
Biomarkers of Neurodegenerative Progression from Spontaneous
Speech Recorded in Mobile Devices: An Approach based on
Articulation Speed Estimation
A Study of Patients Suffering from Amyotrophic Lateral Sclerosis
Ana Londral
1,2
, Pedro Gómez-Vilda
3
and Andrés Gómez-Rodellar
3
1Translational Clinical Physiology Lab, Instituto de Medicina Molecular, University of Lisbon, Lisbon, Portugal
2Escola Superior de Tecnologia de Setúbal, Instituto Politécnico de Setúbal, Portugal
3Neuromorphic Speech Processing Lab, Center for Biomedical Technology, Universidad Politécnica de Madrid,
Campus de Montegancedo, 28223 Pozuelo de Alarcón, Madrid, Spain
Keywords: Mobile Devices, Clinical Remote Monitoring, Speech Signal Processing, Amyotrophic Lateral Sclerosis.
Abstract: A majority of patients with Amyotrophic Lateral Sclerosis (ALS) experiment a rapid evolution of symptoms
related to a progressive decline in movement function that affects different systems. Clinical assessment is
based on measures of progression for identifying the need and the pace of medical decisions, and to measure
also the effects of novel therapies. But assessment is limited to the periodicity of clinical appointments that
are increasingly difficult for patients due to progressive mobility impairments. In this paper, we present a
novel method to assess neurodegeneration process through speech analysis. An articulation kinematic model
is proposed to identify markers of neuromotor functional progression in speech. We analysed speech
samples that were collected with a mobile device, in 3-month intervals, from a group of six subjects with
ALS. Results suggest that the method proposed is sensitive to the symptoms of the disease, as rated by
observational clinical scales, and it may contribute to assist clinicians and researchers with better and
continuous measures of disease progression.
1 INTRODUCTION
Motor Neuron Disease (MND) is a progressive
neurological condition that affects the motor
neurons, present both in the central and spinal neural
systems. The most common MND is Amyotrophic
Lateral Sclerosis (ALS), which is a disease with
rapid progression and unknown cure, affecting both
upper and lower motor neurons. The deterioration of
the neuromotor system involved in respiration,
phonation, swallowing, and lingual and oro-facial
muscle function degenerates in a rapidly progressing
speech dysarthria (Tomik and Guiloff, 2010).
The clinical support of this disease is based on
the management of the symptoms, as they manifest
(Andersen et al., 2012). Clinical scales for
monitoring progression are either invasive, based on
EMG (de Carvalho et al. 2005) or on observational
evaluation and Likert-type functional scales
(Cedarbaum & Stambler, 1997). Assessments are
performed in 3-6 month spaced clinical
appointments; frequently, when the progression of
symptoms is severe, the periodicity of clinical
appointments decreases due to difficulties in
transportation of the patient from their residence to
clinical facilities.
As speech intelligibility decreases, patients often
use mobile devices (often a tablet or a smartphone)
with text-to-speech to support communication
(Londral et al. 2015). The aim of this work is to
explore the potential of those mobile devices to
continuously and remotely monitor ALS progression
through speech collection, by evaluating quantitative
speech parameters, using a methodology as depicted
in Figure 1.
In this study, we explore speech as a biomarker
of disease progression. Voice is a signal that is
easily collected with non-invasive and low-cost
techniques. In fact, modern mobile devices allow to
collect speech with integrated apps that run from
patient’s home, for remote monitoring using e-
Londral, A., Vilda, P. and Gómez-Rodellar, A.
Biomarkers of Neurodegenerative Progression from Spontaneous Speech Recorded in Mobile Devices: An Approach based on Articulation Speed Estimation - A Study of Patients Suffering
from Amyotrophic Lateral Sclerosis.
DOI: 10.5220/0006731302690275
In Proceedings of the 11th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2018) - Volume 4: BIOSIGNALS, pages 269-275
ISBN: 978-989-758-279-0
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
269
Health platforms, as are example the works from
Abad et al. (2013) and Vacher et al. (2006).
Figure 1: A scheme of the system that we aim to
implement, based on speech collection from home, using
regular mobile devices, and remote speech processing.
This paper firstly describes the Articulation
Kinematics Model (AKM) that is proposed, then in
section 3, the methodology of this study is
presented, and the dataset of ALS patients is
described. Results are presented in section 4; the
final section includes the conclusion with the
discussion of results and future work.
2 SYMPTOMS OF NEURO-
-MOTOR DEGENERATION IN
SPEECH
Progressive dysarthria is a symptom in ALS.
Dysarthria is a disorder that results from
neurological impairment of the motor component of
the motor-speech system. The neurological origin of
dysarthria may vary in ALS but ultimately it affects
the intelligibility of patient’s speech causing great
difficulties in communication.
It has been studied that, as ALS progresses,
speech movements become smaller in extent and
slower in speed (Green et al., 2013). Classical
articulation measures define the vowel space area
(VSA) and the Formant Centralization Ratio (FCR)
as valid parameters to estimate the vowel span range
and positioning produced by a given speaker (Sapir
et al. 2011). Absolute span of formants F1 and F2 of
a given utterance has been additionally proposed as
a sensible feature to dysarthria in a longitudinal
study with five persons with ALS (Gómez-Vilda et
al., 2015).
While those parameters are known to be
sensitive to the assessment of dysarthria, its
semantic meaning is unclear. Besides, these features
only express the average values of the most frequent
formant positions, mainly associated to vowels. As
dysarthrias with neuromotor origin express changes
in dynamic activity of the articulation organs
(imprinted in rapid formant changes), we used a
method based on the measure of the kinematic
behavior of formant dynamics, described in the
following section.
3 ARTICULATION KINEMATIC
MODEL (AKM)
Estimated parameters in common speech are
associated to specific neuromuscular complexes
involved in articulation, more specifically the
masseter, the stylo-glosus and the genio-hyo-glosus
muscles – as described in (Gómez-vilda et al., 2013).
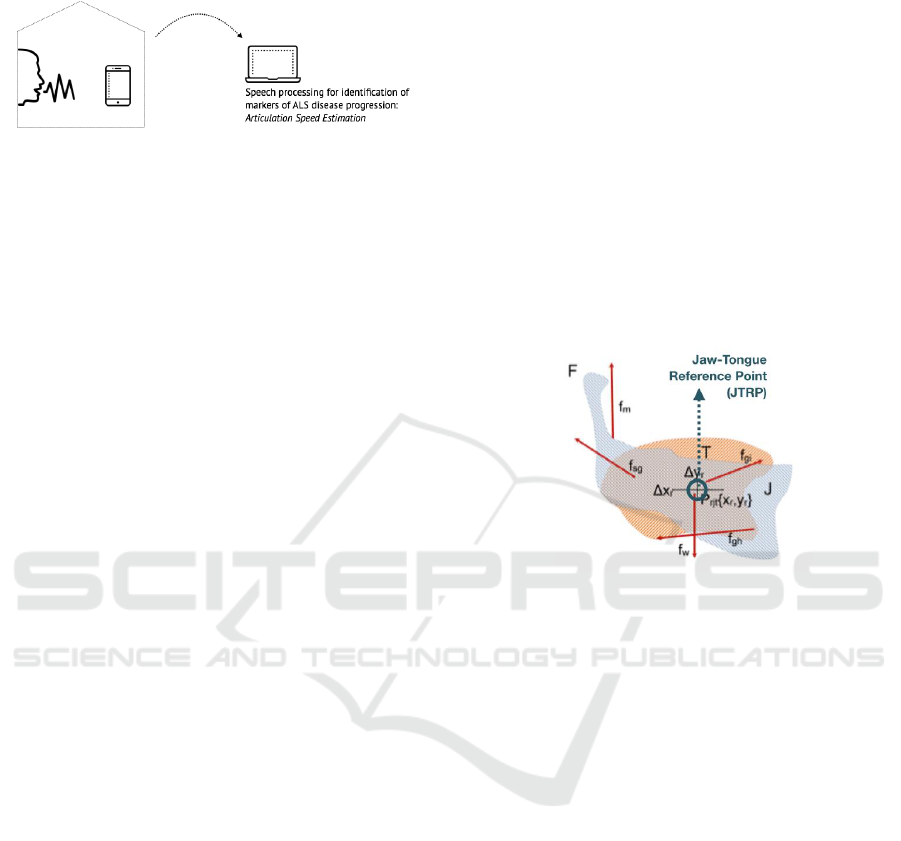
The model presented in this paper allows the
estimation of the articulation positions based on the
indirect inference of vocal tract configuration using
the simplified model depicted in Fig. 2.
Figure 2: Articulation kinematic model used in this study.
A dynamic system of muscle force vectors representing
the motor articulation system can be simplified in one
reference-point (JTRP), which is situated in the action
centre of the oral cavity and moves in the sagittal plane,
that expresses the dynamics of articulation.
The AKM includes the jaw, the tongue and the
facial tissues attached to them in a dynamic system
that can be approximated to a third-order lever fixed
at the skull. Considering only movements in the
sagittal plane, we define the Jaw-Tongue Reference
Point (JTRP) Prjt {x
r
,y
r
}, where different forces act
during speech, related to the neuromuscular system
involved in the masseter, lips and tongue
movements, as described in Gomez et al. (2017). As
a result of multiple muscle forces, the reference
point JTRP will move in the sagittal plane (Δx
r
,
Δy
r
); these movements are related to formant
changes as represented in Equation 1, where a
ij
are
nonlinear time-variant and multi-valued functions
associating Prjt to formants, and t is the time.
F
1
(t)
F
2
(t)
é
ë
ê
ê
ù
û
ú
ú
=
a
11
a
12
a
21
a
22
é
ë
ê
ê
ù
û
ú
ú
x
r
(t)
y
r
(t)
é
ë
ê
ê
ù
û
ú
ú
; A =
a
11
a
12
a
21
a
22
é
ë
ê
ê
ê
ê
ê
ù
û
ú
ú
ú
ú
ú
(1)
3.1 Absolute Kinematic Velocity
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
270
Under the assumption that formant F
1
is related to
vertical kinematics and formant F
2
is related to
horizontal kinematics, the articulation kinematic
velocity (AKV) may be inferred by Equation 2.
|vr (t)|= w21
¶F1(t)
¶t
æ
è
ç
ö
ø
÷
2
+ w12
¶F2(t)
¶t
æ
è
ç
ö
ø
÷
2
(2)
The expected AKV profile would be that of a
decaying curve, with the properties of a χ
2
distribution, illustrating the dynamics of articulation
behavior.
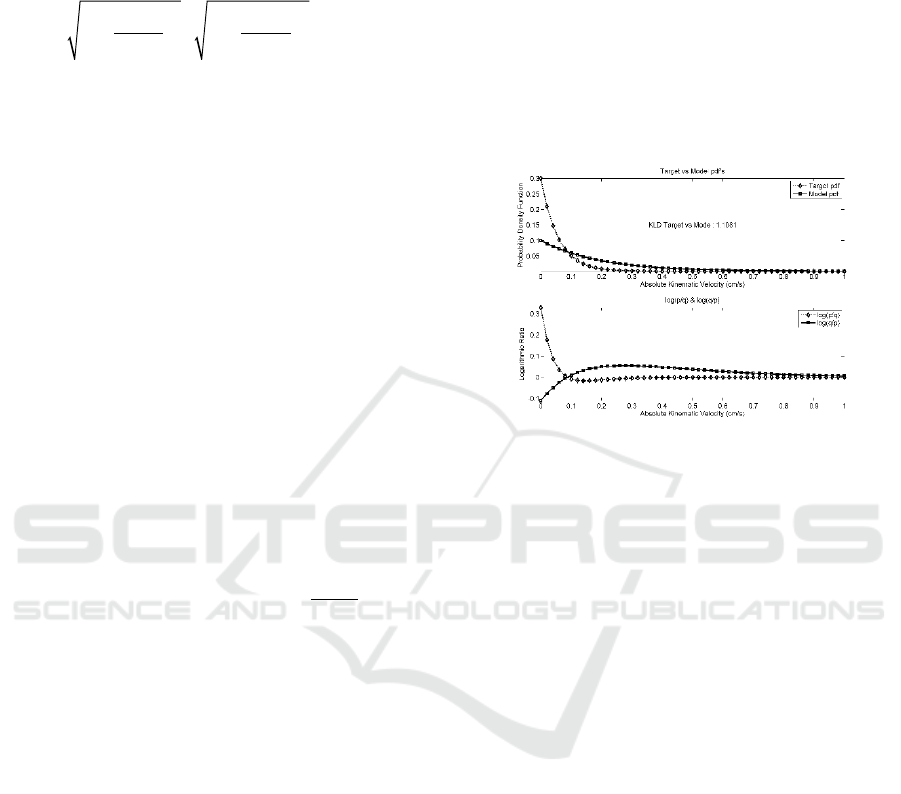
3.2 Kullback-Leibler’s Divergence
(KLD)
As the disease progresses, the dynamic behavior of
patient’s speech is expected to be hampered by the
difficulty in moving the articulation organs with
enough speed. Dysarthria will limit the movements
and the decaying curve of AKV probability density
function (pdf) will be more marked, with higher
probabilities for lower velocities and lower
probabilities for higher velocities. As described by
P.Gomez et al. (2017), we will use the Kullback-
Leibler’s Divergence (KLD) to model the dynamic
behavior differences between healthy controls and
ALS patients, according to Equation 3:
D
KLij
p
Ti
v
r
( )
, p
Mj
v
r
( )
{ }
= p
Mi
z
( )
abs log
p
Ti
z
( )
p
Mj
z
( )
é
ë
ê
ê
ù
û
ú
ú
ì
í
ï
î
ï
ü
ý
ï
þ
ï
d
z
z
=0
¥
ò
(3)
where v
r
is the absolute value of the articulation
kinematic velocity, p
Ti
is the probability density
distribution of the target utterance T
i
, and p
Mj
is the
probability density distribution of the model
utterance T
j
. Figure 3 depicts the comparison
between the probability density functions (pdf) of a
‘unhealthy’ and a ‘healthy’ speech sample.
Figure 3. The model based on the AKV pdf to model the
dynamic behavior differences between healthy controls
(diamonds) and ALS patients (squares).
4 METHODOLOGY
In this work we applied the AKM to a dataset of
sound samples from eight subjects with ALS that
recorded speech in 2 to 5 assessments performed in
periods of 2 to 5 month-intervals during 2 to 19
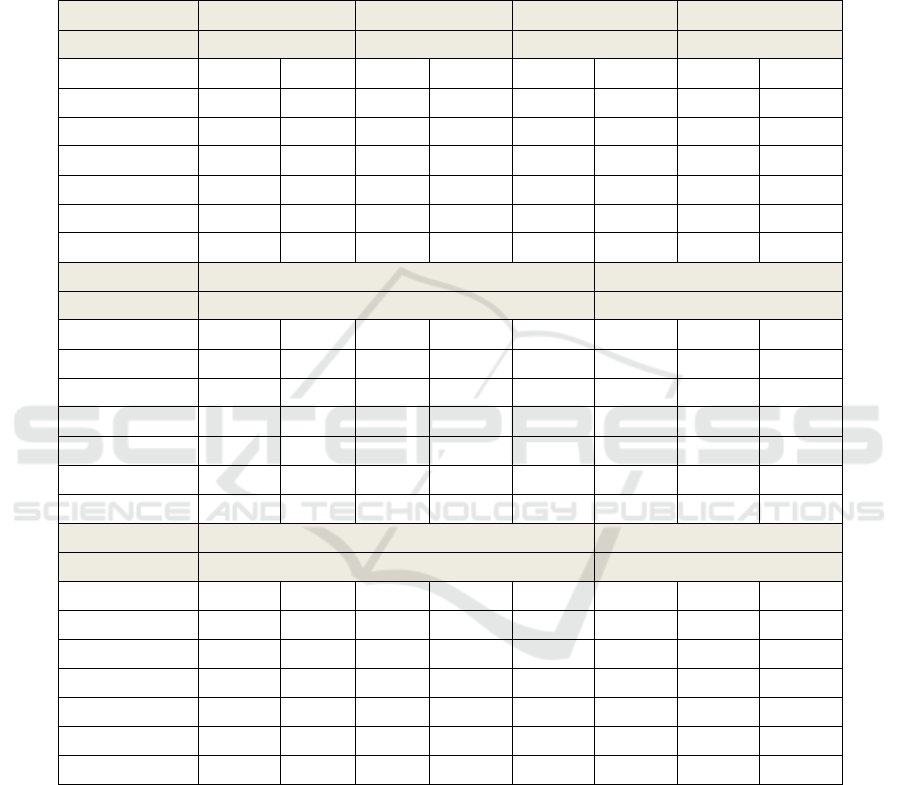
months (as described in Table A1).
4.1 Dataset
We used data from 8 women with ALS, saved in a
dataset of voice samples collected for a longitudinal
study that was approved by the Ethical Commission
in the Hospital of Santa Maria, Lisbon, Portugal. All
participants signed an informed consent to be
included in the study. Speech was recorded using the
microphone of a laptop or a smartphone (2-channel
wav files with sample rate of 44100Hz and 16 bits).
All files have the same sentence recorded by the
patients in consecutive assessments.
Figure 3: The model based on the AKV pdf to model the
dynamic behavior differences between healthy controls
(diamonds) and ALS patients (squares).
The mean age of the 8 subjects is 66.7 (±13.0)
years old, the youngest with 38 and the oldest with
80 years old.
Patients were asked to repeat once, a popular
sentence from the Portuguese writer Fernando
Pessoa, which happens to be very well known by
most people in Portugal: /tudo vale a pena quando a
alma não é pequena/. This sentence was recorded
from the patient in 2 to 5 assessments that were
taken in successive clinical appointments, as
described in Table A1. The same recording was
taken from two female healthy controls of 36 and
63-years old (CF36 and CF63).
4.2 Procedure
The basic methodological protocol consists in the
following steps:
• Recordings are undersampled to 8 kHz.
• The vocal tract transfer function of the speech
segment is evaluated by a 8-pole adaptive
inverse Linear Prediction (LP) filter (Deller et
al., 2000) with a low-memory adaptive step to
grasp fine time variations.
• The first two formants are estimated by
evaluating the maxima and slenderness of the
Biomarkers of Neurodegenerative Progression from Spontaneous Speech Recorded in Mobile Devices: An Approach based on Articulation
Speed Estimation - A Study of Patients Suffering from Amyotrophic Lateral Sclerosis
271
LP spectrogram. The formant estimation
resolution used is 2 Hz every 2 ms.
• The derivatives of the first two formants are
used to estimate the absolute velocity of the
JTRP following (2).
• The values of the AKV in the recording interval
are used to build a histogram.
• The histograms are used to estimate probability
density functions by Kolmogorov-Smirnov
approximations (Webb, 2003).
• Kullback-Leibler’s Divergence between each
patient’s histogram-derived distribution vs that
of the control subject is estimated as by (3).
5 RESULTS
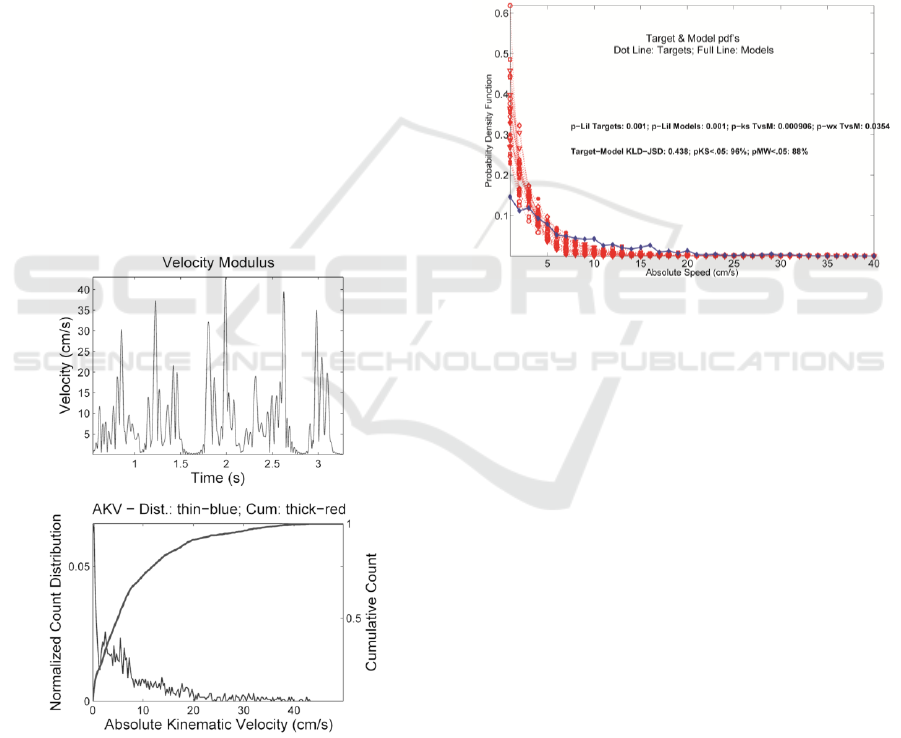
The AKV was calculated for all the sound files of
each subject. Figure 4 represents the velocity that
was dynamically calculated for a 3.5 seconds sample
from the younger control and the respective
histogram of the AKV. From this figure, it is
possible to observe that articulation velocity is zero
in pauses between words and has a maximum that is
approx. 45 cm/s.
Figure 4: (up) the velocity of JTRP calculated dynamically
for a sound sample with 3.5 seconds. (down) the
histogram of the AKV with cumulative count (darker
line).
The comparison between the average
distributions of the ALS samples and the model
probability from older control subject are depicted in
Figure 5, with the example of subject HA. In this
example, the Liljefors tests discard gaussianity
(p<0.05); Kolmogorov-Smirnov (KS) and Wilcoxon
(WX) reject the null hypothesis (H0) of similarity
(p
wx
<0.05) between Targets and Models. The
average Kullback-Leibler distance is 0.483, the 96%
of the cases reject H0 with respect to the model
(CF63) under KS, and 88% reject H0 under Mann-
Whitney test.
When comparing the same subject with the
younger control, the similarity in the average is not
rejected according to WX test, and only 76% and
52% of the files reject similarity to the model under
KS and MW, respectively.
Figure 5: Comparison between the average distributions of
the subject HA samples (dotted lines) and the model
probability from the older control subject (full line). At the
middle of the plot, the two text lines indicate results from
tests of gaussianity of the distributions following Liljefors
test (first text line, Targets and Models), and the
Kolmogorov-Smirnov and Wilcoxon tests of Targets vs
Models for the null hypothesis of similarity (H0); the
Kullback-Leibler distance of the average Targets and
Models, and the percentage of Targets rejecting H0
between Targets and Models according to the
Kolmogorov-Smirnov and Mann-Whitney tests (second
text line).
The KLD and the LLR distance were calculated
between the two control subjects and all the subjects
with ALS are described in Table 1 (Appendix).
From Figure 6, it is clear that we can observe the
disease progression within assessments, for all the
subjects. It is also clear from Figure 5 and Table 1,
that in some assessments, there seems to be a
regression in the symptoms of neurodegeneration, as
the KLD and LLR values decrease. When observing
this behaviour and comparing with respective
ALSFRS-B values, we can observe that these
decreases are related to observed stabilization of
symptoms by the doctor (the value of ALSFRS-B
remains the same of the previous assessment).
Subject AR, the youngest patient with ALS (38
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
272
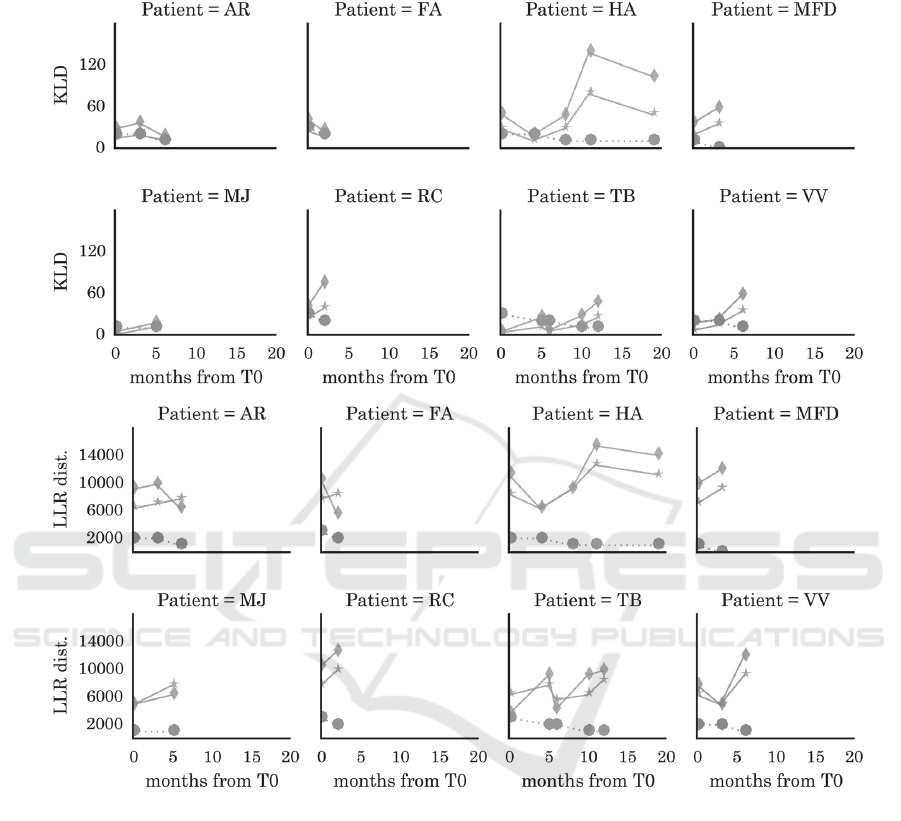
years old), is the exception (values of KLD decrease,
despite the observed symptoms were rated with
lower values of speech functionality).
The LLR distance to the younger control subject
had the best results for demonstrating unceasing
Figure 6: These plots represent the KLD and LLR distance of the 8 subjects with ALS from the younger control subject
(marker *) and older control (marker ). Bullet points indicate the ALSFRS-B evaluation. The x-axis represents the months
from the first assessment (T0).
neurodegeneration in ALS, since LLR increases
along time for all the subjects with ALS (exceptions
are coincident to stability of observed symptoms).
6 DISCUSSION
In this paper, we propose studying the AKV as a
marker of neurodegenerative progression in ALS.
We are interested in evaluating current speech that
can be recorded via a current mobile device to be
used in remote monitoring, outside the clinical
facilities. In particular for ALS, for which standard
clinical assessments are spaced of 3 to 6 months, this
would facilitate:
1. A continuous evaluation of progression that
could be sent to the clinician.
2. Novel markers to study the impact of new
therapies.
3. The remote assessment, in particular when
patient has mobility impairments and the
transportation for the clinical facilities becomes
difficult.
Biomarkers of Neurodegenerative Progression from Spontaneous Speech Recorded in Mobile Devices: An Approach based on Articulation
Speed Estimation - A Study of Patients Suffering from Amyotrophic Lateral Sclerosis
273

We used a database containing sound samples
that were recorded from a mobile phone or a laptop
computer. These samples contain the same common
20-words sentence in Portuguese. For the severity of
ALS disease, a short sentence is an important
requisite of our methodology, to make it valid for
these patients’ context. In fact, as speech becomes
difficult to produce, the more complex is the sample
collection, and the more dropouts will take place.
The results described in previous sections
demonstrate that the KLD from a healthy control is
sensitive to neurodegeneration progression in ALS.
For all ALS subjects, except one, the AKV model
expressed progression of neurodegenerative
symptoms in speech, by increase of KLD, for both
the younger and the older models. The exception
was observed for the youngest subject with ALS (38
years old). In fact, we can hypothesize that the
samples from this patient will fit better to the
younger control model, due to the age proximities.
The LLR distance for the younger model expresses a
continuous increase and correlation to the ALSFRS-
B values attributed to this subject.
In general, the results described in this work
confirm the hypothesis that we can model the speech
dysarthria in ALS as a “freezing” of the articulation
process: the probability of AKV close to 0 increases
as diseases progresses.
Our objective and quantitative measures are
according to the qualitatively assessed clinical rating
for bulbar involvement that is based on clinical
experienced observation. The apparent regression in
neurodegeneration from our results can be
confirmed by the experienced clinical observation of
stabilization of symptoms within assessments. But,
our quantitative measure may have implicit
information that is not observable and needs further
insight on its meaning. By hypothesis, our measures
may be sensible to different therapies that cause
variations observed within assessments. A new
dataset of samples that are collected in shorter time
intervals, from home mobile devices, is needed to
obtain a continuous observation of articulation
measures. A continuous observation will support a
novel insight on progression behaviour.
Our study has some limitations. One is the
heterogeneity of our sample, since some subjects
have 2 and others have 5 assessments. For this
reason, it is not possible to have a solid
demonstration of the progression along time.
Another limitation is that we are using samples
containing the same phonetic material. In all
samples, subjects use the same sentence.
For future work, a larger database containing
spontaneous speech from subjects with ALS will be
used to further test our model and study progression
from symptoms measured in speech signal.
ACKNOWLEDGEMENTS
This work was supported by Calouste Gulbenkian
Foundation and the Portuguese Association of ALS
(APELA), as well as by grant TEC2016-77791-C4-
4-R (Plan Nacional de I+D+i, Ministry of Economic
Affairs and Competitiveness of Spain).
REFERENCES
Abad, A. et al., 2013. Automatic word naming recognition
for an on-line aphasia treatment system. Computer
Speech and Language, 27(6), pp.1235–1248.
Andersen, P.M. et al., 2012. EFNS guidelines on the
Clinical Management of Amyotrophic Lateral
Sclerosis (MALS) - revised report of an EFNS task
force. European Journal of Neurology, 19(3), pp.360–
375.
de Carvalho, M., Costa, J. & Swash, M., 2005. Clinical
trials in ALS: a review of the role of clinical and
neurophysiological measurements. Amyotrophic
Lateral Sclerosis & Other Motor Neuron Disorders,
6(4), pp.202–212. Available at:
http://search.ebscohost.com/login.aspx?direct=true&d
b=a2h&AN=19020138&loginpage=Login.asp&site=e
host-live.
Cedarbaum, J.M. & Stambler, N., 1997. Performance of
the amyotrophic lateral sclerosis functional rating
scale (ALSFRS) in multicenter clinical trials. In
Journal of the Neurological Sciences.
Deller, J.R., Proakis, J.G. & Hansen, J.H.L., 2000.
Discrete-Time Processing of Speech Signals,
Available at:
http://www.library.wisc.edu/selectedtocs/bd429.pdf
Gómez-vilda, P. et al., 2013. Characterization of Speech
from Amyotrophic Lateral Sclerosis by Neuromorphic
Processing. In IWINAC 2013. pp. 212–224.
Gómez-Vilda, P. et al., 2015. Monitoring amyotrophic
lateral sclerosis by biomechanical modeling of speech
production. Neurocomputing, 151(1), pp.130–138.
Londral, A. et al., 2015. Quality of life in ALS patients
and caregivers: impact of assistive communication
from early stages. Muscle and Nerve.
P.Gomez, P. et al., 2017. Articulation acoustic kinematics
in ALS speech. In 2017 International Conference and
Workshop on Bioinspired Intelligence (IWOBI). IEEE,
pp. 1–6. Available at:
http://ieeexplore.ieee.org/document/7985522/.
Sapir, S. et al., 2011. Acoustic metrics of vowel
articulation in Parkinson’s Disease: Vowel Space Area
(VSA) vs. vowel articulation index (VAI). In C.
Manfredi, ed. Proceedings of the MAVEBA 2011.
Florence: Florence University Press, pp. 173–175.
Tomik, B. & Guiloff, R.J., 2010. Dysarthria in
amyotrophic lateral sclerosis: A review. Amyotrophic
Lateral Sclerosis, 11(1–2), pp.4–15.
BIOSIGNALS 2018 - 11th International Conference on Bio-inspired Systems and Signal Processing
274
Vacher, M. et al., 2006. Speech and sound use in a remote
monitoring system for health care,
Webb, A.R., 2003. Statistical pattern recognition,
APPENDIX
Table A1: Description of the results of speech features extracted from all the assessments of the 8 subjects with ALS,
considered in this study.
Patient
FA
MFD
RC
MJ
Age
73
68
64
56
#assessment
0
1
0
1
0
1
0
1
ALSFRS-B
3
2
1
0
3
2
1
1
Months from T0
0
2
0
3
0
2
0
5
Divergence_CF63
40.88
24.76
36.51
57.44
40.65
75.43
4.45
17.30
LLRDistance_CF63
10524.84
5641.64
9837.10
11949.03
10518.08
12625.50
4945.46
6325.12
Divergence_CF36
34.45
33.86
18.89
18.85
13.98
13.90
16.62
14.13
LLRDistance_CF36
9138.28
9118.03
7020.08
7026.55
4916.18
6324.33
8415.68
6337.03
Patient
HA
AR
Age
68
38
#assessment
0
1
2
3
4
0
1
2
ALSFRS-B
2
2
1
1
1
2
2
1
Months from T0
0
4
8
11
19
0
3
6
Divergence_CF63
49.77
18.74
46.92
137.96
103.39
27.50
36.44
17.78
LLRDistance_CF63
11224.19
6334.85
9137.77
15437.21
14033.18
9147.58
9830.65
6340.91
Divergence_CF36
28.44
28.21
24.65
24.57
24.50
78.30
48.36
38.49
LLRDistance_CF36
9120.44
8413.34
7714.18
8428.58
7708.48
12626.64
11222.63
9814.93
Patient
TB
VV
Age
77
80
#assessment
0
1
2
3
4
0
1
2
ALSFRS-B
3
2
2
1
1
2
2
1
Months from T0
0
5
6
10
12
0
3
6
Divergence_CF63
4.21
24.70
6.52
27.18
46.60
17.22
22.02
56.69
LLRDistance_CF63
3526.56
9126.58
4239.57
9134.90
9841.94
7720.34
4933.06
11928.92
Divergence_CF36
11.85
11.43
11.43
10.72
6.32
5.46
3.27
0.37
LLRDistance_CF36
7719.38
7712.00
7709.37
6319.77
6308.00
5621.47
6312.70
4925.75
Biomarkers of Neurodegenerative Progression from Spontaneous Speech Recorded in Mobile Devices: An Approach based on Articulation
Speed Estimation - A Study of Patients Suffering from Amyotrophic Lateral Sclerosis
275
