
Characterization and Modification of Chitosan-reduced Graphene
Oxide Composite Films for Electrochemical Sensor
Rosnani Harahap
1*
, Irwana Nainggolan
1
and Zul Alfian
1
1
Department of Chemistry, Faculty of Mathematics and Sciences, University of Sumatera Utara, Medan, Indonesia
Keywords: Characterization, Chitosan, Electrochemical, Reduced Graphene Oxide, Sensor.
Abstract: Chitosan-reduced graphene oxide composite film was succesfully fabricated by electrodeposition method for
electrochemical sensor. In this regard, we have prepared chitosan-reduced graphene oxide composites using
a simple methodology, where chitosan-reduced graphene oxide composite can coat on the surface of screen-
printed copper electrode via a simple electrodeposition method. In this work, the characterization of the
composite film was intensively investigated by XRD and FT-IR method. The results of the XRD represented
reduced graphene oxide structure in 2θ, appeared at 26.61° with interlayer spacing was about 3.347Å. The
characterization of FT-IR confirmed the successful rGO presence in matrix polymer of chitosan.
1 INTRODUCTION
Electrochemical analysis is a quantitative or
qualitative analysis method based on the electrical
properties of a solution of the analyzed substance in
an electrochemical cell (Hendayana, 1994).
The electrochemical method is the most common
method in various fields of application, in particular
electrochemical sensors and biosensors developed for
pharmaceutical, food, agriculture and environmental
analysis because they have many advantages, such as
high sensitivity, cheap and easy to operate the tool
and quick response (Beitollahi et al., 2009), (Gajdar
et al., 2016) dan (Mazloum-Ardakani et al., 2011).
There are several advantages of electrochemical
sensors because electrodes can sense material present
in a sample without damaging the system with low
detection limits and high specificity. The active
sensing material on the electrode must act as a
catalyst and catalyze the chemical and biochemical
compound reaction in order to obtain a signal for
production. The combination of biosensors and
electrochemical sensors leads to a new type of sensor
called electrochemical biosensors, which applies
electrochemical methods to the construction and
work of biosensors (Kumar & Zou, 2005).
An electrochemical cell generally consists of three
types of electrodes, namely the working electrode, the
reference electrode, and the counter electrode. The
material used for the manufacture of electrochemical
cells must be able to be used over a wide temperature
range, stable form, resistant to solutions, organic
solvents and reagents, durable, and most importantly
is made of transparent material, so that the solution
and the electrodes can be observed (Sawyer, 1995).
Ideal working electrodes are electrodes that have a
reproducible surface area and low background
current.
(Vyskocil & Barek, 2009) have been determinated
of metronidazole based on electrochemical sensors
has many advantages ranging from high sensitivity,
low cost and easy to use and is widely used in
different fields, especially in analytical chemistry.
The growing importance of polymer-coated
nanomaterials from biological polymer sources has
brought chitosan to the fore, especially because of its
biological properties, which have been used in many
engineering and biological fields (Jayakumar et al.,
2010)
Chitosan is a biopolymer that has recognized
properties such as biodegradability and
biocompatibility (Muzzarelli, 2010). Due to its
biocompatibility, hydrophilicity, non-toxicity, good
mechanical stability, cost-effectiveness and
availability of reactive functional groups for chemical
modifications, chitosan has also been brought into
sharp focus as a suitable matrix (Kaur et al., 2019).
Over the past few years, chitosan-based sensor
materials have been widely developed, including in
conduction polymers, metal nanoparticles, and
oxidizing agents (Yang et al., 2010).
20
Harahap, R., Nainggolan, I. and Alfian, Z.
Characterization and Modification of Chitosan-reduced Graphene Oxide Composite Films for Electrochemical Sensor.
DOI: 10.5220/0010132500002775
In Proceedings of the 1st International MIPAnet Conference on Science and Mathematics (IMC-SciMath 2019), pages 20-23
ISBN: 978-989-758-556-2
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

Polymer composites have now developed into one
of the largest groups in material science and offer
significant potential for the production of advanced
materials in a wide variety of applications (G. Singh
et al., 2012). Once nanofillers are distributed on a
molecular scale within a polymer matrix, chemical
bonding interacts with the matrix. These chemical
functionalities have been found to be a practicable
and effective way to improve the dispersion of
graphene materials and the interfacial bonding
between graphene and the polymer matrix (Verdejo
et al., 2008). GO has been reported to be well
dispersed in the chitosan matrix on a molecular scale
because of interactions between the chitosan matrix
and the GO sheets (Yang et al., 2010).
In recent years, many electrochemical techniques
have used electrocatalysts, one of which is graphene.
Graphene is a hexagonal lattice that has a single atom
and has many researchers' attention because of its
new mechanical and electronic features (Ramanathan
et al., 2008). Graphene has unique electrical,
mechanical, and optical properties that researchers
around the world use to create advanced electronic
materials including transparent conductors and
ultrafast transistors (R. Singh et al., 2019). Due to its
extraordinary properties, this novel nanomaterial has
great potential in electrochemical sensor.
Several individuals such as (Muralidharan et al.,
2016) have investigated reduced chitosan graphene
oxide film, stating that it is clear from studies on
mechanical properties that the tensile strength and
module of chitosan composites have improved
drastically with the incorporation of rGO as a filler
reinforcement. The added graphene increases the
chitosan voltage strength from 39.7 MPa to 69.5 MPa.
Based on the description above, the researchers
were interested in conducting a study of chitosan-
graphene oxide reduced modification for
electrochemical sensor.
2 EXPERIMENTAL
2.1 Materials
Chitosan and reduced graphene oxide (RGO) was
obtained from Sigma Aldrich, Co., 3050 Spruce
Street St. Louis, MO 63103 USA 314-771-5765 with
82% deacetylation degree and molecular weight of
190,000-310,000 Da using the viscometer method.
All chemicals have a standard analytical level and are
used when received. Solvents and electrolyte
solutions are prepared using double distilled water
(DD) without further purification.
2.2 Fabrication of Chitosan/rGO
Composite Flm Modified
Screen-printed Copper Electrode
To achieve a mirror-like coating on the electrode, the
bare screen-printed copper electrode surface
(diameter = 5 mm) was washed by polishing with
0.05 μm alumina, and then ultrasonically with
distilled water.
Chitosan solution was prepared by dissolving 1 g
of chitosan powder in 100 mL of 1.0% (v/v) acetic
acid solution. Then, the chitosan solution was stirred
for 24 h and refrigerated at 4°C (Baccarin et al.,
2017). Reduced graphene oxide (rGO) has been
prepared in different concentration (50, 100, 150, 200
and 500 ppm) and has been distributed into 100 mL
of double distilled water.
2.3 Characterization Method
Electrodes were characterized using
Potentiostat/Galvanostat Electrochemical
Workstation Corrtest with model CS-350. X-ray
diffraction (XRD) were observed with a Shimadzu
XRD-6100 to analyze the crystallinity of graphene
oxide reduction. The Fourier Transform Infrared (FT
IR) spectra were performed using Shimadzu Pestige
21 for chitosan and chitosan / rGO.
3 RESULTS
3.1 Preparation of Chitosan-Reduced
Graphene Oxide (rGO) Electrodes
Preparation of chitosan/rGO electrodes was carried
out in several stages, namely stiring, sonication and
fabrication. Chitosan was made with a concentration
of 1% using 1% acetic acid solvent, while reduced
graphene oxide (rGO) under the Sigma Aldrich brand
was dissolved using aquabides and made with several
variations of 50, 100, 150, 200 and 250 ppm.
The reduced graphene oxide (rGO) has been
added to the chitosan solution for 2 h under stirring at
room temperature and sonicated to ensure
homogeneous solution for 30 min. Fabrication of
reduced chitosan-graphene oxide sensors was carried
out using the electrodeposition method. The
electrodes are then applied to the electrochemical cell
as a working electrode.
Characterization and Modification of Chitosan-reduced Graphene Oxide Composite Films for Electrochemical Sensor
21
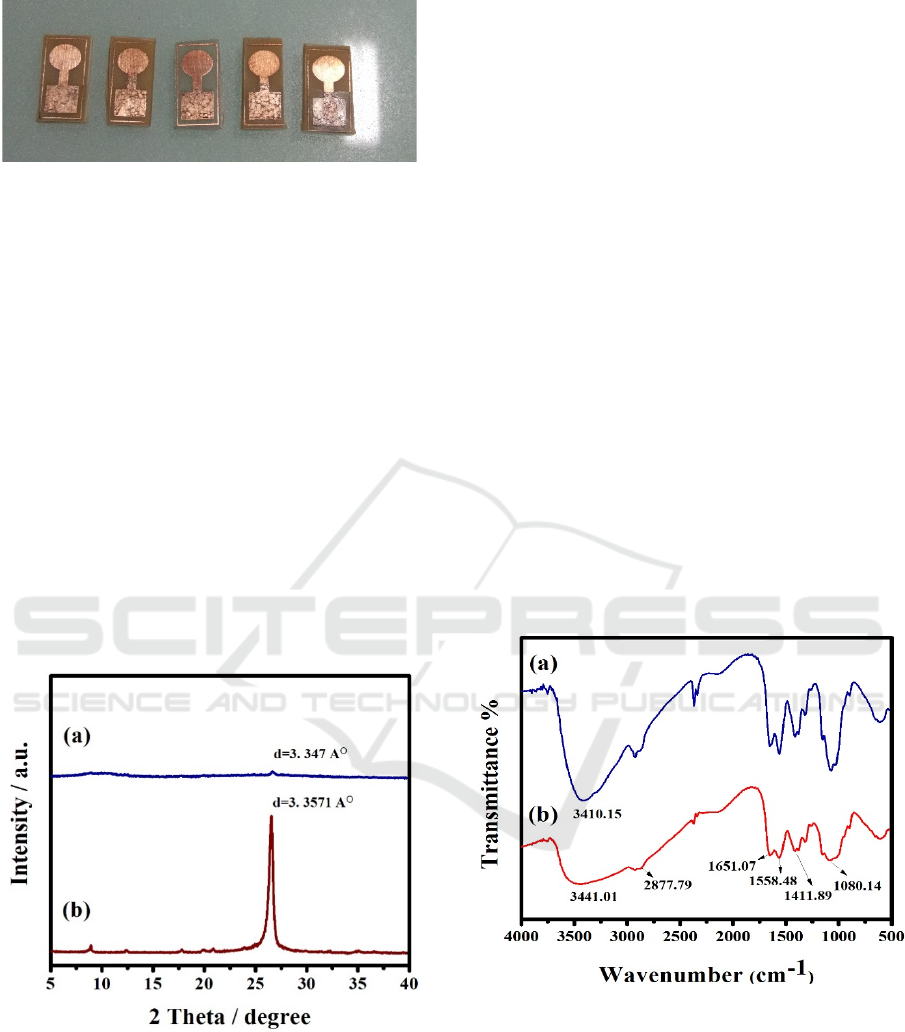
Figure 1: Chitosan/rGO Composite Film.
3.2 Characterization of Graphite and
rGO by X-ray Diffraction (XRD)
The composite film structure was further investigated
with X-ray diffraction (XRD). The typical peaks of
graphite (Fig.2b) observed at 26.53° with a d- spacing
3.3571 Å (Fig.2b). This is in accordance with
previous literature (Sandhya et al., 2018). The sharp
peak produced by graphite shows high crystallinity.
In Fig.1a, the XRD patterns of rGO shows the sharp
peak disappeared and move to higher 2θ angles at
26.61° with a d-spacing of 3.347 Å (Ali Umar et al.,
2013).
Due to the covalently bound oxygen atoms and the
displacement of sp3 hybridized carbon atoms above
and below the original graphene film, rGO films are
thicker than graphite films (Hassan et al., 2009). It has
been confirmed from the XRD patterns that the rGO
has been completely reduced.
Figure 2: XRD patterns of (a) rGO and (b) graphite.
3.3 Characterization of Chitosan and
Chitosan/rGO
Chitosan and rGO can be mixed well and forming a
homogeneously aqueous solution, and at room
temperature stable. And then, chitosan /rGO was cast
into substrates, and the films formed. Fig. 3 described
the chitosan and chitosan / rGO FT-IR spectra.
The chitosan spectrum (Fig.3a), the characteristic
hydroxyl group of OH peaks appeared at 3410.15 cm
-
1
and the absorption spectrum acquired at 1651.07 cm
-
1
and 1558.48 cm
-1
correspond to stretching vibration
of carboxylic group C=O of -NHCO- and the N-H
bending vibration of –NH
2
group (Yang, 2010).
Characteristic of CH
3
and C-H functional groups are
the bands at 1411.89 cm
-1
and 2877.79 cm
-1
. And
chitosan/rGO film, its spectrum shows a combination
of characteristics which includes the absorption peak
at 3441.01 cm
-1
(Fig.3b), assigned to extend NH
2
(amine) vibration group.
In the FT-IR peak of chitosan/rGO, The C-O
stretching chitosan intensity vibration is found to
have faded due to the interaction of chitosan and rGO.
Meanwhile, the intensities of C=C stretching
vibration peak at 1558.48 cm
-1
and the presence of
chitosan-derived N-H bonding. Deformation peak of
N-H from chitosan at 1411.89 cm
-1
. All these result
confirm that rGO presence on the chitosan polymer
matrix. And in the composite films, the chemical
structure of chitosan barely changes with the
increasing content of rGO, indicating that there was
primarily physical interaction but scarcely a chemical
reaction between chitosan and rGO.
Figure 3: FT-IR spectra of (a) chitosan and (b)
chitosan/rGO.
4 CONCLUSION
Fabrication of the chitosan-reduced graphene oxide
(rGO) in screen-printed copper electrode modified
was successfully by electrodeposition method and
was used for electrochemical sensor. The
characterizations of XRD confirmed the successful
IMC-SciMath 2019 - The International MIPAnet Conference on Science and Mathematics (IMC-SciMath)
22

formation of rGO. Furthermore, the spectra of FT-IR
also indicated rGO presence on the chitosan polymer
matrix composite film.
REFERENCES
Ali Umar, M. I., Yap, C. C., Awang, R., Hj Jumali, M. H.,
Mat Salleh, M., & Yahaya, M. (2013). Characterization
of multilayer graphene prepared from short-time
processed graphite oxide flake. Journal of Materials
Science: Materials in Electronics.
https://doi.org/10.1007/s10854-012-0920-5
Baccarin, M., Santos, F. A., Vicentini, F. C., Zucolotto, V.,
Janegitz, B. C., & Fatibello-Filho, O. (2017).
Electrochemical sensor based on reduced graphene
oxide/carbon black/chitosan composite for the
simultaneous determination of dopamine and
paracetamol concentrations in urine samples. Journal of
Electroanalytical Chemistry.
https://doi.org/10.1016/j.jelechem.2017.06.052
Beitollahi, H., Mazloum Ardakani, M., Naeimi, H., &
Ganjipour, B. (2009). Electrochemical characterization
of 2, 2?-[1, 2-ethanediylbis (nitriloethylidyne)]-bis-
hydroquinone-carbon nanotube paste electrode and its
application to simultaneous voltammetric
determination of ascorbic acid and uric acid. Journal of
Solid State Electrochemistry, 13(3), 353–363.
https://doi.org/10.1007/s10008-008-0553-z
Gajdar, J., Horakova, E., Barek, J., Fischer, J., & Vyskocil,
V. (2016). Recent Applications of Mercury Electrodes
for Monitoring of Pesticides: A Critical Review. In
Electroanalysis.
https://doi.org/10.1002/elan.201600239
Hassan, H. M. A., Abdelsayed, V., Khder, A. E. R. S.,
Abouzeid, K. M., Terner, J., El-Shall, M. S., Al-
Resayes, S. I., & El-Azhary, A. A. (2009). Microwave
synthesis of graphene sheets supporting metal
nanocrystals in aqueous and organic media. Journal of
Materials Chemistry. https://doi.org/10.1039/b906253j
Hendayana. (1994). Kimia Analitik Instrumen. IKIP.
Jayakumar, R., Menon, D., Manzoor, K., Nair, S. V., &
Tamura, H. (2010). Biomedical applications of chitin
and chitosan based nanomaterials - A short review. In
Carbohydrate Polymers.
https://doi.org/10.1016/j.carbpol.2010.04.074
Kaur, N., Bharti, A., Batra, S., Rana, S., Rana, S., Bhalla,
A., & Prabhakar, N. (2019). An electrochemical
aptasensor based on graphene doped chitosan
nanocomposites for determination of Ochratoxin A.
Microchemical Journal.
https://doi.org/10.1016/j.microc.2018.08.064
Kumar, S., & Zou, S. (2005). Electrooxidation of carbon
monoxide on gold nanoparticle ensemble electrodes:
Effects of particle coverage. Journal of Physical
Chemistry B. https://doi.org/10.1021/jp051981a
Mazloum-Ardakani, M., Beitollahi, H., Amini, M. K.,
Mirjalili, B. F., & Mirkhalaf, F. (2011). Simultaneous
determination of epinephrine and uric acid at a gold
electrode modified by a 2-(2,3-dihydroxy phenyl)-1, 3-
dithiane self-assembled monolayer. Journal of
Electroanalytical Chemistry, 651(2), 243–249.
https://doi.org/10.1016/j.jelechem.2010.09.020
Muralidharan, M. N., Shinu, K. P., & Seema, A. (2016).
Optically triggered actuation in chitosan/reduced
graphene oxide nanocomposites. Carbohydrate
Polymers, 144, 115–121.
https://doi.org/10.1016/j.carbpol.2016.02.047
Muzzarelli, R. A. A. (2010). Chitins and chitosans as
immunoadjuvants and non-allergenic drug carriers. In
Marine Drugs. https://doi.org/10.3390/md8020292
Ramanathan, T., Abdala, A. A., Stankovich, S., Dikin, D.
A., Herrera-Alonso, M., Piner, R. D., Adamson, D. H.,
Schniepp, H. C., Chen, X., Ruoff, R. S., Nguyen, S. T.,
Aksay, I. A., Prud’Homme, R. K., & Brinson, L. C.
(2008). Functionalized graphene sheets for polymer
nanocomposites. Nature Nanotechnology.
https://doi.org/10.1038/nnano.2008.96
Sandhya, P. K., Jose, J., Sreekala, M. S., Padmanabhan, M.,
Kalarikkal, N., & Thomas, S. (2018). Reduced
graphene oxide and ZnO decorated graphene for
biomedical applications. Ceramics International.
https://doi.org/10.1016/j.ceramint.2018.05.143
Sawyer, D. . et al. (1995). Experimental Electrochemistry
for Chemist (2nd editio). Wiley.
https://www.wiley.com/en-
id/Electrochemistry+for+Chemists,+2nd+Edition-p-
9780471594680
Singh, G., Choudhary, A., Haranath, D., Joshi, A. G.,
Singh, N., Singh, S., & Pasricha, R. (2012). ZnO
decorated luminescent graphene as a potential gas
sensor at room temperature. Carbon.
https://doi.org/10.1016/j.carbon.2011.08.050
Singh, R., Sansare, S., & Shidhaye, S. (2019). Biomedical
application of graphenes. In Biomedical Applications of
Nanoparticles. https://doi.org/10.1016/b978-0-12-
816506-5.00007-3
Verdejo, R., Barroso-Bujans, F., Rodriguez-Perez, M. A.,
De Saja, J. A., & Lopez-Manchado, M. A. (2008).
Functionalized graphene sheet filled silicone foam
nanocomposites. Journal of Materials Chemistry,
18(19), 2221–2226. https://doi.org/10.1039/b718289a
Vyskočil, V., & Barek, J. (2009). Mercury electrodes-
possibilities and limitations in environmental
electroanalysis. Critical Reviews in Analytical
Chemistry.
https://doi.org/10.1080/10408340903011820
Yang, Z., Peng, H., Wang, W., & Liu, T. (2010).
Crystallization behavior of poly(ε-
caprolactone)/layered double hydroxide
nanocomposites. Journal of Applied Polymer Science,
116(5), 2658–2667. https://doi.org/10.1002/app
Characterization and Modification of Chitosan-reduced Graphene Oxide Composite Films for Electrochemical Sensor
23
