
Objective Evaluation of Bradykinesia in Parkinson’s Disease using
Evolutionary Algorithms
Siti Anizah Muhamed
1
, Rachel Newby
2,3,4
, Stephen L. Smith
1
, Jane Alty
3,4
,
Stuart Jamieson
3,4
and Peter Kempster
2,5
1
Department of Electronic Engineering, University of York, York, U.K.
2
Neurosciences Department, Monash Medical Centre, Victoria, Australia
3
Department of Neurology, Leeds General Infirmary, Leeds, U.K.
4
Hull York Medical School, University of York, U.K.
5
Department of Medicine, Monash University, Victoria, Australia
Keywords: Parkinson’s Disease, Evolutionary Algorithms, Cartesian Genetic Programming, Bradykinesia, Finger
Tapping, Hand Pronation-Supination, Hand Opening-closing.
Abstract: Bradykinesia, a slowing of movement, is the fundamental motor feature of Parkinson’s disease (PD) and the
only physical sign that is obligatory for diagnosis. The complex nature of Bradykinesia makes it difficult to
reliably identify, particularly as the early stages of the disease. This paper presents an extension of previous
studies, applying evolutionary algorithms to movement data obtained from the standard clinical finger tapping
(FT) test to characterise Bradykinesia. In this study, hand pronation-supination (PS) and hand opening-closing
(HO) tasks are also considered. Cartesian Genetic Programming (CGP), is the evolutionary algorithm used to
train and validate classifiers using features extracted from movement recordings of 20 controls and 22 PD
patients. Features were selected based on the current clinical definition of Bradykinesia. The results show the
potential of HO and PS to be used as effective classifiers with an accuracy of 84%. Discriminative features
were also investigated with the possibility of informing clinical assessment.
1 INTRODUCTION
Bradykinesia, meaning slowness of movement, is
the only clinical sign that is mandatory for the
diagnosis of Parkinson’s Disease (PD) (Heldman et
al. 2011). The terms akinesia (absence of movement),
bradykinesia (slowness of movement), and
hypokinesia (decreased amplitude), are all used
interchangeably to describe the most prominent
phenomena of Parkinsonism. The conditions they
describe are usually referred to collectively as
bradykinesia (Figure 1). This symptom might have
the highest potential as a motor progression marker of
Parkinson’s disease (Maetzler et al. 2009). The
complex nature of Bradykinesia itself is one of the
reasons that makes it difficult for clinicians and
neurologists to be certain of its existence in the early
stages of Parkinson’s disease. Clinicians look for
signs of bradykinesia by observing a patient’s ability
to perform rapid, repetitive, alternating movements of
the hand using tasks such as finger taps, toe taps, hand
grips and hand pronation–supination (Jankovic
2008). The gold standard for clinical evaluation is the
Unified Parkinson’s Disease Rating Scale, UPDRS,
and its modified version, MDS-UPDRS (Goetz et al.
2008). It remains unclear how slowed movements due
to physiological ageing are different from the
bradykinesia seen in parkinsonian conditions. A
better understanding of characteristics of
bradykinesia and how it differs between these groups
can be used to inform clinical assessments towards
conforming early diagnosis.
Finger tapping (FT) is a popular task that has been
used many times in studies to evaluate Bradykinesia
in PD. Several methods have been used to optimise
FT data recorded by movement sensors in studies that
use statistical tests to compare movement features of
PD patients against healthy controls (Dunnewold et
al. 1997) (Jobbágy et al. 2005) (Yokoe et al. 2009)
(Espay et al. 2011) or with other movement disorders
(Ling et al. 2012). Alternatively, popular statistical
machine learning methods such as support vector
machine (SVM) is claimed to achieve better
classification on FT movement data. (Martinez
Manzanera et al. 2015) (Patel et al. 2009).
Muhamed, S., Newby, R., Smith, S., Alty, J., Jamieson, S. and Kempster, P.
Objective Evaluation of Bradykinesia in Parkinson’s Disease using Evolutionary Algorithms.
DOI: 10.5220/0006601700630069
In Proceedings of the 11th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2018) - Volume 4: BIOSIGNALS, pages 63-69
ISBN: 978-989-758-279-0
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
63

Figure 1: Descriptions of Bradykinesia. (Fernandez et al.
2014).
Our group have successfully used Evolutionary
Algorithm (EA) to evolve high accuracy classifiers
that differentiate Parkinson's disease patients from
healthy controls (Lones et al. 2012) (Smith and
Timmis 2008). Further investigation into classifiers
evolved was able to characterise movement disorder
in PD (Lacy et al. 2013) and inform clinical
assessment (Lones et al. 2013). Based on the success
of using FT data, we believe that EAs can also be used
on other motor tasks to achieve the same if not better
results. Specifically, this study extends our work to
other common clinical motor tasks; pronation-
supination (PS) and hand opening-closing (OC) tasks.
FT was also included in this study for validation and
comparison purposes.
Cartesian Genetic Programming (CGP), a type of
EA was used to train classifiers. CGP was introduced
by Miller and Thomson (Miller and Thomson n.d.)
where the candidate solutions are represented as a
string of integers of fixed length that is mapped to a
non-cyclic directed graph. CGP and its variants have
shown excellent ability in the classification of a range
of medical applications including the classification of
mammograms for the detection of breast cancer
(Hope et al. 2007) and diagnosis of Alzheimer’s
disease (Hazell and Smith 2008). Additionally, there
were also classifications using bio-signals such as
spectral data for evaluation of cancerous thyroid cell
lines (Lones et al. 2010), digital images of the cells to
differentiate benign and malignant breast mass cells
(Ahmad et al. 2012) and electrocardiography (ECG)
signals to classify cardiac arrhythmia types (Ahmad
et al. 2013).
A distinct advantage of EAs is that the classifiers
evolved can be scrutinised to discover which features,
or even, which parts of the movement data were used
in their construction. Although statistical machine
learning methods such as SVM usually able to
generate comparable classifiers, it requires extra steps
to identify most discriminating inputs. A technique
such as forward-selection wrapper approach or other
feature ranking methods had to be integrated to
achieve the same objective.
The main objectives of this paper are to look into
the potential of applying EAs to evolve classifiers
using movement data of PS and HO tasks and
evaluate possible Bradykinesia characteristics that
later can be used to inform clinical assessments.
2 METHODOLOGY
After obtaining informed written consent, 20 controls
and 22 patients with idiopathic Parkinson’s disease
were tested using the Movement Disorders Society
Unified Parkinson’s Disease Rating Scale (MDS-
UPDRS) in a conventional clinical setting at the
Monash Medical Centre, Melbourne, Australia. The
finger tapping, pronation-supination and hand
opening-closing components of the MDS-UPDRS
were assessed both clinically and using an objective
motion tracking system.
2.1 Movement Data Collection
The motion tracking system used for movement
recording employ Polhemus Patriot Electromagnetic
(EM) tracking sensors (Polhemus 2016). The system
consists of electronic system unit (SEU), a magnetic
transmitter and two EM tracking sensors. Each
participant wears the EM sensors on index finger and
thumb when they perform the specified assessments.
The EM sensors record position and orientation
relative to the transmitter in six degrees of freedom
with an update rate of 60 Hz per sensor. The system
returns three Cartesian coordinates (X, Y, and Z) and
three orientation Euler Angles: azimuth, elevation
and roll.
2.2 Movement Features
Features were extracted based on the current clinical
definition of bradykinesia and the nature of the
movement in each task.
2.2.1 Finger Tapping
In this study, patients were asked to perform the
standard clinical finger tapping test as defined by the
Movement Disorders Society Unified Parkinson’s
Disease Rating Scale (MDS-UPDRS). This instructs
patients to perform ten finger taps as fast and as wide
as possible. As one of the final objectives of this study
is to inform clinical assessment, it is important for the
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
64
task to be performed identically with standard clinical
evaluation.
The separation distance between the finger and
the thumb during the finger tapping action was
computed by first calculating the difference between
the x, y and z coordinate values for the respective
sensors, and then, the Euclidean distance, or overall
positional separation, between index finger and
thumb. Speed and acceleration were calculated as
the first and second derivatives of the distance,
respectively. The raw movement data was also
preprocessed to remove noise using Low Pass 5Hz
Butterworth filter. Butterworth filter is most common
filter used in biomechanics data analysis due to its
excellent passband response (Christodoulakis et al.
2010).
Patients often have difficulties in performing the
exact number of the cycles as instructed. Therefore,
cycles frequency is one of the features selected
instead of time taken to finish the task. Other features
were quantified for the opening and closing phases of
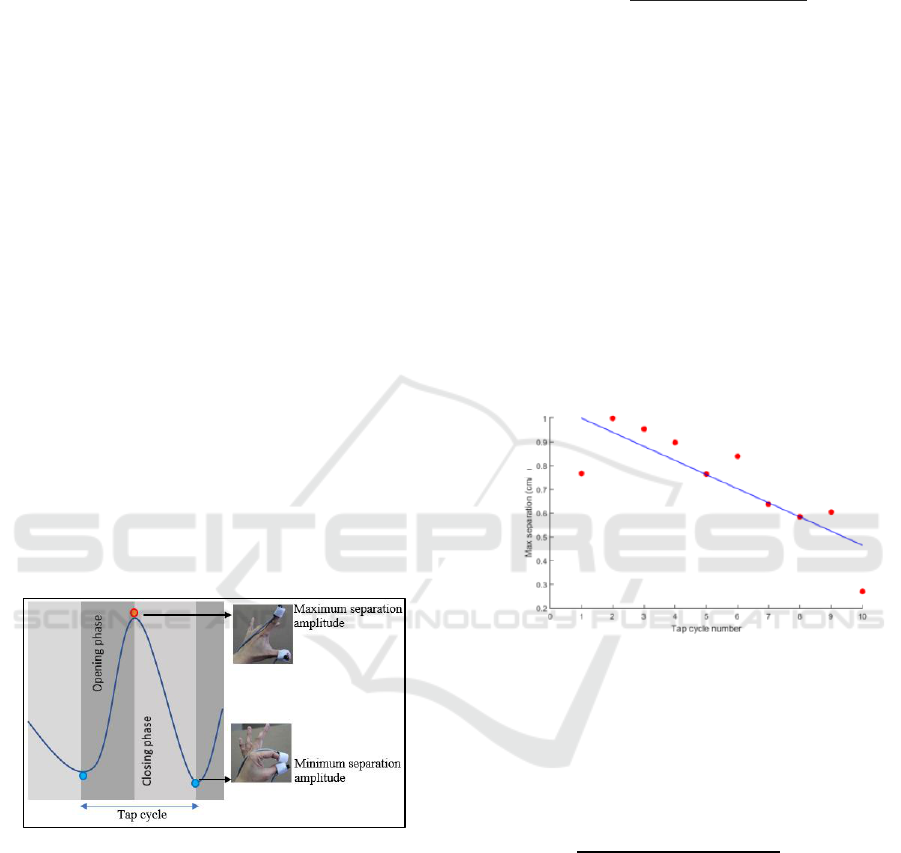
the cycle. The opening phase begins once the fingers
are separated, from an initially closed position –
equating to a minimal distance between the sensors –
to when they are maximally separated; the closing
phase begins once the sensors move towards one
another after the point of maximal separation and
finishes when the sensors have achieved a minimum
separation.
Figure 2: Separation data showing opening and closing
phases of a tapping cycle.
Figure 2 provides a representation of positional
separation data, showing opening and closing phases
of a cycle. Minimum, maximum and average of
normalised speed and acceleration of both cycle
phases were computed according to (Lacy et al.
2013). To measure rhythm, Coefficient of Variation
(COV) was used. COV reflects how much a
movement component measure varies over a defined
period. It may be considered a measure of how
rhythmic the repetitive movements are. COV of
amplitude was calculated over a period of tapping
cycles as follows:
To calculate the decrementing trend, maximum
separation amplitude or speed for each tap cycle was
linearly regressed against the number of cycles. A
negative slope indicates that the overall trend of a
movement component measure is decrementing and a
zero or positive slope indicates that the amplitude is
not decrementing. Figure 3 provides examples of
linear regression plots of maximum amplitude to
obtain the slope indicating a trend of separation
amplitude. Measures of amplitude and speed alone
may not have captured the real movement patterns of
subjects. To capture the relationship between these
components a variable called periodicity was
calculated.
Figure 3: An example of tap decrementing trend for a
patient with slope = -0.6.
Other features extracted are halts, hesitation and
amp*freq. Halts were measured by calculating the
percentage of the tap cycle duration spent at ‘zero’ (<
5% of the maximum) speed:
When the movement showed smaller peaks
between tapping cycle phases (Figure 4), it is treated
as hesitation.
Objective Evaluation of Bradykinesia in Parkinson’s Disease using Evolutionary Algorithms
65

Figure 4: The smaller peaks counted as four hesitations.
Bigger amplitude with greater frequency during
finger tapping means faster finger movement. This is
considered as better performance. Alternatively, the
movement can be executed faster with smaller
amplitude. The amplitude × frequency of tapping is
suggested in (Jobbágy et al. 2005) to characterise the
speed. This feature is determined for each tapping
cycle and then averaged over the whole test. Table 1
summarises all features used as inputs to the CGP
classifier.
Table 1: Finger tapping extracted features.
Feature
(0)
Cycles frequency
(1)
Max overall amplitude
(2)
Mean amplitude
(3)
Maximum overall speed
(4)
Max opening speed
(6)
Max closing speed
(7)
Max opening acceleration
(8)
Max opening deceleration
(9)
Max closing acceleration
(10)
Max closing deceleration
(11)
Periodicity
(12)
COV amplitude
(13)
COV speed
(14)
Decrementing amplitude
(15)
Decrementing speed
(16)
Halts
(17)
Hesitation
(18)
Amp*freq
2.2.2 Hand Pronation-supination
For the hand pronation-supination task (PS), the
MDS-UPDRS requires the participant to extend the
arm out in front of their body with the palms face
down and then turn the palm up and down alternately
10 times as fast and fully as possible.
After some experimentation, it was concluded that
the most useful data in our pronation-supination
recordings came from the movement of the thumb.
Since only one sensor is used, the amplitude is
defined as the Euclidean distance between thumb
sensor and Patriot transmitter.
Velocity was calculated by differentiation of each
Cartesian coordinate component (x, y, z) over the
sampling time period to compute the respective
velocity components
. The total velocity
was computed from the sum of its components and its
magnitude, the speed
Acceleration is obtained by differentiating the
velocity, using the same sampling time. The same
features in Table 1 were used for PS classifiers by
replacing opening and closing phases with pronation
and supination phases respectively.
Since PS involves angular movements, movements
were computed using Euler angles. Average,
minimum and maximum of angular velocity and
angular acceleration values were calculated
according to (Picardi et al. 2010), giving the
additional six angular features shown in Table 2.
Table 2: Hand pronation-supination features.
Feature
(19)
Mean angular speed
(20)
Max angular speed
(21)
Min angular speed
(22)
Mean angular acceleration
(23)
Max angular acceleration
(24)
Min angular acceleration
2.2.3 Hand Opening-closing
For the hand opening-closing task (HO), the MDS-
UPDRS requires the participant to make a tight fist
with the arm bent at the elbow so that the palm faces
the examiner and then requires the participant to open
the hand ten times as fully and as quickly as possible.
Sensors were placed at the same positions as in finger
tapping task. However, unlike finger tapping, which
is a simultaneous movement of thumb and fingers, the
hand-opening task involves two steps movements.
Therefore, the features extracted were also taking into
account the measurements of both sensors separately,
instead of just considering the distance between the
two sensors. The thumb sensor (TS) and finger
sensor (FS) movement data were used to compute the
total of seventeen features. (Table 3).
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
66
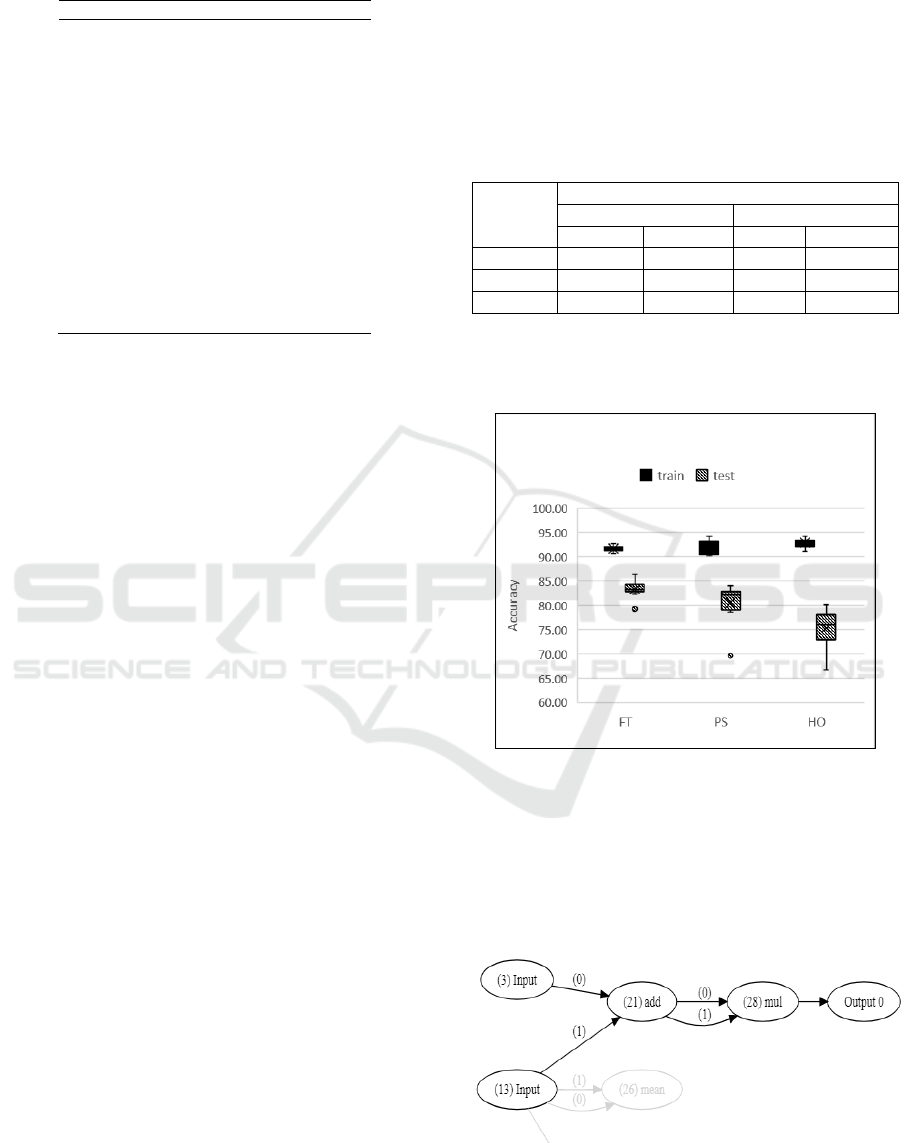
Table 3: Hand opening-closing extracted features.
Feature
(0)
HO frequency
(1)
Maximum opening
(2)
COV opening
(3)
TS average speed
(4)
TS minimum speed
(5)
TS maximum speed
(6)
TS minimum acceleration
(7)
TS maximum acceleration
(8)
TS COV speed
(9)
TS Halts
(10)
FS average speed
(11)
FS minimum speed
(12)
FS maximum speed
(13)
FS minimum acceleration
(14)
FS maximum acceleration
(15)
FS COV speed
(16)
FS Halts
2.3 Classification
Classification used a typical CGP evolutionary
strategy which selects one parent from each
generation and uses mutation to produce four
children. The next generation then comprises the
parent and the four children, giving a population of
size five - four children plus one parent: (1+4) - ES.
Three sets of classifiers were evolved, one for each
movement task. The input data consists of floating
point values representing selected Bradykinesia
features extracted from the patient’s movement (as
defined in section 2.2).
The fitness assigned to each classifier is simply
the proportion of samples correctly classified.
Previous CGP classifiers in FT studies (mentioned in
the introduction) used the area under a ROC Curve
(Fawcett 2006) as fitness function, but in this study,
classification accuracy is used for simplicity and
direct comparison. Through experimentation, the
following CGP parameters values were adopted:
number of nodes available 15, nodes arities of 2,
mutation rate of 0.05 and number of generations
10000. The function set comprised
. Data from each class was
divided into training and test sets. To compensate for
any effect on results caused by small amounts of
training and test data, 5-fold cross validation was
used. Results are averaged over ten runs for statistical
significance. The best classifier model is used to
determine those features that are most discriminative.
3 RESULTS
With numbers of subjects relatively low compared to
our previous FT studies, the classifications accuracies
in this study are surprisingly good. For the finger
tapping task, averaged accuracy of the test set across
ten runs is 82.66%. For the pronation-supination task,
80.54%, and for the hand opening task, 75.32%. Best
and average accuracies of all tasks are summarised in
table 4.
Table 4: Average and best accuracies of classifiers evolved
for all motor tasks.
Task
Accuracy
Averaged ten runs
Best run
train
test
train
test
FT
91.69
83.3
91.79
87.20
PS
92.04
80.54
94.34
84.03
HO
92.92
75.32
94.27
80.21
Figure 5 showing the distribution of cross-
validated classification accuracies for ten runs of each
task.
Figure 5: Distribution of accuracies across ten runs.
As mentioned in the introduction, one of the main
advantages of using GP method is the ability to
recognise which inputs were used to evolve the
strongest classifier. For example, the PS classifier
with 85% accuracy is visualised in figure 6, showing
only the active nodes.
Figure 6: Visualisation of a PS classifier.
In this example, the inputs used are maximum
Objective Evaluation of Bradykinesia in Parkinson’s Disease using Evolutionary Algorithms
67

overall speed and speed rhythm (COV). All features
used to evolve the best classifier of each task are
summarised in table 5.
Table 5: Most discriminating features for each task.
Task
Features
FT
(4)(7)(9) (14) from Table 1
HO
(0)(2)(3)(4)(5)(6) (14) from Table 2
PS
(8) (13) (14) (16) (17) (20) from Table 1*
and Table 2
* replaced opening phase with pronation phase and closing
phase with supination phase.
4 CONCLUSIONS
It is clear from the classification results that hand
opening-closing and hand pronation-supination have
the same potential as finger tapping to be used as a
tool in the characterisation of Bradykinesia using GP
to inform clinical assessment. The overall accuracy
was lower than shown in previous studies of GP
classifications using finger tapping data, but we
believe this is due to smaller numbers of subjects.
Almost all classifiers across ten runs for all tasks are
consistent with good accuracies above 70%.
Although the most discriminative movement features
in this study may not be generalised to inform clinical
assessment because of the small sample numbers, it
was demonstrated that by using GP, it could easily be
acquired.
Movement features are computed based on the
current clinical definition of Bradykinesia. However,
CGP has the ability to accept raw positional or speed
data points and perform an unbiased search that will
not be constrained by pre-defined characteristics.
Future work will process PS and HO data using a
sliding window, similar to FT acceleration data in
continuous time series adopted by (Lones et al. 2014).
By using raw data points to induce classifiers, it opens
the possibility of finding new features of
Bradykinesia from the movement tasks.
REFERENCES
Ahmad, A.M. et al., 2012. Breast cancer detection using
cartesian genetic programming evolved artificial neural
networks. Proceedings of the fourteenth international
conference on Genetic and evolutionary computation
conference - GECCO ’12, p.1031.
Ahmad, A.M., Khan, G.M. & Mahmud, S.A., 2013.
Classification of Arrhythmia Types Using Cartesian
Genetic Programming Evolved Artificial Neural
Networks. , pp.282–291.
Christodoulakis, G. et al., 2010. On the filtering and
smoothing of biomechanical data. Communication
Systems Networks and Digital Signal Processing
(CSNDSP), 2010 7th International Symposium on,
pp.512–516.
Dunnewold, R.J., Jacobi, C.E. & van Hilten, J.J., 1997.
Quantitative assessment of bradykinesia in patients
with Parkinson’s disease. Journal of neuroscience
methods, 74, pp.107–112.
Espay, A.J. et al., 2011. Differential response of speed,
amplitude, and rhythm to dopaminergic medications in
Parkinson’s disease. Movement Disorders, 26(14),
pp.2504–2508.
Fawcett, T., 2006. An introduction to ROC analysis.
Pattern Recognition Letters, 27, pp.861–874..
Fernandez, H.H., Machado, A.G. & Pandya, M., 2014. A
Practical Approach to Movement Disorders: Diagnosis
and Management., Demos Medical Publishing.
Goetz, C.G. et al., 2008. Movement Disorder Society-
Sponsored Revision of the Unified Parkinson’s Disease
Rating Scale (MDS-UPDRS): Scale presentation and
clinimetric testing results. Movement Disorders,
23(15), pp.2129–2170.
Hazell, A. & Smith, S.L., 2008. Towards an Objective
Assessment of Alzheimer â€
TM
s Disease : The
Application of a Novel Evolutionary Algorithm in the
Analysis of Figure Copying Tasks. , pp.2073–2079.
Heldman, D. a et al., 2011. The modified bradykinesia
rating scale for Parkinson’s disease: reliability and
comparison with kinematic measures. Movement
disorders : official journal of the Movement Disorder
Society, 26(10), pp.1859–63.
Hope, D.C., Munday, E. & Smith, S.L., 2007. Evolutionary
Algorithms in the Classification of Mammograms.
2007 IEEE Symposium on Computational Intelligence
in Image and Signal Processing, (CIISP), pp.258–265.
Jankovic, J., 2008. Parkinson’s disease: clinical features
and diagnosis. Journal of neurology, neurosurgery, and
psychiatry, 79(4), pp.368–76.
Jobbágy, A. et al., 2005. Analysis of finger-tapping
movement. Journal of neuroscience methods, 141(1),
pp.29–39.
Lacy, S.E., Lones, M. a & Smith, S.L., 2013.
Characterisation of Movement Disorder in Parkinson 's
Disease using Evolutionary Algorithms Categories and
Subject Descriptors., (August 2009), pp.1479–1485.
Ling, H. et al., 2012. Hypokinesia without decrement
distinguishes progressive supranuclear palsy from
Parkinson’s disease. Brain, 135, pp.1141–1153.
Lones, M.A. et al., 2013. Evolving Classifiers to Inform
Clinical Assessment of Parkinson ’ s Disease. , pp.76–
82.
Lones, M.A. et al., 2012. Evolving Classifiers to Recognise
the Movement Characteristics of Parkinson’s Disease
Patients. , pp.1–18.
Lones, M.A. et al., 2014. Evolving Classifiers to Recognize
the Movement Characteristics of Parkinson’s Disease
Patients. , 18(4), pp.559–576.
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
68

Lones, M. a. et al., 2010. Discriminating normal and
cancerous thyroid cell lines using implicit context
representation Cartesian genetic programming. IEEE
Congress on Evolutionary Computation, pp.1–6.
Maetzler, W., Liepelt, I. & Berg, D., 2009. Progression of
Parkinson’s disease in the clinical phase: potential
markers. The Lancet Neurology, 8(12), pp.1158–1171.
Martinez Manzanera, O. et al., 2015. A method for
automatic and objective scoring of bradykinesia using
orientation sensors and classification algorithms. IEEE
Transactions on Biomedical Engineering, 9294(c),
pp.1–1.
Miller, J.F. & Thomson, P., 2000. Cartesian genetic
programming. Genetic Programming, Proceedings of
the Third European Conference on Genetic
Programming (EuroGP2000)., 1802, pp.121–132.
Patel, S. et al., 2009. Monitoring Motor Fluctuations in
Patients With Parkinson’s Disease Using Wearable
Sensors. IEEE Transactions on Information
Technology in Biomedicine, 13(6), pp.864–873.
Picardi, C. et al., 2010. Objective Assessment of Cognitive
Impairment in Parkinson’s Disease Using Evolutionary
Algorithm C. Di Chio et al., eds., Berlin, Heidelberg:
Springer Berlin Heidelberg.
Polhemus motion tracking company. (2016).
http://polhemus.com.
Smith, S.L. & Timmis, J., 2008. An immune network
inspired evolutionary algorithm for the diagnosis of
Parkinson’s disease. Bio Systems, 94(1–2), pp.34–46.
Yokoe, M. et al., 2009. Opening velocity, a novel
parameter, for finger tapping test in patients with
Parkinson’s disease. Parkinsonism & related disorders,
15(6), pp.440–4.
Objective Evaluation of Bradykinesia in Parkinson’s Disease using Evolutionary Algorithms
69
