
18
O Isotope Concentration Control
Valentin Sita
1
, Vlad Mureşan
1
, Mihail Abrudean
1
, Mihaela-Ligia Ungureşan
2
and Iulia Clitan
1
1
Automation Department, Technical University of Cluj-Napoca, Memorandumului street, no. 28, Cluj-Napoca, Romania
2
Department of Physics and Chemistry, Technical University of Cluj-Napoca, Memorandumului street, no. 28,
Cluj-Napoca, Romania
Keywords:
18
O Isotope, Concentration Control, Mathematical Model, Neural Network, Distributed Parameter Process,
Nonlinear System, Independent Variable.
Abstract: The paper presents a solution to control the
18
O isotope concentration at the output of a separation column.
The proposed mathematical model which describes the work of the separation column is a distributed
parameter one and it approximates with high accuracy the work of the real plant. The isotope separation
process is included in a complex control structure which generates high control performances. In order to
improve significantly the separation column productivity, an original solution for the efficient rejection of
the disturbances effects, is proposed. Also, a solution to determine the instantaneous value of the separation
column length constant is proposed, solution which opens the possibility to implement new control
strategies.
1 INTRODUCTION
The separation column for the
18
O isotope and the
equipment of the refluxing system necessary for its
work is presented in Fig. 1. In this context, the
18
O
isotope separation is made through isotopic
exchange in the system NO, NO
2
-H
2
O, HNO
3
(Abrudean, 1981). In Fig. 1, FSC is the Final
Separation Column, the term “Final” being used due
to the fact that this column works as the final
column of a separation cascade which contains two
separation columns. The main aim of this paper is to
control the FSC work in the case when it works
independently from the separation cascade. The
results of this research activity, consisting from the
proposed control strategy, will be further used in
order to find an appropriate method to control the
separation cascade work.
As it can be remarked in Fig. 1, FSC has the
height h = 10 m, being built from 5 sectors
(S1 – S5), each having the height h/5 = 2 m. The
column diameter is d = 14 mm. The separation of the
18
O isotope is based on the circulation in FSC, in
counter-current, of the nitric oxides (NO, NO
2
)
introduced in the lower part of the column and of the
nitric acid (HNO
3
) – solution introduced in the upper
part of the column. The input flow of nitric oxides is
notated with F
i
and the output flow of nitric acid is
notated to F
W
, representing the isotopic waste of the
separation process. F
o
represents the output flow of
the nitric oxides from FSC which is circulated to the
arc-cracking reactor ACR. At the ACR output, the
flow F
N
of both nitrogen (N
2
) and nitric oxides (NO,
NO
2
) results, with an increased concentration of
NO
2
. In the absorber A, the absorption of the nitric
oxides in water is made, resulting nitric acid solution
which supplies FSC (the flow of nitric acid solution
is notated with F
A
). The absorber A is supplied with
the water flow F
H2O
. The amount of nitrogen and
nitric oxide from the absorber output, under the flow
F
NN
(NO, N
2
) is sent to the catalytic reactor CR. In
CR, the water with which the absorber is supplied
results through the reaction of nitric oxide (NO) with
hydrogen (H
2
) (CR is supplied with the hydrogen
flow F
H
). Also, after the reaction from CR, the
excess of nitrogen and hydrogen results (the flow
F
NH
).
The product can be extracted from the top of
FSC under the form of nitric acid with an increase
concentration of
18
O isotope (HN
18
O
3
). The product
flow is notated with F
P
and the corresponding pipe is
figured with dashed line due to the fact that in this
paper, only the total reflux (F
P
= 0) working regime
is approached. The hachure used in Fig. 1 signifies
Sita, V., Mure¸san, V., Abrudean, M., Ungure¸san, M-L. and Clitan, I.
18
O Isotope Concentration Control.
DOI: 10.5220/0006877403690379
In Proceedings of the 15th International Conference on Informatics in Control, Automation and Robotics (ICINCO 2018) - Volume 1, pages 369-379
ISBN: 978-989-758-321-6
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
369

the fact that the corresponding elements contain
packing (steel packing in the case of FSC and in the
case of the reactors) and ceramic packing (Axente,
1984) in the case of the absorber. In order to
measure the concentration of the
18
O isotope, at the
separation column output, a mass spectrometer is
used.
Figure 1: The separation column for the
18
O isotope and
the equipment of the refluxing system.
2 ISOTOPE SEPARATION
PROCESS MODELLING
The
18
O isotope separation process is a distributed
parameter one (Colosi, 2013; Li, 2011) due to the
fact that during the separation exchange, the main
output signal (the
18
O isotope concentration)
depends on two independent variables: time (t) and
the position in FSC length (s). In order to highlight
the position in FSC length, the (0s) axis is defined
(in Fig. 1). The origin (0) of the (0s) axis represents
the centre of the Column Base Section (CBS). The
independent variable (s) has an increasing evolution
from the column base to the column top and its
maximum value can be obtained for s = h,
corresponding to the Column Top Section (CTS).
The main input signal in the process is considered
the input flow of nitric oxides F
i
(t) and, as it was
previously mentioned, the main output signal from
the process is considered the
18
O isotope
concentration notated with y(t,s). The mathematical
modelling is made in the hypothesis when FSC
works in total reflux regime (F
P
= 0). This regime is
used in the first part of the plant working with the
purpose of increasing the
18
O isotope concentration
until the imposed value, at the column top, is
reached. This working regime is the most relevant
one for the FSC dynamics.
The mathematical model which describes with
high accuracy the work of FSC is proposed in
(Mureşan, 2018). This model is briefly presented in
the following equations. The final form of the output
signal is given by:
s
S
s1 0
0f 0f
s
f
s1 f 0
S
F(s) y
e1
y(t,s) y y (t) y y (t)
F(s s h) y
e1
−
−
=+ ⋅ =+ ⋅
==−
−
(1)
where y
0
= 0.204% represents the natural abundance
of the
18
O isotope. Also, the y
f
(t) function models
the separation process dynamics in relation to time
(t), representing the solution of the ordinary
differential equation:
f
ff
ii
dy (t)
11
y(t) u
dt T(F ) T(F )
=− ⋅ + ⋅
(2)
where T(F
i
) is the isotope exchange process time
constant which depends on the input flow F
i
(t) and
u
f
(t) represents the increase of the
18
O concentration
over the y
0
value, in steady state regime. The
previously presented differential equation is solved
considering the remark that firstly the value of the
time constant is singularized for a certain (F
i
). The
linear variation of the time constant in relation to the
input signal (T(F
i
)) is given by:
i2Ti2
T(F ) T K (F F )=+ ⋅− (3)
where F
2
= 185 l/h (implying the time constant
T
2
= 84.703 h experimentally determined) and
2
T
h
K 3.9262
l
=−
represents the gradient of the
ramp T(F
i
). The u
f
(t) signal has the following form:
fff 00i
n(F)
tp i
0
uy(t,ssh)yy(S(F)1)
y( 1)
===−=⋅−=
=⋅α −
(4)
ICINCO 2018 - 15th International Conference on Informatics in Control, Automation and Robotics
370

where t = t
f
is the final value for time (corresponding
to the steady state regime) and s = s
f
is the final
value for the independent variable (s)
(corresponding to the column top). Also,
n(F)
tp i
i
S(F ) =α represents the separation, being a
function which depends on the input signal. In its
equation, α = 1.018 is the elementary separation
factor of the
18
O isotope for the isotopic exchange
procedure in the system NO, NO
2
-H
2
O, HNO
3
. Also,
the equation of the number of the theoretical plates
is:
tp i
i
h
n(F)
HETP(F )
=
(5)
where HETP (F
i
) is the Height of Equivalent
Theoretical Plate. Both n
tp
and HETP are functions
depending of the input signal (F
i
). The HETP
variation is linear on intervals, but is nonlinear on
the entire domain of F
i
values. The HETP function is
given by the following system of equations (being
linear on intervals, but is nonlinear on the entire
domain of F
i
values):
i0H1i0i
i0H2i0i
HETP(F ) HETP K (F F ), if F 140 l / h
HETP(F ) HETP K (F F ),if F 140l / h
=+⋅− ≤
=+⋅− >
(6)
where HETP
0
= 8.6 cm obtained for the input flow
F
0
= 140 l/h, and K
H1
, respectively K
H2
(
H1
cm h
K0.08
l
⋅
=−
for
i
F 140 l / h≤ ;
H2
cm h
K 0.0333
l
⋅
=
for
i
F 140 l / h> ) are the
gradients of the two ramps from (6) (experimentally
determined). The HETP (F
i
) function is graphically
represented in Fig. 2.
Figure 2: The HETP dependency in relation to F
i.
In order to avoid the usage of the two equations
from (6), a neural network is trained to learn the
HETP(F
i
) function. The structure of the used neural
network is a feed-forward fully connected one
(Haykin, 2009; Vălean, 1996), having one input
signal (F
i
), one hidden layer (containing 20 neurons)
and one output signal (HETP(F
i
)). The neurons of
the hidden layer are nonlinear having as activation
function the hyperbolic tangent and the output
neuron is linear. After training, the neural network
output, more exactly the HETP(F
i
) function, is given
by:
iio
HETP(F ) WL tanh(W F B) b=⋅ ⋅++ (7)
where WL, W, B and b
o
are the training solutions
(WL is the row vector which contains the input
weights, W is the column vector which contains the
layer weights, B is the column vector which contains
the bias values of the neurons from the hidden layer
and b
o
is the output neuron bias value). Also, the
notation “tanh” represents the hyperbolic tangent
function.
The
s
S
s
s
f
S
e1
F(s)
e1
−
=
−
function from (1) highlights the
proportion of the
18
O isotope concentration in a
certain position from the FSC height, in relation to
the
18
O isotope concentration at the FSC top.
Practically, F
s
(s) function models the concentration
evolution on the column height. The F
S
(s) function
is obtained using the
s
S
s1 0
F(s) y e=⋅ function which
approximates the
18
O isotope concentration
distribution on the FSC height, for a certain (F
i
)
(implicitly for a certain HETP(F
i
)). In the F
s1
(s)
function, (S) represents the “length” constant of the
column and depends on the input signal F
i
. The S(F
i
)
function is given by the system of two equations
from (8).
i1S1i1 i
i2S2i2 i
S(F ) S K (F F ), if F 140 l / h
S(F ) S K (F F ), if F 140 l / h
=+ ⋅ − ≤
=+ ⋅− >
(8)
where S
1
= 751.124 cm obtained for F
1
= 80 l/h and
S
2
= 565.5 cm obtained for F
2
= 185 l/h. Also, K
S1
and K
S2
(
S1
cm h
K 4.4804
l
⋅
=−
for
i
F140l/h≤ ;
S2
cm h
K 1.8489
l
⋅
=
for
i
F 140 l / h> ) are the
gradients of the two ramps from (8) (experimentally
80 100 120 140 160 180 200
8.5
9
9.5
10
10.5
11
11.5
12
12.5
13
13.5
Fi [l/h]
HETP [cm]
HETP
F3
F4
18
O Isotope Concentration Control
371

determined). From (8), it can be remarked that S(F
i
)
has an evolution linear on intervals, but nonlinear on
the entire domain of F
i
values. In order to learn the
S(F
i
) function, the neural structure as in the case of
HETP(F
i
) function is used, but having 50 neurons in
the hidden layer. After training, the neural network
output, more exactly the S(F
i
) function, is given by:
iio
S(F ) WL tanh(W F B) b=⋅ ⋅++ (9)
where WL, W, B and b
o
have the same significance
as in the case of HETP(F
i
) function. Obviously, WL,
W, B and b
o
are singularized as values for the S(F
i
)
function, being the solutions of its corresponding
neural network training.
3 THE PROPOSED CONTROL
STRATEGY
The proposed control structure for the
18
O
concentration control is presented in Fig. 3. In
Fig. 3, SDPP represents the Separation Distributed
Parameter Process which has as input signal the flow
of nitric oxides F
i
(t) and its work depending, also, on
the independent variable (s). The output signal from
SDPP is the
18
O concentration y(t,s), signal which is
not yet affected by the disturbance effect. The main
disturbance signal modifies directly the value of
SDPP outputs signal, having the value y
d
(t).
Practically, the final value of the
18
O concentration
(the final output signal) is y
1
(t,s) = y(t,s) + y
d
(t). DD
is the disturbance delay element, modelling the
disturbance propagation into the process and y
d0
is
the steady state value of the disturbance. The main
disturbance signal in the control system is
represented by the product extraction flow (F
P
).
Even, the product extraction is a usefully procedure,
it can be assimilated with a disturbance due to the
fact that extracting (HN
18
O
3
), with an increased
concentration of
18
O, from the column, the
18
O
isotope concentration in the column decreases. The
concentration of
18
O isotope is measured using the
concentration sensor CS which is a mass
spectrometer and generates the feedback signal r
1
(t).
The automation equipment (Golnaraghi, 2009; Love,
2007) from the
18
O isotope control system works
with unified current signals (4 – 20 mA), obviously
r
1
(t) being an unified current signal. The actuating
signal (F
i
(t)) is generated by the actuator, in this case
the nitric oxides pump P. MSFP represents the
reference Model of the System Fixed Part (which
includes the mathematical models of the pump P, of
SDPP and of the sensor CS, serial connected). It is
run on a process computer in parallel with the real
process, having its initial (reference) behaviour (not
affected by the exogenous disturbances (y
d
(t) = 0)
and not affected by parametric disturbances). The
SDPP is integrated in MSFP by implementing the
mathematical model presented in Section 2. Also,
the mathematical models of P and CS are integrated
in MSFP by implementing for each a neural
network. The two neural networks have nonlinear
autoregressive structure with exogenous inputs
(NARX), they contain, each, 9 linear neurons in the
hidden layer (they have only one hidden layer) and
one linear neuron in the output layer, respectively
they have, each,
two unit delays (one on the input
signal and one on the output signal, due to the fact
that P and CS are first order systems).
The main error signal is e
1
(t) = w(t) – r
1
(t), where
w(t) is the concentration setpoint signal. The main
concentration controller CC of PID (Proportional –
– Integral – Derivative) type, processes the signal
e
1
(t) and generates the main control signal c
1
(t). The
secondary error signal e
2
(t) = r
1
(t) – r
2
(t), where r
2
(t)
is the feedback signal generated at the output of
MSFP, represents the measure of the effects of all
disturbances (both exogenous and of parametric
type) which occur in the system. Practically, e
2
(t) is
a measure of the deviation of the output signal value
in relation to its reference value (generated by the
simulation of MSFP). For a correct comparison
between the real plant behaviour (referring to the
fixed part) and its reference model behaviour, at the
input of both entities, the same input signal c
f
(t) is
applied. The mentioned parametric disturbances can
be of two types: the small variations in relation to
time of the separation column structure parameters
or the small variations of (s) independent variable
(due to the change of the CS position and of the
product extraction point; due to this aspect, the
reference model is simulated for s = s
f
). The
disturbances compensator DC of PD type
(Proportional – Derivative) processes the error
signal e
2
(t) and generates the compensation control
signal c
2
(t). The total control signal due to the
control efforts of CC and DC results as
c
3
(t) = c
1
(t) – c
2
(t). The final control signal c
f
(t)
results as c
f
(t) = c
0
– c
3
(t), where c
0
represents the
value, in unified current, proportional with the value
of the reference flow. The reference flow is referring
to the initial flow at which the separation column
starts to work.
From (3), the fact the lowest value of the
separation column time constant is obtained for the
flow F
2
.
ICINCO 2018 - 15th International Conference on Informatics in Control, Automation and Robotics
372
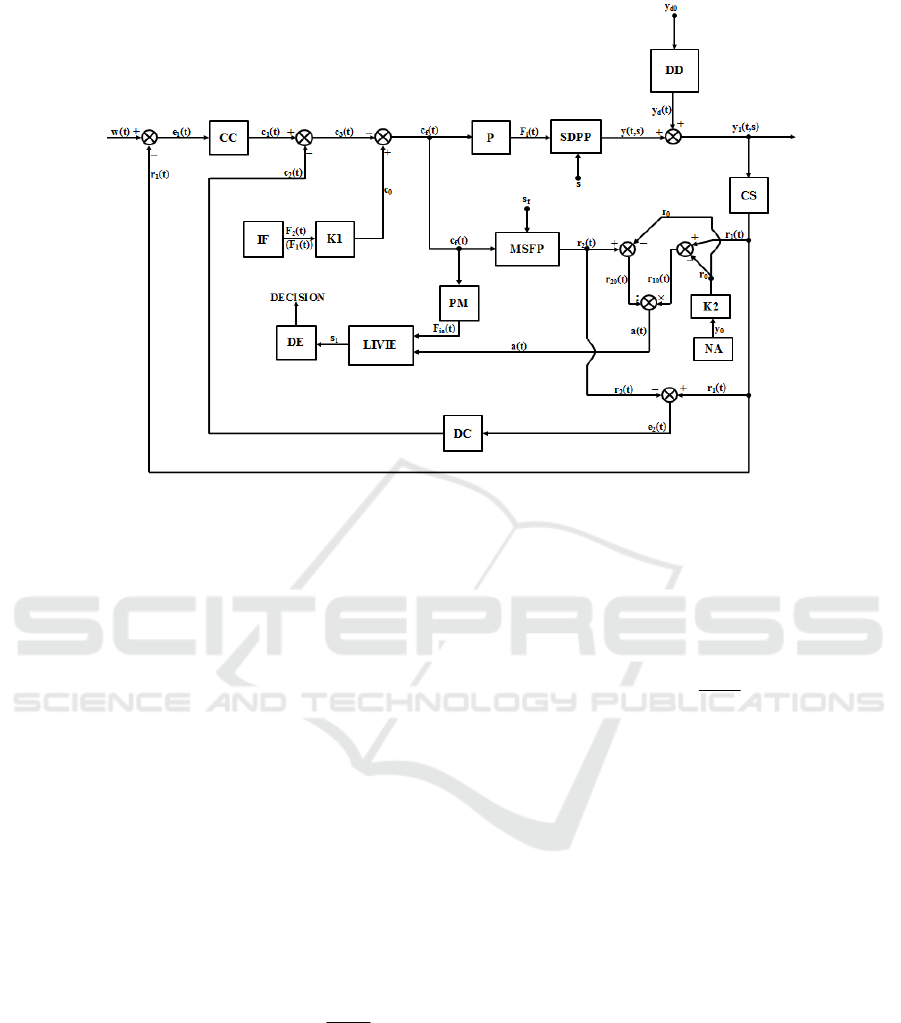
Figure 3: The proposed control structure.
Consequently, in order to obtain an acceptable
value of the concentration control system settling
time, the reference flow is fixed at the value of F
2
.
Also, other values could be chosen for the reference
flow (for example F
1
) but we would obtain a slower
control system. The element IF (Initial Flow)
generates the value F
2
which is scaled to the c
0
value
using the proportionality constant K1.
The instantaneous value of (s) independent
variable is determined using LIVIE (Length
Independent Variable Identification Element), more
exactly the sensor position in the column. This
procedure is necessary in order to determine if the
separation plant can be physically used when the
sensor position in the column is changed. Another
advantage of this procedure is the possibility to
equivalate the disturbances effects with the change
of the CS position in the column. The work of
LIVIE is based on obtaining the F
S
(s) function from
the ratio between the feedback signals r
10
(t) and
r
20
(t), resulting the signal a(t) (
10
20
r(t)
a(t)
r(t)
=
). The
feedback signals r
10
(t) and r
20
(t) result as differences
between the primary feedback signals r
1
(t),
respectively r
2
(t) and r
0
, which is the value in unified
signal corresponding to the
18
O natural abundance
(y
0
). The value of (y
0
) is generated by NA (Natural
Abundance Element), after that it being scaled with
the proportionality constant K2, resulting (r
0
).
Obviously, at the plant work starting, r
20
(t) is
initialized at a value different than 0, in order to
make possible the computation of a(t). The
instantaneous value of the independent variable (s),
notated with (s
1
), can be obtained from the equation
of F
S
(s), being given by:
s
f
S(F (t ))
i
1i
s S(F (t)) ln[a(t) e (1 a)]=⋅⋅+−
(10)
The numerical implementation of (10) is made
by LIVIE. The two input signals in LIVIE are a(t)
and F
ia
(t) which is the approximated value of the
actuating signal F
i
(t). The signal F
ia
(t) is generated
by the simulation of PM (Pump Model) element,
which is implemented using the same procedure as
in the case of MSFP and having as input signal the
c
f
(t) signal. The natural logarithm and the
exponential function from (10) are numerically
implemented using feed-forward fully connected
neural networks, too. The Decision Element (DE)
generates the “DECISION” in relation to the
identified value of (s
1
). The “DECISION” can be the
generation of an alarm if the (s
1
) variable tends to
reach the limit value or the separation plant stop if
(s
1
) variable equals the imposed limit.
In case of the real separation plant, the following
elements from Fig. 3: CC, DC, MSFP, LIVIE, PM,
IF, K1, NA and K2 have to be implemented on
numerical equipment. In order not to increase Fig. 3
complexity in a non-understandable manner, the
analog to digital and the digital to analog converters
18
O Isotope Concentration Control
373

which connect the numerical equipment to the
separation plant are not figured.
4 CONTROLLERS TUNING AND
SIMULATIONS RESULTS
The simulations presented in this paper are made in
MATLAB/Simulink. The CC tuning is made not
considering the disturbances action in the system
(y
d0
= 0 and s = s
f
) and, also, not considering the DC
control action (c
2
(t) = 0). Due to the fact that the
isotope separation process, besides being a
distributed parameter process, is a strong nonlinear
one, the relay method is used for CC tuning. In order
to obtain an oscillatory variation of the output signal
y(t,s), the CC controller is replaced with a two
position relay having the output commutation
between the value b
min
= 0 mA and the value
b
max
= 6.4 mA (without considering the initial value
of 4 mA for the unified current signals), respectively
having the hysteresis set to ±1 mA.
If the simulation is made for a step type setpoint
signal (proportional with the imposed value of 1.5 %
for the output
18
O isotope concentration), the control
system response (the variation in relation to time of
the y(t,s) signal) is presented in Fig. 4.
Figure 4: The control system response in the case of the
relay tuning method application.
In Fig. 4, the fact that the constant amplitude
oscillations are obtained, after 1000 h from the
simulation start, is highlighted. From Fig. 4, the
oscillation period T
OSC
= 552 h and the oscillation
amplitude A
OSC
= A/2 = 0.0907 % result. Using the
values obtained for T
OSC
and A
OSC
, the parameters of
the PID CC controller can be computed using the
Ziegler – Nichols equations. The PID controller
transfer function is:
DCC
CC CC
ICC fCC
Ts
1
H(s)K (1 )
TsTs1
⋅
=⋅+ +
⋅⋅+
(11)
where K
CC
is the controller proportionality constant,
T
ICC
is the controller integral time constant, T
DCC
is
the controller derivative time constant and T
fCC
is the
time constant of the first order filter used to make
the PID controller feasible. After the application of
Ziegler – Nichols equations, the fine tuning of the
controller is made in order to obtain better control
performances. The fine tuning consists in modifying
the initial form of the obtained controller parameters
and in testing the control performances variation,
until the best set of performances is obtained. At the
tuning procedure end, the following parameters are
obtained: K
CC
= 0.358, T
ICC
= 197.143 h,
T
DCC
= 33.12 h and T
fCC
= 0.625 h. If the setpoint
concentration is set to 1.5%, the comparative graph
between the separation process open loop response
and the control system responses for the cases of
using the best PI controller that could be obtained
respectively using the proposed PID controller, is
presented in Fig. 5. In Fig. 5, the notation CSt
signifies the minimum concentration value of the
steady state band near the steady state value of the
output signal (the steady state band is fixed at ±2%,
more exactly CSt = 1.47 %).
Figure 5: The comparative graph between the open loop
separation process response and the control system
responses using two different controllers.
From Fig. 5, it results that in all three simulation
cases, the steady state error e
st
= 0 %, due to the fact
that, in steady state regime the three curves reach the
imposed value for
18
O concentration (y
st
= 1.5 %).
Also, in the case of using both controllers, a small
overshoot occurs, having the value smaller than 1 %
(the maximum imposed limit). The PID controller
generates the overshoot σ
1
= 0.94 %, insignificantly
higher than σ
2
= 0.9 % (generated by the
PI controller). Obviously in the case of the
separation process open loop response the overshoot
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
1.8
TIME [h]
18
O CONCENTRATION [%]
A
TOSC
0 500 1000 1500 2000 2500
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
TIME [h]
18
O CONCENTRATION [%]
Case of PID Controller
Open loop response
Case of PI controller
ts1
CSt
ICINCO 2018 - 15th International Conference on Informatics in Control, Automation and Robotics
374
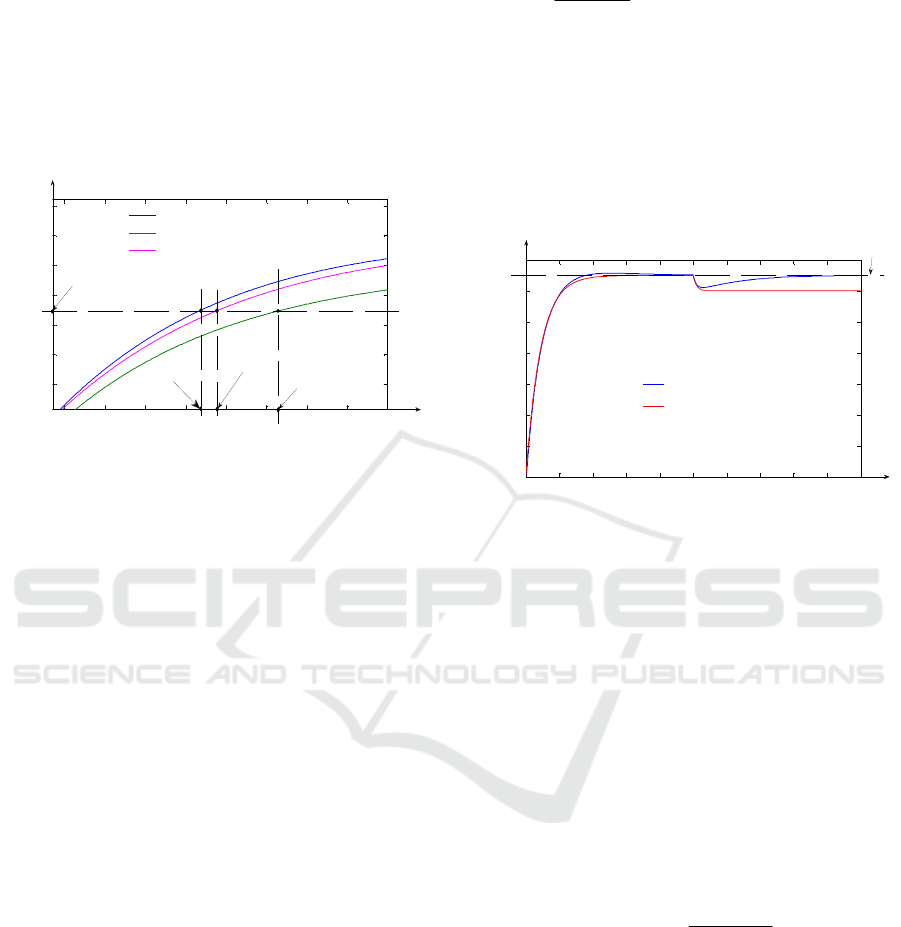
is σ
3
= 0 %. Due to the fact that in all three cases the
overshoot is included in the steady state band, the
settling time (which is a critical performance) is
considered when the curves from Fig. 5 reach the
CSt value. In Fig. 5, the fact that the PID controller
generates the smallest settling time t
s1
, results. In
order to distinguish the three values of the settling
times, Fig. 6 is considered.
Figure 6: The settling times associated to the three curves
from Fig. 5.
From Fig. 6, the fact that the PID controller
generates the best settling time (t
s1
= 719.58 h) is
highlighted. It can be remarked that t
s1
< t
s2
< t
s3
(t
s2
= 740.21 h and t
s3
= 819.92 h). Consequently, the
PID controller usage, improves significantly the
separation plant settling time implying important
economic advantages. Also, using the PID
controller, even better settling times were obtained
in simulations, but only if the actuating signal exits
from allowed variations limits. Practically, t
s1
represents the best settling time that could be
obtained if the actuating signal (F
i
(t)) remains
enclosed between the saturation limits. The open
loop separation process simulation is made
simulating the mathematical model presented in
Section II for the input flow F
5
= 150.255 l/h, value
which generates the steady state value of 1.5 % for
the output
18
O concentration (according to (3) and
(6), the best settling time for the open loop process,
if we impose a certain value of the output signal, is
obtained if F
i
(t) > 140 l/h). The main problem in the
case of usage the separation process in open loop is
the fact that the effect of the disturbances cannot be
rejected, aspect highlighted in Fig. 7. The
simulations from Fig. 7 are made in the same
conditions as the simulations from Fig. 5, but the
exogenous disturbance with the value y
d0
= − 0.1 %
occurs in the system at the moment t
1
= 2500 h.
The y
d0
value is propagated in the system through
the DD element having the transfer function
DD
DD
1
H(s)
Ts1
=
⋅+
, where the time constant
T
DD
= 50 h. Also, the negative value of y
d0
is
physically interpreted as the output
18
O isotope
concentration decrease, due to the exogenous
disturbance effect. From Fig. 7, it results that in the
case of the PID controller usage, the disturbance
effect is rejected, in steady state regime the system
response reaching again the imposed value
(y
st
= 1.5 %).
Figure 7: The exogenous disturbance effect.
The open loop response of the separation
process, after the disturbance occurs in the system,
gets steady to the value y
st1
= 1.4 % (with y
d0
smaller
than y
st
, resulting that the disturbance effect is not
rejected) fact which highlights again the necessity of
using a controller for the concentration control. An
important problem which results from Fig. 7 is the
slow reaction of the control system in disturbance
rejecting regime (the duration of the transitory
regime implied by the disturbance effect is over
800 h). In this context, the necessity of using the DC
element occurs. For the structure of the Disturbance
Compensator (DC) a PD transfer function is
proposed, it having the form:
DDC
DC DC
fDC
Ts1
H(s)K
Ts1
⋅+
=⋅
⋅+
(12)
The tuning procedure of DC follows the stages:
firstly the proportionality constant of DC is
initialized at the value K
DC
= 1, its derivative time
constant is initialized at the value T
DDC
= 10 h,
respectively the time constant of the first order filter
used to obtain the feasible form of DC is fixed at the
value T
fDC
= 1 h; secondly, T
DDC
is iteratively
determined by increasing its value from an iteration
to the next one with ∆T
DDC
= 1 h until the system
response overshoot in disturbance effect rejecting
regime occurs (the overshoot values are determined
550 600 650 700 750 800 850 900
1.42
1.44
1.46
1.48
1.5
1.52
1.54
TIME [h]
18
O CONCENTRATION [%]
Case of PID Controller
Open loop response
Case of PI controller
ts1
ts3
ts2
CSt
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
18
O CONCENTRATION [%]
TIME [h]
The case with PID Controller
The open loop response
yst
18
O Isotope Concentration Control
375

repeating the simulation at each iteration); finally,
K
DC
is iteratively determined by increasing its value
from an iteration to the next one with ∆K
DC
= 0.01
until the system response overshoot in disturbance
effect rejecting regime equals the system response
overshoot in starting regime (σ
1
). After the
application of the presented algorithm, the structure
parameters of DC result: K
DC
= 1.1, T
DDC
= 50 h.
The comparative graph between the system response
in the case of using and the case of not using the DC
element, is presented in Fig. 8.
Figure 8: The effect of using DC.
The simulations from Fig. 8 are made in the
same conditions as the simulations from Fig. 7.
From Fig. 8, it results that the control system
generates much better performances in disturbance
rejecting regime in the case of using DC than in the
case without DC. Relative to the moment t
1
(when
the disturbance signal occurs in the system), in the
case of using DC, the
18
O concentration reaches
again the steady state regime after 219.4 h
(corresponding to the moment t
s4
). This transitory
regime is much shorter than in the context of not
using DC (in this case the
18
O concentration reaches
again the steady state regime after 842.7 h
(corresponding to the moment t
s5
)). Consequently,
the usage of DC implies a consistent improvement
of the system performances in rejecting the
disturbances effects, fact which represents a major
technological and economical advantage. The
control effort is highlighted in Fig. 9.
From Fig. 9, it results that the variation of the
input flow of nitric oxides is enclosed only between
the saturation limits [F
1
,F
2
], aspect which proves that
the usage of the two controllers (CC and DC) is
feasible. Also, in disturbance effect rejecting regime,
due to the action of DC, the actuating signal has a
much faster variation, fact which signifies a much
stronger control effect and implicitly a higher
efficiency of the control system.
Figure 9: The variation, in relation to time, of the actuating
signal.
In Fig. 10, the evolution in relation to time of
HETP(F
i
) function is presented. This evolution is
associated to the system response presented in
Fig. 8, in the case of using DC.
Figure 10: The variation in relation to time of HETP(F
i
)
function.
From Fig. 10, it results that the limit values of
HETP(F
i
) presented in Fig.2 (HETP
min
= 8.6 cm) and
(HETP
max
= 13.4 cm) are almost reached (but not
equalled) only in transitory regimes due to the CC,
respectively DC actions. This aspect proves again
the physical possibility to implement the proposed
control strategy. In Fig. 11, the control system
response is presented in the case of the (s)
independent variable variation. The variation occurs
at the moment t
2
= 2500 h from the simulation start,
when (s) independent variable value is changed from
the initial value s = s
f
(s
f
= 1000 cm) to the value
s = 970 cm. This variation is due to the
concentration sensor position change in relation to
the column height. In Fig. 11, the fact that the two
control elements (CC and DC) reject the effect of the
(s) independent variable value “jump”, is
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
TIME [h]
18
O CONCENTRATION [%]
The case with DC
The case without DC
ts4
ts5
Cst
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
80
100
120
140
160
180
200
TIME [h]
NIT RIC O X IDES IN PU T F LO W [l/h]
The case without DC
The case with DC
F2
F1
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
8.5
9
9.5
10
10.5
11
11.5
12
12.5
13
13.5
TIME [h]
HETP [cm]
HETP(Fi) function in relation to time
HETPmax
HETPmin
ICINCO 2018 - 15th International Conference on Informatics in Control, Automation and Robotics
376
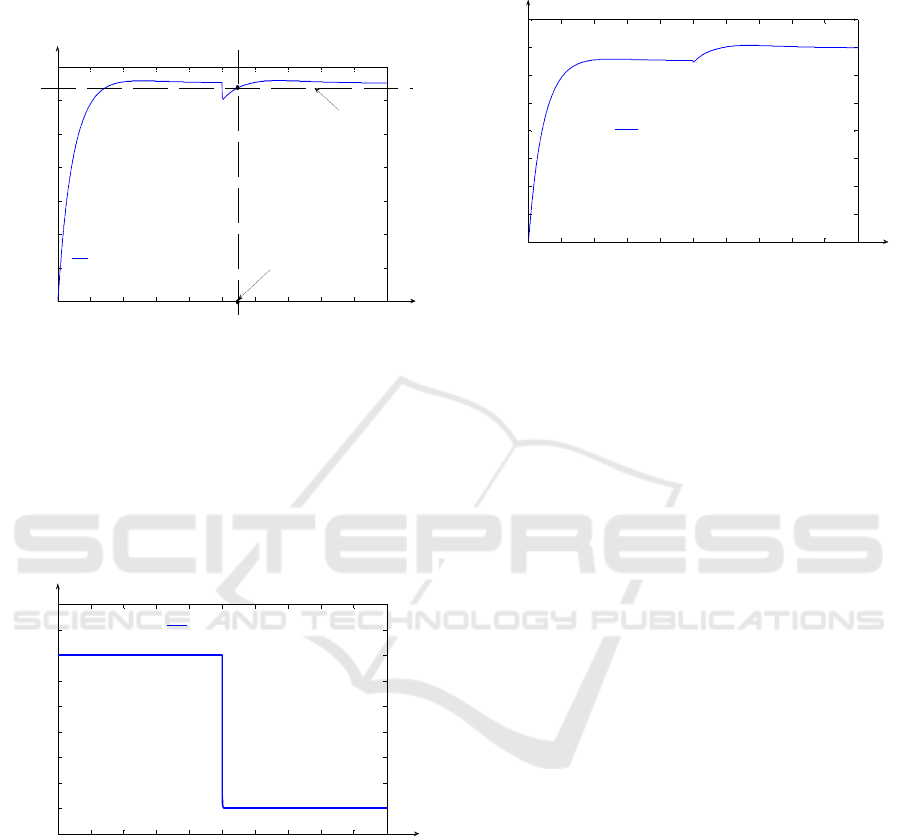
highlighted. Only after 237.8 h (corresponding to the
moment t
s6
) from the moment t
2
, the
18
O
concentration equals the CSt value, the imposed
steady state regime (near the value y
st
= 1.5 %) being
reached again.
Figure 11: The system response, in the case of the (s)
independent variable variation.
This aspect proves the fact that the proposed
control structure can efficiently manage the effect of
the parametric disturbances, too. The (s)
independent variable value change is identified by
the LIVIE element. The variation in relation to time
of the LIVIE output signal (the approximation (s
1
) of
the (s) instantaneous value) is presented in Fig. 12.
Figure 12: The variation in relation to time of the (s
1
)
signal.
It can be remarked the fact that the
approximation of the (s) independent variable is an
exact one both before and after the variation moment
(t
2
).
In the case when the position of the
concentration sensor remains in the position given
by s = 970 cm, the
18
O isotope concentration
evolution, in relation to time, for the value s = s
f
, is
presented in Fig. 13. From Fig. 13, it results that the
18
O isotope concentration increases at the column
top (s = s
f
). This aspect is explained through the
increasing evolution of the output signal in relation
to (s) independent variable (according to (1)).
Figure 13: The output signal evolution, in relation to time,
for s = s
f.
The change of the concentration sensor position
has an important application, more exactly when,
immediately after the extraction of the necessary
18
O
isotope quantity at the imposed concentration (in the
example 1.5 %), the isotope extraction at a higher
concentration is necessary (for example 1.6 %). In
this case, firstly the extraction is made from the
point s = 970 cm, where the control is made and
secondly, when the
18
O isotope concentration has to
be increased, the extraction point is changed to
s = s
f
= 1000 cm (the sensor position can be, also,
changed to the column top). The main advantage
consists in the avoidance of a new transitory regime
and implicitly in a much faster extraction of the
18
O
isotope at an increased concentration. The procedure
of identifying the (s) independent variable value can
be, also, used in order to determine if the separation
plant can be physically used. The constrain
regarding the possibility of usage the separation
plant consists in limiting the maximum deviation of
the (s) independent variable with more than 40 cm in
relation to (s
f
) value. In this context, the equivalent
value of (s) has to be approximated (containing the
superposition of the physical value of (s) and also
the equivalent value of (s) (which represents a
measure of the exogenous disturbances equated as a
position variation in the column height)). In Fig. 14,
the same simulation as in the case of Fig. 11 is
presented (on the first 5000 h), but considering the
fact that at the moment t
3
= 5000 h, an exogenous
disturbance with the value y
d0
= − 0.2 % occurs in
the system.
In Fig. 14, the moment t
2
is highlighted with the
ellipse 1 and the moment t
3
is highlighted with the
ellipse 2. In both cases, the effect of the disturbance
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
TIME [h]
18
O CONCENTRATION [%]
18
O Isotope Concentration
ts6
CSt
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
965
970
975
980
985
990
995
1000
1005
1010
TIME [h]
s1 [cm ]
s1 Independent Variable [cm]
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
1.8
TIME [h]
18
O CONCENTRATION [%]
18
O Concentration for s = sf
18
O Isotope Concentration Control
377

is rejected efficiently, the output signal having in all
steady states the imposed value (1.5 %). The
evolution, in relation to time, of the signal resulted
as the difference (s
f
– s
1
), is presented in Fig. 15.
Figure 14: The output signal evolution in relation to time,
when both parametric and exogenous disturbances occur
in the system.
Figure 15: The evolution of (s) independent variable in
relation to (s
f
).
Practically, the representation from Fig. 15
signifies the equivalent position of the concentration
sensor in relation to the column top. After the
moment t
3
, the value of the signal from Fig. 15
increases with 7 cm (due to the exogenous
disturbances). Due to the fact that the maximum
value of the signal from Fig. 15 is 37 cm (value
smaller than the maximum imposed limit – 40 cm),
the conclusion that the system is physically usable at
the considered disturbances values, results. At the
limit of 40 cm, the effect of the disturbances cannot
be rejected without the increase of the actuating
signal over the saturation limits. At the detection of
the value (s
f
– s = 37 cm) the DE element from
Fig. 3 takes the DECISION of emitting an Alarm
and at the detection of the limit value
(s
f
– s = 40 cm), the DE element takes the
DECISION of stopping the separation plant.
Obviously, the difference (s
f
– s) is always positive
due to the fact that the sensor position can be
changed only in the column volume (and only near
its top).
5 CONCLUSIONS
In this paper, an original control structure used for
the control of the
18
O isotope concentration (isotope
produced using a separation column), is presented.
An important achievement is represented by the fact
that the separation process, which is a distributed
parameter process, is included in a control system.
In order to obtain high control performances in
disturbance rejecting regime, a compensation loop,
based on the process reference model, is designed.
The HETP variation in relation to F
i
is nonlinear
due to the following explanations: at the consistent
decrease of F
i
, the steel packing of FSC is not
properly wet and the contact between the liquid and
the gas from the column is not an efficient one, fact
which implies a high value of HETP; at the
consistent increase of F
i
, the formation of the
flooding phenomenon occurs, fact which implies a
high value of HETP, too; the column optimum
HETP (the smallest value of HETP) is obtained in
the case of the studied separation column for
F
i
= 140 l/h which is an intermediary value; each
separation column has an optimum point given by its
optimum loading.
Considering the previous remarks regarding the
HETP value variation, the saturation limits of the
actuating signal are determined, obtaining the
[F
1
, F
2
] domain.
Another original element proposed in the paper
is the design of an identification system of the exact
concentration sensor position in the neighbourhood
of the column top. Also, a procedure to equivalate
the disturbances effects, with a change of (s)
independent variable value, is proposed. These
mathematical mechanisms can be used in order to
optimize the product extraction from the separation
plant, when the future orders (for
18
O isotope) can be
predicted.
Another application of these mathematical
mechanisms is represented by the possibility to
determine if the separation plant is physically usable
and in the case when it is not, the decision element
can take the decision of stopping the plant work.
A future possibility to improve the control
system performances is given by the possibility to
replace the main CC PID controller with an
advanced controller (Kern, 2015; Zou, 2017). Also,
0 1000 2000 3000 4000 5000 6000 7000 8000
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
TIME [h]
18
O CONCENTRATION [%]
18
O Isotope Concentration
1
2
0 1000 2000 3000 4000 5000 6000 7000 8000
0
5
10
15
20
25
30
35
40
TIME [h]
s VARIATION IN RELATION TO sf [cm]
s Independent Variable in Relation to sf
ICINCO 2018 - 15th International Conference on Informatics in Control, Automation and Robotics
378

the proposed model can be extended in order to
obtain the mathematical model of a separation
cascade, which contains two separation columns in
its structure. Obviously, for the case of a separation
cascade, the proposed control strategy has to be
adapted.
REFERENCES
Abrudean, M., 1981. Ph.D. Thesis.
Axente, D., Abrudean, M., Bâldea, A., 1994.
15
N,
18
O,
10
B,
13
C Isotopes Separation trough Isotopic
Exchange, Science Book House.
Coloşi, T., Abrudean, M., Ungureşan, M.-L., Mureşan, V.,
2013. Numerical Simulation of Distributed Parameter
Processes, Springer.
Golnaraghi, F., Kuo, B. C., 2009. Automatic Control
Systems, 9
th
edition, Wiley Publishing House.
Haykin, S., 2009. Neural Networks and Learning
Machines, Third Edition, Pearson Int. Edition.
Kern, R., Shastri, Y., 2015. Advanced control with
parameter estimation of batch transesterification
reactor, In Journal of Process Control, Vol. 33, pp.
127-139.
Li, H.-X., Qi, C., 2011. Spatio-Temporal Modeling of
Nonlinear Distributed Parameter Systems:
A Time/Space Separation Based Approach, 1st
Edition, Springer.
Love, J., 2007. Process Automation Handbook, 1 edition,
Springer.
Mureşan, V., Abrudean, M., Ungureşan, M.-L. Coloşi, T.,
2018. Modeling and Simulation of the Isotopic
Exchange for 18O Isotope Production, proposed for
publishing to AQTR, Cluj-Napoca, Romania.
Vălean H., 1996. Neural Network for System
Identification and Modelling. In Proc. of Automatic
Control and Testing Conference, Cluj-Napoca,
Romania, 23-24 May, pp. 263-268.
Zou, Z., Wang, Z., Meng, L., Yu, M. Zhao, D., Guo, N.,
2017. Modelling and advanced control of a binary
batch distillation pilot plant, In Chinese Automation
Congress (CAC), 20-22 October.
18
O Isotope Concentration Control
379
