
Aging Treatment on Aluminium AA6063 against Bacterial Corrosion
Resistance in Marine Environment
Herman Pratikno
1
, Harmin Sulistiyaning Titah
2
and Nurika Primiliana
1
1
Department of Ocean Engineering, Faculty of Marine Technology, Institut Teknologi Sepuluh Nopember,
Keputih, Sukolilo, 60111 Surabaya, Indonesia
2
Department of Environmental Engineering, Faculty of Civil, Environmental and Geo Engineering,
Institut Teknologi Sepuluh Nopember, Keputih, Sukolilo, 60111 Surabaya, Indonesia
Keywords: Aging Treatment, Aluminium Aa6063, Bacterial Corrosion.
Abstract: The corrosion is a natural occurrence of metal damage caused by electrochemical interactions with the
environment. One of the causes of corrosion is the attachment of bacteria to the material. The hazard caused
by corrosion, especially on ship, corrosion can cause a failure on ship’s structure which causes the ship to not
operate properly. The purpose of the research was to determine the effects of Aluminium AA6063 aging
treatment on bacterial corrosion. The material was be treated by heat treatment (Aging Treatment) before the
bacterial corrosion test was conducted. The bacterial corrosion test was carried out in artificial seawater with
salinity of 33‰, 35‰ and 37 ‰. The addition of three species of bacteria, i.e. Thiobacillus ferrooxidans,
Pseudomonas fluorescens and Escherichia coli were applied in all salinity with 5% (v/v). The bio-corrosion
rate was determined using weight loss method and the microstructure of material was conducted at pre and
post bacterial corrosion to determine the differences in the microstructure of the material before and after the
bacterial corrosion test. The results showed the non aging treatment material has a higher corrosion rate when
compared with the material with aging treatment. The non aging treatment material has the highest corrosion
rate of 1.189 mmpy with the addition of Thiobacillus ferrooxidans at salinity of 37‰ and the lowest corrosion
rate of 0.186 mmpy at salinity of 33‰. However, the aging treatment material has the highest corrosion rate
of 0.770 mmpy and the lowest corrosion rate of 0.175 mmpy at similary condition. Based on microstructure
results, uniform and pitting corrosion occurred on all specimens with the addition of bacteria or without the
addition of bacteria. However, the bacterial corrosion rates were different. It was identified that aging
treatment on Aluminium AA6063 can reduce the bacterial corrosion rate or increase the bacterial corrosion
resistance.
1 INTRODUCTION
The usage of aluminium in maritime industry as one
of the supporting materials has considerable role.
Aluminium with 6xxx series is aluminium alloys in
which magnesium and silicon are the principal
alloying elements, commonly used for architectural
extrusions and automotive components (Davis,
2001). Aluminium with 6xxx series have a high
strength when used for building structures in the
marine environment and more corrosion resistant
when compared with other aluminium series.
However, many factors can cause the decreasing of
aluminium metal. One of the factor is corrosion.
Alloys in the 6xxx series contain silicon and
magnesium approximately in the proportions
required for formation of magnesium silicide
(Mg2Si), thus making them heat treatable. Although
not as strong as most 2xxx and 7xxx alloys, 6xxx
series alloys have good formability, weldability,
machinability, and corrosion resistance, with medium
strength (Davis, 2001).
Corrosion is considered a significant factor in the
failure and damage of metals (Nuhia, et.al, 2011).
Corrosion is a material damage that was caused by the
influence of the surrounding environment. Corrosion
can cause many losses due to the reduction of
relatively large dimensions per unit time and also
reduce the age of the building. Aluminium with
erosion corrosion can accelerate or increase the
destruction due to relative movement and corrosive
media on metal surface.
Pratikno, H., Titah, H. and Primiliana, N.
Aging Treatment on Aluminium AA6063 against Bacterial Corrosion Resistance in Marine Environment.
DOI: 10.5220/0008649701590164
In Proceedings of the 6th International Seminar on Ocean and Coastal Engineering, Environmental and Natural Disaster Management (ISOCEEN 2018), pages 159-164
ISBN: 978-989-758-455-8
Copyright
c
2020 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
159

In the marine environment, the rate of corrosion
increase rapidly, due to the seawater contain solution
that are able to dissolve other substances in greater
quantities than other liquids. Those substances
include inorganic salts, organic compounds derived
from living organisms (bacteria) and dissolved gases.
One of the causes of corrosion is bacteria. Bacteria
live in the marine environment extensively in their
habitats and form colonies and attach to the metal
surfaces in the form of thin layers. Factors that affect
the occurrence of bio-corrosion are temperature, pH,
and oxygen levels. Based on our previous study, 3
species of bacteria Escherichia coli, Pseudomonas
fluorescens, and Thiobacillus ferroxidans can caused
bio-corrosion on steel structures of ASTM A106 and
A53 in deep seawater (salinity of 33‰), medium
seawater (salinity of 35‰), and shallow seawater
(salinity of 37‰) (Pratikno and Titah, 2016). The bio-
corrosion rate by P. fluorescens on Aluminium Alloy
6063 at salinity of 37‰ increased by one point six-
fold compared with the condition without bacteria
addition at the same salinity (Pratikno and Titah,
2016).
Corrosion can occur rapidly if neither
environmental control or prevention. The usage of
aluminium in the maritime industry is high such as in
ship building. So that aluminium should treat with
suitable treatment to increase the resistance of
corrosion or decrease the corrosion rate. The aim of
the research was to determine the effects of
Aluminium AA6063 aging treatment on bacterial
corrosion. The material was be treated by heat
treatment (Aging Treatment) before the bacterial
corrosion test was conducted.
2 MATERIALS AND METHODS
2.1 Preparation of Specimen
Material cut with area of ± 2 cm2 with diameter ±
1mm with thickness 1-2 mm. After that, the
hardening process was conducted by heating the
specimen with temperature of 535 ºC for 6 h. This
process was called as Solution Heat Treatment. After
that, the specimen was immersed in water or
quenching phase, then re-heat it at 200 ºC for 5 h. This
process was called as Aging Treatment. After the
aging treatment, the hardness test was conducting to
all specimens using Vickers method. Vickers
Hardness Test is the standard method for measuring
the hardness of metals, particularly those with
extremely hard surfaces: the surface is subjected to a
standard pressure for a standard length of time by
means of a pyramid-shaped diamond. The diagonal of
the resulting indention is measured under a
microscope.
2.2 Preparation of Artificial Seawater
This research used a chemical solution instead of sea
water with a salinity of 35 ‰. Salinity of 35‰ is the
salinity in the ocean of medium depths, where in the
microorganism commonly found at this depth,
including bacteria. The chemical composition of
seawater replacement is in accordance with ASTM
D1141-90, 1994 (ASTM, 2004).
2.3 Preparation of Bacteria
The preparation of bacteria was conducted based on
Pratikno and Titah (Pratikno and Titah, 2017). The
pure culture of Thiobacillus ferrooxidans,
Pseudomonas fluorescens and Escherichia coli, were
be inoculated onto nutrient agar (NA) media using
streak plate technique based on Harley and Prescott
(Harley and Prescott, 2002). The age of bacteria for
the test was 24 h. After that, one colony of bacteria
was transferred to nutrient borth (NB) and keep in
shaker incubator of Innova 2000 (New Brunswick-
Eppendorf, Germany) at 150 rpm and room
temperature, 33 oC for 24 h. The cell suspension of
selected bacteria was prepared by harvesting the cells
at the middle of the logarithmic phase, based on the
typical of growth rate graph for the selected bacteria.
At this time, the OD at 600 nm was 1.0 was
determined using UV spectrophotometer Genesys 20
(Thermo, USA). The cells were harvested through
centrifugation of Jouan E82 (Thermo, USA) at 4,000
rpm for 15 min. The obtained pellet was then washed
twice using 8.5 g NaCl/1000 mL solution.The
suspension of bacteria was ready to be used in bio-
corrosion test.
2.4 Immersion Method for
Bio-corrosion Test
The specimen was tested by immersion tehnique in a
prepared seawater solution with salinity 33‰, 35‰
and 37‰ using ASTM G31-72 standard (ASTM,
2004). There were two different treatments, namely
treatment without bacterial addition as a control and
treatment with bacterial addition Testing was carried
out for 4 weeks. Immersion testing was conducted in
beaker glass with size of 300 mL and the artificial
seawater was 250 mL for each beker glass.
ISOCEEN 2018 - 6th International Seminar on Ocean and Coastal Engineering, Environmental and Natural Disaster Management
160
2.5 Calculation of Corrosion Rate
Running rate of corrosion is a rapid propagation of
material quality decline against time. There is a
formula for calculating the corrosion rate based on the
ASTM G1-03 standard (ASTM, 2002) as follows:
𝐶𝑜𝑟𝑟𝑜𝑠𝑖𝑜𝑛 𝑟𝑎𝑡𝑒
𝑚𝑝𝑦
(1)
With:
K = Constanta
T = Time of exposure (h)
A = Surface area (cm2)
W = Weight loss (gram)
D = Material density (gram/cm3)
2.6 Macrostructure and
Microstructure Testing
After corrosion testing was conducted, the
microstructure of specimens were determinated using
a microscope for detailed morphology of the
specimen structure. It was used for documentary
evidence and it can be known that the specimen
differences between before and after testing.
3 RESULTS AND DISCUSSION
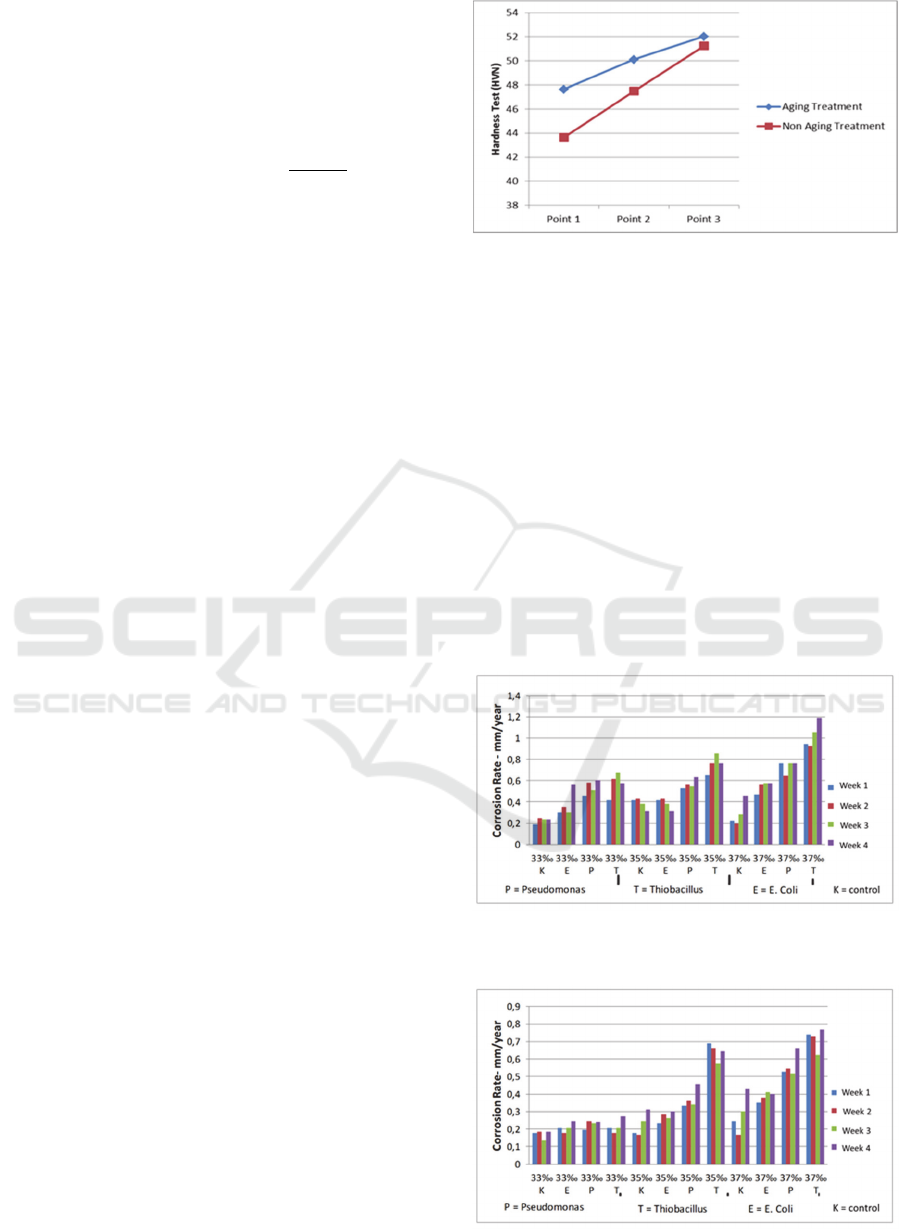
Figure 1 showed the results of hardness test on
AA6063 before and after aging treatment. Based on
the Figure 1, the value of hardness test on AA6063
increased after aging treatment. The high value of
Vickers hardness was 52.02 HVN after aging
treatment. It indicated that aging treatment can
accelerate the hardness value. According to Abel-
Rahman et al. (2010), the Vickers hardness of 6066
alloy has a maximum value of 80 HVN after 10 days
of quenching at 530oC which is the solution
temperature of this alloy. The hardness of 6063 alloy
has a maximum value of 40 HVN after 14 days of
quenching at 520 oC. A tremendous increase in
hardening of the Al-Mg-Si alloys is caused by
precipitates formed from solution with merely 1 wt%
of Mg and Si added to aluminum. During natural or
artificial aging, Al-Mg-Si alloys first produce clusters
of a few nm sizes. These clusters are an enrichment
of the Mg and Si precipitates (Abdel-Rahman, et.al,
2010). Extrudability of the alloy is strongly
influenced by the amount of Mg and Si and the size
and distribution of Al-Mg2Si precipitate particles
(Andersen, et.al, 1998).
Figure 1: Hardness test.
Based on Figure 2 and 3, the higher of salinity can
accelerate the rate of corrosion. The higher of salinity
contain chloride ions due to cause pitting corrosion and
other damage to the material. The corrosion rate on
alumnium alloys with the highest heat treatment at
salinity 33 ‰ with addition of Thiobacillus ferroxidans
was 0.565 mmpy, meanwhile the lowest corrosion rate
in the control specimens was 0.175 mmpy. At salinity
35 ‰, the highest corrosion rate occurred in specimens
with the addition of Thiobacillus ferroxidans (0.692
mmpy), and the lowest corrosion rate in control
specimens was 0.178 mmpy. Similarly, the highest
corrosion rate in specimens with the addition of
Thiobacillus ferroxidans was 0.770 mmpy at salinity
37 ‰ and the lowest corrosion rate on the specimen
with the addition of Escherichia coli (0.175 mmpy).
Figure 2: Corrosion Rate on AA6063 without aging
treatment.
Figure 3: Corrosion Rate on AA6063 with aging treatment.
Aging Treatment on Aluminium AA6063 against Bacterial Corrosion Resistance in Marine Environment
161
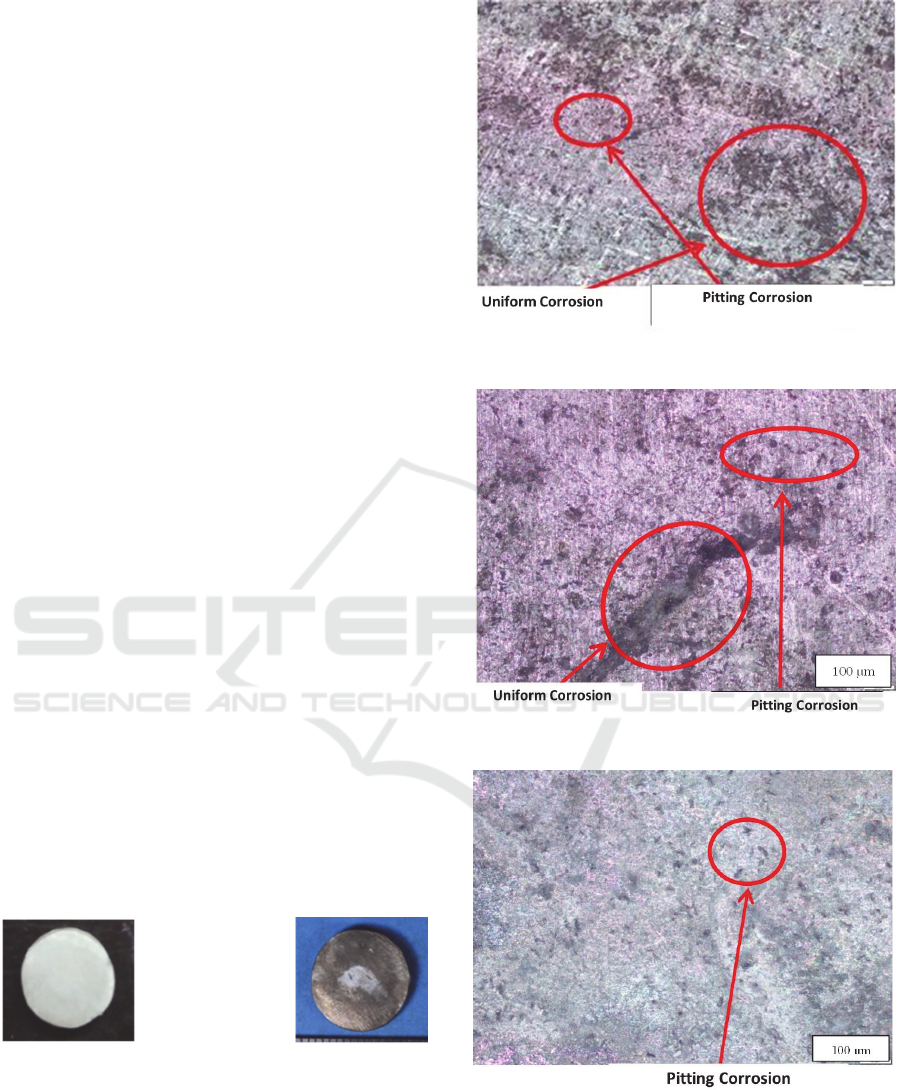
Based on graph, the most corrosive bacteria to test
specimens on corrosion rate of aging material and non
aging treatment was Thiobacillus ferroxidans.
Thiobacillus ferroxidans has the highest corrosion
rate due to this bacteria can produce more organic
acids, pigments, H2S ligands that can remove heavy
metal ions from the material. The non aging treatment
material has a higher corrosion rate when compared
with the material with aging treatment. The non aging
treatment material has the highest corrosion rate of
1.189 mmpy and the lowest corrosion rate of 0.186
mmpy, meanwhile the aging treatment material has
the highest corrosion rate of 0.770 mmpy and the
lowest corrosion rate of 0.175 mmpy.
3.1 Macro Structure and
Microstructure
Macrostructure showed that appearance of specimen
before and after corrosion test. Based on Figure 4 (a),
the specimen was clear, however the apprearance of
specimen was different after corrosion test Figure 4
(b). Microstructure were determined on two sides of
the specimen's surface to detect the characteristics of
corrosion forming on each surface. Based on the
microstructure results, the corrosion on the specimens
test were pitting and uniform corrosion. There was a
difference of pitting corrosion in specimens
immersed at difference of bacteria species. The
observation of the visible side of the specimen soaked
in E. coli bacteria, the pitting corrosion was a small
holes clumped. The pitting corrosion in
Pseudomonas was slightly larger and not clustered.
The last of pitting corrosion in Thiobacillus
ferroxidans addition was hole tends to be large and
not clustered. However, all the shape of corrosion was
a pitting corrosion. The observations on the bottom
side showed no difference, however it have a
relatively similar form of pitting corrosion.
Figure 4: Image of macrostructure aging treatment material
(left) before corrosion test (rigth) after corrosion test.
(a)
(b)
(c)
Figure 5: Microstructure of top of material aging treatment
with (a) kontrol, (c) Escherichia coli (e) Pseudomonas
fluorescens (g) Thiobacillus ferrooxidans, and on bottom of
specimen with (b) kontrol, (d) Escherichia coli (f)
Pseudomonas fluorescens (h) Thiobacillus ferrooxidans.
ISOCEEN 2018 - 6th International Seminar on Ocean and Coastal Engineering, Environmental and Natural Disaster Management
162
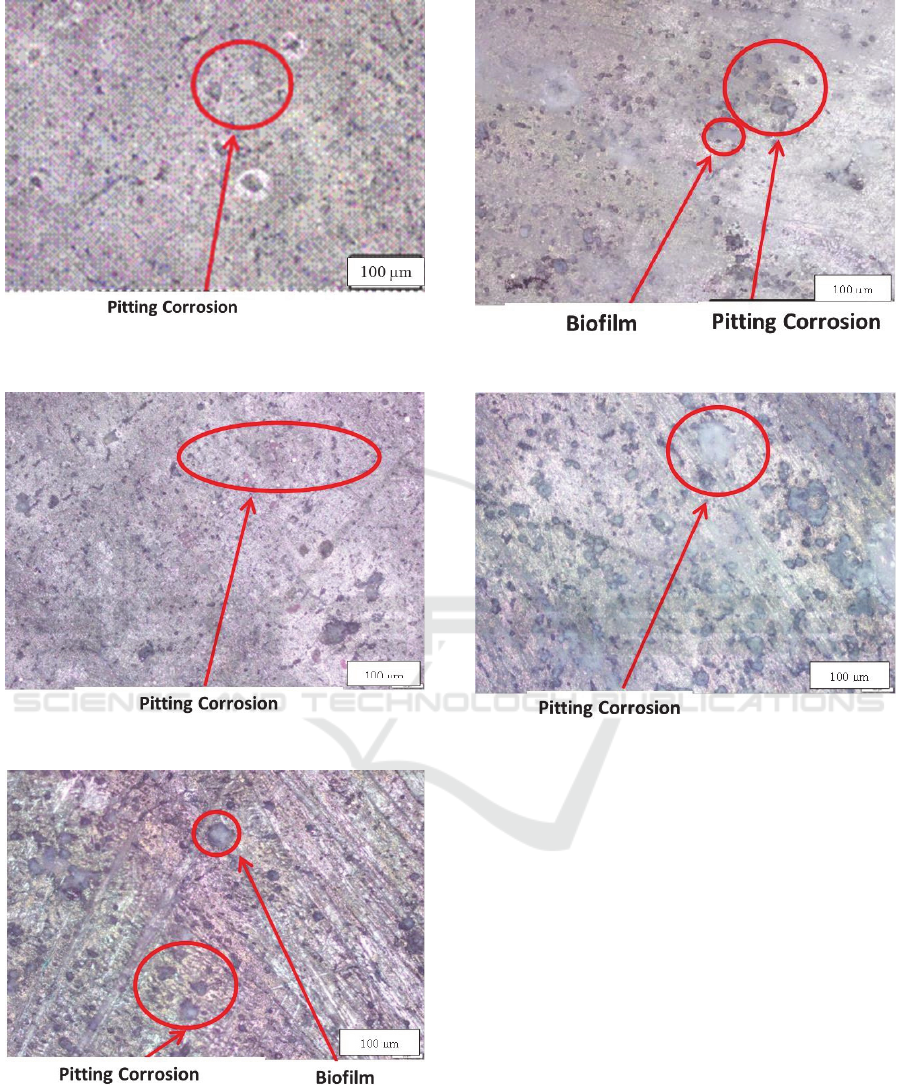
(d)
(e)
(f)
(g)
(h)
Figure 5: Microstructure of top of material aging treatment
with (a) kontrol, (c) Escherichia coli (e) Pseudomonas
fluorescens (g) Thiobacillus ferrooxidans, and on bottom of
specimen with (b) kontrol, (d) Escherichia coli (f)
Pseudomonas fluorescens (h) Thiobacillus ferrooxidans
(cont.).
4 CONCLUSION
The non aging treatment material has the highest
corrosion rate of 1.189 mmpy with the addition of
Thiobacillus ferrooxidans at salinity of 37‰ and the
lowest corrosion rate of 0.186 mmpy at salinity of
33‰. However, the aging treatment material has the
highest corrosion rate of 0.770 mmpy and the lowest
corrosion rate of 0.175 mmpy at similary condition.
Based on microstructure results, uniform and pitting
corrosion occurred on all specimens with the addition
of bacteria or without the addition of bacteria.
However, the bacterial corrosion rates were different.
Aging Treatment on Aluminium AA6063 against Bacterial Corrosion Resistance in Marine Environment
163

It was identified that aging treatment on Aluminium
AA6063 can reduce the bacterial corrosion rate or
increased the corrosion resistance.
REFERENCES
American Society for Testing and Materials. 2004. ASTM
D1141-98 Standard Practice for Artificial Sea Water
ASTM Society.
American Society for Testing and Materials. 2002. ASTM
G1-03 Standard Practice for Preparing, Cleaning, and
Evaluating Corrosion Test. ASTM Society.
American Society for Testing and Materials. 2004. ASTM
G31-72 Standard Practice for Laboratory Immersion
Corrosion Testing of Metals. ASTM Society.
H. Pratikno, H.S. Titah. IPTEK, 2017. The J for Technol
and Sci 28(2) 55-58.
H. Pratikno, H.S. Titah. 2016. Asian J of Appl Sci 9 120-
125.
H. Pratikno, H.S. Titah. 2016. Int J of ChemTech Res 9(12)
600-609.
J. S. J. Andersen, H. W. Zandbergen, J. Jansen, C. Tráholt,
U. Tundal, and O. Reiso, 1998. Acta Mater., 46[9].
J.P. Harley, L.M. Prescott. 2002. Laboratory exercises in
microbiology. Fifth Edition McGraw−Hill Companies
Texas.
J.R. Davis Alloying: Understanding the Basics J.R. Davis,
2001. p351-416 DOI:10.1361/autb2001p351 ASM
International.
M. A. Abdel-Rahman, Alaa Aldeen Ahmeda and Emad A.
Badawi. 2010. Defect and Diffusion Forum 303-304:
107-112.
T. Nuhia, Abu Seerb , A. M. Al Tamimia , M. Modarres, A.
Seibib, 2011. Reliability Analysis for Degradation
Effects of Pitting Corrosion in Carbon Steel Pipes M.
Procedia Engineering 10:1930–1935.
ISOCEEN 2018 - 6th International Seminar on Ocean and Coastal Engineering, Environmental and Natural Disaster Management
164
