
Neural Model for the Influence of Shading on the Multistability of the
Perception of Body Motion
Leonid Fedorov
1,2
, Joris Vangeneugden
3
and Martin Giese
1,2
1
Dept. of Cognitive Neurology, CIN, HIH, University of Tuebingen, Tuebingen, Germany
2
IMPRS for Cognitive and Systems Neuroscience, Tuebingen, Germany
3
School of Mental Health and Neuroscience, Maastricht, The Netherlands
Keywords:
Action Recognition, Multistable Perception, Biological Motion, Neural Fields, Shading.
Abstract:
Body motion perception from impoverished stimuli shows interesting dynamic properties, such as multista-
bility and spontaneous perceptual switching. Psychophysical experiments show that such multistability dis-
appears when the stimulus includes also shading cues along the body surface. Classical neural models for
body motion perception have not addressed perceptual multistability. We present an extension of a classical
neurodynamic model for biological and body motion perception that accounts for perceptual switching, and
its dependence on shading cues on the body surface. We demonstrate that a set of psychophysical observa-
tions can be accounted for in a unifying manner by a hierarchical neural model for body motion processing
that includes an additional shading pathway, which processes luminance gradients within the individual body
segments. The goal of our model is to explain psychophysics and neural mechanism in the brain.
1 INTRODUCTION
The perception of body motion from image sequences
requires the dynamic integration of complex spatio-
temporal visual patterns. This important visual func-
tion is accomplished by processing within a hierar-
chy of cortical areas along the visual pathway. Psy-
chophysical studies suggest depth cues are important
for biological motion perception (Jackson and Blake,
2010). In absence of such depth information, e.g. in
point-light walkers, body motion perception can be-
come multistable (Vanrie and Verfaillie, 2004). Then
the same stimulus can be perceived as alternating ran-
domly between two interpretations that correspond to
two different walking directions (Vanrie and Verfail-
lie, 2006). Multistabile phenomena has been also in-
vestigated in the context of static ambiguous figures
and binocular rivalry (Leopold and Logothetis, 1999),
(Blake and Logothesis, 2001), as well as in structure
from motion (Andersen and Bradley, 1998). An ex-
ample of the body motion stimulus that produces such
multistability is shown in Fig. 1A (panel SILHOU-
ETTE). For this stimulus, an articulating silhouette
without intrinsic shading cues, observers perceive the
walker alternately walking obliquely into or out of the
image plane. The two reported percepts correspond to
the unambiguous walking directions indicated in pan-
els TOWARDS and AWAY. The figure illustrates also
that this perceptual ambiguity disappears when shad-
ing gradients are added to the surface of the walker,
which provide information about the surface orienta-
tion of the body segments and occlusions.
Existing physiologically-inspired neural models
for the processing of body motion and goal-directed
actions (e.g. (Giese and Poggio, 2003), (Lange
and Lappe, 2006), (Escobar and Kornprobst, 2008),
(Jhuang et al., 2007), (Fleischer et al., 2013) and
(Layher et al., 2014)) do not reproduce such multi-
stability, or at least never have investigated this phe-
nomenon. Computer vision and deep learning archi-
tectures for body motion recognition do not address
perceptual multistability. Thus, the study of such phe-
nomena is important for neuroscience, even if such
multistability is often unwanted in technical action
recognition systems.
In the context of low-level vision, perceptual
multi-stability and the underlying neural dynamics
have been extensively studied e.g. in the context of
binocular rivalry (see e.g. (Wilson, 2003)), visual
motion integration (Rankin et al., 2014), or as gen-
eral property of attractor neural networks (Pastukhov
et al., 2013).
The goal of this paper is to extend existing
physiologically-inspired neural models (not computer
Fedorov, L., Vangeneugden, J. and Giese, M.
Neural Model for the Influence of Shading on the Multistability of the Perception of Body Motion.
DOI: 10.5220/0006054000690076
In Proceedings of the 8th International Joint Conference on Computational Intelligence (IJCCI 2016) - Volume 3: NCTA, pages 69-76
ISBN: 978-989-758-201-1
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
69

vision algorithms) in a way that accounts for multi-
stability in action perception, where we use as ex-
ample an established model that has been shown to
account jointly for many experimental results in this
area (Giese and Poggio, 2003). We extend it in two
ways: 1) by introduction of a multi-dimensional neu-
ral field that accounts for multi-stable behavior by lat-
eral interactions between shape-selective neurons; 2)
by addition of a new pathway that realizes robust pro-
cessing of intrinsic luminance gradients along the sur-
face of the body segments.
The paper is structured as follows: after dis-
cussing related work in the following section, we de-
scribe the developed architecture in section 3. In sec-
tion 4 we show simulation results, illustrating that
the model provides a unifying account for several key
psychophysical results, followed by a brief discussion
in section 5.
2 RELATED THEORETICAL
WORK
Body motion recognition has been a core topic in
computer vision and many technical neural architec-
tures for this purpose have been proposed (Edwards
et al., 2016), (Nguyen et al., 2016), (Ziaeefard and
Bergevin, 2015), (Lee et al., 2014). The goal of that
work is typically a maximization of recognition per-
formance, not a reproduction of perceptual dynamics
of humans. This paper does not contribute to com-
puter vision or machine learning and is entirely fo-
cused on modeling of the brain.
We follow the approach in physiologically-
plausible models of body motion perception, such as
(Giese and Poggio, 2003), (Lange and Lappe, 2006),
(Escobar and Kornprobst, 2008), (Fleischer et al.,
2013), (Layher et al., 2014), while other biological
models in this area (e.g. (Thurman and Lu, 2014)
(Thurman and Lu, 2016)) account for experimental
data without direct relationship to neural mechanisms.
Diverse approaches (see (Tyler, 2011)) have been
proposed for the analysis of shape from shading, but
typically not related to the processing of body mo-
tion. Perceptual dynamics and perceptual switching
have been extensively studied in the context of low-
level vision (reviews see e.g. (Leopold and Logo-
thetis, 1999), (Sterzer et al., 2009), (Pastukhov et al.,
2013)). Multistability in the processing of non-rigid
motion has been rarely studied in neural modeling.
While hierarchical technical algorithms in com-
puter vision typically focus on the problem how the
body motion patterns (e.g. the direction of body
movement) might be distinguished, our model tries
to unify this account with a reproduction of the dy-
namics of perceptual organization in humans which
emerges specifically for the SILHOUETTE stimulus,
where for the same stimulus two alternating percepts
emerge. This problem is typically not addressed in
technical recognition systems, and to our knowledge
no account for this phenomenon has been given in
biologically-inspired neural models for motion recog-
nition.
3 MODEL ARCHITECTURE
Our model builds on a previous neural model (Giese
and Poggio, 2003), which has been shown to provide
a unifying account for a variety of experimentally
observed phenomena in body motion perception in-
cluding physiological, psychophysical and fMRI data.
The original model included a motion and a form
pathway, processing shape and optic flow features.
The pathways consist of a hierarchy of feature de-
tectors that mimic properties of real cortical neurons.
For the implementation in this paper we used only the
form-pathway and extended it by a multi-dimensional
neural field, and a new pathway for the processing of
intrinsic luminance gradients. An extension by in-
clusion of an additional motion pathway is straight-
forward, and will be part of future work.
3.1 Silhouette Pathway
The backbone of our model is a ’silhouette pathway’
(Fig. 1B) that is identical to the the form pathway of
the classical model (Giese and Poggio, 2003). Due
to space limitations, we sketch here only some basics
about this pathway and refer to the original publica-
tion (Giese and Poggio, 2003) with respect to details.
In brief, the form pathway consists of a hierarchy of
layers that process form features of increasing com-
plexity along the hierarchy. More complex features
are formed by combination of the features from pre-
vious layers. Levels that increase feature complex-
ity are interleaved by layers that increase position and
scale invariance by MAX pooling. The highest level
of this shape processing hierarchy is formed by radial
basis function units (called ’snapshot neurons’) that
have been trained with the feature vectors that corre-
spond to keyframes from training movies showing the
recognized action. Each snapshot neuron responds se-
lectively to the body posture that corresponds to time
instance θ (within the gait cycle). In addition, con-
sistent with physiological data (Vangeneugden et al.,
2011), we assume these neurons are view-specific,
where the variable φ specifies the preferred view an-
NCTA 2016 - 8th International Conference on Neural Computation Theory and Applications
70
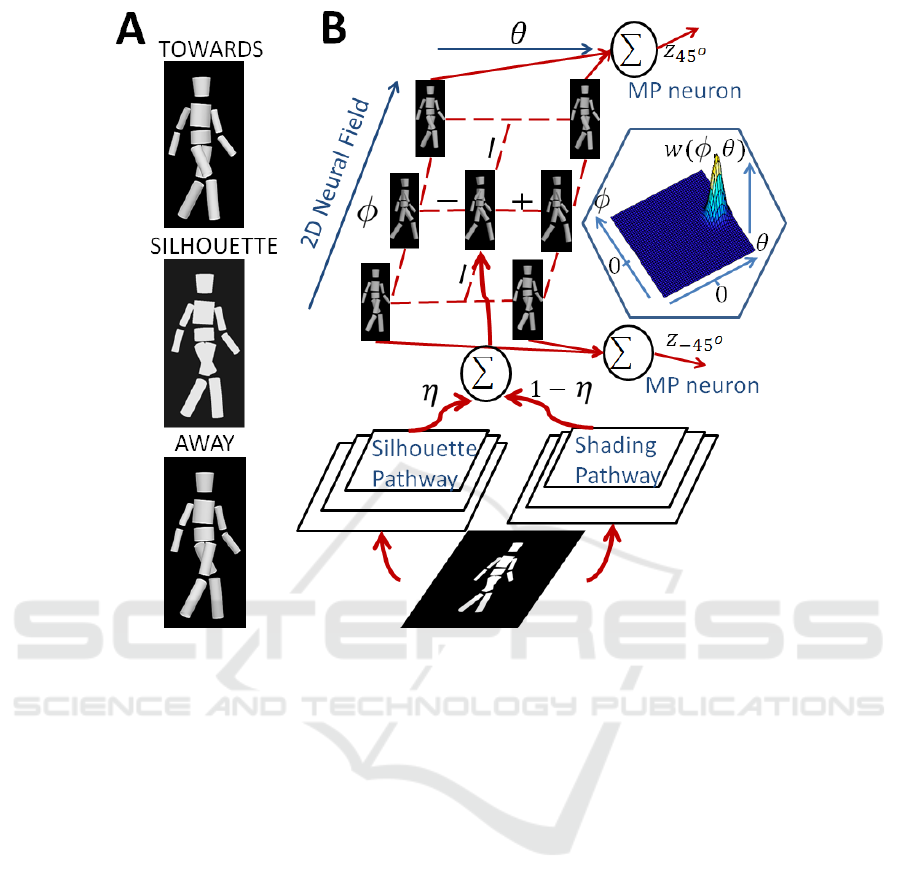
Figure 1: A. Snapshots from movies showing dynamic walker: TOWARDS shaded walker, walking direction 45 deg; SIL-
HOUETTE bistable silhouette walker and AWAY shaded walker, walking direction -45 deg. B. Model architecture. Stimulus
is analyzed by Silhouette and Shading pathways. Their outputs are linearly combined and mapped linearly onto the input of
a 2D dynamic neural field that consists of laterally coupled snapshot neurons. Inset shows the lateral interaction kernel of the
field. The field activity is read out by Motion Pattern (MP) neurons that encode the perceived walking directions ±45 deg.
gle of the neuron. (We assume that the side view of
a walker walking to the right in the image plane de-
fines the view direction φ = 0). Very similar architec-
tures underlie many other classical and modern neu-
ral and deep models for object recognition, where the
popular deep architectures are typically trained with
much more data and often include many more lay-
ers. Since the goal of this paper is to model the per-
ceptual dynamics, and not to maximize recognition
rate, we used this simple hierachical model, where ex-
tension with modern deep architectures as front-end
seem straight-forward.
3.2 Shading Pathway
The described simple form pathway recognizes body
shape on backgrounds with sufficient contrast. How-
ever, it turned out that with small amounts of training
data it is difficult to accomplish with this architecture
a robust recognition of the silhouette shape together
with a high sensitivity for the luminance shading gra-
dients that disambiguate the depth structure. As one
possible solution to this problem we implemented a
second pathway that is specialized for the processing
of intrinsic shading gradients using physiologically-
plausible operations (Fig. 1B). We do not claim this
is the only possible solution, but it is one that works
with small amounts of training data.
The first level of this new pathway overlaps with
the first hierarchy level of the silhouette pathway,
described above. It consists of Gabor filters that
are selective for local orientation features at differ-
ent positions, and for different spatial scales. Let
G
e,u
(x,y, α, σ) signify the output signal of the even
(e) or uneven (u) Gabor filter with preferred posi-
tion (x,y), preferred orientation α (we used 8 orienta-
tions), and scale σ (we used 1 scale for the given small
stimuli set). The activations of the uneven Gabor fil-
ters provide a population code for the local luminance
gradients.
By pooling of the responses of the Gabor fil-
ters with the same preferred position over all orien-
tations we obtain position-specific detectors for con-
tours with the output signals:
Neural Model for the Influence of Shading on the Multistability of the Perception of Body Motion
71

C(x, y) = max
{e,u},α,σ
|G
e,u
(x,y, α, σ)|. (1)
This output signal was used to suppress the re-
sponses of the uneven Gabor filters along the external
contour of the body, exploiting multiplicative gating.
The outer contour of the body typically creates strong
local contrast that dominates the detector responses,
so that the weak intrinsic gradients that signal the 3D
structure cannot be reliably estimated from the neural
responses. A population vector signaling the intrinsic
luminescence gradients is given by the gated signal:
L(x,y, α, σ) = [G
u
(x,y, α, σ) ·H(λ
1
−C(x,y))]
+
. (2)
Here λ
1
is a positive constant, and the function
H(x) is the Heaviside function, thus H(x) = 1 for x >
0 and H(x) = 0 otherwise.
The next level of the shading pathway consists
of (partially) position-invariant detectors for local lu-
minance gradients. Their responses are computed
by pooling of the gated responses of gradient detec-
tors for the same preferred gradient direction α over
all positions and scales in a quadratic neighborhood
U(x
0
,y
0
) of the point (x
0
,y
0
) using a maximum opera-
tion, providing the output signals:
D(x,y, α) = max
(x,y)∈U(x
0
,y
0
),σ
L(x,y, α, σ). (3)
These position-invariant detectors were defined
for substantially less spatial positions, resulting in
a strong spatial down-sampling (6,480,000 position-
and scale-specific detectors vs. 648 position-invariant
detector units).
In order to make recognition robust against fluctu-
ating weak features, we selected the strongest features
that provide input to the radial basis function units.
We selected those features that showed the maxi-
mum variance over the training data (where clearly
much more sophisticated feature selections are avail-
able that might lead to better results). We computed
the circular variance of the detectors at position (x,y),
exploiting the (complex) circular mean:
The (complex) circular mean of these responses is
given by:
m(x,y) = (1/K)
K
∑
k=1
∑
α
D
(k)
(x,y, α) exp(iα), (4)
where K is the number of training patterns. A cir-
cular variance measure is then given by the formula:
V (x, y) =
K
∑
k=1
∑
α
D
(k)
(x,y, α) exp(iα) − m(x,y)
.
(5)
We selected the direction-specific responses
D(x,y, α) that fulfilled the relationship:
V (x, y) > λ
2
, (6)
where λ
2
> 0 is a threshold parameter. In total 9
out of 81 feature vectors were selected according to
this criterion.
The next level of the shading pathway is formed
by Gaussian radial basis functions, whose centers
were trained with the feature vectors p
l
(including
only the selected features) that were generated by in-
dividual keyframes from the training movies. For the
results shown here, the shading pathway was trained
with movies of fully shaded walkers, shown with view
directions −45 deg and 45 deg. In other implementa-
tions, we have realized such models with a continuum
of different views (Fleischer et al., 2013).
The RBF network returns an 50-dimensional out-
put vector R
SH
(t) for each keyframe at time t, where
the components of this vector are given by:
R
l
SH
(t) = exp(−λ
3
||p(t) − p
l
||
2
), (7)
where p(t) is the feature vector for the actual input
frame, and where the components correspond to the
different keyframes and associated training views.
In order to link the shape recognition pathway to
dynamic neurons that reproduce the perceptual dy-
namics, the outputs of the RBF units were mapped
linearly onto a discretely sampled two-dimensional
input activity distribution s
SH
(θ,φ;t) that provides in-
put to the neural field that is described below. Signi-
fying by s
SH
(t) the appropriately reordered sampling
points, the linear mapping was given by the equation:
s
SH
(t) = W(t)R
SH
(t). (8)
The weight matrices W(s) were learned by ridge
regression from a training set that consisted of pairs
of vectors R
SH
(t) for each training keyframe, and a
corresponding vector s
SH
(t) that was computed from
an idealized two-dimensional input activity distribu-
tion s
SH
(θ,φ;t). The idealized activity distribution
was given by a Gaussian peak that was centered at
the keyframe number θ and the corresponding view
φ of the walker (s.b.). A similar input distribution
s
SL
(θ,φ;t) was computed by a corresponding linear
mapping in the silhouette pathway. The total input
distribution of the neural field was then computed
by ’cue fusion’, modeled by a convex combination
of two input distribution functions according to the
equation:
s(θ,φ;t) = ηs
SL
(θ,φ;t) + (1 − η)s
SH
(θ,φ;t), (9)
with 0 ≤ η ≤ 1. Choosing η = 1 one can eliminate
the influence of the shading pathway.
NCTA 2016 - 8th International Conference on Neural Computation Theory and Applications
72

3.3 Dynamic Neural Field of Snapshot
Neurons
The core of our model is a dynamic recognition layer
that is implemented as a two-dimensional neural field
of Amari type (Amari, 1977), which consists of body
shape-selective neurons that are laterally connected
(Fig. 1B). Consistent with physiological data (Van-
geneugden et al., 2011), we assume that such neurons
encode body shapes that emerge during actions in a
view-specific manner. In the spatial continuum limit,
we can describe the activity of neurons encoding the
body shape that corresponds to the normalized time θ
(0 ≤ θ ≤ 2π) during the gait cycle and the view angle
φ by the function u(φ,θ,t). The network dynamics is
given by the equation (? signifying a spatial convolu-
tion):
τ
u
d
dt
u(φ,θ,t) = −u(φ,θ,t)+ w(φ,θ) ? H(u(φ,θ,t))
+s(φ,θ,t) − h + ξ(φ,θ,t) − c
a
a(φ,θ,t).
(10)
The input signal s was described above. For the
trained stimulus movies it corresponds to an activity
maximum that moves in θ-direction along the field.
The lateral connectivity is specified by the interac-
tion kernel w(φ,θ) (whose shape is indicated by the
inset in Fig. 1B). It stabilizes a traveling pulse so-
lution in θ-direction and realizes a winner-takes-all
competition in the φ-direction. As consequence, if
multiple views are consistent with the stimulus, one
view is selected by competition. The positive param-
eters τ
u
and h define the time scale and the resting
potential of the field. The variable ξ(φ, θ,t) defines a
Gaussian noise process whose statistics was coarsely
adapted to the noise correlations from cortical data
(Giese, 2014). These fluctuations essentially drive the
perceptual switching in the model. Since action per-
ception shows adaptive properties, such as high-level
after-effects and fMRI adaptation, we also included
a neural adaptation process in the model, which re-
duces the activity of snapshot neurons after extended
firing. The corresponding adaptation variable follows
the dynamical equation:
τ
a
d
dt
a(φ,θ,t) = −a(φ,θ,t)+ H(u(φ,θ,t)). (11)
The positive constant c
a
determines the strength of
adaptation(τ
α
is the time constant). The parameters of
this adaptation dynamics were fitted to experimental
data (Giese, 2014).
The activity of the neurons in the neural field was
read out by motion pattern (MP) neurons, which sig-
nal the walking directions perceived in this case as
AWAY from and TOWARDS the observer. These
neurons compute the maximum of the neural field ac-
tivity function u(φ,θ,t) over the domains φ > 0 and
φ < 0 in the (φ,θ) space, producing the output signals
z
45
and z
−45
.
4 SIMULATION RESULTS
Testing the model after training with a non-shaded
walker as illustrated in Fig. 1A and 1B, the output
of the shading pathway remained silent because of
the absence of intrinsic luminance gradients in this
stimulus. The silhouette pathway was activated in an
ambiguous way by this stimulus because the stimulus
is consistent with walking in the directions ±45 deg
relative to the image plane. Consistent with simula-
tions described in (Giese, 2014), this stimulus leads
to a bistable solution of the neural field that alternates
between two traveling pulse solutions that encode the
spontaneous perceptual switching of a traveling pulse
between the view angles φ = ±45 deg (perception
of TOWARDS or AWAY from the observer). In this
case, the probabilities of the two percepts are almost
identical (Fig. 2B). More detailed simulations show
that the model coarsely reproduces also the switching
time statistics of human perception, comparing it with
experimental data (not yet published (Vangeneugden
et al., 2012)). Fig. 2G shows a histogram of the per-
cept times for the model, and Fig. 2H the percept
times estimated in the psychophysical experiment.
For shaded stimuli (see Figs. 1A TOWARDS and
AWAY), when both pathways are included (η = 0.5),
the model successfully disambiguates the walking di-
rection: For the AWAY stimulus (direction -45 deg)
the output neuron for AWAY remains always acti-
vated while the output neuron for TOWARDS re-
mains silent. If an TOWARDS stimulus is shown (di-
rection 45 deg) the situation is reverse and the TO-
WARDS output neuron is always active (Fig.2 C and
D).
If however the shading pathway is deactivated
(η = 0) again perceptual switching occurs, since the
output of the silhouette pathway is ambiguous, result-
ing in equal percept probabilities for either direction.
The silhouette pathway is not sufficiently sensitive to
disambiguate the stimulus robustly based on the avail-
able luminance gradients intrinsic to the body seg-
ments (Fig.2 E-F). This demonstrates the necessity of
the shading pathway in the chosen architecture for the
disambiguation of the percept.
The model makes several verifiable experimental
predictions in relation to the time course of the adap-
tation process.
Neural Model for the Influence of Shading on the Multistability of the Perception of Body Motion
73

Figure 2: A. Time courses of the activity of motion pattern neurons for depth-ambiguous walker stimulus. B-F. Percept
probability of the motion pattern neurons for the percepts TOWARDS and AWAY for (B) depth-ambiguous walker for model
with both pathways ; (C) shaded −45 deg (AWAY) walker for model with both pathways; (D) same for shaded 45 deg
(TOWARDS) walker; (E) shaded 45 deg (TOWARDS) walker for model without shading pathway; (F) same for shaded 45
deg (TOWARDS) walker; G-H. Histogram of percept times (PT) from experimental data (Vangeneugden et al., 2012) and from
the model. I. Paradigm for testing after-effects in action perception which is compatible with our model. After presentation
of an unambiguous adaptor stimulus (AWAY or TOWARDS), and a fixed Inter-stimulus Interval, an ambiguous test stumulus
(SILHOUETTE) is presented. J. Probability that test stimulus is perceived as walking in the adaptor direction as a function of
the duration of the adaptor.
NCTA 2016 - 8th International Conference on Neural Computation Theory and Applications
74

An example is illustrated in Fig. 2I that shows
a diagram of a typical adaptation experiment to
demonstrate after-effects in action perception. First,
an unambiguous adaptation stimulus (TOWARDS or
AWAY) is presented to participants, where the dura-
tion of the adaptor (2, 6, 10, 14, 18 or 22 gait cycles)
was varied over different blocks of the experiment.
After this stimulus (and a fixed Inter-stimulus Inter-
val of 2.8 s) an ambiguous test stimulus (SILHOU-
ETTE) is presented for 3 gait cycles, asking for the
perceived walking direction. The predicted results for
such an experiment (from 20 repeated simulations)
are presented in Fig. 2J, which shows the probabil-
ities of the percept for the ambiguous test stimulus
(which was identical in all cases). With increasing
the duration of the adaptor stimulus the probability
that participants perceive the test stimulus as walking
in the same direction as the adaptor decreases. A sig-
nificant decrease of the percept probability (from 0.5
without adaptator presentation) is already perceived
for the shortest adaptor duration of 2 gait cycles, and
we observed a further decrease with longer adaptor
durations (where 1 gait cycle corresponds to 1.4 sec-
onds of stimulus duration). This behavior is consis-
tent with after-effects, as investigated previously for
many modalities (motion, lightness, etc) in low-level
vision. Such after-effects for action perception with a
similar time course have been shown for other types
of action stimuli in the literature (see (Barraclough
and Jellema, 2011), (de la Rosa et al., 2014)), and we
are presently running psychophysical experiments to
verify this prediction of the model in detail.
A further set of experiments that we are presently
running, and for which the model provides quantita-
tive predictions, investigates the interdependence of
the stability of action percepts and the switching times
between the different percepts (which depend on the
mean-first passage times of the corresponding attrac-
tors). This extends studies that have been made for
muti-stability of low-level motion perception (Hock
et al., 1993) to the domain of action perception.
5 CONCLUSIONS
To our knowledge, we have described the first
biologically-inspired neural model that accounts si-
multaneously for the following properties of body
motion perception: (i) perceptual multi-stability and
switching, (ii) switching time statistics and (iii) the in-
fluence of shading information on the perceptual dy-
namics. We showed that the model reproduces the
psychophysically observed phenomenology and dis-
tributions of the percept times. Since the model is
based on learned templates, these results would trans-
fer trivially to other action patterns with the similar
form of bistability in the view domain.
It is important to stress that the goal of this pa-
per was the modeling of the perceptual dynamics, and
neither the proposal of novel deep shape or action
recognition architecture, nor the claim that the pro-
posed two-pathway architecture is significantly bet-
ter for shape recognition. Testing this claim would
require additional experiments with larger data sets,
and was not the focus of this paper. Also it remains to
be shown whether any of the popular recurrent deep
architectures reproduce the details of the human per-
ceptual dynamics.
Future work will have to extend the model for
more stimuli and include more accurate fits of exper-
imental data.
ACKNOWLEDGEMENTS
The first author thanks Tjeerd Dijkstra for his insight-
ful commentary on the analysis of the Amari field
behavior. Funded by: BMBF, FKZ: 01GQ1002A,
ABC PITN-GA-011-290011, CogIMon H2020 ICT-
644727; HBP FP7-604102; Koroibot FP7-611909,
DFG GZ: KA 1258/15-1.
REFERENCES
Amari, S. (1977). Dynamics of pattern formation in lateral
inhibition type neural fields. Biological Cybernetics.
Andersen, R. and Bradley, D. (1998). Perception of three-
dimensional structure from motion. Trends in Cogni-
tive Sciences.
Barraclough, N. and Jellema, T. (2011). Visual aftereffects
for walking actions reveal underlying neural mecha-
nisms for action recognition. Psychological Science.
Blake, R. and Logothesis, N. (2001). Visual competition.
Nature Review Neuroscience.
de la Rosa, S., Streuber, S., Giese, M., Buelthoff, H., and
Curio, C. (2014). Putting actions in context: Visual
action adaptation aftereffects are modulated by social
contexts. PLOS One.
Edwards, M., Deng, J., and Xie, X. (2016). From pose to
activity. In Computer Vision and Image Understand-
ing. Elsevier Science Inc.
Escobar, M. and Kornprobst, P. (2008). Action recogni-
tion with a bioinspired feedforward motion processing
model: the richness of center-surround interactions.
In ECCV’08. 10th European Conference on Computer
Vision. Springer Berlin Heidelberg.
Fleischer, F., Caggiano, V., Thier, P., and Giese, M. (2013).
Physiologically inspired model for the visual recog-
Neural Model for the Influence of Shading on the Multistability of the Perception of Body Motion
75

nition of transitive hand actions. Journal of Neuro-
science.
Giese, M. (2014). Skeleton model for the neurodynamics
of visual action representations. In Artificial Neu-
ral Networks and Machine Learning ICANN 2014,
Lecture Notes in Computer Science. Springer Interna-
tional Publishing.
Giese, M. and Poggio, T. (2003). Neural mechanisms for
the recognition of biological movements and action.
Nature Reviews Neuroscience.
Hock, H., Kelso, J., and Schoener, G. (1993). Bistability
and hysteresis in the perceptual organization of appar-
ent motion. Journal of Experimental Psychology: Hu-
man Perception and Performance.
Jackson, S. and Blake, R. (2010). Neural integration of in-
formation specifying human structure from form, mo-
tion, and depth. Journal of Neuroscience.
Jhuang, H., Serre, T., Wolf, L., and Poggio, T. (2007). A bi-
ologically inspired system for action recognition. In
2007 IEEE 11th International Conference on Com-
puter Vision. IEEE.
Lange, J. and Lappe, M. (2006). A model for biological
motion perception from configural form cues. Journal
of Neuroscience.
Layher, G., Giese, M., and Neumann, H. (2014). Learning
representations of animated motion sequencesa neu-
ral model. In Topics in Cognitive Science. Topics in
Cognitive Science.
Lee, T., Belkhatir, M., and Sanei, S. (2014). A compre-
hensive review of past and present vision-based tech-
niques for gait recognition. In Multimedia Tools and
Applications. Kluwer Academic Publishers.
Leopold, D. and Logothetis, N. (1999). Multistable phe-
nomena: changing views in perception. Trends in
Cognitive Science.
Nguyen, D., Li, W., and Ogunbona, P. (2016). Human de-
tection from images and videos. In Pattern Recogni-
tion. Elsevier Science Inc.
Pastukhov, A., Garca-Rodrguez, P., Haenicke, J., Guilla-
mon, A., Deco, G., and Braun, J. (2013). Multi-stable
perception balances stability and sensitivity. Frontiers
in Computational Neuroscience.
Rankin, J., Meso, A., Masson, G. S., Faugeras, O., and Ko-
rnprobst, P. (2014). Bifurcation study of a neural field
competition model with an application to perceptual
switching in motion integration. Journal of Computa-
tional Neuroscience.
Sterzer, P., Kleinschmidt, A., and Rees, G. (2009). The
neural bases of multistable perception. Trends in Cog-
nitive Science.
Thurman, S. and Lu, H. (2014). Bayesian integration of po-
sition and orientation cues in perception of biological
and non-biological forms. Frontiers in Human Neuro-
science.
Thurman, S. and Lu, H. (2016). A comparison of form pro-
cessing involved in the perception of biological and
nonbiological movements. Journal of Vision.
Tyler, C. (2011). Computer Vision: From Surfaces to 3D
Objects. Chapman & Hall/CRC, London, 1st edition.
Vangeneugden, J., de Maziere, P., van Hulle, M., Jaeggli, T.,
van Gool, L., and Vogels, R. (2011). Distinct mecha-
nisms for coding of visual actions in macaque tempo-
ral cortex. Journal of Neuroscience.
Vangeneugden, J., van Ee, R., Verfaillie, K., Wagemans, J.,
and de Beeck, H. (2012). Activity in areas mt+ and
eba, but not psts, allow prediction of perceptual states
during ambiguous biological motion. In Society for
Neuroscience Meeting. Society for Neuroscience.
Vanrie, J. and Verfaillie, K. (2004). Perception of biological
motion: A stimulus set of human point-light actions.
Behavior Research Methods, Instruments, and Com-
puters.
Vanrie, J. and Verfaillie, K. (2006). Perceiving depth in
point-light actions. Perception and Psychophysics.
Wilson, H. (2003). Computational evidence for a rivalry
hierarchy in vision. Proceedings of the National
Academy of Sciences.
Ziaeefard, M. and Bergevin, R. (2015). Semantic human
activity recognition: A literature review. In Pattern
Recognition. Elsevier Science Inc.
NCTA 2016 - 8th International Conference on Neural Computation Theory and Applications
76
