Stephenson and O’Conor, 2004).
The neurogenic bladder dysfunction is defined as
a neurological disease produced by nerve damage
that interferes with the mechanisms of voluntary and
involuntary urination, thus causing changes in nor-
mal bladder function. The neurogenic bladder cor-
responds to the overactive and/or underactivity of the
Figure 1: Neurophysiology of urination: 1) Motor cortex;
2) afferent nerve; 3) Parasympathetic ganglion; 4) Pontine
“continence” center (L-region) – 4a: Nerve impulses stimu-
late contraction of muscle-hypogastric nerve and 4b: Nerve
impulses stimulate relaxation of internal urethral sphincter
muscle-pudendal nerve.
detrusor (Azevedo et al., 1990).
The underactive bladder retention or overflow is
characterized by urinary loss that occurs when intra-
vesical pressure exceeds urethral pressure. This is as-
sociated with bladder distention, but in the absence
of detrusor activity. This overflow happens when one
reaches the limits of distensibility or compliance of
the bladder (Miltrano, 2009).
According to the International Continence Soci-
ety, the overactive bladder is defined as a neurogenic
injury due to the presence of involuntary detrusor con-
tractions during the filling phase (Coelho, 2009). This
is characterized by urinary incontinence, urinary fre-
quency, nocturia and urgency (Fischer-Sgrott et al.,
2009; Jones III et al., 1988; O’Conor et al., 1998;
Guidi et al., 2005).
3 PHARMACHOLOGICAL
TREATMENT
Several drugs have been developed for treating the
overactive bladder. However, in our clinical practice
we observed that most become innocuous at the end,
leading to a symptom reduction that only lasts during
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
276

drug usage. Other drugs with proven clinical effects,
on the other hand, must be used for long periods and
show severe adverse effects, resulting in high dropout
rates.
3.1 Anticolinergics: The Most Used
Drugs
Stimulation of muscarinic receivers M2 and M3 cause
bladder contractions. Anticholinergic drugs inhibit
these receivers, causing the decrease of contractions’
amplitude and the increase of the first contraction vol-
ume, thus resulting in a higher bladder functional ca-
pacity.
These drugs’ parasympatholytic action occurs se-
lectively on these receptors. Yet, they have uncom-
fortable system effects, since the inhibition of mus-
carinic receivers do not occur only in the bladder,
where M2 and M3 receivers predominate. In the blad-
der, the M2 muscarinic are predominant, but the M3
receivers are more functionally important, mediating
direct contraction of detrusor muscle (Chapple et al.,
2002).
This prevents the interaction of acetylcholine with
receptors and inhibits the release of this neurotrans-
mitter in the synaptic cleft post-ganglionic.
There are two types of anticholinegic: (i) I-Mixed
(antispasmodics) — combine action antimuscarinic
with relax direct muscule (receiver independent) and
local anesthetic. The main representative of these
groups and cloridrat oxibutina. (ii) II-Pure — repre-
sented by tolterodine, propantelina, darifenacin, and
vamicamide. They are classified also with aminoter-
ciarios or aminoquaternarios. The difference between
the two groups refers to the ability of the blood brain
barrier crossing aminioterciarios.
The use of anthicholinergic is not free of side ef-
fects. Its major side effects are: dry mouth, consti-
pation and blurred vision, headache, dyspepsia and
even diarrhea, which corroborates with a dropout rate
of treatment during the first three months around
25,5% (Diokno and Ingber, 2006).
A survey showed that only 18% patients remained
taking anticholinergic by one period exceeding six
months. The activation of M3 receivers is due to the
detrusor contraction, but also to the contraction of the
smooth muscles of the gut and salivary glands (Dio-
kno and Ingber, 2006; Chapple et al., 2002).
The ability of the blood brain barrier to cross
the antimuscarinic agents can lead to cognitive alter-
ations, especially in the elderly. These side effects
occur by the relative loss of selectivity for the bladder
over other organs (Appell, 2003).
Other central effects include dizziness, memory
loss and drowsiness, the presence of receptors M1 in
the neocortex, hippocampus and neostratum (Appell,
2003).
Muscarinic receptor antagonists are formally not
recommended for patients with closed angle glau-
come and should be used with care in case of in-
fravesical obstruction, dur to the possibility of pre-
cipitating urinary retention.
4 TREATMENTS BASED ON
ELECTRICAL STIMULATION
The existing electrical stimulation techniques for UI
treatment are based on applying electrical current di-
rectly over the perineum muscle. This approach uses,
in the case of male patients, internal anal electrodes
and, in the case of female patients, internal or surface
vaginal electrodes. These techniques are embarrass-
ing, invasive (in the case of internal electrodes), and
may cause discomfort and burns in patients with ab-
normal sensibility (Marques, 2008).
Treatments with transcutaneous electrical stimu-
lation in the posterior tibial nerve aim at reducing UI
and assume that bladder neural projections exist in
that nerve’s path (Fischer-Sgrott et al., 2009).
The TENS current is used for the treatment of
urinary incontinence by bladder hyperactivity (BH).
The electrodes are placed bilaterally in the medial re-
gion of the legs, causing motor and sensory stimu-
lation as the current is applied (Fischer-Sgrott et al.,
2009). During each session, the patient’s neurologi-
cal physiotherapist or urological physiotherapist ob-
serves the stimulation caused by the motor current,
and the sensory way is not changed to modulate the
current flow. This technique promotes the reduction
of involuntary detrusor contractions (Marques, 2008).
Regarding the TENS current for the treatment of BH,
some researchers propose a sequence of pulses with
a frequency of around 20 Hz and with a duration of
around 200 milliseconds per pulse (Amarenco et al.,
2003). The therapies based on electrical currents can
be used in neurological patients with abnormal sensi-
tivity, because applying electrical stimulation results
in rhythmic flexing of the hallux, thus indicating the
correct placement of electrodes and confirming this
to be intact innervation (Maciel and Souto, 2009;
Fischer-Sgrott et al., 2009). However, in individu-
als with Babinski’s reflex, it is difficult to apply the
current therapeutic modulation due to incorrect motor
response from these individuals, so it becomes impos-
sible to control the current intensity and the electro-
motor response (Perez, 2011).
In the case of hyposensitivity, the dose should be
Use of Electrical Stimulation of the Posterior Tibial Nerve in Patients with Bladder Hyperactivity as a Substitute for Pharmacological
Therapy based on Solifenacin Succinate and Oxybutynin Hyloridrate
277
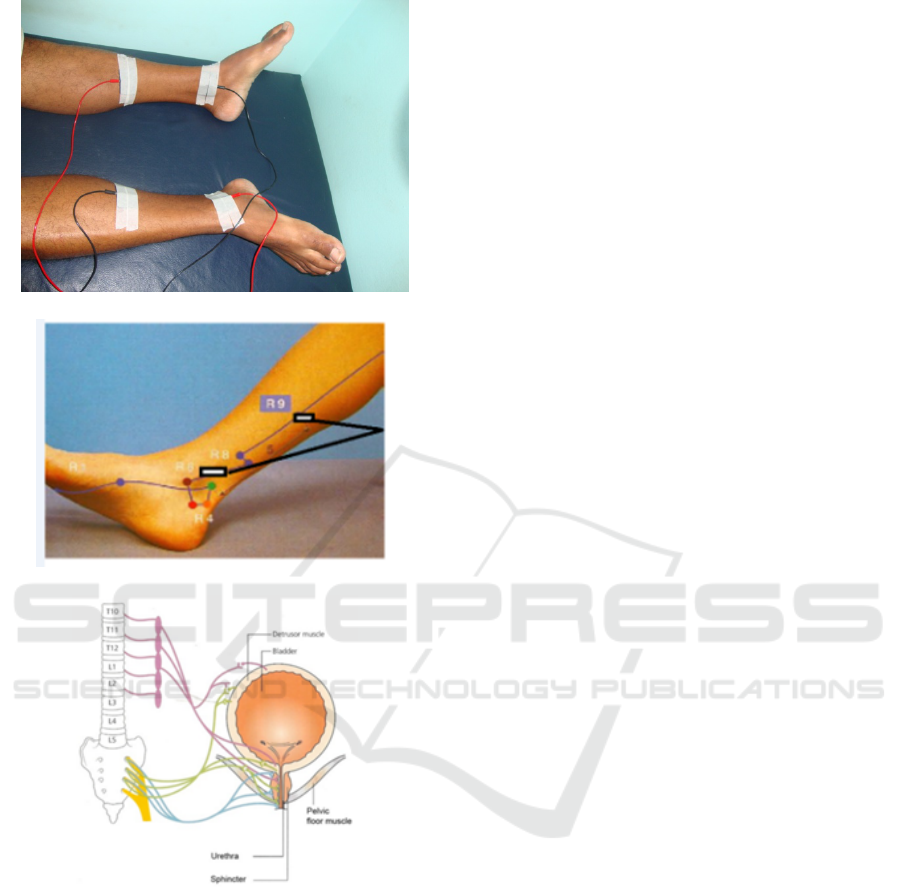
(a)
(b)
(c)
Figure 2: (a) Positioning of the 4 electrodes, two for
each channel, used to apply the electrical currents during
the TENS sessions. (b) Detail of the electrode position-
ing. Source: (Marques, 2008). (c) The connection be-
tween the posterior tibial nerve and the detrusor muscle;
source: http://www.bladderclinic.com.au/images/bladder-
conditions/neurogenic-bladder.png.
applied until it causes rhythmic inflections of the big
toe, and it should then be reduced until the motor ac-
tion disappears. The provided dose agrees with sev-
eral studies arguing that the ideal intensity must be
maintained according to the threshold of each patient
and below the motor threshold (Fischer-Sgrott et al.,
2009; Maciel and Souto, 2009; Amarenco et al., 2003;
C.Kabay et al., 2009).
The use of TENS in the current technique of pos-
terior tibial nerve can reduce the uninhibited detrusor
contractions and improve the quality of life of patients
with DO due to a reduction of urinary incontinence
and of the number of times that the patient urinates.
This can result in a better quality of sleep, humor, and
personal relationships, as well as in less embarrass-
ment and in reduction of stress. It can also potentially
reduce the use of medication while reducing or elim-
inating the sings of anticholinergic syndrome.
5 METHODS
In order to collect preliminary data, we started with
an assessment of patient’s urological history, using
validated questionnaires applied at Unifesp. The pa-
tients’ selection and inclusion criteria included over-
active bladder and concomitant use of anticholiner-
gics.
We then performed the Babinski test in the case of
patients with hyposensitivity and hyperreflexia (a pre-
requisite for patients to remain in the study was the
presence of a neurological dysfunction such as multi-
ple sclerosis). We only started the Electrotherapy in
the case of patients with absence of the Babinski re-
flex in at least one lower limb.
The patients who were selected to participate in
the study were then submitted to a physio-therapeutic
protocol. The main procedure consisted in applying
in an electrical current to the posterior tibial nerve,
according to the following steps.
On the first evaluation day, we applied the phys-
iotherapy assessment protocol forms, which the pa-
tient completed in the Urogynecology laboratory at
Unifesp. We instructed the patient to complete a void-
ing diary for three days after treatment.
During the neurological assessment, a physical
examination showed no sensory deficit and bilateral
Babinski’s reflex. Given that no pathological reflex
existed, we positioned the electrodes in the path of
the posterior tibial nerve, to detect whether innerva-
tion was intact, by using electrical stimulation (we
used the TENS NEURODYN/FES portable device, by
Ibramed Ltda). This stimulation was based on a se-
quence of 200-millisecond pulses, with a frequency of
20 Hz, following the recommendation in (Amarenco
et al., 2003).
The treatment protocol consisted of 10 sessions,
twice a week and lasting 20 minutes each. We applied
the TENS current through two channels, using four
electrodes positioned transcutaneously and bilaterally
in the lower limb (2 electrodes per channel). For each
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
278

channel, one electrode was fixed to the posterior me-
dial malleolus and the other 10 cm above. The inten-
sity parameter due to hyposensitivity was measured
through the signal engine rhythmic inflections of the
hallux. A maximum intensity of 30 mA was applied,
for safety reasons. In the case of patients with nor-
mal sensibility, the current intensity was adjusted for
comfort.
6 RESULTS AND DISCUSSION
This paper presents the results of 21 case studies mon-
itored by descriptive assessments from the Unifesp
Physiotherapeutic Protocol in Urogynecology and the
voiding diary for three days. The patients were sub-
mitted to physiotherapeutic treatment with transcuta-
neous electrical stimulation in order to attenuate uro-
logical clinical complaints. This justifies the choice
of a treatment by elective electrostimulation separated
from other techniques such as kinesiotherapy. We ap-
plied both evaluation procedures before treatment and
after 10 TENS sessions.
According to (C.Kabay et al., 2009), when ap-
plied to people suffering from multiple sclerosis, the
technique noticeably decreased nocturia in 75% of pa-
tients. Marques reports decrease in nocturia with 38%
of symptoms relief (Marques, 2008). Another study
found improvement in nighttime urination in 21% of
cases (F. E. Govier et al., 2001). Tables 1 and 2 show
that before and after 10 sessions of electrical stimula-
tion of the posterior tibial nerve, there was a decrease
in the signs of enureses, as well as a reduction in the
need for urine-loss protection, and of symptoms asso-
ciated to the cholinergic syndrome.
We can explain the improvement of urinary ur-
gency conditions based on a study in which urody-
namic evaluation with electrical stimulation of the
posterior tibial nerve revealed that the maximum blad-
der capacity can increase together with a decrease of
involuntary detrusor contractions during standard cys-
tometry (Amarenco et al., 2003).
This research aimed at verifying the efficacy of the
electrical stimulation as applied to the posterior tib-
ial nerve in order to control the detrusor hyperactiv-
ity (while reducing the symptoms related to the non-
inhibitted detrusor contractions and the anticholiner-
gic syndrome).
We analyzed five conditions and symptoms before
and after applying the proposed electrical stimulation
protocol: i) number of enuresis cases per night; ii)
number of needed urine-protection devices (UPD) per
day; iii) dry mouth; iv) intestinal constipation; and
v) blurred vision. Tables 1 and 2 describe the obtained
results.
We then conducted a statistical analysis of the
measured conditions and symptoms. The null hy-
potheses refer to each condition or symptom being the
same or even worse after the stimulation sessions, as
compared to the values measured before them. The
preliminary normality tests we performed indicated
that the measured values do not follow a normal dis-
tribution, and therefore we used the non-parametric
Wilcoxon test in order to evaluate the main null hy-
potheses.
Table 3 shows the p-values obtained for the null-
hypothesis associated to each of the considered con-
dition or symptom. Note that the low values (p < 0.1)
indicate that the null hypothesis can be rejected. This
suggests that the applied electrical stimulation proto-
col significantly reduces the corresponding condition
or symptom.
Hence, the applied electrical stimulation can in-
hibit or reduce the adverse effects of the anticholin-
ergic drugs. This conservative and non-invasive tech-
nique was hence beneficial to the evaluated patients
suffering from detrusor hyperactivity.
These results are the beginning of an intense
planned study of the use of electrical stimulation of
the posterior tibial nerve in patients with bladder hy-
peractivity as a substitute for pharmacological ther-
apy based on solifenacin succinate and oxybutinin hy-
drochloride. By increasing the sample size and by
evaluating the patients for longer periods, we want to
further evaluate other impacts of the proposed proto-
col, including long-term effects.
7 CONCLUSION
Patients with hyperactive bladder that use anticholin-
ergic drugs may present the cholinergic syndrome,
characterized by dry mouth, intestinal constipation,
and blurred vision. These symptoms add to the prob-
lems caused by the non-inhibited detrusor contrac-
tions, including loss of urine and enuresis. By replac-
ing the pharmacological approach by the electrical
stimulation of the posterior tibial nerve, we observed
an improvement of the hyperactive bladder condition
(p < 0.01), without the inconvenience associated to
the cholinergic syndrome.
We also emphasize that the proposed approach is
noninvasive, does not cause pain, and is cheaper than
the continuous use of solifenacin succinate and oxy-
butynin hydrochloride. It therefore has an important
effect on the patients’ quality of life, in terms of social
and economical aspects.
Use of Electrical Stimulation of the Posterior Tibial Nerve in Patients with Bladder Hyperactivity as a Substitute for Pharmacological
Therapy based on Solifenacin Succinate and Oxybutynin Hyloridrate
279

Table 1: Cases of enuresis per night and number of used urine-loss protection per day for all male and female participants, as
measured before and after the electrical stimulation (ES) sessions. We interrupted the pharmacological treatment (medication
A or B) in all cases, after the ES started.
Patient ID Genre Age NIC
Medication Before Electrical Stimulation After Electrical Stimulation
type Enuresis/night ULP/day Enuresis/night ULP/day
1 Female 46 NO A 3 1 0 0
2 Female 55 YES A 4 3 3 0
3 Male 22 YES A 6 0 0 0
4 Female 67 YES A 3 4 0 1
5 Male 25 YES A 3 3 0 1
6 Female 76 CE A 3 6 1 1
7 Female 56 YES B 2 5 0 0
8 Male 64 CE B 2 6 0 1
9 Male 79 YES B 3 3 0 0
10 Female 50 CE A 1 0 0 0
11 Female 67 YES A 4 0 0 0
12 Male 72 YES – 3 8 1 3
13 Male 67 YES A 0 3 0 0
14 Male 63 YES A 1 4 0 1
15 Female 43 YES B 2 2 0 0
16 Male 70 CE B 7 1 2 0
17 Female 41 YES B 0 0 0 0
18 Female 57 YES – 3 2 0 0
19 Female 52 YES B 1 0 0 0
20 Female 41 YES A 0 0 0 0
21 Male 89 YES A 4 3 1 0
Table 2: Symptoms of the cholinergic syndrome.
Patient ID
Before Electrical stimulation After Electrical Stimulation
Dry mouth Intestinal constipation Blurred Vision Dry mouth Intestinal constipation Blurred Vision
1 NO YES NO NO NO NO
2 NO YES NO NO NO NO
3 YES YES NO NO YES NO
4 YES NO NO NO NO NO
5 YES YES NO NO NO NO
6 YES NO YES NO NO NO
7 NO YES NO NO NO NO
8 YES NO NO NO NO NO
9 YES NO NO NO NO NO
10 YES NO NO NO NO NO
11 YES YES NO NO YES NO
12 NO NO NO NO NO NO
13 YES YES NO NO NO NO
14 YES YES YES NO YES NO
15 NO YES NO NO NO NO
16 YES NO NO NO NO NO
17 YES YES YES NO NO NO
18 NO YES NO NO NO NO
19 YES NO NO NO NO NO
20 YES NO NO NO NO NO
21 YES YES YES NO NO NO
ACKNOWLEDGEMENTS
The authors thank Doctor Denise Sistorelli Diniz, a
neurologist at the Hospital das Cl
´
ınicas, at the Fed-
eral University of Goi
´
as, for her assistance and use-
ful comments and suggestions. We also thank the Al-
fredo Nasser School (Unifan).
BIODEVICES 2016 - 9th International Conference on Biomedical Electronics and Devices
280

Table 3: Results, in terms of p-values, of the statistical tests
regarding the null hypothesis that each symptom or condi-
tion after the electrical stimulation (EE) is equal or worse
than before the EE.
Condition or symptom p-value
Enuresis 7.9 · 10
−6
Need for urine-loss protection 2.9 · 10
−4
Dry mouth
∗
9.8 · 10
−7
Intestinal constipation
∗
0.002
Blurred vision
∗
0.021
∗
Symptoms associated to the cholinergic syndrome.
REFERENCES
Amarenco, G., S., S., et al. (2003). Urodynamic effect of
acute transcutaneous posterior tibial nerve stimulation
in overactive bladder. J Urol, 169(6):2210–5.
Appell, R. A. (2003). Overactive bladder in special patient
populations. Rev Urol, 5 Suppl 8:S37–41.
Azevedo, M. A. J., Maria, M. L. S., and Soler, L. M. A.
(1990). Promovendo o auto-cuidado: treinamento
e assist
ˆ
encia de enfermagem a pacientes portadores
de bexiga neurog
ˆ
enica. Revista Brasileira de Enfer-
magem, 43:52–57.
Chapple, C. R., Yamanishi, T., et al. (2002). Muscarinic
receptor subtypes and management of the overactive
bladder. Urology, 60(5 Suppl 1):82–88.
C.Kabay, S., Yucel, M., et al. (2009). The clinical and uro-
dynamic results of a 3-month percutaneous posterior
tibial nerve stimulation treatment in patients with mul-
tiple sclerosis-related neurogenic bladder dysfunction.
Neurourol Urodyn, 28(8):964–8.
Coelho, M. M. (2009). Avaliac¸
˜
ao urodin
ˆ
amica na esclerose
m
´
ultipla. Acta Urologica, 26(3).
Coyne, K. S., Zhou, Z., et al. (2003). The impact on health-
related quality of life of stress, urge and mixed urinary
incontinence. BJU Int, 92(7):731–5.
Diokno, A. and Ingber, M. (2006). Oxybutynin in detrusor
overactivity. Urol Clin North Am, 33(4):439–45, vii.
F. E. Govier, F. E., Litwiller, S., et al. (2001). Percutaneous
afferent neuromodulation for the refractory overac-
tive bladder: results of a multicenter study. J Urol,
165(4):1193–8.
Fischer-Sgrott, F. O., Manffra, E. F., et al. (2009).
Qualidade de vida de mulheres com bexiga hiperativa
refrat
´
aria tratadas com estimulac¸
˜
ao el
´
etrica do nervo
tibial posterior. Revista Brasileira de Fisioterapia,
13:480–6.
Guidi, H. G. C., Silveira, S. R. B., et al. (2005). Reabilitac¸
˜
ao
do assoalho p
´
elvico nas disfunc¸
˜
oes urin
´
arias e anorre-
tais. Segmentofarma, pages 57–66.
Jones III, H. W., Wentz, A. C., and Burnett, L. S.
(1988). Novak – Tratado de ginecologia, relaxamen-
tos, distopias, f
´
ıstulas e incontin
ˆ
encia. Guanabada-
Koogan, Rio de Janeiro, Brazil.
Maciel, L. C. and Souto, S. (2009). Estimulac¸
˜
ao
do Nervo Tibial Posterior (PTNS) no Tratamento
da Bexiga Hiperativa (chapter 20), pages 223–7.
Unicamp, S
˜
ao Paulo, Brazil. In: Urofisioterapia:
Aplicac¸
˜
oes Cl
´
ınicas das T
´
ecnicas Fisioterap
ˆ
euticas
nas Disfunc¸
˜
oes Miccionais e do Assoalho P
´
elvico.
Marques, A. A. (2008). A eletroestimulac¸
˜
ao do nervo
tibial posterior no tratamento da bexiga hiperativa.
Unicamp, Campinas, S
˜
ao Paulo, Brazil.
Miltrano, P. (2009). Fisiopatologia e classificac¸
˜
ao da in-
contin
ˆ
encia urin
´
aria, pages 29–37. Manole, Barueri.
In: Moreno: Fisioterapia em Uroginecologia.
Monteiro, E. S. et al. (2009). Queixas urin
´
arias em mulheres
com infarto cerebral. Revista Neuroci
ˆ
encias, 17:103–
7.
O’Conor, R. M., Johannesson, M., et al. (1998). Urge in-
continence. quality of life and patients’ valuation of
symptom reduction. Pharmacoeconomics, 14(5):531–
9.
Oliveira, K. A. C. et al. (2007). T
´
ecnicas fisioterap
ˆ
euticas
no tratamento e prevenc¸c¸ao da incontin
ˆ
encia urin
´
aria
de esforc¸o na mulher. Revista Eletrnica F@pci
ˆ
encia,
1:31–49.
Stephenson, R. G. and O’Conor, L. J. (2004). Fisioterapia
Aplicada
`
a Ginecologia e Obstetr
´
ıcia. Manole, S
˜
ao
Paulo, Brazil, 2
nd
edition.
van der Pal, F., van Balken, M. R., et al. (2006). Correla-
tion between quality of life and voiding variables in
patients treated with percutaneous tibial nerve stimu-
lation. BJU Int, 97(1):113–6.
Use of Electrical Stimulation of the Posterior Tibial Nerve in Patients with Bladder Hyperactivity as a Substitute for Pharmacological
Therapy based on Solifenacin Succinate and Oxybutynin Hyloridrate
281
