
Analysis and Classification of Voice Pathologies using Glottal Signal
Parameters with Recurrent Neural Networks and SVM
Leonardo Forero Mendoza
1
, Manoela Kohler
2
, Cristian Muñoz
2
, Evelyn Conceição Santos Batista
2
and Marco Aurélio Pacheco
2
1
Universidade do Estado do Rio de Janeiro, Rio de Janeiro, Brazil
2
Pontifícia Universidade Católica do Rio de Janeiro (PUC-Rio), Rio de Janeiro, Brazil
Keywords: Classification of Vocal Folds Pathologies, Glottal Signal Parameters, Neural Network, Deep Learning.
Abstract: The classification of voice diseases has many applications in health, in diseases treatment, and in the design
of new medical equipment for helping doctors in diagnosing pathologies related to the voice. This work uses
the parameters of the glottal signal to help the identification of two types of voice disorders related to the
pathologies of the vocal folds: nodule and unilateral paralysis. The parameters of the glottal signal are obtained
through a known inverse filtering method and they are used as inputs to an Artificial Neural Network, RNN,
LSTM, a Support Vector Machine and also to a Hidden Markov Model, to obtain the classification, and to
compare the results, of the voice signals into three different groups: speakers with nodule in the vocal folds;
speakers with unilateral paralysis of the vocal folds; and speakers with normal voices, that is, without nodule
or unilateral paralysis present in the vocal folds. The database is composed of 248 voice recordings (signals
of vowels production) containing samples corresponding to the three groups mentioned. In this study a larger
database was used for the classification when compared with similar studies, and its classification rate is
superior to other studies, reaching 99.2%.
1 INTRODUCTION
The diagnosis of voice pathologies currently requires
invasive endoscopy procedures, such as laryn-
gostroboscopy or surgical microlaryngoscopy.
However, one wants to aid the pre-diagnosis of the
vocal folds pathologies with computer-based,
decision support diagnostic tools using voice signals.
Two pathologies related to the vocal folds will be
considered here: nodules and unilateral paralysis
(Roy N et al., 2017) (Francis D. O. At al., 2014).
Vocal cord nodules are growth on both vocal folds
caused by their repeated and incorrect usage, which
permits the developing of swollen spots on them. The
nodules will become larger and stiffer the longer the
vocal incorrect usage continues. Singers, teachers and
announcers are examples most probably to have this
kind of pathology in the vocal folds (Francis D. O. At
al., 2014). Unilateral vocal fold paralysis (UVFP)
occurs from a dysfunction of the recurrent laryngeal
or vagus nerve innervating the larynx. It causes a
characteristic breathy voice often accompanied by
swallowing disability, a weak cough, and the
sensation of shortness of breath. This is a common
cause of neurogenic hoarseness. When this paralysis
is properly evaluated and treated, normal speaking
voice is typically restored (Steffen N Pedrosa V. V.
and Kazuo R., Pontes P, 2009) (Behlau M, Pontes PP,
1995).
The aim here is to evaluate the use of glottal
signals (signal obtained just after the vocal folds and
before the vocal tract) for providing better
classification models of the pathologies discussed
above. The most common method for extracting
voice features is directly from the voice signal (Roy
N et al., 2017).
However, many researchers have looked for some
characteristics extracted from the glottal signal, not
only for identifying pathologies related to the vocal
folds, but also to other applications, as to synthesize
voice (Henrich, N., 2001)( Henrich N, d'Alessandro
C., 2014) or identifying vocal aging (Mendonza L.,
Vellasco M., Cataldo E., 2014).
The process of obtaining the glottal signal, from
the voice signal, has been facilitated due to the
development of algorithms which can perform an
Mendoza, L., Kohler, M., Muñoz, C., Batista, E. and Pacheco, M.
Analysis and Classification of Voice Pathologies using Glottal Signal Parameters with Recurrent Neural Networks and SVM.
DOI: 10.5220/0007250700190028
In Proceedings of the 11th International Conference on Agents and Artificial Intelligence (ICAART 2019), pages 19-28
ISBN: 978-989-758-350-6
Copyright
c
2019 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
19

inverse filtering from the voice signal, eliminating the
influence of the vocal tract (Software Aparat).
Different methods have been used to classify
diseases related to the voice, such as Hidden Markov
Models (HMM) (Francis D. O. At al., 2014),
Gaussian Mixture Models (GMM) and Artificial
Neural Networks (Steffen N Pedrosa V. V. and
Kazuo R., Pontes P, 2009), all of them using as inputs
Mel-Frequency Cepstral Coefficients (MFCC) and
parameters such as jitter and shimmer. However, as
most voice disorders are due to some disorder on the
vocal folds dynamics, it is best to work with
parameters extracted from the glottal signal, since the
signal is produced by the vocal folds.
In (Rosa I. S., 2005) (Londoño J., Llorente J.,
2010), (Wang X, Zhang J, Yan Y, 2009) the Mel-
frequency cepstral coefficients (MFCC) were used as
input parameters to classify pathologies. A database
composed of 12 recordings for men and women,
resulting in a maximum performance of 80%
accuracy (Londoño J., Llorente J., 2010). MFCC have
also been proved to be effective in speaker
recognition problems. However, their performance is
not as effective in the classification of voice
pathologies. In (Rosa I. S., 2005), several models for
the classification of voice pathologies are compared.
The best performance has been provided by a neural
network based model, differing from speaker
recognition applications where best results are
usually obtained with GMM and HMM. This is
probably because classification of voice pathologies
does not fully depend on temporal features of the
voice, and the pathology causes change in the voice
signal (Hariharan, M., 2009).
Therefore, the main objective of this work is to
evaluate the performance of voice pathologies
classification models based on parameters extracted
from the glottal signal. Additionally, a new database
was created, with a larger number of voice
recordings, which allows a better evaluation of the
influence of each parameter in the classification
performance.
This paper is organized as follows. Methods
section explains how the glottal signal is obtained and
how the features, extracted from the glottal signal, are
used. Proposed Methodology section presents the
three classifiers evaluated in this paper, so their
performance can be evaluated in voice pathologies
classification: Neural Network, Support Vector
Machine and Hidden Markov Model. Results section
presents the database details, results obtained and
their analysis. Lastly, Conclusions are outlined in the
final section.
2 METHODS
2.1 The Glottal Signal
The voice signal production, particularly the one
related to voiced sounds, e.g. vowels, starts with the
contraction-expansion of the lungs, generating a
pressure difference between the air in the lungs and
the air near the mouth. The airflow created passes
through the vocal folds, which oscillate in a frequency
called the fundamental frequency of the voice. This
oscillation modifies the airflow coming from the
lungs, changing it into air pulses. The pressure signal
formed by the air pulses is quasi-periodic and it is
called the glottal signal (M. D. O. Rosa., 2000).
2.2 Features Extracted from the
Glottal Signal
The glottal signal is obtained performing an inverse
filtering on the voice signal, which consists on
eliminating the influence of the vocal tract and the
voice radiation caused by the mouth, preserving the
glottal signal characteristics (Pulakka H., 2005). The
inverse filtering algorithm used here is the so-called
PSIAIF (Pitch Synchronous Iterative Adaptive
Inverse Filtering) (Mendonza L., Vellasco M.,
Cataldo E., 2014) (Pulakka H., 2005). It was chosen
due to its high performance and ease development.
There is a toolbox implementation in Matlab®, called
Aparat (Software Aparat), which was constructed
especially based on the PSIAIF method to obtain the
glottal signal and to extract its main features or
parameters. The parameters that will be used can be
divided into three groups: time domain, frequency
domain, and the ones that represent the variations of
the fundamental frequency. More details about these
parameters can be found in (Pulakka H., 2005).
2.2.1 Time-domain Parameters of the
Glottal Signal
The time domain parameters which can be
extracted from the glottal signal are described below
(Wang X, Zhang J, Yan Y, 2009) (Pulakka H., 2005).
Closing phase (Ko): describes the interval
between the instant of the maximum opening of
the vocal folds and the instant where they close
(M. D. O. Rosa., 2000);
Opening phase (Ka): describes the interval
between the instant where the vocal folds start
the oscillation up to their maximum opening
(M. D. O. Rosa., 2000);
ICAART 2019 - 11th International Conference on Agents and Artificial Intelligence
20

Open quotient (OQ): The ratio between the
total time of the vocal folds opening and the
total time of a cycle (or period) of the glottal
signal (T). It is inversely proportional to the
intensity of the voice. The smaller it is, the
higher the voice intensity (Wang X, Zhang J,
Yan Y, 2009) (Pulakka H., 2005);
Close quotient (CIQ): The ratio between the
closing phase parameter (Ko) and the total
length of a glottal pulse (T) (Pulakka H., 2005).
It is inversely proportional to the voice
intensity. The smaller it is, the higher the voice
intensity;
Amplitude quotient (AQ): The ratio between
the glottal signal amplitude (Av) and the
minimum value of the glottal signal derivative
[16]. It is re-lated to the speaker phonation
(Pulakka H., 2005);
Normalized amplitude quotient (NAQ): It is
calculated by the ratio between the amplitude
quotient (AQ) and the total time length of the
glottal pulse (T) (Pulakka H., 2005);
Open quotient defined by the Liljencrants-Fant
model (OQa): This is another opening quotient
but calculated by the Liljencrants-Fant model
for inverse filtering. Details about this model
can be found in (Wang X, Zhang J, Yan Y,
2009);
Quasi open quotient (QoQ): It is the
relationship between the glottal signal opening
at the exact instant of the oscillation and the
closing time. It has been used in some works to
classify emotions (Wang X, Zhang J, Yan Y,
2009);
Speed quotient (SQ): defined as the ratio of the
opening phase length to the closing phase
length (Pulakka H., 2005);
2.2.2 Frequency Domain Parameters
Difference between harmonics (DH12): Also
known as H1-H2, it is the difference between
the values of the first and second harmonics of
the glottal signal (Wang X, Zhang J, Yan Y,
2009) (Pulakka H., 2005). This parameter has
been used to measure vocal quality;
Harmonics richness factor (HRF): relates the
first harmonic (H1) with the sum of the energy
of the other harmonics (Hk) (Pulakka H.,
2005). It has also been used to measure vocal
quality;
2.2.3 Parameters that Represent Variations
and Perturbations in the Fundamental
Frequency
Jitter: variations in fundamental frequency
between successive vibratory cycles (Wang X,
Zhang J, Yan Y, 2009) (Pulakka H., 2005).
Changes in jitter may be indicative of
neurological or psychological difficulties (Roy
N et al., 2017);
Shimmer: variations in amplitude of the glottal
flow between successive vibratory cycles
(Wang X, Zhang J, Yan Y, 2009) (Pulakka H.,
2005). Changing the shimmer is found mainly
in the presence of mass lesions in the vocal
folds, such as polyps, edema, or carcinomas
(Roy N et al., 2017);
3 PROPOSED METHODOLOGY
FOR VOICE PATHOLOGIES
CLASSIFICATION
The proposed model used has two stages: the first
stage is the features extraction, where all the above
mentioned parameters from the glottal signal are
obtained; the second stage is the classification
module, where four algorithms have been selected to
classify different pathologies of the voice - a
multilayer perceptron (MLP) neural network, a
support vec-tor machine, Long short-term memory
(LSTM) and a Hidden Markov Model (HMM), for
comparison reasons. The proposed methodology is
illustrated in Figure 1 and each model is described in
the following sub-sections. A similar methodology
has been already applied for classifying voice aging
(Mendonza L., Vellasco M., Cataldo E., 2014), with
very good results.
Figure 1: Methodology used for the classification of voice
pathologies.
3.1 Inverse Filtering
For each vocal utterance a corresponding glottal
signal is obtained by inverse filtering (PSIAIF meth-
od) and the parameters are extracted using the Aparat
(Software Aparat) and Praat (Software Praat)
software. The following parameters are obtained:
Analysis and Classification of Voice Pathologies using Glottal Signal Parameters with Recurrent Neural Networks and SVM
21

fundamental frequency (fo), jitter, shimmer, Ko, Ka,
NAQ, AQ, CIQ, OQ1, OQ2, Oqa, Qoq, SQ1, SQ2,
DH12, and HRF. The parameters are separated
according to the groups to which they belong. In
particular, OQ was divided into OQ1 and OQ2, the
open quotients calculated from the so-called primary
and secondary openings of the glottal flow. The
difference between OQ1 and OQ2 is that OQ1 is
calculated from the closure of the glottal flow until
the closure of the next glottal flow, and OQ2 is
calculated from de opening until the closure of the
glottal flow; SQ, as well, was divided into speed
quotients calculated from the primary and secondary
openings of glottal signal. It is important to mention
that some parameters provide similar information,
but, in this phase, all of them will be considered.
3.2 Classification Module
For the classification of voice pathologies four
classifiers have been used: Artificial Neural
Networks (ANN), Support Vector Machine (SVM),
LSTM and Hidden Markov Models (HMM).
For the ANN classifier, a multi-layer perceptron
(MLP) structure, trained with the back-propagation
algorithm, was chosen, since it is a universal
approximator. Different topologies were examined
with different numbers of neurons in the hidden layer
to seek the best generalization performance. For the
SVM classifier, different kernels (polynomial, radial
basis function (RBF), and sigmoid) and different
values for the normalization coefficient (C) were
evaluated to determine the optimal settings. Final-ly,
an Estimate-Maximize (Baum Welch) approach was
used to train three HMM models (one HMM for each
output class), each one to maximize the likelihood of
the training data with respect to the unknown
parameters. To classify a sequence into one of the
three classes, the log-likelihood given by each model
is computed, and the most likely model defines the
class that the test sequence belongs to. Left-to-right
HMM models with five states and three Gaussian
mixtures were trained in order to obtain an optimal
classification rate.
4 RESULTS
4.1 Database
Most of the works on vocal folds diseases
classification just classify speakers into two groups:
speakers with disease (all kinds of disease) and
speakers with normal voices (Rosa I. S., 2005),
(Londoño J., Llorente J., 2010). In this work the type
of disease is also identified, helping in the indication
if the patient has nodule or paralysis on the vocal
folds, or neither one.
The developed database is composed of 248
records consisting of voices of both genders, women
and men, with different ages, and it is divided into
three groups: 12 speakers with nodule on the vocal
folds; 8 speakers with vocal folds paralysis; and 11
speakers with normal voices. Eight voice records
were taken from each speaker. This database was
obtained from a speech therapist in Rio de Janeiro
among people in treatment.
For the recordings is used a computer, the Doctor
speech software and an omnidirectional microphone.
The voices were recorded in a doctor's office.
The speakers belonging to the pathology groups
(nodule and paralysis) have different categories of the
disease in each group, as described in Tables 1 and 2.
The following tables describe the speakers in more
details.
Table 1: Speakers with Nodule on the Vocal Folds (F –
Female, M – Male).
Speaker
Gender
Age
Description of
the disease
Speaker 1
F
42
Bilateral nod-
ules causing a
small irregular
vocal cord
chink
Speaker 2
F
38
Bilateral nod-
ules with mid-
posterior chink
Speaker 3
F
24
Vocal nodules
with moderate
and severe
anterior and
posterior
irregular chinks
Speaker 4
F
53
Vocal nodules
with an
irregular vocal
cord chink
Speaker 5
F
53
Vocal nodules
with an
irregular vocal
cord chink
ICAART 2019 - 11th International Conference on Agents and Artificial Intelligence
22

Table 1: Speakers with Nodule on the Vocal Folds (F –
Female, M – Male) (Cont.).
Speaker 6
F
38
Vocal nodules
with mid-
posterior chink
Speaker 7
F
34
Vocal nodules
with mid-
posterior chink
Speaker 8
F
32
Fibrous nodules
- mid-posterior
chink - great
vocal effort
Speaker 9
F
29
Vocal nodules
with mid-
posterior chink
Speaker 10
F
33
Vocal
nodules with
an irregular
vocal cord
chink
Speaker 11
F
28
Vocal
nodules with
a slight
irregular
vocal cord
chink
Speaker 12
F
28
Vocal
nodules with
mid-posterior
chink
Table 2: Speakers with Vocal Folds Paralysis (F – Female,
M – Male).
Speaker
Gender
Age
Description
of the disease
Speaker 13
M
50
Right vocal
fold paralysis
with scar re-
traction in the
middle 1/3 -
anterior spin-
dle chink
(lar-yngeal
trauma
sequel)
Speaker 14
M
50
Right
hemilarynx
idiopathic
paralysiswith
slight vocal
cord bowing
Speaker 15
M
24
Right vocal
cord paralysis
with spindle
chink
Speaker 16
F
69
Right vocal
cord paralysis
in
paramedian
posi-tion with
a slight
bowing and a
slight
spindle chink
- paralytic
falsetto
Speaker 17
F
45
Left vocal
cord paralysis
in the left
median and
paramedian
positions –
no chinks
Speaker 18
F
43
Right
hemilarynx
idio-pathic
paralysis -
para-median
position
Speaker 19
M
66
Left vocal
cord paralysis
with a slight
bow-ing
(intubation
trauma)
Speaker 20
M
53
Right vocal
cord paralysis
in
paramedian
position - left
vocal fold
stiffness
Analysis and Classification of Voice Pathologies using Glottal Signal Parameters with Recurrent Neural Networks and SVM
23
Table 3: Speakers with No Disease (F - Female, M – Male).
Speaker
Gender
Age
Speaker 21
F
56
Speaker 22
M
30
Speaker 23
F
41
Speaker 24
M
46
Speaker 25
F
61
Speaker 26
M
35
Speaker 27
M
63
Speaker 28
M
48
Speaker 29
M
26
Speaker 30
F
56
Speaker 31
F
56
4.2 Analysis of the Parameters for
Classification
In order to evaluate the influence of each input
parameter in the classification of voice diseases, the
box-plot [24] function was used. The boxplot was
constructed for each of the parameters extracted from
the glottal signal, in order to see their influence in
each type of pathology or normal voices and to
compare their behavior. The three boxplots for each
group are related to Nodule, Paralysis and Normal
Voices, respectively. To facilitate the analysis and
better understand the parameters variation, their
boxplots were grouped and analyzed by each type of
parameter: Time-domain parameters, Frequency-
domain parameters and Parameters that Represent
Variations and Perturbations in the Fundamental
Frequency, as described in the following sub-sections
and in Figures 2 to 15.
4.2.1 Time-domain Parameters of the
Glottal Signal
The following figures shows the corresponding
boxplots for the so-called time-domain parameters
extracted from the glottal signal, where some
interesting observations can be extract-ed. The
parameter Ko, which shows the closing phase of the
vocal folds, is higher in normal voices than in voices
with the pathologies considered (Figure 2). OQ1,
OQ2, CIQ, AQ and NAQ parameters (Figure 4 to 8)
imply that normal voices have more intensity and
better voice quality when compared with pathologies.
The values of the parameters SQ1 and SQ2 are lower
in normal voices, which indicate a shortening in the
structure of the vocal folds when one has these
diseases, especially paralysis (Figures 11 and 12).
Figure 2: Closing phase(Ko).
Figure 3: Opening phase(Ka).
ICAART 2019 - 11th International Conference on Agents and Artificial Intelligence
24
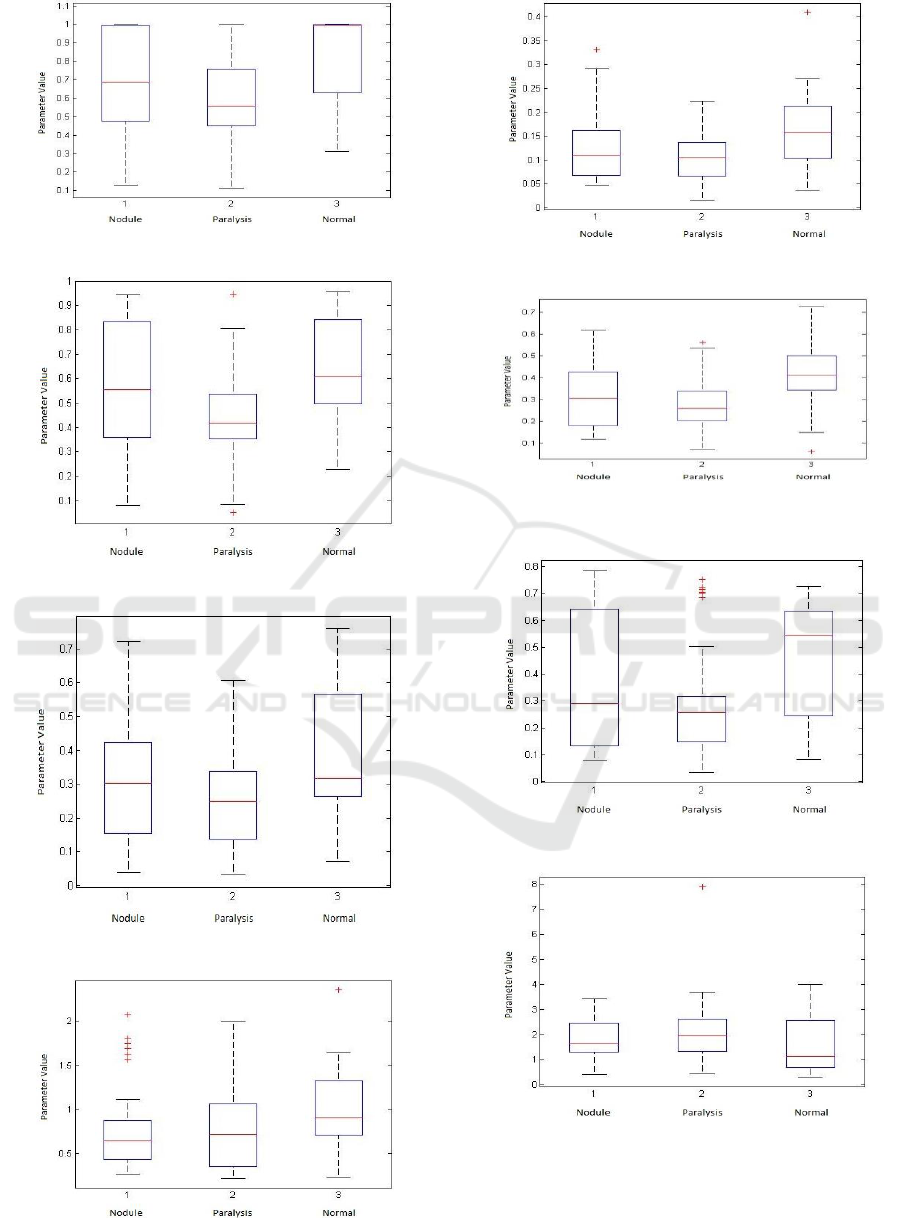
Figure 4: Open quotient(OQ1).
Figure 5: Open quotient (OQ2).
Figure 6: Close quotient(CIQ).
Figure 7: Amplitude quotient (AQ).
Figure 8: Normalized amplitude quotient (NAQ).
Figure 9: Open quotient defined by the Liljencrants-Fant
model (OQa).
Figure 10: Quasi opening quotient (QoQ).
Figure 11: Speed quotient (SQ1).
Analysis and Classification of Voice Pathologies using Glottal Signal Parameters with Recurrent Neural Networks and SVM
25
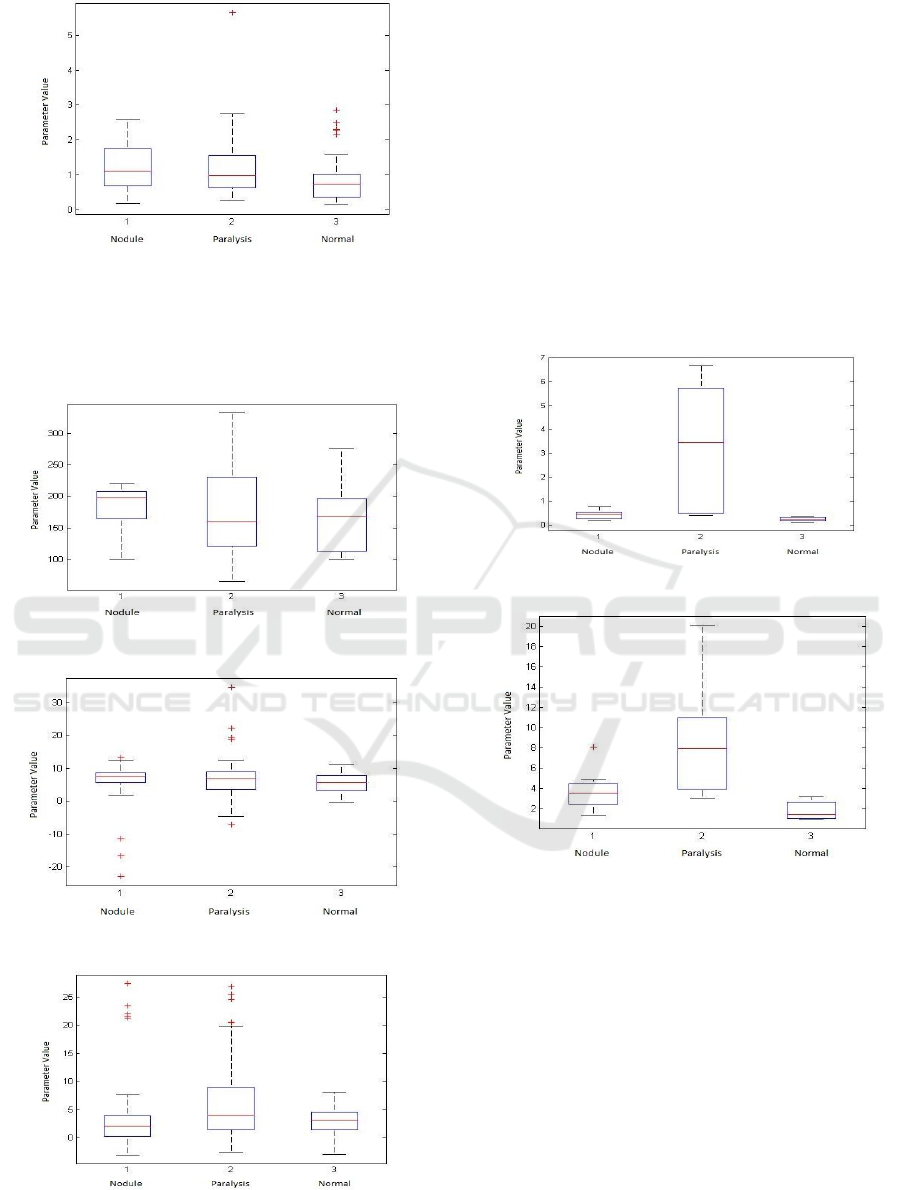
Figure 12: Speed quotient (SQ).
4.2.2 Frequency Domain Parameters
Figures 13 to 15 show the corresponding boxplots
related to the frequency domain parameters.
Figure 13: Fundamental Frequency (F0).
Figure 14: Difference between harmonics (DH12).
Figure 15: Harmonics richness factor (HRF).
The fundamental frequency (Fig. 13) has a wide
variation for voices with unilateral paralysis, showing
a greater disturbance in the vocal folds. Voices with
nodules have less variation of fundamental frequency
when compared with normal voices. Harmonics
richness factor (Fig. 15) changes a lot for unilateral
paralysis.
4.2.3 Parameters That Represent Variations
and Perturbations in the Fundamental
Frequency
Figures 16 and 17 present the boxplots of the
parameters directed related to the variations and
perturbations of the fundamental frequency.
Figure 16: Jitter.
Figure 17: Shimmer.
As can be seen from these boxplots, in the
pathologies cases, the function of the vocal folds is
greatly compromised, which is indicated by jitter and
shimmer parameters, as shown in Figures 16 and 17.
Jitter and shimmer parameters vary the most in the
voice when paralysis occurs. Jitter and Shimmer are
very high in voices with paralysis, proving to have
affected the most the vocal folds.
4.2.4 Analysis of the Classification Results
Classification of the pathologies was performed using
four different classifiers: ANN, SVM, LSTM and
HMM. For each classifier, three cases were
considered for the input parameters: (i) only the
ICAART 2019 - 11th International Conference on Agents and Artificial Intelligence
26

parameters extracted from the glottal signal, (ii) only
the MFCCs, and (iii) a combination of (i) and (ii). The
results for each input configuration are presented in
the following sub-sections.
Classification Results with the Parameters of the
Glottal Signal. In this case, the inputs of the
classifiers are 16 parameters of the glottal signal. The
original database was divided into training, validation
and test sets, where 70% of the database was used for
training, 20% for validation, and 10% for testing. For
ANN (after performing lots of tests varying the
number of the neurons in the hidden layer) the best
result was obtained with 8 processors in the hidden
layer.
Considering the SVM as the classifier, the best
result was achieved when a RBF kernel was used with
a regularization constant of C=1 and a Gaussian
standard deviation of σ=1.
Our model is a deep recurrent neural network with
two layers of 100 LSTM cells each. The bottommost
layer is the input layer where we inject each time
frame of an individual example at each time step. The
layer contains 13 units that would contain the
coefficients of the time frames. The next layer is two
layers are LSTM recurrent layers.
An Estimate-Maximize (Baum Welch) approach
was used to train three HMM models (one HMM for
each class), each one to maximize the likelihood of
the training data with respect to the unknown
parameters. To classify a sequence into one of the
three classes, the log-likelihood given by each model
is computed, and the most likely model defines the
class that the test sequence belongs to. Left-to-right
HMM models with five states and three Gaussian
mixtures were trained in order to obtain an optimal
classification rate.
The classifiers has three outputs: speakers with
nodule on the vocal folds, containing 93 voice
records, speaker with vocal folds paralysis,
containing 67 records, and speaker with normal voice,
containing 88 records.
4.2.5 Classification Results with
Mel-Frequency Cepstral Coefficients
(MFCCS)
Mel-Frequency Cepstral Coefficients (MFCCs) are
coefficients that collectively make up an MFC and are
derived from a type of cepstral representation of the
audio clip (a nonlinear "spectrum-of-a-spectrum").
MFCCs are common in speaker recognition, which is
the task of recognizing people from their voices. 12
MFC coefficients were used, the number most often
used in the literature (Rosa I. S., 2005) (Londoño J.,
Llorente J., 2010).
The inputs of the classifiers are, therefore, 12
MFC coefficients in this case. The original database
was divided into training, validation and test sets,
where 70% of the database was used for training, 20%
for validation, and 10% for testing, as in the previous
case. After lots of tests varying the number of the
neurons, the best result was achieved with 6
processors in the hidden layer. Considering the SVM
as the classifier, the best result was achieved when a
RBF kernel was used with a regularization constant
of C= 0,8 and a Gaussian standard deviation of σ=1.
HMM configuration is the same as above.
LSTM had the best performance with 100 cells.
4.2.6 Classification Results with Combining
MFCCs and Glottal Signal Parameters
In this third configuration, the input vector of the
classifiers is composed of 12 MFC coefficients and
16 parameters of the glottal signal. The original
database was also divided into training, validation
and test sets, where 70% of the database was used for
training, 20% for validation, and 10% for testing.
After lots of tests, the best ANN configuration was
obtained with 9 processors in the hidden layer.
Considering the SVM as the classifier, the best result
was achieved when a RBF kernel was used with a
regularization constant of C= 2 and a Gaussian
standard deviation of σ=1. HMM and LSTM
configuration is the same as above.
4.2.7 Discussion
Table 4 presents a summary of the results obtained
with all three classifiers and all four configurations of
input signals. As can be seen from the results in Table
4, the classification was successful with the glottal
signal parameters, despite having an imbalanced
database (fewer samples for voices with paralysis)
and factors such as gender and age difference
between speakers, reaching the conclusion that these
parameters are good discriminators for classifying
voice disorders.
When using only MFCC parameters, the best
result is obtained with the LSTM classifier, since its
stochastic behaviour can better handle temporal
samples.
The combination of MFCCs and glottal signal
parameters provided the best classification results,
with an increase of 1% in the average performance
when compared with the results with only glottal
signal parameters. The best classification
performance was obtained with the LSTM classifier,
Analysis and Classification of Voice Pathologies using Glottal Signal Parameters with Recurrent Neural Networks and SVM
27

with over 98,3% accuracy. The results were obtained
by Intel® Optimization for TensorFlow. The LSTM
network was run on a gold Xeon processor showing
faster speed than on a 1080 nvidea graphics board in
30% under the same conditions.
Table 4: Classification of Voice Pathologies.
Parameters
ANN
HMM
LSTM
SVM
Parameters of
the glottal
signal
95.8%
82%
97%
96.2%
MFCCs
75.2%
87%
94%
80%
Glottal signal
parameters
and MFCCs
96.6%
92%
98.3%
97.2%
LSTM
Xeon
1080 Nvidia
527 sec
742 sec
Training
time
5 CONCLUSIONS
The aim of this work was the classification of two
voice diseases: nodule and unilateral paralysis and the
evaluation of the impact of parameters from the
glottal signal on this identification. Three different
classifiers have been used, to compare their
performance: an Artificial Neural Network, a Support
Vector Machine, LSTM and a Hidden Markov
Model.
From the results obtained, it can be verified that
glottal signal parameters are more relevant to
discriminate pathologies of the vocal folds than
MFCC’s, when they are evaluated individually. This
is the case even when the database is composed of
individuals with different genders and ages, providing
an average accuracy over 99%.
ACKNOWLEDGMENTS
This work was supported by Intel Corporation.
REFERENCES
Roy, N., Holt, K. I., Redmond, S., Muntz, H., 2007,
Behavioral characteristics of children with vocal fold
nodules. J Voice. 21(2):157-68.
Francis, D. O.; McKiever, M. E.; Garrett, G., Jacobson, B.;
Penson, D. F., 2014, Assessment of Patient Experience
with Unilateral Vocal Fold Immobility: A Preliminary
Study, Journal of voice 28 (5), 636-643.
Steffen, N., Pedrosa, V. V., Kazuo, R., Pontes, P., 2009,
Modifications of Vestibular Fold Shape from
Respiration to Phonation in Unilateral Vocal
FoldParalysis, Journal of Voice, Vol. 25, No. 1, pp.
111-113.
Behlau, M., Pontes, P. P., 1995, Avaliação e tratamento das
disfonias. São Paulo: Lovise, in unilateral vocal fold
paralysis, Journal of Voice 13(1):36-42.
Henrich, N., 2001, Étude de la source glottique en voix
parlée et chantée modelisation et estimation, mesures
acoustiques et electroglottographiques, perception,
Thèse de doctorat de l'Université Paris 6 (PhD Thesis).
Henrich, N., d'Alessandro, C., Doval, B., Castellengo, M.
2005, Glottal Open quotient in singing: Measurements
and correlation with laryngeal mechanisms, vocal
intensity, and fundamental frequency, Journal of the
Acoustical Society of America 117(3), pp 1417-1430.
Mendonza, L., Vellasco, M., Cataldo, E., 2014,
Classification of Vocal Aging Using Parameters
Extracted From the Glottal Signal J Voice. 21(2):157-
68.
Software Aparat, http://aparat.sourceforge.net/index.php/
Main_Page, Helsinki University of Technology
Laboratory of Acoustics and Audio Signal Processing.).
Rosa, I. S., 2005, Análise acústica da voz de indivíduos na
terceira idade, Tese de mestrado Universidade de São
Paulo, São Carlos (in portuguese).
Londoño, J., Llorente, J., 2010, An improved method for
voice pathology detection by means of a HMM-based
feature space transformation Pattern Recognition,
Volume 43, Issue 9, September 2010.
Wang, X., Zhang, J., Yan, Y., 2009, Glottal Source
biometrical signature for voice pathology detection,
Speech Communication, 51 759-781.
Hariharan, M., Paulraj, M. P., Yaacob, S., 2009,
Identification of vocal fold pathology based on Mel
Frequency Band Energy Coefficients and singular
value decomposition Signal and Image Processing
Applications (ICSIPA), volume 514 – 517.
Rosa, M. D. O., Pereira, J. C., Grellet, M., 2000, Adaptive
Estimation of Residue Signal for Voice Pathology
Diagnosis, IEEE Trans. Biomedical Eng., Vol. 47, No.
1, Jan. 2000.
Pulakka H., 2005, Analysis of Human Voice Production
Using Inverse Filtering, High-Speed Imaging, and
Electroglottograph,. University of Technology
Helsinki.
Software Praat, http://www.fon.hum.uva.nl/praat/,
University of Amsterdam.
ICAART 2019 - 11th International Conference on Agents and Artificial Intelligence
28
