
ECG-derived Blood Pressure Classification using Complexity
Analysis-based Machine Learning
Monika Simjanoska
1
, Martin Gjoreski
2
, Ana Madevska Bogdanova
1
, Bojana Koteska
1
,
Matja
ˇ
z Gams
2
and Jurij Tasi
ˇ
c
3
1
Faculty of Computer Science and Engineering, Ss. Cyril and Methodius University,
Rugjer Boshkovikj 16, 1000 Skopje, Macedonia
2
Department of Intelligent Systems, Jozef Stefan Institute, International Postgraduate School,
Jamova cesta 39, 1000 Ljubljana, Slovenia
3
Faculty of Electrical Engineering, University of Ljubljana, Trzaska cesta 25, 1000 Ljubljana, Slovenia
Keywords:
Blood Pressure, ECG-derived, Complexity Analysis, Machine Learning, Stacking, Classification.
Abstract:
The recent advancement on wearable physiological sensors supports the development of real-time diagnosis
in preventive medicine that demands various signal processing techniques to enable the extraction of the vital
signs (e.g., blood pressure). Blood pressure estimation from physiological sensors data is challenging task
that usually is solved by a combination of multiple signals. In this paper we present a novel complexity
analysis-based machine-learning perspective on the problem of blood pressure class estimation only from
ECG signals. We show that high classification accuracy of 96.68% can be achieved by extracting information
via complexity analysis on the ECG signal followed by applying a stack of machine-learning classifiers. In
addition, the proposed stacking approach is compared to a traditional machine-learning approaches and feature
analysis is performed to determine the influence of the different features on the classification accuracy. The
experimental data was gathered by daily monitoring of 20 subjects with two different ECG sensors.
1 INTRODUCTION
Real-time diagnosis in preventive medicine can sig-
nificantly reduce the costs of expensive medical treat-
ments. The real-time preventive diagnosis and on-
time alarming in case of abnormal events (Lehocki
et al., 2014), demand various signal processing tech-
niques to enable continuous monitoring of vital signs
(heart rate, blood pressure, respiratory rate, oxygen
saturation), which usually are extracted from wear-
able sensors data (electrocardiogram - ECG, pho-
toplethysmogram - PPG, phonocardiogram - PCG,
breathing sensor, SPO2 sensor). The identification of
morphologic characteristics and time intervals within
the physiological signals allow continuous monitor-
ing of vital signs and providing medical diagnosis
(Cosoli et al., 2015). Blood pressure (BP) is consid-
ered to be one of the most valuable vital sign, however
it still requires dedicated equipment for its estimation.
Moreover, the related research on BP estimation fo-
cuses on using several sources of data (e.g., ECG and
PPG) due to the complexity of the problem. In this
paper we present a method for BP classes estimation
by using only ECG as a single source of data.
The relationship between the BP and the morpho-
logical changes of the physiological signals is widely
discussed in the related work. A huge study encom-
passing more than 11,000 individuals has been con-
ducted by (Schroeder et al., 2003) where it has been
examined whether the hypertension leads to changes
in heart rate variability (HRV). The study reports re-
sults within 9 years period showing that even though
the subjects with hypertension had decreased HRV,
there was no significant difference in the HRV be-
tween the groups with and without hypertension 9
years after one group was predicted to have a higher
risk of developing a hypertension. This confirms
the complexity of the problem and suggests that new
methods for deriving BP should be considered. (Has-
san et al., 2008) investigate the relationship between
the heart rate (HR) and the systolic blood pressure
(SBP) at 10 healthy subjects. Even though the prelim-
inary results showed positive correlation, the physi-
cians do not completely agree that they proportionally
rise together, e.g., in an event of dangerous situation,
both the HR and BP might increase; however, the rise
of HR, does not automatically trigger the rise of BP
- appropriate example are the patients with coronary
282
Simjanoska, M., Gjoreski, M., Bogdanova, A., Koteska, B., Gams, M. and Tasi
ˇ
c, J.
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning.
DOI: 10.5220/0006538202820292
In Proceedings of the 11th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2018) - Volume 5: HEALTHINF, pages 282-292
ISBN: 978-989-758-281-3
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

artery disease who have normal HR (Heart and Vas-
cular Team, 2016).
ECG alone has never been used for deriving BP,
instead most of the published literature refers to the
combination of both ECG and PPG. (Nye et al., 2015)
performed a deep survey categorizing the methods for
deriving BP into three major classes relying on: Pulse
Wave Velocity (PWV), Pulse Arrival Time (PAT) and
Pulse Transit Time (PTT). PWV is the rate at which
the pressure wave moves down the vessel and PTT
is the time at which the pressure wave propagates
through the arterial tree (Grenwis et al., 2012). PAT
is equal to the sum of PTT and the pre-ejection pe-
riod, however a study (Zhang et al., 2011) indicates
that PAT is not an adequate for PTT calculation since
it is unable to detect the challenging BP changes.
BP is measured in terms of systolic blood pres-
sure (SBP) and diastolic blood pressure (DBP), the
maximum and the minimum value of an arterial pres-
sure tracing, correspondingly. Assuming the PWV is
a reliable measure for estimation of the central BP,
(i Car
´
os, 2011) provides a comprehensive analysis of
the PWV-based techniques for continuous and non-
invasive monitoring of BP; even though it is claimed
that no clear information is provided on whether PWV
is related to SBP, DBP or mean BP. Alternative tech-
niques that do not rely on PWV are also discussed
such as those for multi-parametric processing of car-
diovascular variables, or those that consider parame-
ters within the heart sounds (PCG analysis). One ex-
ample is the analysis of R2 - the time interval from
a particular R peak in ECG, to the peak of the sec-
ond heart sound in PCG (Wong et al., 2006). Close
inverse correlation between the timing of the sec-
ond heart sound has been found by SBP(Zhang and
Zhang, 2006) . (Goli and T., 2014) used PTT derived
from ECG R-wave and the maximum first derivative
PPG in correlation analysis at 11 healthy subjects.
The reported results show PTT to be strongly corre-
lated with SBP (0.734) and the DBP (0.731).
In another study, (Wong et al., 2009) perform a
half year study on the relationship between BP and
PTT, showing the PTT is highly correlated with SBP,
but not with DBP - probably due to the small varia-
tions of the DBP. Even though PPG signals are widely
used for the purpose of BP estimation and present
high prediction accuracy, there are also some experi-
ments that withstand those statements. (Nitzan, 2011)
compares multiple methods indicating that PPG is not
applicable for the estimation of DBP since no sub-
stantial change of blood flow or PPG pulse is found
when the cuff pressure decreases. Similarly, (Payne
et al., 2006) proves that PTT extracted from ECG and
PPG is unreliable marker of beat-to-beat BP.
This research approaches the problem from a
completely different perspective of deriving BP from
ECG only. The rule presented by (Najarian and
Splinter, 2012) states that the healthy biomedical sys-
tems are of high complexity and once an abnormal-
ity occurs this complexity drops. The complexity de-
crease has been experimentally proven in the litera-
ture for problems related to brain activity in presence
of Alzheimers disease (G
´
omez et al., 2006); compar-
ative EEG study on pathological and healthy groups
(Bhattacharya et al., 2000); ECG analysis of ventric-
ular tachycardia and fibrillation (Zhang et al., 2000);
complexity loss theory of aging and disease (Costa
et al., 2005); complexity analysis of respiratory sys-
tem (Raoufy et al., 2017), etc.
In order to explore the idea of using ECG com-
plexity analysis, 332 ECG signals were obtained dur-
ing normal physical activities from 20 subjects of dif-
ferent age groups. During the ECG acquisition, cuff-
based BP measurements at different timestamps were
manually performed, which later were used to label
the ECG signals with one of the BP classification
groups reported by (Program et al., 2004): hypoten-
sion, normal, prehypertension, stage 1 hypertension,
stage 2 hypertension, isolated systolic hypertension
(ISH), and hypertensive crisis. Having extracted fea-
tures using the complexity analysis later discussed in
the Sect. 2, two types of Machine-Learning (ML) so-
lutions, a flat (simple) design and a stacking design
were developed and compared.
The rest of the paper is organized as follows. Sect.
2 presents the methods and materials used in the ECG
signal processing system. Sect. 3 presents the ex-
periments and the obtained results. Sect. 4 and Sect.
5 present the discussion and the conclusions of the
study.
2 THE PROPOSED APPROACH
When developing the novel methodology we followed
the steps of a typical biomedical signal-processing
system (Najarian and Splinter, 2012) presented in Fig-
ure 1. The following subsections provide a detailed
explanation of the methods and techniques applied in
each step.
2.1 The Biological System
The ECG signal presents the electrical activity of the
heart. It is recorded by placing electrodes on the in-
dividual’s chest or limb. The heart system consists of
two atria for collecting the blood and two ventricles
for pumping the blood. There are two main phases, a
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning
283

The Biological
System (ECG + BP)
The Sensors
(ECG + BP)
ECG Signals
Preprocessing
Features Extraction
(Complexity Analysis)
Machine Learning
(Classification)
Figure 1: The ECG signal processing system.
filling (resting) phase called diastole, and a pumping
(contracting) phase called systole. The deoxygenated
blood is collected in the right atrium and therefrom is
passed to the right ventricle. While the ventricular is
in a systole phase, the deoxygenated blood is pumped
to the lungs where it will be oxygenated. The oxy-
genated blood is received by the left atrium and dur-
ing the atrial systole it is passed to the left ventricle.
The left ventricle is the largest and strongest chamber
since it has to pump the oxygenated blood to the rest
of the body. A normal HR during resting is about 70
bpm (beats per minute - measure for the rhythm of the
heart) (Rangayyan, 2015).
The pressure that is caused by the blood flow
on the blood vessels walls is referred to as blood
pressure. BP is measured in millimeters of mercury
(mmHg), and depending on the different values of
the maximum pressure during one heart beat (SBP)
and the minimum pressure in between two heart beats
(DBP), there are seven categories published online
and in the literature (Program et al., 2004; AHA,
2016) presented in Tab. 1.
2.2 The ECG Sensors
The ECG signals used in this research were obtained
using two different ECG sensors and the reference BP
values were measured by using an electronic sphyg-
momanometer. Sixteen participants at age 16 - 72
were recorded by using ECG sensor as a part of
the Cooking hacks e-Health Sensor Platform (Cook-
ing Hacks, 2016). All the measurements were per-
formed in a sitting position in duration up to 60 sec-
onds at 125Hz sample rate, and a single measurement
of the SBP and DBP has been done for each ECG
signal. Additionally, 4 participants at age 25 - 27
were recorded using the 180°eMotion FAROS (Bit-
tium Biosignals, 2016) ECG sensor. In this case, the
sensor was attached to the participants for at least 4
hours, continuously capturing the electrical activity of
the heart at sampling rate of 1000Hz during the daily
activities. The participants were required to measure
the BP at particular times ranging from 30 minutes to
1 hour. All of the 20 participants had no history of
heart problems.
2.3 ECG Signals Preprocessing
A valid ECG information is considered to exist in
range of 0.05 - 100Hz (Rangayyan, 2015). The ap-
plication of the 180 eMotion FAROS sensor pro-
duced a high-resolution ECG sampled at frequency
of 1000Hz. Those ECG signals were filtered by ap-
plying a low-pass Butterworth filter of 18th order and
cutoff frequency of 100Hz. The order of the filter was
optimally determined based on the filter design speci-
fications. All the signals were trimmed by taking only
the last 30 seconds before the BP measurement. Ex-
perimentally is observed that 30 seconds of ECG car-
ries the right information for BP class prediction. As
a result from the preprocessing, we created a database
that consists of 332 signals labeled with the appropri-
ate BP class as described in Tab. 1.
2.4 Feature Extraction from ECG -
Complexity Analysis
The ML approach in BP estimation from ECG signal
requires building distinguishing feature vectors that
will represent the created database. In this novel ap-
proach the feature extraction is based on complexity
analysis rather than on extraction of the morphologi-
cal characteristics, the conservative way of processing
the ECG signals as described in the literature (i Car
´
os,
2011; Wong et al., 2006; Zhang and Zhang, 2006;
Goli and T., 2014; Payne et al., 2006; Wong et al.,
2009; Nitzan, 2011).
As mentioned in the Introduction, a normal and
healthy biomedical system (the ECG signal) is con-
sidered to be a very complex one and once a dramatic
change occurs (as is the sudden change in BP), the
complexity drops (Najarian and Splinter, 2012). This
relationship is already proven in the literature pre-
sented (G
´
omez et al., 2006; Bhattacharya et al., 2000;
Zhang et al., 2000; Costa et al., 2005; Raoufy et al.,
2017). Considering the complexity features perfor-
mance presented in the related work (McBride et al.,
2014; Morabito et al., 2012; Eke et al., 2002; Cancio
et al., 2008), in this research the BP changes are mod-
eled by inspecting complexity measures of the ECG
signals to extract six features that define the feature
vectors:
HEALTHINF 2018 - 11th International Conference on Health Informatics
284

Table 1: Blood pressure categorization.
Category SBP (mmHg) Logical DBP (mmHg)
Hypotension ≤ 90 OR ≤ 60
Normal 90-119 AND 60-79
Prehypertension 120-139 OR 80-89
Stage 1 hypertension 140-159 OR 90-99
Stage 2 hypertension ≥ 160 OR ≥ 100
Isolated systolic hypertension ≥ 140 AND < 90
Hypertensive crisis ≥ 180 OR ≥ 110
2.4.1 Signal Mobility
Signal mobility represents the first-order variations in
the signal. Let x
i
,i = 1,...,N be the ECG signal of
length N, and d
j
, j = 1,...,N − 1 be the first-order
variation calculated as given in Eq. 1:
d
j
= x
j+1
− x
j
(1)
Then the first-order factors S
0
and S
1
are obtained
as follows:
S
0
=
s
∑
N
i=1
x
2
i
N
(2)
S
1
=
s
∑
N−1
j=2
d
2
j
N − 1
(3)
Hereupon the signal mobility is simply calculated
as the ratio between the factors S
1
and S
0
:
Mobility =
S
1
S
0
, (4)
providing a quantitative measure of the level of varia-
tion along the signal.
2.4.2 Signal Complexity
Signal complexity represents the second-order vari-
ations in the signal. Given the first-order variation
of the ECG signal d
j
, j = 1,...N − 1, the second-
order variation of the signal is presented by g
k
,k =
1,...,N − 2, or:
g
k
= d
k+1
− d
k
(5)
Consequently, the second-order factor is calcu-
lated as:
S
2
=
s
∑
N−2
k=3
g
2
k
N − 2
, (6)
and the signal complexity is calculated according to
the equation:
Complexity =
s
S
2
2
S
2
1
−
S
2
1
S
2
0
(7)
To compute both the signal mobility and com-
plexity, we used the Hjorth parameters method
(Kugiumtzis and Tsimpiris, 2010).
2.4.3 Fractal Dimension
Fractal dimension is a measure of self-similarity and
describes the fundamental patterns hidden in the sig-
nal. It works like a magnifier, zooming and compar-
ing different portions of the signal with the entire sig-
nal. Higuchi algorithm is one of the most efficient
methods for calculating the fractal dimension and we
use it as implemented by (Alvarez, 2015). For choos-
ing the maximum number of subseries, k
max
, we fol-
lowed the advice by (Doyle et al., 2004). The Higuchi
algorithm forms a set of k subseries with different res-
olutions. For m = 1,...,k, the new time series X
k
are
formed as follows:
X
m
k
: x(m),x(m + k),x(m + 2k),...,x(m + b
N − m
k
ck)
(8)
Consequently, the length of the curve X
m
k
, l(k) is
calculated as:
l(k) =
(
∑
bN−m/kc
i=1
|x(m + ik) − x(m + (i − 1)k)|(N − 1))
(b
N−m
k
c)k
(9)
For each k in range 1 to k
max
, the average length
is calculated as the mean of the k lengths l(k) for m =
1,...k. The fractal dimension is the estimation of the
slope of the plot ln(l(k)) vs. ln(1/k).
2.4.4 Entropy
Entropy measures the amount of information (the ran-
domness) in the signal. The reduction of entropy con-
sidering a signal as ECG is often associated with a
disease. The information can be represented through
the concept of probability. Assuming X is the ECG
signal and p
i
is the probability of each outcome x
i
within X for i = 1,...,N − 1. Then, by addressing all
the zero/infinity problems, the equation for entropy
calculation gets the following form:
Entropy =
N−1
∑
i=0
p
i
log(
1
p
i
) (10)
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning
285
Low-pass filtering
Feature Extraction Class Labeling
DBP
DBP
DBP
SBP
SBP
SBP
Classifier
1
Classifier
2
Classifier
K
Feature Vector F = (f
1
,…,f
m
, label)
P
11
(F) P
12
(F) P
1N
(F) P
21
(F) P
22
(F) P
2N
(F) P
K1
(F) P
K2
(F) P
KN
(F)
. . .
. . . . . . . . .
EF = (P
11
(F), P
12
(F), …, P
1N
(F), P
21
(F), P
22
(F), …, P
2N
(F), P
K1
(F), P
K2
(F), …, P
KN
(F), label)
Meta Classifier
BP Class Prediction
Preprocessing
Feature
Extraction
Stacking
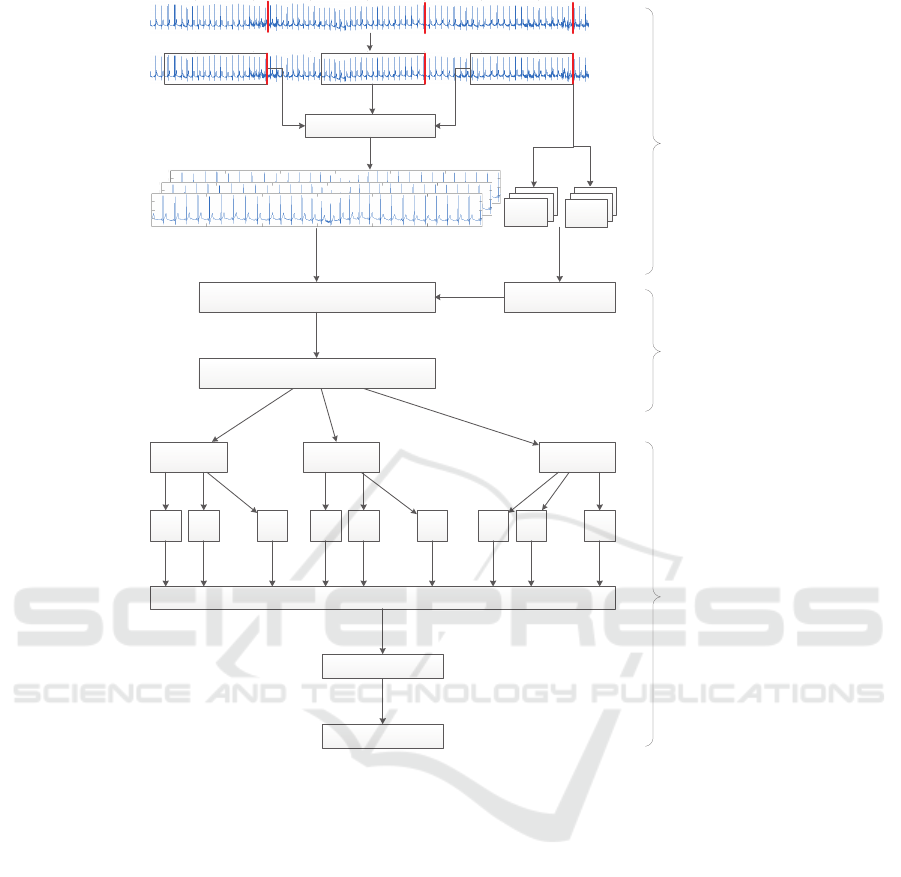
Figure 2: Stacking Design.
2.4.5 Autocorrelation
Autocorrelation calculates the similarity of the signal
when it is compared to its shifted version - a similar-
ity will exhibit a peak which is captured and quanti-
tatively measured. Given τ is the amount of shift, the
autocorrelation function is calculated as follows:
r
xx
(τ) =
Z
+inf
−inf
x(t)x(t −τ)p
xx
(x(t), x(t −τ))dt, (11)
where p
xx
(x(t), x(t − τ)) presents the joint proba-
bility density function of x(t) and x(t − τ).
2.4.6 Age
A simple mathematical model that represents the
blood flow in the arteries is made by (Labadin and Ah-
madi, 2006). It proves that the blood flow is affected
by the size of the blood vessel. When the left ventri-
cle contracts to push the blood into the aorta (during
systole), a pressure wave is generated along the ar-
terial tree. Affected mostly by the age or some other
changes in the arterial wall, the vessels become stiffer,
meaning the pressure wave velocity increases. As a
result, the reflected pressure waves that move back to
the heart will also move faster, causing greater sys-
tolic pressure to handle the load. Having in mind that
the age affect the changes in the arterial wall, partic-
ipant’s age as one of the features is included in the
feature vectors.
2.5 The Machine Learning Approach
Fig. 2 presents a detailed scheme of the proposed
methodology by following the steps of a biomedi-
cal processing system as depicted in Fig. 1. On the
top of Fig. 2 an example of raw ECG signal is pre-
HEALTHINF 2018 - 11th International Conference on Health Informatics
286

sented with red marks (lines) that represent the mo-
ment when the subject’s BP has been measured. In the
next step, the raw ECG signals are segmented by tak-
ing windows of 30 seconds before each BP measure-
ment. Then, the low-pass filtering is applied. Having
established the database of the ECG signals and the
corresponding SBP and DBP values, we proceed with
the feature extraction procedure that encompass both
the complexity analysis described in Sect. 2.4 and the
BP class labeling according to Tab. 1. Hereupon, two
ML approaches have been applied to train a model
that is able to predict the BP class for a given ECG
signal. The first approach is a flat ML design, includ-
ing the evaluation of seven different classification al-
gorithms as described in Sect. 2.5.1. The second ap-
proach is stacking ML design, visually described in
Fig. 2 and comprehensively explained in Sect. 2.5.2.
2.5.1 The Flat Design
In the flat design, ML models are trained by using
seven different classifiers, which model the data from
various aspects:
1. J48 - to rank the features according to the infor-
mation gain;
2. Naive Bayes (NB) - to consider strong indepen-
dence between the features;
3. KNN - to consider inter-instance similarity;
4. SVM - to recognize the most distinguishable fea-
ture vectors;
5. Random Forest (RF) - to combine multiple mod-
els built on varying features set in an ensemble;
6. Bagging (BGG) - to introduce dataset subsam-
pling as a way of reducing the variance of the de-
cision tree-based J48 algorithm, and
7. Boosting (BST) - to introduce instance weighting
in the dataset for addressing the mis-recognized
instances.
2.5.2 The Stacking Design
Assuming that different algorithms model different
structures in the data, we implemented a stacking
module presented in the third phase of Fig. 2. The im-
plementation mainly consists of tree phases. The first
phase is the application of the seven different classi-
fiers described in the flat design 2.5.1. Each of the
classifiers, K = 1, ...,7, is adapted to produce predic-
tion probabilities (Hall et al., 2009) for a given fea-
ture vector, F, to belong in each of the BP classes,
N = 1,...,6. The goal is to ensemble the probabilities,
p
11
(F),..., p
KN
(F), produced by each of the applied
classifiers and create new feature vectors (the second)
Table 2: ECG instances categorization.
Category Instances Label
Hypotension 3 0
Normal 198 1
Prehypertension 110 2
Stage 1 hypertension 15 3
Stage 2 hypertension 2 4
Isolated systolic hypertension 4 5
Hypertensive crisis 0 6
phase. In the final phase, a meta-classifier (Random
Forest) is trained with the new feature vectors.
Leave-one-instance-out principle has been used
for both the creation of the probability vectors and
the meta-classifier phase. The probability vector for
each instance was created when the instance is being
left out for testing. Having created the new proba-
bility vectors for all instances, the meta-classifier has
also been evaluated by the same principle, leaving-
one-instance-out. Thus it means that in a particular
iteration, one instance is a test instance in both phases
at the same time, and the overfitting is avoided.
3 RESULTS
After the preprocessing phase, a database consisting
of 332 instances was created. The distribution of the
instances is provided in Tab. 2. The majority class
is the Normal class consisting of 198 ECG recordings
(59.6% of the signals). The more extreme the classes
are, the less instances are included since the partici-
pants involved in the measurements were random in-
dividuals with no diagnosis related to heart or blood
pressure problems. The most extreme class, Hyper-
tensive crisis, is completely empty and has been dis-
missed in the following analysis.
The experiments were performed by using a leave-
one-instance-out cross-validation, i.e., the ML models
are built by using all the instances except one that is
left out for testing. The procedure is repeated 332
times (the number of instances in the database) and
the results are averaged. We present three metrics: F-
measure as a balanced mean between precision and
recall, ROC area to measure the trade-off between the
recall and the false positive rate, and the overall ac-
curacy of each classifier. In addition, we performed a
10-fold cross-validation and obtained similar results.
3.1 Flat ML Results
Tab. 3 presents the results for each of the seven
distinct classifiers used as part of the flat ML ap-
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning
287

Table 3: Flat ML results. Leave-one-instance-out evaluation.
Metric Class J48 NB KNN SVM RF BGG BST
F-
Measure
0 0.00 0.00 0.00 0.00 0.00 0.00 0.00
1 0.86 0.74 0.85 0.78 0.85 0.88 0.80
2 0.76 0.12 0.71 0.34 0.71 0.75 0.43
3 0.58 0.00 0.34 0.00 0.37 0.53 0.00
4 0.00 0.00 0.00 0.00 0.00 0.00 0.00
5 0.85 0.14 0.25 0.00 0.66 0.00 0.00
ROC
Area
0 0.62 0.53 0.49 0.06 0.40 0.21 0.33
1 0.80 0.61 0.81 0.61 0.90 0.87 0.31
2 0.80 0.55 0.78 0.59 0.87 0.83 0.30
3 0.75 0.68 0.65 0.48 0.85 0.81 0.06
4 0.29 0.06 0.49 0.53 0.44 0.25 0.56
5 0.86 0.95 0.62 0.94 0.86 0.95 0.86
Accuracy (%) all 81.32 54.21 76.80 66.26 78.01 81.32 68.67
Accuracy 10-fold (%) all 79.81 55.12 76.50 66.26 80.42 78.31 68.67
Table 4: Stacking ML results. Leave-one-instance-out evaluation.
Metric/Class 0 1 2 3 4 5
Precision 1.00 0.97 0.94 1.00 0.00 1.00
Recall 0.33 0.99 0.99 0.73 0.00 0.75
F-measure 0.50 0.98 0.96 0.84 0.00 0.85
ROC Area 0.94 0.99 0.99 0.98 0.42 0.99
Accuracy (%) 96.68
Accuracy 10-fold (%) 96.68
Table 5: Confusion matrix. Leave-one-instance-out evalua-
tion.
Real/Predicted 0 1 2 3 4 5
0 1 2 0 0 0 0
1 0 197 1 0 0 0
2 0 1 109 0 0 0
3 0 1 3 11 0 0
4 0 1 1 0 0 0
5 0 0 1 0 0 3
Correctly classified 321
Incorrectly classified 11
proach. From the results it can be observed that the
least recognized classes are the ones with only a few
instances available. However, even for the sparse
class ”Isolated systolic hypertension (label 5)”, J48
has achieved an F-measure of 0.86, and Bagging (also
NB) has achieved ROC area over 0.85. For the classes
0 and 4 (”Hypotension” and ”Stage 2 hypertension”)
with the least number of instances, all of the mod-
els perform worse in comparison to the performance
for the rest of the classes. However, the ROC area
measure, even though in range between random and
poor prediction, provides promising results for Boost-
ing and J48, and we expect this results to be im-
proved when more instances will be provided. Class
3, ”Stage 1 hypertension”, is also considered to be
sparse class; however, Random Forest and J48 show
slightly better results compared to class 0 and class
4. The classes 1 and 2, ”Normal” and ”Prehyperten-
sion”, contain the majority (92.77%) of the all the in-
stances. Random Forest, J48 and Bagging provide the
best results, but also KNN and to some extent SVM
show promising performance. The results from the
overall accuracy ranks the classifiers as follows: J48,
Bagging, Random Forest, KNN, Boosting, SVM and
Naive Bayes.
3.2 Stacking ML Results
Since all the classifiers in the ML experiments per-
formed well at different cases, we chose to combine
their outputs for the purpose of improved classifica-
tion performance. As meta-classifier we used Ran-
dom Forest. The stacking design produced the results
presented in Tab. 4. It can be seen that this approach
improved the prediction even for the sparse classes.
The overall accuracy is 96.68%, which is for 15.36
percentage points better than the highest accuracy ob-
tained by the flat ML design.
Tab. 5 presents the confusion matrix for the stack-
ing design. It is a good indicator showing the ten-
dency of the classifier to classify the sparse classes
HEALTHINF 2018 - 11th International Conference on Health Informatics
288

Table 6: Classification with ”Age” elimination.
Classifier Accuracy (with ”Age”) (%) Accuracy (without ”Age”) (%)
J48 81.32 80.72
NB 54.21 54.82
KNN 76.80 73.49
SVM 66.26 65.96
RF 78.01 76.81
BGG 81.32 76.2
BST 68.67 68.67
Table 7: Feature’s information gain.
Feature Average merit
Age 0.54
Fractal dimension 0.28
Complexity 0.25
Entropy 0.15
Autocorrelation 0.13
Mobility 0.01
Table 8: Variable ECG length accuracy comparison from
10-fold cross-validation.
Accuracy (%) 10s 30s 50s
J48 79.81 79.81 70.27
NB 58.43 55.12 24.32
KNN 74.39 76.50 54.05
SVM 65.96 66.26 70.27
RF 79.21 80.42 62.16
BGG 77.71 78.31 70.27
BST 68.67 68.67 70.27
Mean 72.02 72.15 60.23
0
2
4
6
8
10
12
0.00 0.66 1.99 3.32
Number of participants
Variance
Class variance distribution
Figure 3: Distribution of the classes variance among the
participants.
into the classes 1 and 2 that encompass the majority
of the instances.
4 DISCUSSION
The premise of using 30 seconds ECG signals is ex-
perimentally proved by using the proposed method-
ology in the paper considering variable-length ECG
windows starting from 10 up to 50 seconds. The
flat design produced the accuracy results presented in
Tab. 8. The results indicate that the feature extrac-
tion methodology is less informative for longer ECG
signals. The accuracies for 10 and 30 seconds ECG
signals provide nearly same average result; however,
the 30s length performs better for KNN, SVM, RF
and BGG, thus is considered to be used in the further
analysis.
Observing the experimental results in Sect. 3, it
can be seen that for the sparse class Isolated systolic
hypertension (label 5), J48 has achieved an F-measure
of 0.86, and both Bagging and NB have achieved
ROC area over 0.95. This results lead to a conclusion
that the class itself is easy to be recognized because of
its nature (characterized by high SBP and low DBP).
Additional analysis is done on the feature’s influ-
ence by measuring the information gain (entropy) for
each feature with respect to the class labels. Con-
sidering the feature ranking presented in Tab. 7, age
is found to be the top feature, fractal dimension and
complexity is the second and the third most influen-
tial attribute, correspondingly. Then follows the en-
tropy, the autocorrelation and mobility at last. It is
interesting that the second-order variations (complex-
ity) are found to be more informative than the first-
order variations (mobility). Obtaining the top most
in influential features does not mean they are enough
for highly accurate classification, instead the experi-
ments show that the elimination of each of the fea-
tures ranked bottom-up degrades the accuracy.
Given the fact that the feature age has highest rank
in Tab. 7 and also the fact that we performed multiple
measurements for each subject, we suspected that age
might act as a kind of ”ID” and therefore to intrigue
person-specific overfitting. In order to eliminate this
possibility, we performed additional testings by ex-
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning
289
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
1.20 2.41 3.61 4.81 6.02
Entropy
Class 1 Class 2 Class 3
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
0.04 0.22 0.40 0.58 0.76 0.94 1.12 1.30
Mobility
Class 1 Class 2 Class 3
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
1.20 2.41 3.61 4.81 6.02
Complexity
Class 1 Class 2 Class 3
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
1.13 1.37 1.61 1.86 2.10
Fractal dimension
Class 1 Class 2 Class 3
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
0.14 0.28 0.42 0.56 0.69 0.83 0.97 1.11
Autocorrelation
Class 1 Class 2 Class 3
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
1.00 2.00 3.00 4.00
Age
Class 1 Class 2 Class 3
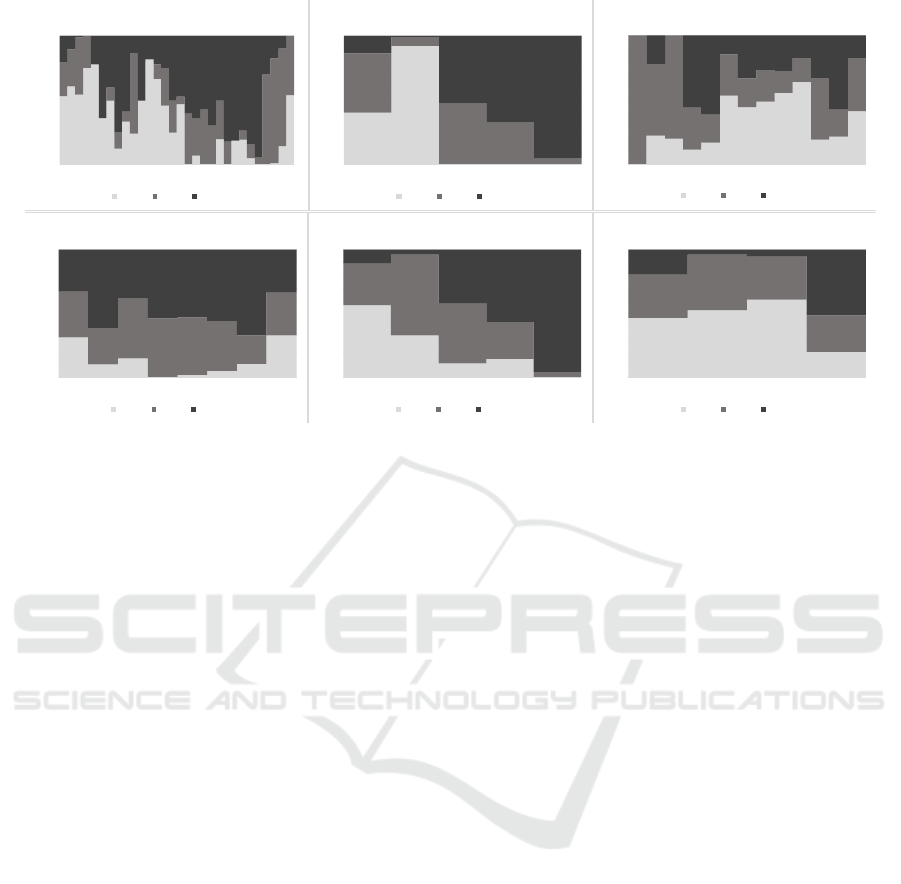
Figure 4: Features absolute values distributions over classes.
cluding the age from the feature vectors.
The results of the overall accuracy for each clas-
sifier are presented in Tab. 6 and are compared to the
old classification accuracy with the age included in
the feature vectors. The results show that the elimina-
tion of the age feature degrades the classification for
at most 5% at the Bagging classifier and for all the
rest there is only minor degradation in the classifiers
performance. This eliminates the suspicion of overfit-
ting and actually confirms the assumptions for the age
influence provided in Sect. 2.4.
Another suspicion is the person-specific overfit-
ting that might appear due to a small class vari-
ance. Fig. 3 presents the classes variance distribution
among the different participants. The figure shows
that for 14 out of 20 participants the class variance is
in range from 0.66 to 3.32, meaning they have suf-
fered variability in their BP measurements.
Fig. 4 presents the absolute values ranges of the
features in each of the BP classes. The figure shows
the amount of values (as % on the y-axis) correspond-
ing to the given range on the x-axis. Observing the en-
tropy as best visual representative, it can be seen that
most of the entropy absolute values obtained from the
instances in class 1 range from 1.20 up to 2.40; for
class 2 this range tends to be from 2.40 to 3.60, and in
class 3 most of the values are in range around 6. For
the autocorrelation, it can be concluded that the limits
are not so strict for the different classes, as well as for
the mobility feature. This is not surprising since both
of them have small average merit obtained from the
feature’s information gain results; however they still
influence the prediction results. For the complexity
and the fractal dimension feature, the ranges are more
clear as in the entropy case. The age feature distri-
bution over classes speaks that participants from all
the age groups (x-axis) can be found in each class.
However, observing the chart it can be concluded that
older population suffers from higher BP and belongs
to class 2 and 3. The age groups are created accord-
ing to the age ranges proposed in the literature (Arm-
strong, 2008).
5 CONCLUSION
The recent efforts on BP estimation mainly focus on
the morphological analysis of the physiological sig-
nals. The presented novel approach for BP classifi-
cation takes into consideration complexity analysis of
the ECG signal for the feature vectors creation. The
experimental results from the flat ML approach con-
firm that the features are suitable for BP classification,
i.e., J48 and Bagging have achieved accuracy of 81%,
which is for 21 percentage points better than the ma-
jority class.
In addition, we proposed a stacking scheme by
combining the output of different ML algorithms. The
stacking scheme achieved an accuracy of 96.68%,
which is for 15.36% improvement when compared
to the best accuracy obtained by the flat ML design,
or 36.36 percentage points improvement compared to
the majority class.
Compared to the newest related work, the pre-
sented results show significant improvement in terms
of simplicity and accuracy, avoiding the complex pro-
cedures when multiple types of signals and devices
are involved for the BP estimation. We performed
HEALTHINF 2018 - 11th International Conference on Health Informatics
290

several analyses to explain the accuracy improve-
ments and our current hypothesis is that the complex-
ity analysis on the ECG signals provides enough in-
formation for accurate BP class estimation. If that is
indeed so, future BP measurements may be performed
using only an ECG sensor. However, to completely
confirm this hypothesis, our method should be evalu-
ated on a bigger dataset with a leave-one-subject-out
evaluation technique, including the publicly available
physiological signals from the Physionet databases
(Goldberger et al., 2000).
Our future work is towards the collection of ECG
signals encompassing various ECG sensors and dif-
ferent target groups, since the research community is
missing this kind of data. The goal is to create a bal-
anced database, covering the critical BP classes (e.g.,
hypertensive crisis), and to develop sensor indepen-
dent methodology for BP estimation. Finally, we plan
to improve the methodology to be able to estimate the
real SBP and DBP values (e.g., using regression tech-
niques), and thus to contribute to the ”single-sensor
fits all” paradigm of using as least equipment to de-
rive as much vital signs as possible.
ACKNOWLEDGEMENTS
This research is supported by SIARS, NATO multi-
year project NATO.EAP.SFPP 984753.
REFERENCES
AHA (2016). Understanding blood pressure readings.
Alvarez, J. M. (2015). Higuchi and katz fractal dimension
measures.
Armstrong, T. (2008). The Human Odyssey: Navigating the
Twelve Stages of Life. Sterling, New York.
Bhattacharya, J. et al. (2000). Complexity analysis of
spontaneous eeg. Acta neurobiologiae experimentalis,
60(4):495–502.
Bittium Biosignals (2016). emotion faros.
Cancio, L. C., Batchinsky, A. I., Salinas, J., Kuusela,
T., Convertino, V. A., Wade, C. E., and Holcomb,
J. B. (2008). Heart-rate complexity for prediction
of prehospital lifesaving interventions in trauma pa-
tients. Journal of Trauma and Acute Care Surgery,
65(4):813–819.
Cooking Hacks (2016). e-health sensor platform v2. 0 for
arduino and raspberry pi [biometric/medical applica-
tions].
Cosoli, G., Casacanditella, L., Pietroni, F., Calvaresi, A.,
Revel, G., and Scalise, L. (2015). A novel approach
for features extraction in physiological signals. In
Medical Measurements and Applications (MeMeA),
2015 IEEE International Symposium on, pages 380–
385. IEEE.
Costa, M., Goldberger, A. L., and Peng, C.-K. (2005). Mul-
tiscale entropy analysis of biological signals. Physical
review E, 71(2):021906.
Doyle, T. L., Dugan, E. L., Humphries, B., and Newton,
R. U. (2004). Discriminating between elderly and
young using a fractal dimension analysis of centre of
pressure. Int J Med Sci, 1(1):11–20.
Eke, A., Herman, P., Kocsis, L., and Kozak, L. (2002). Frac-
tal characterization of complexity in temporal physio-
logical signals. Physiological measurement, 23(1):R1.
Goldberger, A. L., Amaral, L. A., Glass, L., Hausdorff,
J. M., Ivanov, P. C., Mark, R. G., Mietus, J. E., Moody,
G. B., Peng, C.-K., and Stanley, H. E. (2000). Phys-
iobank, physiotoolkit, and physionet. Circulation,
101(23):e215–e220.
Goli, S. and T., J. (2014). Cuff less continuous non-invasive
blood pressure measurement using pulse transit time
measurement. International Journal of Recent Devel-
opment in Engineering and Technology, 2(1):86–91.
G
´
omez, C., Hornero, R., Ab
´
asolo, D., Fern
´
andez, A., and
L
´
opez, M. (2006). Complexity analysis of the magne-
toencephalogram background activity in alzheimer’s
disease patients. Medical engineering & physics,
28(9):851–859.
Grenwis, J., Bogie, H., and Main, B. (2012). A chronic
method for measuring real-time pulse wave veloc-
ity in conscious rodents. The FASEB Journal, 26(1
Supplement):870–10.
Hall, M., Frank, E., Holmes, G., Pfahringer, B., Reutemann,
P., and Witten, I. H. (2009). The weka data min-
ing software: an update. ACM SIGKDD explorations
newsletter, 11(1):10–18.
Hassan, M. K. B. A., Mashor, M., Nasir, N. M., and Mo-
hamed, S. (2008). Measuring of systolic blood pres-
sure based on heart rate. In 4th Kuala Lumpur Interna-
tional Conference on Biomedical Engineering 2008,
pages 595–598. Springer.
Heart and Vascular Team (2016). Busting 6 myths about
blood pressure and heart rate.
i Car
´
os, J. M. S. (2011). Continuous non-invasive
blood pressure estimation. PhD thesis, Universitat
Polit
`
ecnica de Catalunya.
Kugiumtzis, D. and Tsimpiris, A. (2010). Measures of anal-
ysis of time series (mats): A matlab toolkit for compu-
tation of multiple measures on time series data bases.
arXiv preprint arXiv:1002.1940.
Labadin, J. and Ahmadi, A. (2006). Mathematical mod-
eling of the arterial blood flow. In Proceedings
of the 2nd IMT-GT Regional Conference on Mathe-
matics, Statistics and Applications, Universiti Sains
Malaysia, Penang.
Lehocki, F., Kossaczky, I., Homola, M., Skalicky, D., My-
dliar, M., and Thurzo, A. (2014). Yet another hyper-
tension telehealth solution? the rules will tell you.
In Biomedical Engineering and Sciences (IECBES),
2014 IEEE Conference on, pages 510–515. IEEE.
McBride, J. C., Zhao, X., Munro, N. B., Smith, C. D.,
Jicha, G. A., Hively, L., Broster, L. S., Schmitt, F. A.,
ECG-derived Blood Pressure Classification using Complexity Analysis-based Machine Learning
291

Kryscio, R. J., and Jiang, Y. (2014). Spectral and com-
plexity analysis of scalp eeg characteristics for mild
cognitive impairment and early alzheimer’s disease.
Computer methods and programs in biomedicine,
114(2):153–163.
Morabito, F. C., Labate, D., La Foresta, F., Bramanti, A.,
Morabito, G., and Palamara, I. (2012). Multivariate
multi-scale permutation entropy for complexity anal-
ysis of alzheimers disease eeg. Entropy, 14(7):1186–
1202.
Najarian, K. and Splinter, R. (2012). Biomedical Signal and
Image Processing. CRC Press.
Nitzan, M. (2011). Automatic noninvasive measurement
of arterial blood pressure. IEEE Instrumentation &
Measurement Magazine, 14(1):32–37.
Nye, R., Zhang, Z., and Fang, Q. (2015). Continuous
non-invasive blood pressure monitoring using pho-
toplethysmography: A review. In Bioelectronics
and Bioinformatics (ISBB), 2015 International Sym-
posium on, pages 176–179. IEEE.
Payne, R., Symeonides, C., Webb, D., and Maxwell, S.
(2006). Pulse transit time measured from the ecg:
an unreliable marker of beat-to-beat blood pressure.
Journal of Applied Physiology, 100(1):136–141.
Program, N. H. B. P. E. et al. (2004). The seventh report of
the joint national committee on prevention, detection,
evaluation, and treatment of high blood pressure.
Rangayyan, R. M. (2015). Biomedical signal analysis, vol-
ume 33. John Wiley & Sons.
Raoufy, M. R., Ghafari, T., and Mani, A. R. (2017). Com-
plexity analysis of respiratory dynamics. American
Journal of Respiratory And Critical Care Medicine,
(ja).
Schroeder, E. B., Liao, D., Chambless, L. E., Prineas, R. J.,
Evans, G. W., and Heiss, G. (2003). Hypertension,
blood pressure, and heart rate variability. Hyperten-
sion, 42(6):1106–1111.
Wong, M., Poon, C., and Zhang, Y. (2006). Can the timing-
characteristics of phonocardiographic signal be used
for cuffless systolic blood pressure estimation? In
Engineering in Medicine and Biology Society, 2006.
EMBS’06. 28th Annual International Conference of
the IEEE, pages 2878–2879. IEEE.
Wong, M. Y.-M., Poon, C. C.-Y., and Zhang, Y.-T. (2009).
An evaluation of the cuffless blood pressure estima-
tion based on pulse transit time technique: a half year
study on normotensive subjects. Cardiovascular En-
gineering, 9(1):32–38.
Zhang, G., Gao, M., Xu, D., Olivier, N. B., and Mukka-
mala, R. (2011). Pulse arrival time is not an adequate
surrogate for pulse transit time as a marker of blood
pressure. Journal of applied physiology, 111(6):1681–
1686.
Zhang, H. X., Zhu, Y. S., and Wang, Z. M. (2000).
Complexity measure and complexity rate information
based detection of ventricular tachycardia and fibrilla-
tion. Medical and Biological Engineering and Com-
puting, 38(5):553–557.
Zhang, X.-Y. and Zhang, Y.-T. (2006). A model-based
study of relationship between timing of second heart
sound and systolic blood pressure. In Engineering in
Medicine and Biology Society, 2006. EMBS’06. 28th
Annual International Conference of the IEEE, pages
1387–1390. IEEE.
HEALTHINF 2018 - 11th International Conference on Health Informatics
292
