
Characterization of Geopolymer Paste based on Fly Ash and Bottom
Ash in PLTU Kaltim Teluk using Sodium Hydroxide (NaOH)
Andika Ade Indra Saputra, Adrian Gunawan, Latu Anggary Putri Sukandar, Yoel Gallaran Salurante,
Intan Dwi Wahyu Setyo Rini
Institut Teknologi Kalimantan
intan@lecturer.itk.ac.id
Keywords: Bottom Ash, Fly Ash, Geopolymer Paste, Replacement Cement
Abstract: Cement Industry in Indonesia has grown significantly every year. Limestone is one of the raw cement
substitution material which has rapidly grown. The cement production process released carbon dioxide (CO2),
which resulted in global warming. As an alternative material substitution, fly ash (FA) and bottom ash (BA)
as the remained coal was generally used for cement production. PLTU Kaltim Teluk, one of the steam power
plant which located in Balikpapan, East Kalimantan, produced 150 tons of FA in a day. However, FA and BA
were laid out in the landfill and had not managed well. Actually, FA and BA had potential material as a
substitution of cement in the paving block ingredients. Adding an alkaline activation, which consists of
sodium silicate (Na2SiO3) and sodium hydroxide (NaOH), could be increased the compressive strength,
especially for paste using 100% FA. The property of FA was characterized by Scanning Electron Microscope
(SEM) and X-Ray Fluorescence (XRF). A compressive strength test was carried out on paste at the age of 3
and 14 days. The result showed that the highest compressive strength at 14 days was 13.93 MPa with 12 M
NaOH. This research was conducted as an alternative solution to the waste of PLTU Kaltim Teluk as a
substitute for cement in Balikpapan.
1 INTRODUCTION
Cement is a binder that is able to bind solid materials
such as sand and stone into a compact unit when it
mixed with water. The main raw materials in the
process of making cement are limestone and clay; the
main compounds in cement come from both of the
materials (Rahadja, 2010). The ancillary materials of
cement are silica sand, contributing to correct low
level of silica (SiO2) in clay and iron sand as
correction material of low-level Fe2O3 in main raw
material (Farhan, 2016). The components of cement
are lime (CaO), SiO2, alumina (Al2O3), ferric oxide
(Fe2O3), magnesite (MgO), and a small number of
other oxides (Rahadja, 2010).
The cement industry in Indonesia has experienced
rapid development. The Ministry of Industry (2016)
states that currently, the consumption of domestic
cement is 62,4 million tons, with a total of national
cement production reaching 68,7 million. The
increased cement production causes cement raw
materials needs also increasing; one of them is lime.
Nur et al. (2015) stated that the use of lime to produce
cement is the largest use of raw material from total
raw material for 87,4% or 5.047.263,31 tons. The
increasing consumption of limestone affects mining
in the karst area, such as reduced water supply
sources and environmental damage to the needs of the
surrounding communities. On the other hand, the
production process or forming karst to become
cement results or releases carbon dioxide (CO2),
where the CO2, itself, gas can cause the increase of
global warming.
As an alternative to reduce mining in karst areas
and to cope with increasingly high CO2 gas, other
materials are required for replacing cement as a
solution. There are various solutions or alternatives to
substitute cement, including copper slag (Kadhafi,
2015), Lapindo mud (Susanto and Nugroho, 2012),
rice husk ash (Anam and Sumarno, 2018), eggshell
powder (Hibur, 2017), fly ash (FA), and bottom ash
(BA). From all of those alternative materials, FA is
able to become the best one because of the content of
pozzolan as cement replacement with the highest
level of SiO2, Al2O3, and Fe2O3.
252
Saputra, A., Gunawan, A., Sukandar, L., Salurante, Y. and Rini, I.
Characterization of Geopolymer Paste based on Fly Ash and Bottom Ash in PLTU Kaltim Teluk using Sodium Hydroxide (NaOH).
DOI: 10.5220/0009445802520258
In Proceedings of the 1st International Conference on Industrial Technology (ICONIT 2019), pages 252-258
ISBN: 978-989-758-434-3
Copyright
c
2020 by SCITEPRESS – Science and Technology Publications, Lda. All r ights reserved

Furthermore, FA and BA able to be used in this
research are the waste from the coal combustion
process at Kaltim Teluk Steam-electric Power Station
(PLTU). Everyday PLTU Kaltim Teluk produces FA
of 150 tons (Rini et al., 2018). According to
Government Regulation (PP), Number 101, the Year
2014 large amount of resulted waste in a period is
categorized as specific hazardous and toxic material
waste (B3). Therefore, FA and BA from PLTU
Kaltim Teluk are categorized as specific hazardous
and toxic waste.
So far, FA and BA of PLTU Kaltim Teluk are
managed by using the third party. The increasing total
amount of FA and BA in every year requires
management in order to not causing environmental
issues, for example, air or water pollution and
decreased quality of the ecosystem. Due to that, one
solution or alternative measure that is able to be
conducted is to process FA and BA as substitution or
replacement of cement in a mixture of pozzolan
concrete, causing them to become good additive
mineral for concrete (Adibroto et al., 2018).
2 MANUSCRIPT PREPARATION
2.1 Materials
Materials used in this research are FA and BA wastes,
which were collected from PLTU Kaltim Teluk on
February 4, 2019, solid Sodium Hydroxide (NaOH)
pro analyst (PA) type from Ensure ISO brand, and
solution of Sodium Silicate (Na2SiO3) from Pudak
Scientific brand.
2.2 FA Characterization
FA raw material is tested by using a Scanning
Electron Microscope (SEM) of FEI type from
Inspect-S50 brand and X-Ray Fluorescence (XRF)
from Panalytical brand with Minipal 4 type. SEM is
intended to determine shape, size, and surface
structure of FA grain, meanwhile XRF is intended to
analyze chemical composition and element
concentrate in FA.
2.3 The Making and Vicat Test on
Portland Cement Paste
In this procedure, making and Vicat test is conducted
on the cement paste. The purpose of this procedure is
to find out the effect of setting the time on the cement
paste. In order to figure out the setting time of a
cement paste, the normal consistency of a cement test
should be carried out first so that the need for water is
able to be determined. Steps of implementation in
normal consistency of a cement paste test are in
accordance with SNI 03-6827-2002 (Normal
Consistency Test Method of Portland Cement Using
Vicat Apparatus for Civil Work) that the object of
cement test is prepared with the composition of
cement and water in table 1. Afterward, cement is put
in, and water is added into the mixing pan to form
cement paste. After mixing has been completed, it is
formed into ball-shaped by using a hand. Then, the
cement paste is put into Vicat mold through its base
hole until fully filled and place it on the glass plate.
Next, the Vicat mold filled with cement paste is
positioned on the Vicat apparatus, and then the tip of
the Vicat rod is contacted to the center of cement
paste surface, and the position of the Vicat rod is
tightened.
Table 1: Composition of portland cement paste for the Vicat
test.
Cement (gram) Water (mL)
300 76,95
Steps of setting time tests are in accordance with
SNI 03-6827-2002 (Initial Setting Time Test Method
Using Vicat Apparatus for Civil Work) that 300
grams of cement and volume of water required for
normal consistency in accordance with normal
consistency of a cement paste test method are
prepared. Next, cement is put in, and water is added
into the mixing pan to form cement paste. Afterward,
a vicat mold filled with cement paste is stored along
with the glass plate in a humid cabinet, and the
concrete thermometer is positioned on the Vicat mold
and counted for 30 minutes by using a stopwatch.
After that, the vicat mold filled with cement paste is
positioned on the Vicat apparatus, then the tip of the
Vicat rod is contacted on the center of the cement
paste surface, and the Vicat rod is tightened. This
process is repeated every 15 minutes for other
different holes on the cement paste surface. The space
between penetration points is no less than 5 mm to the
inner sidewall of mold, and the space between 2
penetration points is no less than 10 mm.
2.4 The Making and Vicat Test on
Geopolymer Paste
In this research, the used composition of FA and
alkaline activation to form geopolymer paste is
determined by the Vicat test result. The mass ratio
between Na
2
SiO
3
solution and NaOH solution used
Characterization of Geopolymer Paste based on Fly Ash and Bottom Ash in PLTU Kaltim Teluk using Sodium Hydroxide (NaOH)
253
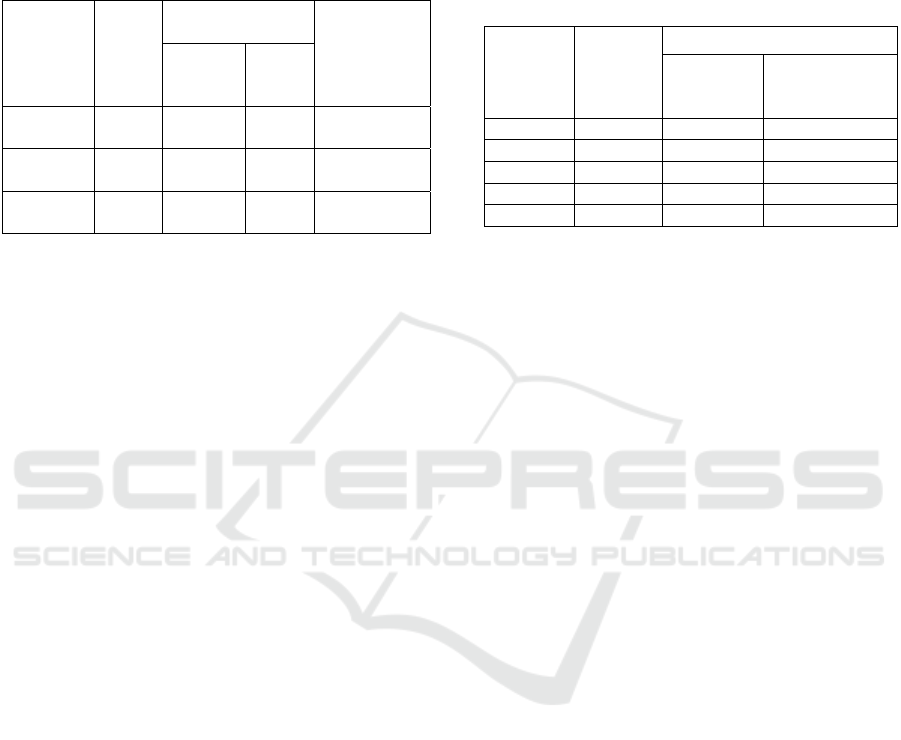
for alkaline activator is 3: 2. The composition of FA
and alkaline activation used on the Vicat test are
shown in table 2.
Table 2: Composition of FA and alkaline activation for
Vicat test
Variable
FA
(gram
)
Alkaline
Activation
FA:
Alkaline
Activation
Ratio
Na
2
SiO
3
(gram)
NaOH
(gram
)
100% FA 300 159,62
106,4
2
53% : 47%
100% FA 300 166,15
110,7
7
52% : 48%
100% FA 300 172,94
115,2
9
51% : 49%
In this procedure, forming and Vicat test on the
geopolymer paste are conducted. The purpose of this
is to figure out the effect of setting the time on
geopolymer paste. Steps of implementing setting time
tests are in accordance with SNI 03-6827-2002
(Initial Setting Time Test Method on Portland
Cement Using Vicat Apparatus for Civil Work) FA
and alkaline activation mass are prepared in
accordance with table 2. Afterward, FA is put in, and
alkaline activation is added into mixing pan to form a
paste. Then, the geopolymer paste is put into Vicat
mold. The top surface of geopolymer paste is leveled
with one movement using the scoop and smooth off
the surface of geopolymer paste using the tip of scoop
without applying pressure on geopolymer paste. After
that, the vicat mold filled with a geopolymer paste is
stored in the humid cabinet along with a glass plate,
and the concrete thermometer is positioned on the
Vicat mold and count for 30 minutes using the
stopwatch. And after that, Vicat mold filled with
geopolymer paste is positioned on the Vicat
apparatus, then the tip of the Vicat rod is contacted on
the center of the geopolymer paste surface, and Vicat
rod is tightened. This process is repeated every 15
minutes for other different holes on the geopolymer
paste surface. The space between penetration points
is no less than 5 mm on the inner sidewall of mold,
and the space between 2 penetration points is no less
than 10 mm. Every time this penetration is carried
out, the Vicat needle shall be cleaned and always
align, and there should be no vibration. The steps are
repeated for Vicat mold with a composition ratio of
FA and alkaline activation.
After the Vicat test on geopolymer paste is
conducted, the result which is close to the result of the
Portland cement Vicat test, the ratio of FA and
alkaline activation are acquired with the percentage
of 51 %: 49%. The making of paste is carried out to
find out the maximum compressive strength acquired
on concentrate variation of NaOH solution. The
composition of making geopolymer paste for six
Vicat molds is shown in table 3.
Table 3: Composition of making geopolymer paste
Vicat
Mould
Code
FA
(51%)
(gram)
Alkaline Activation (49%)
Na2SiO3
(gram)
NaOH solution
(gram)
(molarity)
P-8 600 345,87 230,58 (8 M)
P-9 600 345,87 230,58 (9 M)
P-10 600 345,87 230,58 (10 M)
P-11 600 345,87 230,58 (11 M)
P-12 600 345,87 230,58 (12 M)
2.5 Curing Geopolymer Paste
In this research, after a geopolymer is removed from
the mold, it is continued by curing the geopolymer
paste. The type of curing that is used is a process of
treatment by placing a geopolymer paste in open
space with the specified time. Curing is conducted on
normal curing temperatures for 3 days and 14 days.
2.6 Compressive Test
On the paste compressive strength test, a compressive
test machine is used. The first step of the compressive
paste test is to clean the test object from any attached
dirt using a wet cloth. Afterward, the paste is weighed
to determine the mass of the test object. After that, it
is placed on a compressive test machine centrally.
Then, the compressive test machine is turned on and
compress the test object until it is cracked or splinter.
After that, record the maximum load of the test
objects displayed on the compressive test machine.
And then, calculate the paste compressive strength,
which is the load per metric unit in N/mm2 or MPa.
3 RESULTS AND DISCUSSION
3.1 XRF Test on FA
XRF test is conducted to find out the chemical
composition in FA.
ICONIT 2019 - International Conference on Industrial Technology
254

Table 4: The result of the XRF test on FA from PLTU
Kaltim Teluk
Element
Concentrate
(%)
Compound
Concentrate
(%)
Al 5,60 Al2O3 7,80
Si 11,50 SiO2 18,00
S 1,60 SO3 2,80
Ca 27,10 CaO 24,70
Fe 42,30 Fe2O3 35,80
In research, Akinyemi and Mugera (2018)
specified that class C FA has SiO
2
+ Al
2
O
3
+ Fe
2
O
3
with the amount of 50% - 70%, a maximum of 5% of
SO
3
, and more than 10% CaO. In Table 5, the result
of the XRF test on FA of PLTU Kaltim Teluk has
SiO
2
of 18%, 7,8% of Al
2
O
3
, and 35,8% Fe
2
O
3
. This
indicates the total of SiO
2
+ Al2O
3
+ Fe
2
O
3
for 61,6%,
2,8% of SO
3
, and 24,7% of CaO. Thus, it can be
inferred that the FA at Kaltim Teluk can be classified
as class C FA.
3.2 SEM Test on FA
SEM is conducted to acquire an overview of
morphological conditions on FA, which is used as a
basic material in synthetizing geopolymer paste
activated by an alkaline activation solution. In
research conducted by Akinyemi and Mugera (2018),
it is stated that generally, the FA particle has irregular
and porous microscopic structures. The result of
observation on the FA particle of PLTU Kaltim Teluk
using the SEM test in figure 1 with a magnification
value of 250x and 10.000x shows that FA of PLTU
Kaltim Teluk has an irregular particle. In research
conducted by Sinaga (2018), it is said that irregular
FA particles will decrease workability because FA is
unable to enter through the paste, causing the
occurrence of pores or tiny holes on paste and slowing
down the hydration process.
Figure 1: SEM of FA (100 wt%) with magnification value
of (a) 250x and (b) 10.000x (Writer, 2019)
Figure 2: SEM of FA (Ilmiah, 2017)
In research conducted by Rommel, et al. (2014),
as well as Ilmiah (2017), displayed in figure 2, also
specified that the smoothness of the particle is able to
affect the hydration process and setting time.
Reactions between pozzolan and water are
commenced from the pozzolan particle surface, so the
more area of particle surface is, the quicker is its
hydration process. This means that a smooth particle
will strengthen and generate faster hydration heat
than the rougher one. In addition, the spherical
particle will increase workability resulting in the
reduced need for water, easily bind each other, and
also narrow the space between mixed materials. In
conclusion, the best shape of FA farticle to be used
for making geopolymer paster is spherical and
possess a smooth surface.
3.3 Normal Consistency Test and Vicat
Test
The steps of normal consistency test of a cement paste
are in accordance with SNI 03-6827-2002 (Normal
Consistency Test Method of Portland Cement Using
Vicat Apparatus for Civil Work) which is conducted
in order to make the requirement of water in forming
cement paste on setting time test is able to be
determined. Cement paste reaches normal
Characterization of Geopolymer Paste based on Fly Ash and Bottom Ash in PLTU Kaltim Teluk using Sodium Hydroxide (NaOH)
255

consistency when the depth of cement paste
penetration is 10 ± 1 mm.
Figure 3: Graphic of normal consistency on cement paste
(Writer, 2019)
It is showed in figure 3 that the linear equation that
can be used as a measurement of required water to
reach normal consistency of a cement paste with a
penetration of 10 mm. In normal consistency, cement
paste, when penetrated 10 mm, is 25,7%. The total
amount of water required in setting the time test of a
cement paste is 76,9 grams. In accordance with SNI
03-6827-2002 (Initial Setting Time Test Method
Using Vicat Apparatus for Civil Work), the setting
time test is conducted to find out the effect of initial
setting time and final setting time of a cement paste.
Initial setting time is the time required for a cement
paste to change its property from liquid to solid, while
final setting time is the time where penetrated Vicat
needle is visually visible. The initial setting time is at
least 45 minutes, and the final setting time is 375
minutes in maximum. Initial setting time is reached
when the Vicat needle enters into the paste in 30
seconds with penetration of 25 mm. Meanwhile, the
final setting time is reached when the Vicat needle
does not come into the paste in 30 seconds or does not
penetrate the surface of paste at all.
Figure 4: Graphic of Vicat test result on cement paste
(Writer, 2019)
In figure 4, during the penetration of 25 mm, the
initial setting time is acquired in the 74th minute, and
the final setting time is in the 135th minute. Setting
the time on a cement paste is used as a reference in
creating geopolymer paste (Manuahe et al., 2014).
3.4 Compressive Strength Test on
Geopolymer Paste
The purpose of this compressive strength test on the
geopolymer test is to find out the concentrate of
NaOH solution optimally required. In figure 5, it is
showed that the compressive strength of geopolymer
paste with the concentrate of 12 M NaOH has a
maximum compressive strength value of 9,51 MPa on
the 3rd day and 13,93 MPa at the 14th day. It means
that there is an increased value of 31,78% for 14 days.
From the compressive strength test on geopolymer
paste showed in figure 5, then the optimal NaOH
concentrate required is 12 M with the ratio of alkaline
activation solution of 3: 2.
Similarly with researches conducted by Setiadji
(2011), Yuanda, et al. (2015), Qomaruddin, et al.
(2018), Abdullah, et al. (2011), and Irawan, et al.
(2015) stated that the higher is the concentrate of
NaOH solution, and the increased age of the paste is,
the higher is the resulted compressive strength. And
also, the concentrate of the NaOH solution produces
the highest compressive strength of 12 M. These are
caused because of the effect of FA on the paste
compressive strength. Reactions between calcium
hydroxide (Ca(OH)2) with SiO2 in FA form calcium
silicate hydrate (CSH) compound with the property of
increasing strength.
3Ca(OH)
2
+ 2SiO
2
+ H
2
O 3CaO.2SiO
2
.3H
2
O
Figure 5: Graphic of NaOH solution concentrate on
compressive strength (Writer, 2019)
ICONIT 2019 - International Conference on Industrial Technology
256

Ferdy (2010) and Ren (2015) explained that on
FA geopolymer and alkaline activation solution
would synthesize, forming solid material through the
polymerization process. In this polymerization
process, there are 3 phases, where there is the
dissolution of Si and Al at the first phase.
In accordance with the reactions, NaOH solution
in polymerization has a role in dissolution Si and Al
of FA, and as a supplier of Na+ to make the charge
remains balance. On high concentrate, NaOH
solution has more Na+ and is able to cause flash
setting, because of high alkalinity and prompt
dissolution of Ca-Si. The Na+ cation strikes the
surface of the solid phase to dissolve Si and Al. High
OH concentrate in NaOH solution is able to
accelerate the dissolve of Si and Al in FA to make it
more optimal. Alkaline activation solution of
aluminosilicate involves chemical reaction where an
atom of bridging oxygen (BO) in the structure of
aluminosilicate is transformed into non-bridging
oxygen (NBO), which leads to Si and Al isolation.
Adding Na
2
SiO
3
also helps to slow the setting on
a paste, providing more time for the Si and Al
dissolution. Zhang et al. (2015), said that Si on FA is
dissolved by NaOH solution and becomes one of Si
sources. Then, other sources of Si are provided by
Na
2
SiO
3
directly. Moreover, Na
2
SiO
3
in alkaline
activation solution accelerates the polymerization.
The second phase is the hydrolysis of s and Al,
where this process causes the forming of Si-OH and
Al-OH bond.
Al
2
O
3
+ 3H
2
O + 2OH
-
2[Al(OH)
4
]
-
SiO
2
+ H
2
O + OH
-
[SiO(OH)
3
]
-
SiO
2
+ 2OH
-
[SiO
2
(OH)
2
]
2-
In this stage, there is the decomposition of Si and
Al in FA to become monomer, then followed by a
polycondensation process from monomer into
polymer structure with a three-dimensional molecule
network. On a high ratio of Si/Al, there will be
condensation between silica and will form polymer
silicate with Al. Adding Na
2
SiO
3
into a geopolymer
mixture will help increase the ratio of Si/Al
inactivation phase. When the polycondensation
process is ongoing, which is endothermic, where the
heat is acquired from the curing process determined
by temperature. In this process, a geopolymer will
absorb the aluminosilicate compound.
4 CONCLUSIONS
This research shows that the highest compressive
strength on geopolymer paste is acquired when the
alkaline activation solution is used with a NaOH
solution of 12 M concentrate with a compressive
strength of 13,93 MPa at 14 days.
ACKNOWLEDGMENTS
The authors gratefully acknowledge for Kalimantan
Institute of Technology and LPPM Kalimantan
Institute of Technology for the 2019 research grant to
support this research.
REFERENCES
Abdullah, M.M.A., Kamarudin, H., Mohammed, H., Nizar,
I. Khairul, Rafiza, A.R., dan Zarina, Y. (2011), “The
relationship of NaOH Molarity, Na2SiO3/NaOH Ratio,
Fly Ash/Alkaline Activator Ratio, and Curing
Temperature to the Strength of Fly Ash-Based
Geopolymer”, Advanced Materials Research, Vol. 328-
330, hal. 1475-1482.
Adibroto, Fauna, Suhelmidawati, Etri, dan Zade, Azri
Azhar Musaddiq. (2018), “Eksperimen Beton Mutu
Tinggi Berbahan Fly Ash Sebagai Pengganti Sebagian
Semen”, JIRS, Vol. 15, No. 1, hal. 11-16.
Akinyemi, Segun A., dan Mugera, W Gitari. (2018), “Coal
Fly Ash Beneficiation – Treatment of Acid Mine
Drainage With Coal Fly Ash”, National and University
Library in Zagreb, Croatia.
Anam, Syaiful, dan Sumarno, Agung. (2018), “Pengaruh
Kombinasi Abu Sekam Padi Sebagai Binder Alternatif
Dan Fly Ash Pada Tinjauan Kuat Tekan Beton
Geopolimer”, Universitas Mercubuana, Bekasi.
Badan Standarisasi Nasional. (2002), “Metode Pengujian
Waktu Ikat Awal Semen Portland dengan
Menggunakan Alat Vicat Untuk Pekerjaan Sipil”, BSN,
Jakarta, SNI 03-6827-2002.
Farhan, Muhammad. (2016), “Penambahan Abu Batubara
Sebagai Bahan Campuran Untuk Proses Pembuatan
Semen”, Politeknik Negeri Sriwijaya, Palembang.
Ferdy. (2010), “Pengaruh Temperatur dan Waktu Curing
Terhadap Kuat Tekan Pasta Geopolimer Berbahan
Dasar Abu Terbang”, Universitas Indonesia, Depok.
Hibur, Yohanes Berkhemans. (2017), “Pengaruh Serbuk
Cangkang Telur Substitusi Semen Terhadap
Karakteristik Beton”, Universitas Atma Jaya
Yogyakarta, Yogyakarta.
Ilmiah, Rihnatul. (2017), “Pengaruh Penambahan Abu
Sekam Padi Sebagai Pozzolan Pada Binder Geopolimer
Menggunakan Alkali Aktivator Sodium Silikat
(Na
2
SiO
3
) serta Sodium Hidroksida (NaOH)”, Institut
Teknologi Sepuluh Nopember, Surabaya.
Characterization of Geopolymer Paste based on Fly Ash and Bottom Ash in PLTU Kaltim Teluk using Sodium Hydroxide (NaOH)
257

Irawan, Ranastra Rulli, Hardono, Setyo, Budiman, Yanu
Ikhtiar, Soeherman, Ogi, Triani, Desak Nyoman Deasy,
dan Gunawan, Gugun. (2015), “Beton Dengan Sedikit
Semen Portland dan Tanpa Semen Portland
Memanfaatkan Abu Terbang Dari PLTU Batu bara”,
Badan Penelitian dan Pengembangan, Bandung.
Kadhafi, Muhammad. (2015), “Pemanfaatan Copper Slag
Sebagai Substitusi Semen Pada Campuran Beton Mutu
K-225”, Jurnal Teknik Sipil dan Lingkungan, Vol. 3,
No. 1, hal. 734-740.
Kementerian Perindustrian. (2016), “Pasokan Semen
Nasional 102 Juta Ton pada 2017”,
http://www.kemenperin.go.id/artikel/12223/pasokan-
semen-Nasional-102-juta-ton-pada2017.
Manuahe, Riger, Sumajouw, Marthin D. J., dan Windah,
Reky S. (2014), “Kuat Tekan Beton Geopolymer
Berbahan Dasar Abu Terbang (Fly Ash)”, Jurnal Sipil
Statik, Vol. 2, No. 6, hal. 277-282.
Nur, Rasdiana Rahma, Hartanati, Firda Dwi, dan Sutikno,
Juwari Purwo. (2015), “Studi Awal Desain Pabrik
Semen Portland dengan Waste Paper Sludge Ash
Sebagai Bahan Baku Alternatif”, Jurnal Teknik ITS,
Vol. 4 No. 2, hal. F164-F168.
Qomaruddin, Mochammad, Munawaroh, Tri Hanafiah, dan
Sudarno. (2018), “Studi Komparasi Kuat Tekan Beton
Geopilimer Dengan Beton Konvensional”, Universitas
Islam Nahdlatul Ulama, Jepara.
Rahadja, Hasan. (2010), “Kursus Eselon III Produksi
Teknologi Semen”. Indonesia Cement Institute,
Padang.
Ren, Xin. (2015), “Complete Recycling and Utilization Of
Waste Concrete Through Geopolymerization”,
Universitiy of Arizona, Arizona.
Rini, Intan Dwi W. S., Sholikah, Umi, Saputra, Andika Ade
I., Fansuri, Hamzah, dan Gunawan, Adrian. (2018),
“Utilization of Hazardous Waste Fly Ash Coal from
Kariangau Thermal Power Plant as Substitution of
Portland Cement on Concrete”, 2nd Borneo
International Conference on Applied Mathematics and
Engineering (BICAME), hal. 253-257.
Rommel, Erwin, Kurniawati, Dini, dan Pradibta, Arman
Putra. (2014), “Perbaikan Sifat Fisik dan Reaktifitas Fly
Ash Sebagai Cementitious Pada Beton”, Media Teknik
Sipil, Vol. 12, No. 2, hal 111-118.
Setiadji, Rudi. (2011), “Sifat Mekanik Beton Geopolimer
Dengan Agregat Tailing”, Universitas Gadjah Mada,
Yogyakarta.
Susanto, Agus dan Nugroho, Prasetyo Agung. (2012),
”Pemanfaatan Lumpur Lapindo Sebagai Pengganti
Agregat Kasar Beton”, Universitas Muhammadiyah
Surakarta, Surakarta.
Yuanda, Dedi, Fadli, Ahmad, dan Drastinawati. (2015),
“Pemanfaatan Limbah Abu Terbang (Fly Ash)
Batubara Sebagai Bahan Pembuatan Beton
Geopolimer”, JOM FTEKNIK, Vol. 2, No. 2, hal. 1-7.
Zhang, Lianyang. (2015), “Molecular Dynamics Study of
Sodium Aluminosilicate Geopolymer Containing
Calcium”, University of Arizona, Arizona.
ICONIT 2019 - International Conference on Industrial Technology
258
