
Classification of Alzheimer's Disease using Machine Learning
Techniques
Muhammad Shahbaz
1
, Shahzad Ali
2 a
, Aziz Guergachi
3
, Aneeta Niazi
1
and Amina Umer
1
1
Department of Computer Science and Engineering, University of Engineering and Technology, Lahore-54890, Pakistan
2
Department of Information Technology, University of Education Lahore, Multan Campus, Multan, Pakistan
3
Department of Information Technology Management, TRS, Ryerson University, Toronto, ON, Canada
aminaumer@gmail.com
Keywords: Alzheimer’s Disease, Data Mining, Machine Learning, Healthcare, Classification, Neurodegeneration.
Abstract: Alzheimer's disease (AD) is a commonly known and widespread neurodegenerative disease which causes
cognitive impairment. Although in medicine and healthcare areas, it is one of the frequently studied diseases
of the nervous system despite that it has no cure or any way to slow or stop its progression. However, there
are different options (drug or non-drug options) that may help to treat symptoms of the AD at its different
stages to improve the patient’s quality of life. As the AD progresses with time, the patients at its different
stages need to be treated differently. For that purpose, the early detection and classification of the stages of
the AD can be very helpful for the treatment of symptoms of the disease. On the other hand, the use of
computing resources in healthcare departments is continuously increasing and it is becoming the norm to
record the patient’ data electronically that was traditionally recorded on paper-based forms. This yield
increased access to a large number of electronic health records (EHRs). Machine learning, and data mining
techniques can be applied to these EHRs to enhance the quality and productivity of medicine and healthcare
centers. In this paper, six different machine learning and data mining algorithms including k-nearest
neighbours (k-NN), decision tree (DT), rule induction, Naive Bayes, generalized linear model (GLM) and
deep learning algorithm are applied on the Alzheimer's Disease Neuroimaging Initiative (ADNI) dataset in
order to classify the five different stages of the AD and to identify the most distinguishing attribute for each
stage of the AD among ADNI dataset. The results of the study revealed that the GLM can efficiently classify
the stages of the AD with an accuracy of 88.24% on the test dataset. The results also revealed these techniques
can be successfully used in medicine and healthcare for the early detection and diagnosis of the disease.
1 INTRODUCTION
Neurodegenerative, continuous deterioration of
neurons, diseases are usually considered as a group of
disorders that damage the working competence of the
human nervous system intensely and progressively
(Scatena et al., 2007). Alzheimer’s disease (AD) is
the key public health concern throughout the world
and one of the most widespread neurodegenerative
disorder (Small, 2005). The AD is a cureless disease
because it has no diagnosis and treatment methods to
slow its progression or stop its onset (Unay et al.,
2010). The median survival duration of the patients
suffering from AD has been estimated to be only 3.1
years for the initial diagnosis of the probable AD, and
a
https://orcid.org/0000-0002-0608-9515
only 3.5 years for the initial diagnosis of possible AD
(Wolfson et al., 2001).
The frequency of AD occurrence is becoming
more common in late life, especially the people at the
age of greater than 65 are at high risk (Cummings et
al., 2014). At present, there are approximately 44
million victims of AD dementia throughout the
world. If the breakthroughs fail to identify the
prevention and diagnosis of the AD, it is expected to
be increased to a number of more than 100 million by
the year 2050 (Association et al., 2013; Touhy et al.,
2014). During the AD, degeneration of the brain
progresses with time. Therefore, the patients
suffering from the AD should be categorized into
different subgroups, depending upon the stage of the
296
Shahbaz, M., Ali, S., Guergachi, A., Niazi, A. and Umer, A.
Classification of Alzheimer’s Disease using Machine Learning Techniques.
DOI: 10.5220/0007949902960303
In Proceedings of the 8th International Conference on Data Science, Technology and Applications (DATA 2019), pages 296-303
ISBN: 978-989-758-377-3
Copyright
c
2019 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

disease. This division is critical because the patients
at different stages of the AD are required to be treated
differently, and the same medication cannot be used
for all of them (Gamberger et al., 2017). For that
purpose, the classification of different stages of the
AD can be very helpful for the treatment of symptoms
of the disease to improve the patient’s quality of life.
The use of computing resources in healthcare
departments is continuously increasing and it is
becoming the norm to record the patient data
electronically that was traditionally recorded on
paper-based forms. This yield increased access to
many electronic health records (EHRs) but 80% of
the data is unstructured. As a result, the processing of
the unstructured data is challenging and difficult
using database management software and other
traditional methods. Machine learning and data
mining tools and techniques can be applied to these
EHRs to enhance the quality and productivity of
medicine and healthcare centers (Alonso et al., 2018).
Data mining or knowledge discovery is the practice
of finding the unknown and useful patterns from
many pre-existing datasets. Such patterns are used to
understand the historical dataset, to classify new data
and generate summaries of data. In this way, data
mining helps in the discovery of the deeper patterns
in data, as well as classify or group records on the
basis of similarities or dissimilarities between them
(Sumathi and Sivanandam, 2006).
Since the last few decades, data mining has been
extensively used in many areas such as marketing,
retail, banking, stock market prediction, and medicine
and healthcare, etc. (Agarwal et al., 2018; Canlas,
2009; Thenmozhi and Deepika, 2014; Yang et al.,
2018). The algorithms used for data mining can be
applied to group different subjects based on the
similarities in their attributes (Eapen, 2004). Many
studies (Soni and Gandhi, 2010; Dipnall et al., 2016;
Ni et al., 2014 have shown that machine learning, and
data mining techniques have been significantly used
in medicine and healthcare research. The application
of data mining in medicine helps in the efficient
prognosis of several diseases, understanding the
classification of disease, specifically in neuroscience,
and biomedicine. The main aim of this research work
is to apply machine learning and data mining
techniques on the Alzheimer's Disease Neuroimaging
Initiative (ADNI) dataset to classify the different
stages of the AD. Moreover, the sub-objective of this
research work is to identify the most distinguishing
attribute for each of the different stages of the AD
among ADNI dataset.
2 DATA
2.1 Data Description
In this research work, the dataset has been collected
from The Alzheimer’s Disease Prediction of
Longitudinal Evolution (TADPOLE) challenge
which is available at https://tadpole.grand-
challenge.org. It is the dataset recorded from the
North American individuals, who participated in
ADNI. ADNI is the multicentre study aimed to
improve the clinical, genetic, biochemical, and
imaging biomarkers for early diagnosis of the AD. A
standard set of procedures and protocols is followed
during ADNI data collection, to avoid any
inconsistencies in the data (Weiner et al., 2013). The
TADPOLE data has been recorded for both male and
female participants, including mild cognitive
impairment subjects, old aged individuals, and AD
patients. The data contain records of participants’
examination carried out at 62 different sites, at
different monthly intervals, ranging from baseline
(i.e. 0 months) to 120 months, from July 2005 to May
2017.
2.2 Data Exploration
The original TADPOLE dataset contains 1,907
attributes for 1,737 participants. On the basis of
diagnosis, these participants have been divided into
five different classes namely Cognitively Normal
(CN), Early Mild Cognitive Impairment (EMCI),
Late Mild Cognitive Impairment (LMCI), Subjective
Memory Complaint (SMC), and Alzheimer’s Disease
(AD). The number of participants and their
examination records for each of the five stages of AD
included in the TADPOLE dataset are illustrated in
Table 1.
Table 1: Original TADPOLE dataset details.
Class
No. of Participants
No. of Records
CN
417
3,821
EMCI
310
2,319
LMCI
562
4,644
SMC
106
389
AD
342
1,568
TADPOLE dataset contains 12,741 examination
records of 1,737 participants. The dataset is unevenly
distributed among the five classes of participants, i.e.
LMCI and CN have more data as compared to AD,
EMCI, and SMC. The class SMC has the least data,
with just 106 participants having 389 examination
records (Table 1).
Classification of Alzheimer’s Disease using Machine Learning Techniques
297

2.3 Data Pre-Processing and Feature
Selection
In this research work, a subset of TADPOLE dataset
has been used. The examination records of 530
participants (106 participants from each AD stage)
have been selected out of 1,737 participants, to
perform experiments using different data mining
algorithms for the classification of five different
stages of the AD. In data mining techniques, the
completeness of data is very critical for precise
modeling. While the TADPOLE dataset contains a lot
of sparseness. Therefore, we have selected only those
attributes, which have maximum data coverage. An
attribute will have complete data coverage if it will
have a value for all the 12,741 instances of
examination records. But, there are only 41 attributes,
out of 1,907 attributes, which have a value for more
than 10,000 instances of examination records.
Furthermore, an analysis has been carried out
to remove redundancy from the 41 attributes having
enough data. There are 6 attributes in the TADPOLE
dataset, that indicate the time of participant’s
examination, including EXAMDATE (date of
examination), EXAMDATE_bl (date of first
examination), M (months of examination, to nearest
6 months as continuous), Month (months of
examination, to nearest 6 months as a factor),
Month_bl (fractional value of months of
examination), and VISCODE (participant’s visit
code, indicating months of examination). As all these
attributes indicate the time of the participant’s
examination. In this way, only one attribute namely
VISCODE has been selected for indicating the
participant’s examination date, and all other five
redundant attributes have been removed. Similarly,
the dataset contains two attributes for the participant’s
identification, including RID (participant Roaster ID)
and PTID (Participant ID). The attribute PTID has
been removed from the dataset to avoid redundant
IDs. After data analysis and pre-processing, 28
attributes have been selected for this investigation.
These attributes are divided into three
categories. The first category is demographics
attributes which include the general quantifiable
characteristics of participants. The second category is
cognitive assessment attributes which include the
attributes representing the cognitive behavior of a
participant. For that purpose, different cognitive
assessment tests are carried out and scores are
assigned to the participant based on their cognitive
abilities. Finally, the third category is clinical
assessment attributes which include the significant
biomarkers of the AD. These attributes have been
recorded after the clinical examination of all
participants. The list of demographic, cognitive
assessment and clinical assessment attributes with
their description that have been extracted for analysis
is illustrated in Table 2, 3 and 4, respectively.
Table 2: List of demographic attributes in the dataset used in this analysis.
Label
Description
Data Type
Units
RID
Participant’s Roaster ID.
Numeric
NA
AGE
Participant’s age.
Numeric
Years
PTGENDER
Participant’s gender
Nominal
NA
PTEDUCAT
Participant’s education
Numeric
Years
PTETHCAT
Participant’s ethnicity
Nominal
NA
PTRACCAT
Participant’s race
Nominal
NA
PTMARRY
Participant’s marital status.
Nominal
NA
VISCODE
Participant’s Visit code.
Nominal
NA
Years_bl
Participant’s year of examination.
Numeric
Years
SITE
A code indicating the site of a participant’s examination
Numeric
NA
Table 3: List of cognitive assessment attributes in the dataset used in this analysis.
Label
Description
Data Type
Units
CDRSB_bl
Clinical Dementia Rating Sum of Boxes (core)
Numeric
NA
ADAS11_bl
11 item-AD Cognitive Scale (score)
Numeric
NA
ADAS13_bl
13 item-AD Cognitive Scale (score)
Numeric
NA
MMSE_bl
Mini-Mental State Examination (score)
Numeric
NA
RAVLT_immediate_bl,
RAVLT_learning_bl,
RAVLT_forgetting_bl,
RAVLT_perc_forgetting_bl
Rey's Auditory Verbal Learning Test (scores for immediate
response, learning, forgetting and percentage forgetting)
Numeric
NA
FAQ_bl
Functional Activities Questionnaire
Numeric
NA
DATA 2019 - 8th International Conference on Data Science, Technology and Applications
298

Table 4: List of clinical assessment attributes in the dataset used in this analysis.
Label
Description
Data Type
Units
APOE4
APOE4 gene presence
Binary
NA
Hippocampus_bl
Volume of hippocampus
Numeric
mm
3
Ventricles_bl
Volume of ventricles
Numeric
mm
3
WholeBrain_bl
volume of Brain
Numeric
mm
3
Fusiform_bl
The volume of the fusiform gyrus.
Numeric
mm
3
Entorhinal_bl
The volume of the entorhinal cortex.
Numeric
mm
3
MidTemp_bl
The volume of the middle temporal gyrus.
Numeric
mm
3
ICV
Intra Cranial Volume
Numeric
mm
3
2.4 Data Partitioning
The dataset is divided into a training dataset (70% of
the entire dataset) and test datasets (30% of the entire
dataset). According to this division, the 2,164
examination records are used for training of the data
mining models, and the 927 examination records are
used for model testing. The data counts of training
and test dataset after pre-processing are given in
Table 5. It illustrates that all the five classes have a
different number of examination records. Because in
TADPOLE dataset, the number of examination
records is not the same for all participants.
Table 5: Data Counts of each AD stage for the Training and
Test Dataset.
AD Stage
Training Samples
Test Samples
CN
632
270
EMCI
448
192
LMCI
566
243
SMC
217
93
AD
301
129
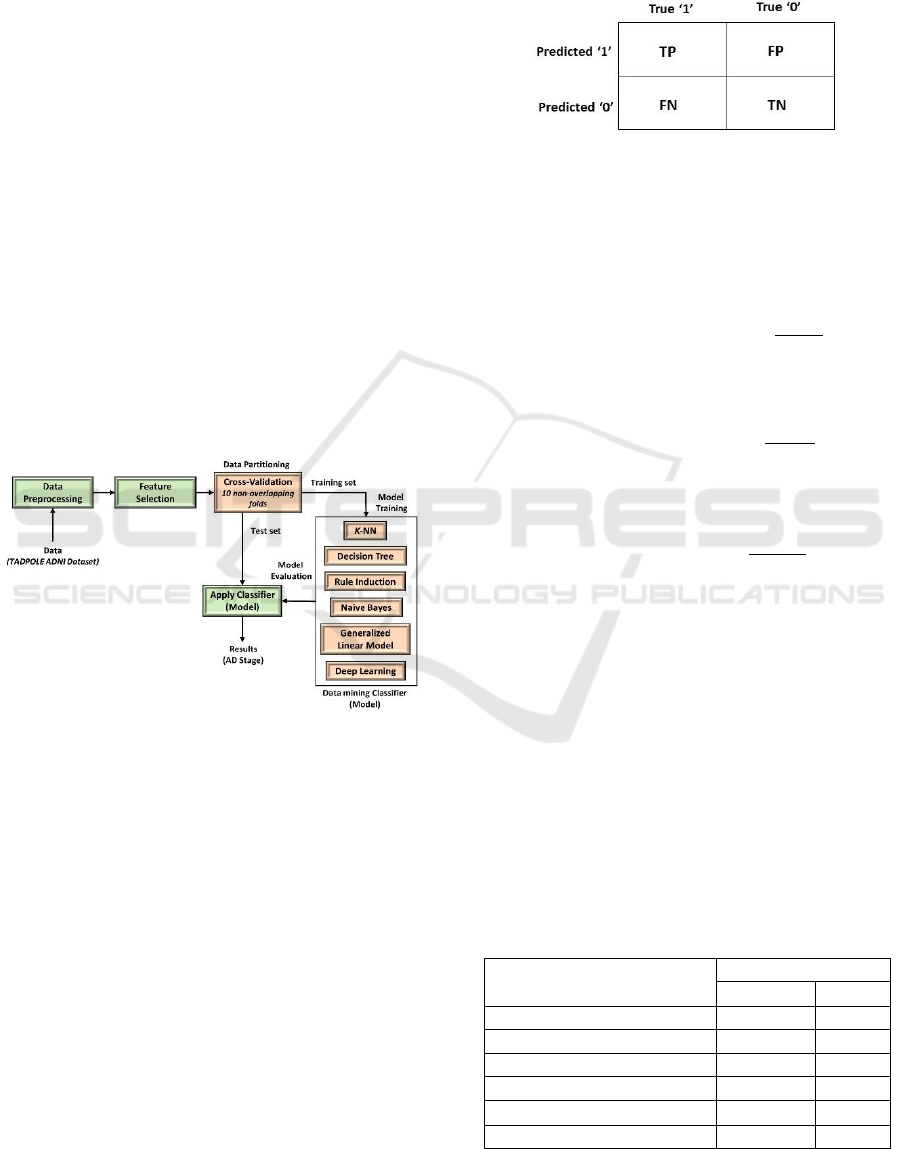
3 METHODOLOGY
In this research work, six different machine learning
and data mining algorithms including K-nearest
neighbors (K-NN), decision tree (DT), rule induction,
Naive Bayes, generalized linear model (GLM) and
deep learning algorithm are applied on ADNI dataset
in order to classify the five different stages of the AD.
In this investigation, rapidminer studio, one of the
famous data mining tools, is used for implementing
all these algorithms.
3.1 K-Nearest Neighbours (K-NN)
K-nearest neighbor algorithm is a simple data mining
technique used for both classification and regression
problems. K-NN classification algorithm assigns an
object to a specific class based on the majority classes
of its K neighbors. The value of K, a positive integer,
defines the number of neighbors to be considered for
polling (Zhang and Zhou, 2005). In this analysis, the
value of K is set to 11 which is selected using the trial
and error method.
3.2 Decision Tree (DT)
The decision tree algorithm is a predictive modeling
technique commonly used for classification in data
mining, statistics, and machine learning applications.
It classifies the dataset by computing the information
gain values for all attributes of a dataset. The leaf
nodes in a tree denote a class label while the branches
to these leaf nodes denote the combination of input
variables that lead to those class labels (Shahbaz et
al., 2013).
3.3 Rule Induction
The rule induction is a data mining algorithm, in
which a pruned set of rules is extracted from the
training data, based on maximum values of
information gain. The rules are in the form of ‘if-then’
statements (see Appendix B). Rule sets have an
advantage over decision trees, as they are simpler to
comprehend and can be represented in first-order
logic (Stepanova et al., 2018).
3.4 Naive Bayes Algorithm
The Naive Bayes algorithm is one of the data mining
and machine learning classification technique which
is based on the Bayesian theorem. It used to find the
probability of an attribute based on other known
probabilities that are related to the attribute. It uses
Gaussian probability densities for data modeling
(Thomas and Princy, 2016).
Classification of Alzheimer’s Disease using Machine Learning Techniques
299
3.5 Generalized Linear Model (GLM)
The generalized linear model (GLM) is a supervised
machine learning approach used for both
classification and regression problems. It is an
extension of traditional linear models. GLM classifies
the data based on the maximum likelihood between
attributes. It performs parallel computations and is an
extremely fast machine learning approach and works
very well for models with a limited number of
predictors (Guisan et al., 2002).
3.6 Deep Learning
Deep learning models are one of the machine learning
techniques. It is based on the multilayer feedforward
artificial neural networks, which are vaguely inspired
by the working of the biological nervous system or
the human brain. In this investigation, a deep neural
network with two hidden layers is trained using
backpropagation learning algorithm. The detailed
working of deep neural networks is given in (Levine
et al., 2018). The schematic representation of the
proposed methodology is illustrated in Figure 1.
Figure 1: Schematic representation of the proposed
methodology.
3.7 Model Evaluation – Confusion
Matrix
The performance of the classifier is usually evaluated
using a confusion matrix. It is a specific table which
is used to presents the true classes and classifier
predicted classes and illustrates the type of errors
made by the classifier. Confusion matrix for binary
classification (class ‘0’ and class ‘1’) is shown in Fig.
2. There are four different terminologies used in the
confusion matrix which are given as follows:
True Positive (TP) means classifier predicted
positive (‘1’) and it is true (‘1’).
True Negative (TN) means classifier
predicted negative (‘0’) and it is true (‘0’).
False Positive (FP) means classifier predicted
positive (‘1’) and it is false (‘0’).
False Negative (FN) means classifier
predicted negative (‘0’) and it is false (‘1’).
Figure 2: Confusion matrix for binary classification.
Accuracy, precision, and recall are the important
performance metrics that are calculated from a
confusion matrix for a classification model.
Accuracy means how often the classification
model correctly classifies the data samples?
(1)
Precision is the number of TP classes over the
sum of both the TP classes and FP classes.
(2)
Recall is the number of TP classes over the sum
of both the TP classes and FN classes.
(3)
4 RESULTS AND DISCUSSIONS
In this investigation, all the classification models are
trained with the 10 folds cross-validation of models
on the training dataset. Cross-validation is performed
with the training dataset, to avoid overfitting of the
models. The performance of the classifiers is
evaluated using the unseen test dataset. The
classification accuracy of the classifiers during the
validation period and the test period is illustrated in
Table 6.
Table 6: The classification accuracy of data mining
classifiers during Validation and test period.
Classifier
Accuracy (%)
Validation
Test
K-NN
73.10
43.26
Decision Tree
76.43
74.22
Rule Induction
92.47
69.69
Naive Bayes
79.44
74.65
Generalized Linear Model
92.75
88.24
Deep Learning
78.79
78.32
DATA 2019 - 8th International Conference on Data Science, Technology and Applications
300
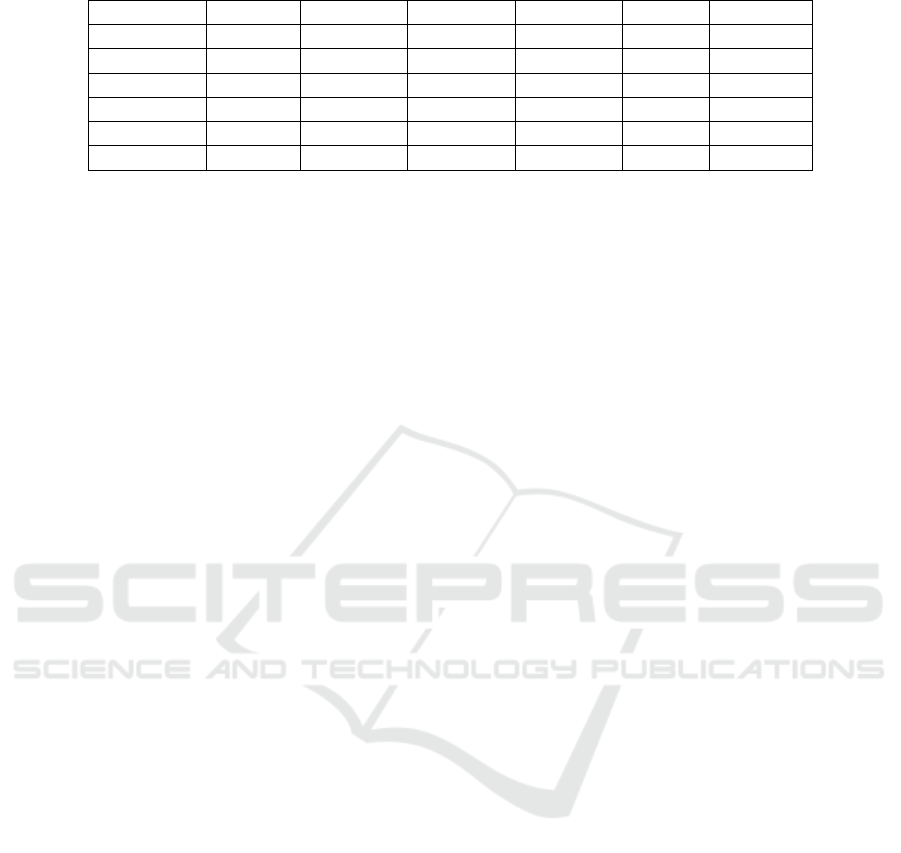
Table 7: Confusion matrix obtained by applying a generalized linear model to the test dataset.
True CN
True LMCI
True EMCI
True SMC
True AD
Precision
Pred. CN
255
6
0
25
0
89.16%
Pred. LMCI
0
209
4
0
0
98.12%
Pred. EMCI
0
28
174
17
0
79.45%
Pred. SMC
15
0
14
51
0
63.75%
Pred. AD
0
0
0
0
129
100 %
Recall
94.44%
86.01%
90.62%
54.84%
100 %
The results analysis indicates that the generalized
linear model outperforms other classifiers and gives
an accuracy of 88.24% during the test period.
Reasonable accuracies are obtained for deep learning
and Naive Bayes algorithms i.e. 78.32% and 74.65%
respectively. It can also be observed that the results
obtained on test data are quite close to the results
obtained from cross-validation period. This shows
that the developed models are not overfitted during
their training period. From the decision tree and rule
induction models, it has been observed that the most
distinguishing attribute for the five stages of the AD
is the CDRSB cognitive test, as it appears at the top
of the decision tree. The most distinguishing clinical
assessment attribute is the volume of the whole brain,
whereas the most distinguishing demographic
attribute is the age of the patient (see Appendices A
and B). The detailed results obtained by applying the
generalized linear algorithm during the test period is
illustrated in Table 7.
It can be observed that generalized linear model
correctly classified most of the unseen instances of
AD, CN, EMCI and LMCI classes, with the class
recall of 100.00%, 94.44%, 90.62%, and 86.01%
respectively, and class precision of 100.00%, 89.16%,
98.12%, and 79.45% respectively. The results of the
SMC class show misclassification of testing
instances. It can be observed from the above table that
25 out of 93 instances of SMC class have been
misclassified with the CN class. This is because the
patients belonging to SMC class have very similar
values of clinical assessment attributes as those who
belong to CN class. As well as, 28 out of 243
instances of the LMCI class have been misclassified
as EMCI. This is because the attribute values of
EMCI and LMCI classes overlap with each other.
5 CONCLUSION AND FUTURE
DIRECTIONS
Machine learning, and data mining techniques are
very helpful in medicine and healthcare studies for
early detection and diagnosis of several diseases. The
results of the research work illustrate that accuracy of
the GLM is 88.24% for the test period. The results
also indicate that the most distinguishing attributes
for the different stages of AD include the CDRSB
cognitive test among the cognitive assessment
attributes, the volume of the whole brain among the
clinical assessment attributes and age of the patient
among the demographic attributes. Furthermore, the
results proved that the machine learning, and data
mining techniques can be successfully used in the
early detection, prediction, and diagnosis of several
diseases. The accuracy of the AD stages classification
could be further improved by increasing the number
of instances for EMCI and SMC classes so that the
model can be trained with sufficient and balanced
data for all classes.
ACKNOWLEDGMENTS
The authors are thankful to the Alzheimer's Disease
Neuroimaging Initiative (ADNI) data committee for
providing access to their AD dataset.
REFERENCES
Agarwal, N., Koti, S. R., Saran, S., and Kumar, A. S.
(2018). Data mining techniques for predicting dengue
outbreak in geospatial domain using weather
parameters for new delhi, india. CURRENT SCIENCE,
114(11):2281–2291.
Alonso, S. G., De La Torre-D´ıez, I., Hamrioui, S., L´opez-
Coronado, M., Barreno, D. C., Nozaleda, L. M., and
Franco, M. (2018). Data mining algorithms and
techniques in mental health: a systematic review.
Journal of medical systems, 42(9):161.
Association, A. et al. (2013). 2013 alzheimer’s disease facts
and figures. Alzheimer’s & dementia, 9(2):208–245.
Canlas, R. (2009). Data mining in healthcare: Current
applications and issues. School of Information Systems
& Management, Carnegie Mellon University,
Australia.
Cummings, J. L., Morstorf, T., and Zhong, K. (2014).
Alzheimer’s disease drug-development pipeline: few
Classification of Alzheimer’s Disease using Machine Learning Techniques
301

candidates, frequent failures. Alzheimer’s research &
therapy, 6(4):37.
Dipnall, J. F., Pasco, J. A., Berk, M., Williams, L. J., Dodd,
S., Jacka, F. N., and Meyer, D. (2016). Fusing data
mining, machine learning and traditional statistics to
detect biomarkers associated with depression. PloS one,
11(2): e0148195.
Eapen, A. G. (2004). Application of data mining in medical
applications. Master’s thesis, University of Waterloo.
Gamberger, D., Lavraˇc, N., Srivatsa, S., Tanzi, R. E., and
Doraiswamy, P. M. (2017). Identification of clusters of
rapid and slow decliners among subjects at risk for
alzheimer’s disease. Scientific reports, 7(1):6763.
Guisan, A., Edwards Jr, T. C., and Hastie, T. (2002).
Generalized linear and generalized additive models in
studies of species distributions: setting the scene.
Ecological modelling, 157(2-3):89–100.
Levine, S., Pastor, P., Krizhevsky, A., Ibarz, J., and Quillen,
D. (2018). Learning hand-eye coordination for robotic
grasping with deep learning and large-scale data
collection. The International Journal of Robotics
Research, 37(4-5):421–436.
Ni, H., Yang, X., Fang, C., Guo, Y., Xu, M., and He, Y.
(2014). Data mining-based study on sub-mentally
healthy state among residents in eight provinces and
cities in china. Journal of Traditional Chinese
Medicine, 34(4):511–517.
Scatena, R., Martorana, G. E., Bottoni, P., Botta, G.,
Pastore, P., and Giardina, B. (2007). An update on
pharmacological approaches to neurodegenerative
diseases. Expert opinion on investigational drugs,
16(1):59–72.
Shahbaz, M., Guergachi, A., Noreen, A., and Shaheen, M.
(2013). A data mining approach to recognize objects in
satellite images to predict natural resources. In IAENG
Transactions on Engineering Technologies, pages 215–
230. Springer.
Small, D. H. (2005). Acetylcholinesterase inhibitors for the
treatment of dementia in alzheimer’s disease: do we
need new inhibitors? Expert opinion on emerging
drugs, 10(4):817–825.
Soni, N. and Gandhi, C. (2010). Application of data mining
to health care. International Journal of Computer
Science and its Applications.
Stepanova, D., Gad-Elrab, M. H., and Ho, V. T. (2018).
Rule induction and reasoning over knowledge graphs.
In Reasoning Web International Summer School, pages
142–172. Springer.
Sumathi, S. and Sivanandam, S. (2006). Introduction to
data mining principles. Introduction to data mining and
its applications, pages 1–20.
Thenmozhi, K. and Deepika, P. (2014). Heart disease
prediction using classification with different decision
tree techniques. International Journal of Engineering
Research and General Science, 2(6):6–11.
Thomas, J. and Princy, R. T. (2016). Human heart disease
prediction system using data mining techniques. In
2016 International Conference on Circuit, Power and
Computing Technologies (ICCPCT), pages 1–5. IEEE.
Touhy, T. A., Jett, K. F., Boscart, V., and McCleary, L.
(2014). Ebersole and Hess’ Gerontological Nursing and
Healthy Aging, Canadian Edition-E-Book. Elsevier
Health Sciences.
Unay, D., Ekin, A., and Jasinschi, R. S. (2010). Local
structure-based region-of-interest retrieval in brain mr
images. IEEE Transactions on Information technology
in Biomedicine, 14(4):897–903.
Weiner, M. W., Veitch, D. P., Aisen, P. S., Beckett, L. A.,
Cairns, N. J., Green, R. C., Harvey, D., Jack, C. R.,
Jagust, W., Liu, E., et al. (2013). The alzheimer’s
disease neuroimaging initiative: a review of papers
published since its inception. Alzheimer’s & Dementia,
9(5):e111–e194.
Wolfson, C., Wolfson, D. B., Asgharian, M., M’lan, C. E.,
Østbye, T., Rockwood, K., and Hogan, D. f. (2001). A
reevaluation of the duration of survival after the onset
of dementia. New England Journal of Medicine,
344(15):1111–1116.
Yang, J., Li, J., and Xu, Q. (2018). A highly efficient big
data mining algorithm based on stock market.
International Journal of Grid and High Performance
Computing (IJGHPC), 10(2):14–33.
Zhang, M.-L. and Zhou, Z.-H. (2005). A k-nearest
neighbour based algorithm for multi-label
classification. In Granular Computing, 2005 IEEE
International Conference on, volume 2, pages 718–721.
IEEE.
APPENDIX
Decision Tree Model
CDRSB_bl>0.250
| CDRSB_bl>2.750
| | MMSE_bl>26.500
| | | AGE>75.300: LMCI {AD=0, CN=0, EMCI=0,
LMCI=13, SMC=0}
| | | AGE≤75.300: EMCI {AD=0, CN=0, EMCI=8,
LMCI=0, SMC=0}
| | MMSE_bl≤26.500: AD {AD=254, CN=0,
EMCI=5, LMCI=0, SMC=0}
| CDRSB_bl≤2.750
| | MMSE_bl>23.500
| | | RAVLT_perc_forgetting_bl>-9.167
| | | | AGE>88.850: AD {AD=5, CN=0, EMCI=0,
LMCI=0, SMC=0}
| | | | AGE≤88.850
| | | | | Fusiform_bl>23133.500
| | | | | | PTETHCAT = Hisp/Latino: EMCI
{AD=0, CN=0, EMCI=2, LMCI=0, SMC=0}
| | | | | | PTETHCAT = Not Hisp/Latino: SMC
{AD=0, CN=0, EMCI=0, LMCI=0, SMC=5}
| | | | | Fusiform_bl≤23133.500
| | | | | | Ventricles_bl>9770.500
| | | | | | | ICV_bl>1268370
DATA 2019 - 8th International Conference on Data Science, Technology and Applications
302

| | | | | | | | RAVLT_immediate_bl>52.500:
EMCI {AD=0, CN=0, EMCI=78, LMCI=0, SMC=5}
| | | | | | | | RAVLT_immediate_bl≤52.500:
LMCI {AD=23, CN=32, EMCI=344, LMCI=553,
SMC=3}
| | | | | | | | SITE≤64.500: SMC {AD=0, CN=0,
EMCI=0, LMCI=0, SMC=4}
| | | | | | Ventricles_bl≤9770.500
| | | | | | | SITE>67.500: EMCI {AD=0, CN=0,
EMCI=6, LMCI=0, SMC=0}
| | | | | | | SITE≤67.500: CN {AD=0, CN=14,
EMCI=0, LMCI=0, SMC=0}
| | | RAVLT_perc_forgetting_bl≤-9.167: SMC
{AD=0, CN=0, EMCI=0, LMCI=0, SMC=5}
| | MMSE_bl≤23.500: AD {AD=19, CN=0,
EMCI=0, LMCI=0, SMC=0}
CDRSB_bl≤0.250
| AGE>69.550
| | SITE>147: SMC {AD=0, CN=0, EMCI=0,
LMCI=0, SMC=6}
| | SITE≤147
| | | WholeBrain_bl>1234525: SMC {AD=0,
CN=0, EMCI=0, LMCI=0, SMC=6}
| | | WholeBrain_bl≤1234525
| | | | FAQ_bl>2.500: SMC {AD=0, CN=0,
EMCI=0, LMCI=0, SMC=5}
| | | | FAQ_bl≤2.500
| | | | | ADAS13_bl>19.500: SMC {AD=0, CN=0,
CN=1, EMCI=0, LMCI=0, SMC=5}
| | | | | | | | ICV_bl≤1814440: CN {AD=0,
CN=566, EMCI=0, LMCI=0, SMC=77}
| | | | | | | RAVLT_learning_bl≤0.500: SMC
{AD=0, CN=0, EMCI=0, LMCI=0, SMC=3}
| | | | | | MMSE_bl≤25: SMC {AD=0, CN=0,
EMCI=0, LMCI=0, SMC=4}
| AGE≤69.550
| | PTEDUCAT>10.500
| | | AGE>64.050: SMC {AD=0, CN=0, EMCI=0,
LMCI=0, SMC=83}
| | | AGE≤64.050
| | | | PTGENDER = Female: SMC {AD=0, CN=0,
EMCI=0, LMCI=0, SMC=2}
| | | | PTGENDER = Male: CN {AD=0, CN=12,
EMCI=0, LMCI=0, SMC=0}
| | PTEDUCAT≤10.500: CN {AD=0, CN=7,
EMCI=0, LMCI=0, SMC=0}
Rule Induction Model
if CDRSB_bl≤0.750 and Fusiform_bl≤16443.500
and ADAS13_bl≤15.500 and
RAVLT_immediate_bl>32 then CN (0/282/7/0/16)
if RAVLT_perc_forgetting_bl>86.607 and
MMSE_bl>26.500 and ADAS13_bl>19.500 then
LMCI (0/0/4/131/0)
if MidTemp_bl>20054 and CDRSB_bl>0.250 and
ICV_bl≤1697385 and RAVLT_forgetting_bl>3.500
then EMCI (0/0/169/4/1)
if Years_bl>2.471 and FAQ_bl>0.500 and SITE≤55
then LMCI (6/0/6/121/0)
if CDRSB_bl≤0.750 and ICV_bl>1489935 and
Years_bl>0.990 and RAVLT_immediate_bl>41.500
then CN (0/137/0/0/14)
if Hippocampus_bl≤7242.500 and FAQ_bl≤4.500
and Entorhinal_bl≤3037 and Fusiform_bl≤16003.500
then LMCI (0/0/0/66/0)
if MMSE_bl≤26.500 and FAQ_bl>4.500 then AD
(285 / 0 / 0 / 25 / 0)
fif CDRSB_bl≤0.750 and ICV_bl>1562475 and
WholeBrain_bl≤1121605 and PTGENDER = Male
then CN (0/106/0/1/0)
if CDRSB_bl>0.750 and ADAS11_bl≤8.500 then
LMCI (0/0/3/42/0)
if ADAS13_bl≤13.500 and SITE≤34.500 and
MidTemp_bl>20055.500 then SMC (0/5/0/0/48)
if ADAS13_bl>13.500 and
RAVLT_learning_bl>4.500 and FAQ_bl >0.500 then
EMCI (0/0/40/0/0)
if ADAS11_bl>12.500 then LMCI (0/3/0/37/0)
if Ventricles_bl≤28863.500 and MMSE_bl>28.500
and AGE>70.600 then CN (0/56/0/0/3)
if Fusiform_bl>18434 and Fusiform_bl≤18888.500
then EMCI (0/0/36/0/2)
if FAQ_bl>1.500 and MidTemp_bl≤19842 then
LMCI (0/0/0/36/0)
if MidTemp_bl≤19923.500 and
WholeBrain_bl>977838 then SMC (0/1/0/0/41)
if Ventricles_bl≤8152 then CN (0/19/0/0/2)
if PTEDUCAT≤14.500 and ADAS13_bl>17.500
then EMCI (0/0/24/0/0)
if Ventricles_bl≤30608 and CDRSB_bl>0.250 then
LMCI (0/0/0/32/0)
if AGE≤70.250 then SMC (0/1/0/0/17)
if ADAS13_bl≤11.500 and Ventricles_bl>40723
then CN (0/17/0/0/0)
if Hippocampus_bl>8041 then EMCI (0/0/13/0/0)
if ADAS13_bl>17.500 then AD (10/0/0/0/0)
if AGE≤78 then SMC (0/0/0/0/8)
else LMCI (0/0/0/1/0)
Classification of Alzheimer’s Disease using Machine Learning Techniques
303
