
A Procedural Model for Snake Skin Texture Generation
Jefferson Magalh
˜
aes Pinheiro
1,2
and Marcelo Walter
1
1
Instituto de Inform
´
atica, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil
2
Faculdade de Inform
´
atica, Centro Universit
´
ario Ritter dos Reis - UniRitter, Porto Alegre, Brazil
Keywords:
Texturing, Procedural Generation, Texture Synthesis, Snakes, Serpents, Mathematical Biology.
Abstract:
There are thousands of snake species in the world, many with intricate and distinct skin patterns. This diversity
becomes a problem for users who need to create snake skin textures to apply on 3D models, as the difficulty
for creating such complex patterns is considerable. We present a procedural model capable of synthesizing a
wide range of texture skin patterns from snakes. Our model was derived from a visual assessment of a large
number of snakes, and uses image processing as well as cellular automata to generate textures. Our results
show good visual similarity with real skin found in snakes. The resulting textures can be used not only for
computer graphics texturing, but also in education about snakes and their visual characteristics. We have also
performed a user study to assess the usability of our tool. The score from the System Usability Scale was 85.8,
suggesting a highly effective texturing tool.
1 INTRODUCTION
Snakes (reptiles of the Serpentes suborder) are numer-
ous in species. They are present on every continent,
except Antarctica and some islands (notably Ireland,
Iceland, Greenland, Hawaii and New Zealand) (Bau-
chot, 2006). Some aquatic species live in the Indian
and Pacific oceans as well. Being so widespread, it
is natural that snakes are typically depicted in digi-
tal media – such as computer animations and video
games – where these animals must be digitalized,
which means creating a 3D model and their textures.
When we consider textures, a complicating factor
comes into play: there are over 3600 snake species,
according to The Reptile Database (Uetz et al., 2016),
which means a large variety of colors and patterns.
Creating such textures manually, from scratch, may
prove to be a very time-consuming task. Extract-
ing textures from photos will be difficult because un-
wrapped snake skin textures are not easily found, and
collecting a specimen for photography could also be
a complicating factor. Procedural texture generation
comes as a possible solution to this problem. Another
advantage of procedural generation is that, since the
end result is driven by a mathematical formula, cer-
tain parameters (such as colors and random seed) can
be modified to generate different patterns, and a col-
lection of visually diverse population. In addition, it
demands no art skills from the user.
This paper introduces a model to procedurally
generate snake skin textures, with the main purpose
of using them in computer graphics applications, such
as animations and games. We also discuss the advan-
tages of this technique over traditional texturing meth-
ods. We have implemented a tool for our model and
we have also assessed the usability of this tool.
2 RELATED WORK
We were unable to find any work in the related litera-
ture that focuses on snakes (or any reptilian) skin tex-
ture generation, to apply on a 3D model and use on
a modern renderer. However, this field was already
studied in the Mathematical Biology field by (Murray
and Myerscough, 1991) and (Cocho et al., 1987a).
These authors discuss how snake skin patterns can
be generated using a mathematical model, which is
precisely the basis for procedural texture generation.
Cocho uses cellular automata, which are further ex-
plained on section 4.1.4, and Murray uses reaction-
diffusion, which is further explained on section 4.1.5.
One drawback of these models for our goals is
that they can generate textures that are visually too
simple to use on many modern applications. For
instance, typically only black-and-white images are
synthesized, and in the case of Cocho’s work, the res-
olution is extremely low (about a dozen pixels wide).
Regardless, these works serve as a good basis for a
Pinheiro, J. and Walter, M.
A Procedural Model for Snake Skin Texture Generation.
DOI: 10.5220/0006626401330144
In Proceedings of the 13th International Joint Conference on Computer Vision, Imaging and Computer Graphics Theory and Applications (VISIGRAPP 2018) - Volume 1: GRAPP, pages
133-144
ISBN: 978-989-758-287-5
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
133

more elaborate (detailed) texture generation method.
Other works focus on snakes in computer graph-
ics, but not specifically on texture generation. One
of these focuses on snakes (and other materials) iri-
descence (Dhillon et al., 2014). Another two studies,
(Panagiotakis and Tziritas, 2006) and (Miller, 1988),
focus on their movement patterns.
Regarding procedural texturing, there are many
works available in literature. Some works worth men-
tioning include (Turk, 1991) and (Witkin and Kass,
1991), who also used reaction-diffusion. However,
these methods are too generic, and it is unclear how
to apply them to our domain (snakes) such that it cov-
ers all, or most of, snake skin patterns. In addition,
another goal of our procedural model is to be very
fast, allowing for an iterative texture creation process,
and these methods are too computationally intensive.
Other texture synthesis methods such as (Efros
and Leung, 1999) and (Gilet et al., 2014) (the latter
even included a snake as an example) rely on a good
amount and quality of sample input images, therefore,
would be impractical in the case of snakes (due to the
difficulty to obtain good photographies for texturing).
(Hendrikx et al., 2013) offers a recent survey on gen-
erating procedural content for games, including tex-
tures.
3 SNAKES BIOLOGY
Snakes are numerous in species and in skin pattern
variation. Some have only a single plain color, while
others have patterns composed of two or more colors.
The pattern can be as simple as stripes or spots, or it
can be so complex that it is even difficult to describe.
The Serpentes suborder is split into 26 fam-
ilies, the most important (numerous) being the
Boidae (Boas), Colubridae (Colubrids), Dipsadi-
dae, Pythonidae (Pythons), Elapidae, and Viperidae
(Vipers and Pit Vipers).
3.1 Skin Pattern
We are mostly interested in the snake’s visual appear-
ance, that is, pattern and colors. When taxonomists
discover a new snake species, they describe, among
other things, their appearance. However, there is not
an international standard for categorization of snake
skin pattern, such as, “if it looks like this then it
should be categorized as Spots; if it looks like that
then it should be categorized as Horizontal Stripes”.
Instead, taxonomists verbally describe the species’
pattern.
The first step in our study was therefore to create
a categorization of snakes’ visual pattern, and assign
each snake species to one of these categories. This
will be useful to help determining the pattern synthe-
sis method, and also to determine what pattern types
are mostly present in nature. Since it is very hard to
find images of a snake’s ventral side (the underside,
or “belly”), and it is typically not visible, we decided
to consider only the visible dorsal side (“back” and
sides). Also, some species have a significantly differ-
ent pigmentation color or pattern on their head or tail
(such as the Apostolepis assimilis, which has no pat-
tern on its body, but its head is black); again, we did
not take this into consideration. Both topics will be
explored in future work.
With the help of a herpetologist, we defined the
following six snake pattern categories. Please note
that when we say “body”, we mean only the dorsal
side of a snake. We show two figures for each cate-
gory, as examples.
1. No Pattern. The snake shows no pigmentation
pattern, consisting only of a single solid color.
Note that we do not consider head or tail pig-
mentation, thus the Apostolepis assimilis is cat-
egorized as having no pattern.
Figure 1: Left: Philodryas aestiva (source: (Nogueira,
ndb)). Right: Apostolepis assimilis (source: (Nogueira,
nda)).
2. Longitudinal Stripes. The snake shows one or
more stripes aligned along its body, that go from
neck to tail or from head to tail.
Figure 2: Left: Phalotris lemniscatus (source: (Borges-
Martins, 2007b)). Right: Philodryas olfersii (source:
(Sawaya, nd)).
3. Transversal Stripes. The snake shows many
stripes aligned perpendicularly to its body axis,
also known as “rings”. These rings may be in two
alternating colors, or may be in multiple colors.
Typical examples include corals and false corals.
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
134

Figure 3: Left: Micrurus altirostris (coral, source: (Borges-
Martins, 2007a)). Right: Rhinobothryum lentiginosum
(false coral, source: (de Albuquerque, nd)).
4. Spots. The snake shows regular (similar) shapes
on its body, often elliptical.
Figure 4: Left: Liophis miliaris (source: (de Aguiar Pas-
sos, 2013)). Right: Ptychophis flavovirgatus (source: (Di-
Bernardo, nd)).
5. Other Simple Pattern. A few snakes show a
pattern that is easily described and repeats along
its body, but doesn’t fall on any of the categories
above. Examples include the Bothrops alternatus,
which has many C-shaped patterns along its body
(see Figure 5).
Figure 5: Left: Oxyrhopus rhombifer (source: (Timm,
2014)). Right: Bothrops alternatus (source: (Borges-
Martins, 2007c)).
6. Complex. When the pattern is none of the above,
it is categorized as “Complex”. Typically, this in-
cludes intricate (or abstract) camouflage patterns,
that would be hard to describe verbally, or with
numerous different elements on its body.
Figure 6: Left: Sibynomorphus turgidus (source: (May,
2010)). Right: Python regius (source: (Campbell, 2005)).
Our goal is to assign each species to one category,
so that we can determine the relative proportions be-
tween different categories. This was done by means
of a visual assessment of each species’ photos.
3.2 Scope Limitation
Naturally, it would be tremendous work to analyze the
skin pattern of all 3600+ snake species. Scope had to
be limited somehow.
For this study, we decided to limit the scope ge-
ographically. In the state of Rio Grande do Sul,
Brazil, there are dozens of snake species, at least 81
of which have been identified and catalogued in the
book (Abegg and Entiauspe Neto, 2012). In this book,
these 81 species have been described as accurately as
possible, however, in text format (instead of a well-
structured data table).
It is important to note that although we limited the
scope to the snakes in this specific region, all major
snake families are present in it, with the exception of
Pythonidae. Therefore, we deemed our subset as a
good representation of the whole and use the book as
a guide.
3.3 Skin Pattern Analysis Results
In the studied region the most expressive family is
Dipsadidae, with 59 species (72.8%), followed by
Viperidae (7 species, 8.6%), Colubridae (5 species,
6.1%) and Elapidae (5 species, 6.1%).
After analyzing pictures of these 81 species, we
categorized them as the following pattern types (as
discussed on section 3.1):
Table 1: Pattern types distribution.
Pattern Count % of Total
No Pattern 19 23.46%
Longitudinal Stripes 19 23.46%
Transversal Stripes 7 8.64%
Spots 8 9.88%
Other Simple Pattern 9 11.11%
Complex Pattern 19 23.46%
From this analysis, we have learned that 76.54%
of snake species from the sample region have some
type of skin pattern and would benefit from the cre-
ation of a procedural model.
4 THE PROCEDURAL MODEL
The goal of our procedural model is to generate high
resolution textures (e.g. 1024x1024 to 4096x4096
A Procedural Model for Snake Skin Texture Generation
135

pixels) for use in a modern renderer. As such, it
should generate at least a Color map and a Height
map. We will also generate a Roughness map, which
describes how rough or glossy the surface looks, and
is explained further in Section 4.3. The generated tex-
tures will be applied to a previously-made 3D mesh
and should yield a realistic result. It should also be
very fast, outputting the resulting textures in less than
a second, allowing for an iterative texture creation
process.
In our model, all texture sizes are normalized in
the [0.0;1.0] range, with [0.0;0.0] being the top-left
corner, and [1.0;1.0] the bottom-right corner. The tex-
ture generator can synthesize any texture size, as long
as there is processing power. We now proceed to ex-
plain how the color map is generated, followed by the
height map and roughness map.
4.1 Color Map
The final texture’s color map begins with a single
solid color, which forms the “background” of the tex-
ture. This is the snake’s predominant skin color. On
top of this, the specific pattern type is drawn. We now
proceed to explain how each pattern type is generated,
except “No Pattern”.
4.1.1 Transversal Stripes Pattern
Our model supports up to 4 distinct stripes colors and
sizes. For each color we can define where it starts and
ends using a two-dimensional vector parameter, with
values ranging from 0.0 to 1.0.
For instance, if the four input colors are red,
green, blue and yellow and the begin/end vectors are
[0.0;0.25], [0.3;0.45], [0.6;0.65] and [0.8;1.0] respec-
tively for each color, the resulting image would be as
seen in Figure 7. Small gray-and-white squares mean
transparency, which will be filled by the background
color.
Figure 7: Example of generated Transversal Stripes Pattern.
We do not know of any snake species that has
more than 5 colors in its pattern (the background plus
the 4 stripes color), so we did not deem necessary to
allow more colors on the transversal pattern, though
that could be easily implemented. Some of the most
colorful snake are the corals, with 3 colors, typically
red, black and yellow.
4.1.2 Longitudinal Stripes Pattern
The longitudinal pattern implementation can be de-
scribed by the following steps. Figure 8 shows an ex-
ample of each step’s result.
1. A single white stripe is generated with the width
set by the user;
2. The result from step 1 is repeated a number of
times set by the user. If greater than 1, the stripe’s
width will be divided a number of times equal to
the parameter;
3. The result from step 2 is squeezed according to
a parameter set by the user. Texture tiling is dis-
abled at this step;
4. Stripe and background colors are blended with
stripes color (a single color chosen by the user)
and transparency.
Figure 8: Example of generated Longitudinal Stripes Pat-
tern.
4.1.3 Spots Pattern
The spots pattern is generated by the following steps:
1. A single circumference is synthesized. Its inten-
sity is 1.0 at the center and 0.0 at the edges and
decays linearly;
2. Its mid-levels are adjusted so that it turns more
opaque;
3. The spots color (set by the user) is applied to it;
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
136
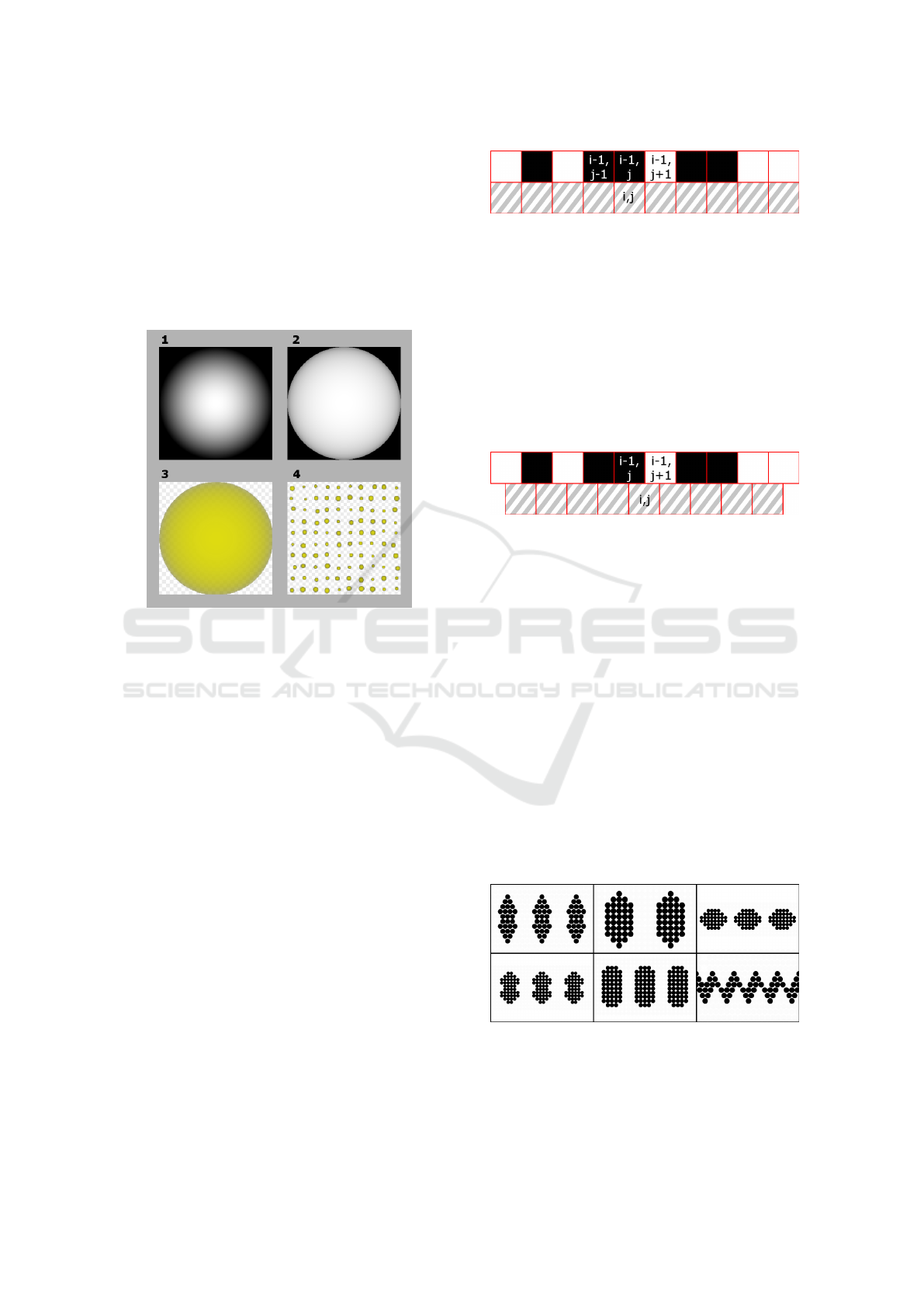
4. It is tiled in a 10x10 grid, and other parameters are
then applied to control the various characteristics
of the pattern, such as tiling, random position vari-
ation, size and random size variation.
Figure 9 shows an example of the generation pro-
cess. The resulting texture has spots that are slightly
different in size and not precisely aligned in the grid
because parameters that control randomness in size
and location have been set.
Figure 9: Example of generated Spots Pattern.
4.1.4 Other Simple Pattern
For the simple pattern group, which includes repeat-
ing geometrical patterns, we took inspiration from the
works of Cocho (Cocho et al., 1987a) (Cocho et al.,
1987b), who used cellular automata to create simple
patterns, including snake skin.
As in (Cocho et al., 1987b), we have two types
of cellular automata: rectangular and triangular, and
both work similarly. The automaton is initialized with
a single row of bits (usually 10 to 25 bits, depending
on the desired pattern size), defined by the user. This
will be the texture’s first row: each bit is a pixel, with
zeros being white, and ones being black. Then, for
a predefined number of loops (called “Epochs”), the
next rows are calculated as follows.
For rectangular automata, there is a transition rule
that is defined by four bits. Assume we are calculat-
ing the value for pixel [i,j]. The algorithm takes the
previous row’s pixel value, [i-1,j], and sums it with its
adjacencies, [i-1,j-1] and [i-1,j+1]. See Figure 10.
If the result of this sum is 0, the last (rightmost)
bit of the transition rule is the new pixel value. If the
sum is 1, the 3rd bit is used. If the sum is 2, the 2nd
bit is used, and if the sum is 3, the first bit is used.
In case there are no adjacent cells (for calculating the
Figure 10: Values used for calculating position i,j in a rect-
angular automaton.
left-most or right-most cells), the value from the other
end is used (i.e. wrap around).
The triangular automaton works similarly, how-
ever, each odd row is displaced 50% of each element’s
diameter to the right, and only the two cells above
it are used for the sum (see Figure 11). Since the
maximum sum is 2, the transition rule has only 3 bits
(for sums of 0, 1, and 2), following the same logic as
above.
Figure 11: Values used for calculating position i,j in a trian-
gular automaton.
Once all rows have been calculated (given by the
number of epochs), an image is synthesized using the
resulting bit matrix. We paint white circles for zeros
and black circles for ones, and the diameter of these
circles is adjustable (50 pixels by default). While we
could have used the bits as vertices and drawn using
vectors, we deem the circles approach to be simpler
and faster to compute. Afterwards, the resulting im-
age is duplicated above the original and mirrored ver-
tically, to create a symmetrical texture. This mirror
may be displaced horizontally to create interleaving
patterns.
Figure 12 shows some examples of generated im-
ages using this algorithm. All these images represent
a specific snake species’ pattern. Although simple,
cellular automata can generate many patterns visually
similar to real snakes.
Figure 12: Sample results obtained using cellular automata.
From top-left to bottom-right: Bothrops neuwiedii, Crotalus
viridis, Daboia russelii, Eunectes murinus, Python molurus
and Vipera berus.
After generating the image, additional steps are
A Procedural Model for Snake Skin Texture Generation
137

taken to remove the circles pattern. The image is
blurred and then its contrast is increased. See Figure
13 for an example.
Figure 13: Cellular automaton’s resulting image post pro-
cess. Left: the generated image. Middle: blurred image.
Right: contrast increased.
4.1.5 Complex Pattern
Snakes categorized as having Complex pattern
present spots of different shapes and sizes, often
forming a camouflage pattern as seen in many mili-
tary vehicles. This pattern’s form is apparently ran-
dom, being hard to even describe it.
We believe that Murray’s reaction-diffusion sys-
tems would perform well to generate these patterns.
Indeed, complex snake skin patterns have been gen-
erated, as discussed in his paper (Murray and Myer-
scough, 1991). However, the computational cost of
running a reaction-diffusion simulation is high com-
pared to our other methods and therefore Murray’s
method was not implemented and is left as a future
improvement.
4.1.6 Pattern Post-process
One problem up to this point is that the generated pat-
tern is very artificial. Lines are perfectly straight and
curves are perfectly round. To counter this, we ap-
ply a distortion effect, using two Perlin noises (Per-
lin, 2002) of different granularities: one of low fre-
quency to provide a macro variation, and one of high
frequency to provide a micro variation. The intensity
of these distortions are adjustable. See Figure 14 for
an example.
4.1.7 Pattern Outline
Some snakes, such as the eastern milk snake (Lampro-
peltis triangulum triangulum) and the burmese python
(Python bivittatus) also present a strong color contrast
on the borders of their pattern (see Figure 15). For this
reason, we implemented a pattern outliner.
The outline is created by the following steps:
1. Perform an edge detection on the pattern image;
2. Distort it using the height map from the scales
(scales generation is explained on section 4.2).
Figure 14: Left: input image. Middle: low frequency dis-
tortion applied (above) and the noise used (below). Right:
high frequency distortion applied (above) and the noise used
(below).
Figure 15: Left: Lampropeltis triangulum triangulum
(source: (Rada, 2008)). Right: Python bivittatus (source:
(Andrews II, 2010)).
This is done so that the outline doesn’t matches
perfectly the contour of the pattern, but is ad-
justable;
3. A blur is applied, and contrast is increased. This
is to remove some of the irregularities introduced
in step 2. Both the blur and the contrast intensities
are adjustable.
Figure 16 shows an example of the outline being
created step by step.
Figure 16: Outliner steps. (a) Input image. (b) Edge detec-
tion. (c) Distortion. (d) Blur. (e) Contrast.
4.2 Height Map Generation
Snakes are scaled reptilians; as such, it is important
for our model to reproduce them, and the height map
is used for rendering this. Scales generation begins
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
138

with first synthesizing a single scale, which will later
be tiled. We can notice from reference images (see
Figures 1 to 6) that most snake scales are leaf-shaped,
and also that some scales are smooth and others have
a protruding ridge along its longitudinal direction.
Those are called keeled scales, an evolution of smooth
scales seen in some snakes that causes light to dis-
perse through the surface, increasing surface rough-
ness.
Scale generation is performed by the following
steps. See Figure 17 for a visual depiction of each
step’s result.
a A grayscale circumference is generated. Its inten-
sity is 1.0 at the center and linearly decays to 0.0
at the edges;
b Mid-levels are adjusted to make it more solid;
c One copy is created and translated to the left...
d ... to the right...
e ... and blended together by taking the minimal
value between these two. This is to generate the
scale’s leaf shape;
f A blur is applied...
g ... and contrast is increased to make it smoother,
particularly on the top and bottom ends;
h It is multiplied with a linear gradient, ranging
from black to white, to create the scale’s height
map;
i If the scale is keeled, then a thin linear gradient
ranging from black-white-black is blended into
the center of the scale.
Figure 17: Scale generation steps.
Once the single scale is generated, it is dupli-
cated horizontally and vertically a number of times
adjustable by parameters (i.e. the number of scales).
It is also possible to add a random variation to size,
location and rotation for each duplicated scale, so the
result looks more natural. On the left and right ends,
we stretch the texture to create ventral (underside)
scales. See Figure 18.c for an example.
We can also enhance fine details on this texture
by using a high frequency noise. We can notice from
reference images that scales’ surface has some irreg-
ularities, which we model by adding a Fractal Sum
noise texture (Mandelbrot and Pignoni, 1983) to the
height map. The intensity should be very low as its
purpose is only to add micro details.
4.3 Roughness Map
We also synthesize another texture to describe how
glossy or rough the surface looks. This is important
because scales on some snake species have a glossier
appearance than others. The Roughness map is a
grayscale texture where black means the surface is not
rough (i.e., glossy or polished) and white means it is
rough (diffuse). It is used in certain game engines
that use a physically-based renderer, such as Unreal
Engine 4. For more details on physically-based ren-
dering, we recommend (Pharr et al., 2016).
While we could have used a single solid color for
the Roughness map, we chose to use the same frac-
tal sum (Mandelbrot and Pignoni, 1983) noise texture
from the Height map, to add fine details. Output lev-
els are adjustable as a means to adjust the minimal
and maximum surface roughness. Figure 18.d shows
an example of generated Roughness map.
Figure 18 shows all generated textures for the Mi-
crurus altirostris, as an example.
Figure 18: Generated textures for the Micrurus altirostris.
From left to right: color, height and roughness maps.
5 RESULTS
We implemented our model to allow the end-user to
create snake skin textures by changing all available
parameters. It also displays what the generated tex-
tures look like, shows a 3D render of the textures ap-
A Procedural Model for Snake Skin Texture Generation
139

plied on a manually modelled snake mesh, allows to
load and save presets (all parameters values), change
generated textures resolution, and export textures for
use in another software.
To implement the texture generation process,
we used the software Substance Designer version
2017.1.0, by Allegorithmic SAS. It is a node-based
procedural texture generator, which performs many
image processing routines such as blur, levels adjust-
ment (brightness, contrast, minimal output and max-
imal output), 2D transformations and distortion; and
also has several built-in noise generators, such as Per-
lin noise (PERLIN, 1985). We chose this software be-
cause it is easy to use, has most routines we’ll need al-
ready implemented, ready for use, and is well known
and widely used by professional texture artists.
The tool itself was implemented using Unreal En-
gine 4, by Epic Games Inc., which is a real-time game
engine. We chose this game engine because it has in-
tegration with Substance Designer, and also because
it has a user interface designer and a renderer to dis-
play the preview snake mesh. The software’s main
interface can be seen in Figure 19.
Figure 19: The software’s interface.
Our model can generate a multitude of snake skin
textures. Some examples can be seen on Figures 20,
21, 22 and 23. These figures show snakes with tex-
tures synthesized by our generator in an example en-
vironment, rendered in real-time in Unreal Engine 4.
Background scenario assets (foliage and rocks) were
provided by Epic Games, Inc. To generate these sam-
ples, we looked at reference photos and reproduced
them by manually tweaking parameters, such as pat-
tern type and size, colors, and specific pattern param-
eters. There are 59 parameters in total.
Figure 24 better shows smooth scales detail, and
Figure 25 shows keeled scales detail.
Figure 26 shows a side-by-side comparison of ref-
erence photos and generated textures. Background on
rendered images was copied from the reference pho-
tos for better comparison.
We can see that our model is capable of generating
Figure 20: Generated Micrurus altirostris textures rendered
on a snake mesh.
Figure 21: Generated Elapomorphus quinquelineatus tex-
tures rendered on a snake mesh.
Figure 22: Generated Philodryas olfersii textures rendered
on a snake mesh.
Figure 23: Generated Crotalus viridis textures rendered on
a snake mesh.
textures that are quite similar to their real-world coun-
terparts, as well as generating fine details, as seen in
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
140

Figure 24: Generated smooth scales.
Figure 25: Generated keeled scales.
Figures 24 and 25.
Although we based our study on real-world
snakes, it is entirely possible to use our model to cre-
ate unrealistic, or fantasy, snake textures. Some ex-
amples can be seen in Figures 27, 28 and 29.
In our test system, equipped with an Intel Core
i5-4670K @ 4.1 GHz, 16GB RAM and NVIDIA
GeForce GTX 1070, it takes an average of 82.73ms
to compute all 4 textures (base color, normal map,
height map and roughness map) at 1024x1024 reso-
lution. Thus the user can change parameters and see
the result in real-time, allowing to work iteratively.
Table 2 shows the average time taken to compute the
4 textures in other resolutions.
Table 2: Average computing times for all 4 textures at dif-
ferent texture resolutions.
Resolution Average time
256x256 51ms
512x512 56.7ms
1024x1024 82.73ms
2048x2048 163.06ms
4096x4096 554.82ms
5.1 User Tests
In order to identify how useful and friendly our tool
is for the average designer, we ran user tests. We se-
lected as subjects students from the 4th semester of
an undergraduate Game Design course at UniRitter,
located in the city of Porto Alegre, Brazil. 13 subjects
participated in the test, most of them between 21 and
26 years old, and most of them males (12 males, 1
female). The test involved 4 tasks:
1. Freely explore the tool;
2. Attempt to reproduce the snake Elapomorphus
quinquelineatus (see Figure 21);
3. Attempt to reproduce any coral snake of their
choice;
4. Attempt to reproduce any snake of their choice.
For each task, users had a maximum working time
of 7 minutes. We chose this time limit because we
take an average time of 3 minutes to create each of
these textures, and added 4 minutes for unexperienced
users.
Users had to look for photos of these snakes on
the internet. The time they had to browse for images
was free (it was not counted in each task’s time limit).
After taking this test, they answered a questionnaire
anonymously.
Among the questions, we asked whether they had
any prior experience in texturing. 30.8% said they had
no prior experience at all, 69.2% said they had some
experience, and no one said they had good experience
or worked with texturing frequently. We also asked if
they were able to reproduce each of the three snakes,
with 3 options: “Yes”, “Yes, partially” and “No”. No
user answered “no” for any snake.
• For the first snake (Elapomorphus quinquelinea-
tus), most users managed to reproduce it faithfully
(61.5% “Yes” against 38.5% for “Yes, partially”).
• For the second snake (the coral), again most users
managed to reproduce it faithfully (92.3% “Yes”
against 7.7% for “Yes, partially”).
• For the third snake (user’s choice), most users
only managed to reproduce it partially (30.8%
“Yes” against 69.2% for “Yes, partially”). We
believe this comes from the fact that some users
chose a snake with a complex pattern, which our
method is currently unable to reproduce.
When asked how easy it was to reproduce each
snake, most users reported the Elapomorphus quin-
quelineatus as easy, followed by the coral with mixed
results; and lastly their chosen snake as the most dif-
ficult, likely because some users chose a snake with
a complex pattern, which is not supported by our
model.
Thus, we asked if users realized they couldn’t re-
produce all patterns in the tool (the complex patterns).
30.8% answered “No” (meaning they think all pat-
terns can be reproduced), 38.5% answered “Yes, just
A Procedural Model for Snake Skin Texture Generation
141

Figure 26: Photo reference (left) and textures obtained using our texture synthesizer (right), applied on a 3D mesh. In
reading order: Crotalus Viridis (photo source: (Stuart, 2011)), Liophis miliaris (source: (de Aguiar Passos, 2013)), Elapomor-
phus quinquelineatus (source: (Martins, 2011)), Micrurus altirostris (source: (Borges-Martins, 2007a)), Philodryas olfersii
(source: (Sawaya, nd)) and Mussurana bicolor (source: (Smith, 2015)).
Figure 27: Generated textures for a fictional snake.
Figure 28: Generated textures for a fictional snake.
Figure 29: Generated textures for a fictional snake.
a few”, 30.8% answered “Yes, several”, and 0% an-
swered “Yes, many”.
Lastly, we asked the 10 questions from the Sys-
tem Usability Scale (SUS) by John Brooke (Brooke,
1986). We only adapted the first question. The origi-
nal “I think that I would like to use this system fre-
quently” was changed to “I would like to use this
tool in the future to create snake textures”. We also
changed all mentions of “system” to “tool”. The SUS
test result was 85.8 points on average, on a scale that
goes from 0 (bad usability) to 100 (good usability).
This indicates a very good usability, and that users
are likely to use the tool in the future to generate
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
142

snake skin textures. The full test results can be seen
at https://goo.gl/GDEs56.
We’ve also shown our texture generator to a her-
petologist to gather feedback. Although originally in-
tended to use in the game or animation industry, the
herpetologist said the tool is useful to them as well. It
could be used to easily create a visual representation
of a new snake species: according to her, biologists
currently use photos (which aren’t always available
or of good quality) or manually draw using a vec-
tor graphics tool such as CorelDraw. The herpetolo-
gist has also tested our tool, and the System Usability
Scale score was 80.0 (however, keep in mind we had
only one test subject).
6 CONCLUSIONS
From the results we obtained and the positive feed-
back on user tests, we believe our tool proves to be
a quite useful texture synthesizer for snake skin pat-
terns. Even though we based our study only on snakes
found in the Rio Grande do Sul region, we found it
can be used for snakes from other regions as well
without any problem (save the complex patterns we
are unable to reproduce), as well as fictional snakes.
Our tool was made available to download for
free at https://goo.gl/tmGxZB. Within the tool are
the presets (all parameters values) used for all
snakes shown in the results. The Substance De-
signer source file was also made available for free at
https://share.allegorithmic.com/libraries/3214. Feed-
back from other users is positive (4 reviews, all with
5 stars) and it was downloaded 90 times as of October
31, 2017 (33 days after it was uploaded).
It should also be mentioned that generated tex-
tures can easily be adapted for other scaled reptilians
as well, or indeed, any other animal if the normal map
and height map aren’t used (scales shape go into these
textures only). Some preliminary results can be seen
in Figure 30.
Figure 30: Color textures generated using our method for
different animals. From left to right: Cheetah (Acinonyx
jubatus), cow (Bos taurus) and five-lined skink (Plestiodon
fasciatus).
Our tool, however, is unable to reproduce textures
of snakes categorized as having the “Complex” pat-
tern type, which was not implemented as discussed
on section 4.1.5, and snakes that combine multiple
pattern types (such as both longitudinal stripes and
spots). We also do not generate head and tail textures
for two reasons. First, these would be dependent on
how the mesh’s UV coordinates were mapped. Sec-
ond, due to the much higher complexity. Scales on the
head often have a specific amount for each species,
and many different shapes. These improvements are
left for a future work.
Finally, we would like to test our tool with experi-
enced users, that is, professional texture artists.
ACKNOWLEDGEMENTS
We would like to thank Marluci M
¨
uller Rebelato, of
the Biosciences Institute at Universidade Federal do
Rio Grande do Sul, for her advice on snakes biology.
REFERENCES
Abegg, A. D. and Entiauspe Neto, O. M. (2012). Serpentes
do Rio Grande do Sul. Livraria e Editora Werlang
Ltda., Tapera, RS, Brazil.
Andrews II, M. J. (2010). Caramel burmese python.
Bauchot, R. (2006). Snakes: A Natural History. Sterling.
Borges-Martins, M. (2007a). Micrurus altirostris (cope,
1860).
Borges-Martins, M. (2007b). Phalotris lemniscatus trilinea-
tus (boulenger, 1889).
Borges-Martins, M. (2007c). Rhinocerophis alternatus
(dum
´
eril, bibron and dum
´
eril, 1854).
Brooke, J. (1986). System usability scale (sus): a quick-
and-dirty method of system evaluation user informa-
tion. Reading, UK: Digital Equipment Co Ltd.
Campbell, C. (2005). Pastel ball python.
Cocho, G., P
´
erez-Pascual, R., and Rius, J. L. (1987a). Dis-
crete systems, cell-cell interactions and color pattern
of animals. i. conflicting dynamics and pattern forma-
tion. Journal of Theoretical Biology, 125:419–435.
Cocho, G., P
´
erez-Pascual, R., Rius, J. L., and Soto, F.
(1987b). Discrete systems, cell-cell interactions and
color pattern of animals ii. clonal theory and cellular
automata. Journal of Theoretical Biology, 125:437–
447.
de Aguiar Passos, M. (2013). Erythrolamprus miliaris (lin-
naeus, 1758).
de Albuquerque, S. (n.d.). Falsa-coral (rhinobothryum
lentiginosum.
Dhillon, D. S., Teyssier, J., Single, M., Gaponenko, I.,
Milinkovitch, M. C., and Zwicker, M. (2014). Inter-
active diffraction from biological nanostructures. In
Computer Graphics Forum, volume 33, pages 177–
188. Wiley Online Library.
A Procedural Model for Snake Skin Texture Generation
143

Di-Bernardo, M. (n.d.). Ptychophis flavovirgatus gomes,
1915.
Efros, A. A. and Leung, T. K. (1999). Texture synthesis by
non-parametric sampling. In Computer Vision, 1999.
The Proceedings of the Seventh IEEE International
Conference on, volume 2, pages 1033–1038. IEEE.
Gilet, G., Sauvage, B., Vanhoey, K., Dischler, J.-M.,
and Ghazanfarpour, D. (2014). Local random-phase
noise for procedural texturing. ACM Transactions on
Graphics (TOG), 33(6):195.
Hendrikx, M., Meijer, S., Van Der Velden, J., and Iosup, A.
(2013). Procedural content generation for games: A
survey. ACM Transactions on Multimedia Computing,
Communications, and Applications (TOMM), 9(1):1.
Mandelbrot, B. B. and Pignoni, R. (1983). The fractal ge-
ometry of nature, volume 173. WH freeman New
York.
Martins, P. H. (2011). Elapomorphus quinquelineatus. serra
do cip
´
o, minas gerais, brazil.
May, P. S. (2010). Figure 1 - (fprep393ph) adult, encar-
naci
´
on, departamento itap
´
ua (paul smith may 2010).
Miller, G. S. P. (1988). The motion dynamics of snakes and
worms. SIGGRAPH Comput. Graph., 22(4):169–173.
Murray, J. and Myerscough, M. (1991). Pigmentation pat-
tern formation on snakes. Journal of Theoretical Bi-
ology, 149:339–360.
Nogueira, C. (n.d.a). R
´
epteis squamata do cerrado - apos-
tolepis assimilis.
Nogueira, C. (n.d.b). R
´
epteis squamata do cerrado -
philodryas aestiva.
Panagiotakis, C. and Tziritas, G. (2006). Snake terrestrial
locomotion synthesis in 3d virtual environments. The
Visual Computer, 22(8):562–576.
Perlin, K. (2002). Improving noise. In Proceedings of the
29th Annual Conference on Computer Graphics and
Interactive Techniques, SIGGRAPH ’02, pages 681–
682, New York, NY, USA. ACM.
Pharr, M., Jakob, W., and Humphreys, G. (2016). Phys-
ically based rendering: From theory to implementa-
tion. Morgan Kaufmann.
Rada, T. (2008). Lampropeltis triangulum triangulum (east-
ern milksnake).
Sawaya, R. (n.d.). Philodryas olfersii (lichtenstein, 1823).
Smith, P. (2015). Bicoloured mussurana mussurana bicolor.
Stuart, J. N. (2011). Prairie rattlesnake (crotalus viridis),
adult.
Timm, C. D. (2014). Falsa-coral (oxyrhopus rhombifer).
Turk, G. (1991). Generating textures on arbitrary surfaces
using reaction-diffusion. In ACM SIGGRAPH Com-
puter Graphics, volume 25, pages 289–298. ACM.
Uetz, P., Freed, P., and (eds.), J. H. (2016). The reptile
database.
Witkin, A. and Kass, M. (1991). Reaction-diffusion tex-
tures. ACM Siggraph Computer Graphics, 25(4):299–
308.
GRAPP 2018 - International Conference on Computer Graphics Theory and Applications
144
