
Adopting Semantic Information of Grayscale Radiographs for Image
Classification and Retrieval
Obioma Pelka
1,2
, Felix Nensa
3
and Christoph M. Friedrich
1
1
Department of Computer Science, University of Applied Sciences and Arts Dortmund,
Emil-Figge-Strasse 42, 44227 Dortmund, Germany
2
Faculty of Medicine, University of Duisburg-Essen, Germany
3
Department of Diagnostic and Interventional Radiology and Neuroradiology,
University Hospital Essen Hufelandstrasse 55, 45147 Essen, Germany
Keywords:
Biomedical Imaging, Deep Learning, Keyword Generation, Machine Learning, Multi-modal Representation,
Transfer Learning, Radiographs.
Abstract:
As the number of digital medical images taken daily rapidly increases, manual annotation is impractical,
time-consuming and prone to errors. Hence, there is need to create systems that automatically classify and
annotate medical images. The aim of this presented work is to utilize Transfer Learning to generate image
keywords, which are substituted as text representation for medical image classification and retrieval tasks.
Text preprocessing methods such as detection and removal of compound figure delimiters, stop-words, special
characters and word stemming are applied before training the keyword generation model. All images are
visually represented using Convolutional Neural Networks (CNN) and the Long Short-Term Memory (LSTM)
based Recurrent Neural Network (RNN) Show-and-Tell model is adopted for keyword generation. To improve
model performance, a second training phase is initiated, where parameters are fine-tuned using the pre-trained
deep learning network Inception-ResNet-V2. For the image classification tasks, Random Forest models trained
with Bag-of-Keypoints visual representations were adopted. Classification prediction accuracy was higher for
all classification schemes and on two distinct radiology image datasets using the proposed approach.
1 INTRODUCTION
Due to advances in software, hardware, and digital
imaging in the medical domain, the number of images
taken per patient scan has rapidly increased (Rah-
man et al., 2007; Tagare et al., 1997). To decrease
the burden on radiologists and maintain the maxi-
mum interpretation of these radiology images, there
is need to create automatic computer-aided interpre-
tation, which can be further applied for image anno-
tation and semantic information extraction.
An important criteria for creating an effective
classification system, is the selection and combina-
tion of features for an adequate representation of the
images. As shown in (Pelka and Friedrich, 2015;
Codella et al., 2014; Valavanis et al., 2016; Kalpathy-
Cramer et al., 2015; Pelka and Friedrich, 2016),
multi-modal representation achieves higher classifica-
tion rates in biomedical annotation tasks. This is the
combination of visual and text representations which
sufficiently represents these biomedical images.
However, for real clinical cases and some image
classification tasks such as ImageCLEF2009 Medical
Annotation Task (Tommasi et al., 2009) and Image-
CLEF 2015 Medical Clustering Task (Amin and Mo-
hammed, 2015), corresponding text representations
are not available. In this paper, Transfer Learning
(Pan and Yang, 2010) is utilized to generate keywords
(Pelka and Friedrich, 2017), which are combined with
visual features to obtain multi-modal image represen-
tations. These text features are further adopted for the
medical image classification tasks mentioned above
and semantic tagging.
As deep learning techniques (LeCun et al., 2015)
have improved prediction accuracies in object detec-
tion (Huang et al., 2017), speech recognition (Hinton
et al., 2012) and in domain application such as med-
ical imaging (Abrao et al., 2007; Xu et al., 2014), a
deep learning architecture is used to create the key-
word generation model. Deep Convolutional Neu-
ral Networks (dCNN) (Szegedy et al., 2017) are ap-
plied to encode the medical images to a feature rep-
Pelka, O., Nensa, F. and Friedrich, C.
Adopting Semantic Information of Grayscale Radiographs for Image Classification and Retrieval.
DOI: 10.5220/0006732301790187
In Proceedings of the 11th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2018) - Volume 2: BIOIMAGING, pages 179-187
ISBN: 978-989-758-278-3
Copyright © 2018 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
179
resentation which is decoded using a Long Short-
Term Memory (LSTM) (Hochreiter and Schmidhu-
ber, 1997) based Recurrent Neural Network (RNN)
(Bengio et al., 1994) to generate appropriate key-
words for a given image.
For the image classification tasks, Random For-
est (Breiman, 2001) models trained with visual and
text representations were adopted. Bag-of-Keypoints
(Csurka et al., 2004; Lazebnik et al., 2006) computed
with dense SIFT descriptors (Dalal and Triggs, 2005)
were combined with text representations obtained by
vector quantization on a Bag-of-Words (Salton and
McGill, 1983) codebook. The codebook was created
with words from the keyword generation model.
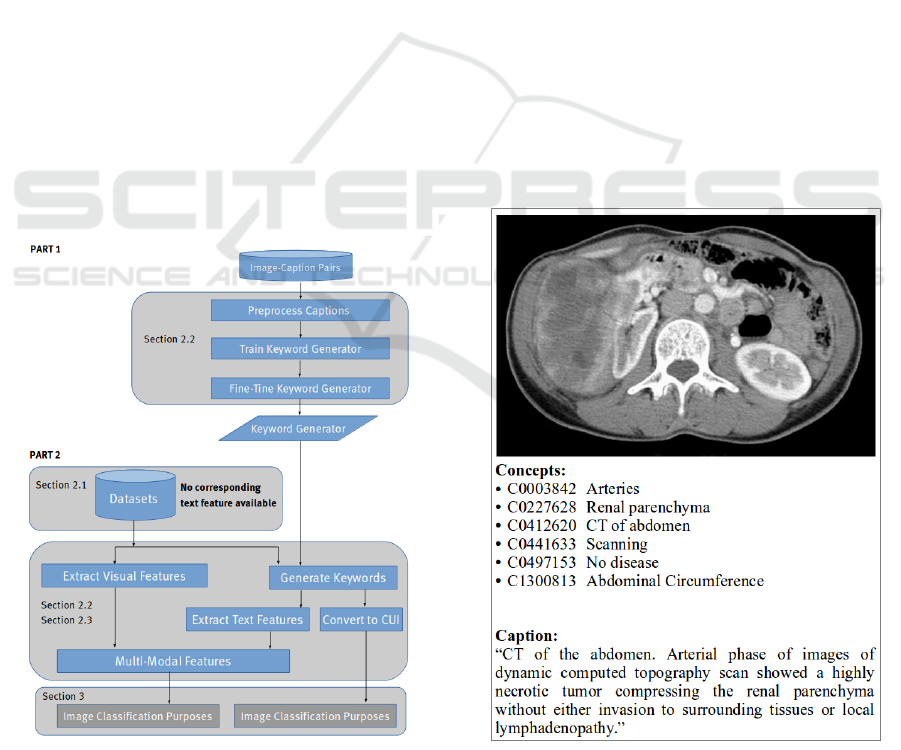
Figure 1 shows the complete workflow for the ap-
proach presented in this paper. PART 1 displays nec-
essary steps for creating a keyword generator. This
is a distinct and stand-alone process which does not
need PART 2 for application. The keyword genera-
tor can be further adopted for several purposes, such
as image classification and retrieval. In PART 2,
the keyword generator is used to create keywords for
medical datasets that lack text representations. This
second part is dependable on the first part. Two
datasets containing grayscale radiographs were uti-
lized in PART 2, however there are no restrictions,
as the keyword generator was created using biomed-
ical literature figures. The rest of this paper is struc-
Figure 1: Overview of the proposed approach workflow.
tured as follows: Section 2.1 lists the adopted dataset
for keyword generation and the two datasets used
for evaluating the proposed approach. In subsec-
tions 2.2 and 2.3, applied deep learning networks, vi-
sual representation and machine learning methods for
keyword generation and image classification are de-
scribed. The achieved results are stated in section 3.
Finally, results are discussed in section 4 and conclu-
sions are drawn in section 5.
2 MATERIAL AND METHOD
2.1 Datasets
Three datasets are applied for this proposed work:
• ImageCLEFcaption Prediction 2017: For key-
word generation model
• ImageCLEF 2009 Medical Image Annotation
Task: For image classification and evaluation
• ImageCLEF 2015 Medical Clustering Task: For
image classification and evaluation
ImageCLEFcaption Prediction 2017. This dataset
was distributed at the ImageCLEFcaption 2017 Task
(Eickhoff et al., 2017). ImageCLEFcaption 2017
Figure 2: Example of a medical image with corresponding
caption and Concept Unique Identifiers (CUI). The com-
puter tomography scan was randomly chosen from the vali-
dation set of the ImageCLEFcaption 2017 Task.
KALSIMIS 2018 - Special Session on Knowledge Acquisition and Learning in Semantic Interpretation of Medical Image Structures
180

consisted of two subtasks: Concept Detection and
Caption Prediction. All distributed figures originate
from open access biomedical journal articles pub-
lished in PubMed Central (PMC) (PubMed Central,
2017). The objective of the concept detection task
was to retrieve clinical concepts present in the med-
ical images whereas for the caption prediction task,
meaningful captions to the images had to be predicted
(Eickhoff et al., 2017). Figure 2 shows an example
of an image with the corresponding information pro-
vided in the distributed dataset. The same datasets
were distributed for both Concept Detection and Cap-
tion Prediction tasks and includes a variety of content
and situations, ranging from ultrasound images and x-
rays to charts and clinical photographs, which can be
seen in figure 3.
For the Caption Prediction Task, a training set
containing 164,614 image - caption pairs and an ad-
ditional validation set of 10,000 biomedical image -
caption pairs for evaluation purposes in the develop-
ment stage were distributed. Official evaluation was
computed using BLEU scores (Papineni et al., 2002)
on a test set with 10,000 biomedical images. For the
Figure 3: Examples of biomedical images showing variety
of content and situation. All images were randomly cho-
sen from the validation set of the ImageCLEFcaption 2017
Task.
Concept Detection Task, a set of UMLS
R
(Unified
Medical Language System) Concept Unique Identi-
fiers (CUIs) (Bodenreider, 2004) were provided for
each of the 164,614 biomedical images. These UMLS
concepts were identified using the QuickUMLS li-
brary (Soldaini and Goharian, 2016) from the original
captions published with the images (Eickhoff et al.,
2017). F
1
-Score was used as evaluation metric.
ImageCLEF 2015 Medical Clustering Task. This
dataset was distributed at the Medical Clustering Task
held at ImageCLEF 2015 and contains high resolu-
tion x-ray images collected from a hospital in Dhaka,
Bangladesh (Amin and Mohammed, 2015). X-rays
of both male and female patients aged 6 months to 72
years were present in the distributed dataset (Amin
and Mohammed, 2015). The training set included
500 images and the test set distributed for evalua-
tion contained 250 images. For each of the classes,
’Body’, ’Head-Neck’, ’Upper-Limb, ’Lower-Limb’
and ’True-Negative’, 100 images were present in the
training set. An example of the x-rays is shown in
figure 4.
Figure 4: Examples of x-rays with corresponding classifi-
cation. All images were randomly chosen from the training
set of the ImageCLEF 2015 Medical Clustering Task.
ImageCLEF 2009 Medical Image Annotation
Task. The dataset was distributed at the Image-
CLEF 2009 Medical Annotation task (Tommasi et al.,
2009). The training set consists of 12,671 grayscale x-
rays and the official evaluation set has 1,732 grayscale
x-rays. Each radiograph in the training set is anno-
tated with a 13 character string denoting the Image
Retrieval in Medical Applications (IRMA) (Lehmann
et al., 2004) classification code.
The IRMA-code describes the modality of the im-
ages, orientation of the image, examined body re-
gion and the biological system investigated. This
Adopting Semantic Information of Grayscale Radiographs for Image Classification and Retrieval
181
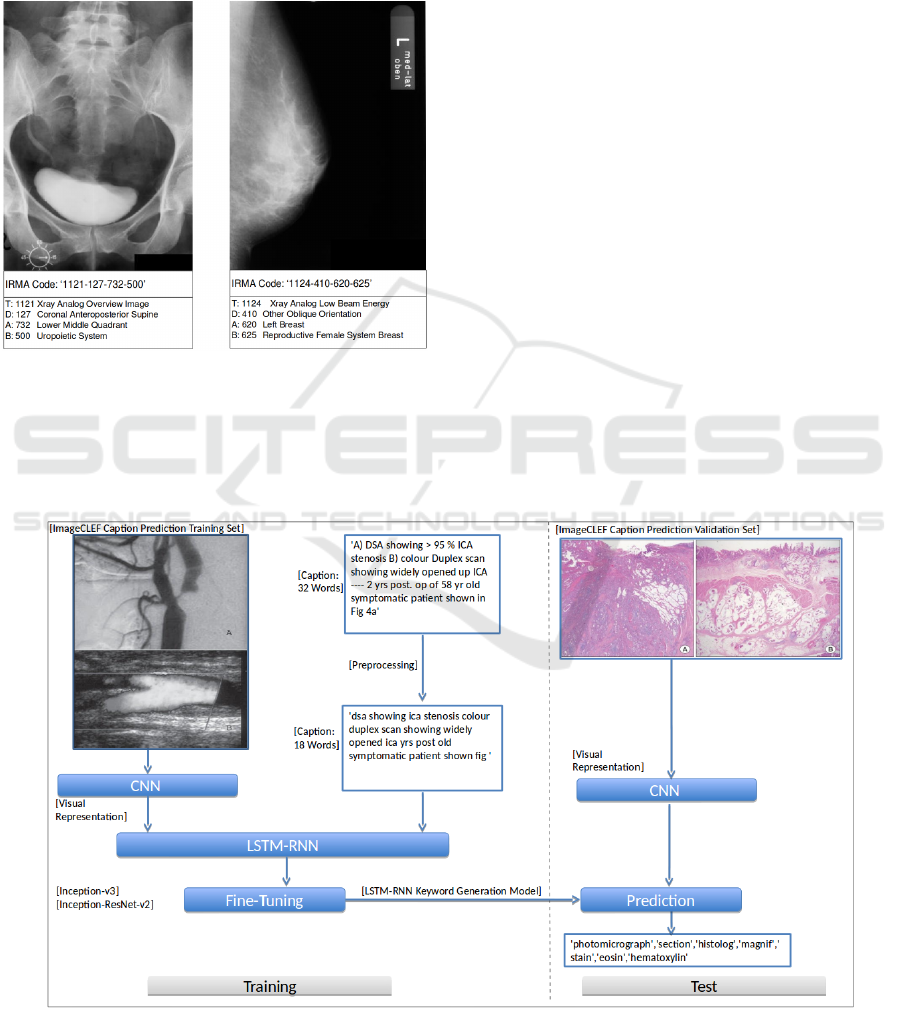
classification scheme contains 193 distinct classes.
Figure 6 shows two radiographs with annotations
1121-127-732-500 and 1121-410-620-625, represent-
ing ”xray overview image; coronal anteroposterior;
middle right abdomen; gastrointestinal system” and
”xray analog low beam energy; other oblique orienta-
tion; left breast; Reproductive female system breast”.
Figure 6: Example of two grayscale radiographs annotated
with the 13-digit IRMA classification code. Both images
are from the ImageCLEF 2009 Medical Annotation Task
Training Set.
2.2 Keyword Generation
For keyword generation, a combination of encod-
ing and decoding using Convolutional Neural Net-
works (CNN) and Long Short-Term Memory (LSTM)
(Hochreiter and Schmidhuber, 1997) based Recur-
rent Neural Networks (RNN) (Bengio et al., 1994) is
adopted. This approach, also known as Show-And-
Tell model was proposed in (Vinyals et al., 2015) and
further improved in (Vinyals et al., 2017).
To produce rich visual representations of the im-
ages, CNN is used as an image encoder by pre-
training it for an image classification task. The
LSTM-RNN utilized as caption decoder generates the
image keywords, using the CNN last hidden layer as
input (Vinyals et al., 2015).
Figure 5 shows the keyword generation model
training setup. In the first training phase, the LSTM
is trained using a corpus of paired image and cap-
tions generated from the biomedical figures in the
ImageCLEF 2017 Caption Prediction Task Training
Set (Eickhoff et al., 2017). No further dataset was
used for training. In the second phase, parameters
of the image model and LSTM are fine-tuned us-
ing the deep learning network Inception-ResNet-V2
(Szegedy et al., 2017). The parameters for the image
keyword generation model are:
• Batch size = [1. Trainingphase = 32; 2. Training-
Figure 5: Overview of Long Short-Term Memory based Recurrent Neural Network Model applied for biomedical image
keyword generation.
KALSIMIS 2018 - Special Session on Knowledge Acquisition and Learning in Semantic Interpretation of Medical Image Structures
182
phase = 32]
• Number of Epochs = [1. Trainingphase = 194; 2.
Trainingphase = 583]
• Vocabulary size = 23,000 {Minimum word occur-
rence ≥ 4}
• Initial learning rate = 2
• Model optimizer = stochastic gradient descent
• Learning rate decay factor = 0.5
• Number of epochs per decay = 8
• Inception learning rate = 0.0005
• Inception model initialization = Inception-
ResNet-V2
• LSTM embedding size = 512
• LSTM units number = 512
• LSTM initializer scale = 0.08
• LSTM dropout keep probability = 0.7
For all other parameters not mentioned above, the de-
fault values as proposed in (Vinyals et al., 2015) and
implemented in the Tensorflow-Slim im2txt-model
(Abadi et al., 2015; Shallue, 2017) were adopted.
Several text preprocessing methods such as reduc-
tion of image captions to nouns and adjectives, re-
moval of stopwords (Bird et al., 2009) and special
characters, and word stemming (Porter, 1980) were
performed. These text preprocessing steps are further
detailed in (Pelka and Friedrich, 2017).
2.3 Classification
Visual Representation. For whole-image classifi-
cation tasks, the Bag-of-Keypoints (BoK) (Csurka
et al., 2004) approach has achieved high classifica-
tion accuracy results (Lazebnik et al., 2006; Zhang
et al., 2006). BoK is based on vector quantization of
affine invariant descriptors of image patches (Csurka
et al., 2004). The simplicity and invariance to affine
transformation are advantages that come with this ap-
proach.
All functions applied for visual representation
computation are from the VLFEAT library (Vedaldi
and Fulkerson, 2010). Dense SIFT (dSIFT) (Li and
Perona, 2005) applied at several resolutions were uni-
formly extracted with an interval of 4 pixels using the
VL-PHOW function. Computational time was sped
up by computing k-means clustering with Approxi-
mated Nearest Neighbor (ANN) (Indyk and Motwani,
1998) on randomly chosen descriptors using the VL-
KMEANS function. This partitions the observations
into k clusters so that the within-cluster sum of square
is minimized.
A maximum number of 20 iterations was defined
to allow the k-means algorithm converge and clus-
ter centers were initialized using random data points
(Hartigan and Wong, 1979). A codebook containing
1,000 keypoints was generated as k = 1,000. Using
the VL-KDTREEBUILD function, the codebook was
further optimized by adapting a kd-tree with metric
distance L
2
for quick nearest neighbor lookup.
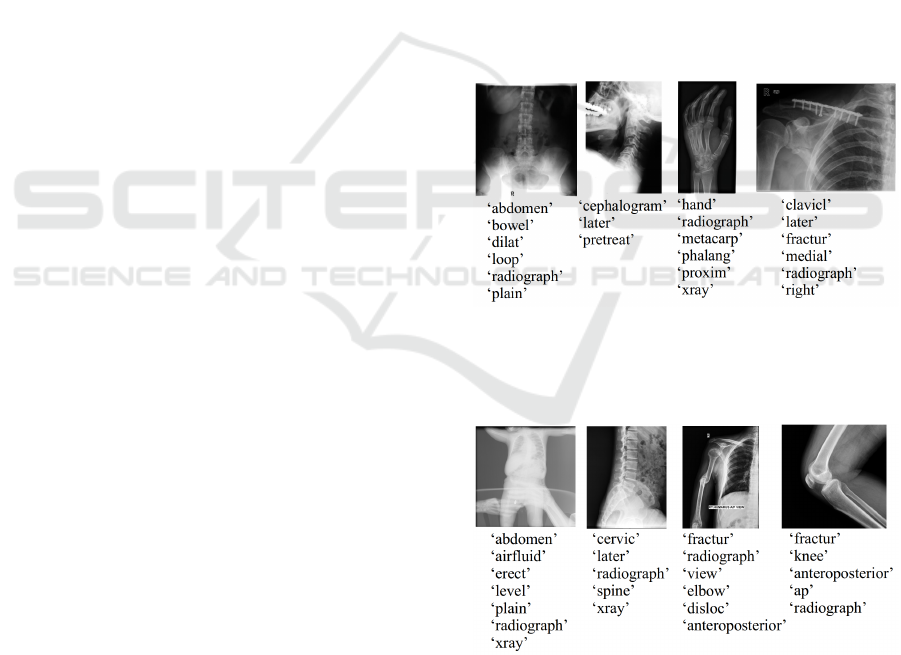
Text Representation. Utilizing the keyword gener-
ation model described in subsection 2.2, keywords
were generated for all radiology images in both Im-
ageCLEF 2009 Medical Annotation Task and Image-
CLEF 2015 Medical Clustering Task datasets. Fig-
ures 7 and 8 show generated keywords for randomly
chosen radiographs from the ImageCLEF 2009 Med-
ical Annotation Task Training Set and ImageCLEF
2015 Medical Clustering Task Training Set, respec-
tively. No further text preprocessing methods were
applied to the generated keywords, as this was done
before creating the keyword generation model.
Figure 7: Examples of keywords generated. All images are
from the ImageCLEF 2009 Medical Annotation Task Train-
ing Set. Keyword generation model was created using the
ImageCLEFcaption Prediction Task Training Set.
Figure 8: Examples of keywords generated. All images are
from the ImageCLEF 2015 Medical Clustering Task Train-
ing Set. Keyword generation model was created using the
ImageCLEFcaption Prediction Task Training Set.
For text representation, the Bag-of-Words (BoW)
(Salton and McGill, 1983) approach was adopted.
The basic concept is to extract features by counting
Adopting Semantic Information of Grayscale Radiographs for Image Classification and Retrieval
183
the frequency or presence of words in the text to be
classified. These words have to be first defined in
a dictionary or codebook. The fifty words with the
highest occurrence were used as dictionary, hence ob-
taining a feature vector size of 50. Several dictionary
sizes as well the benefit of Information Gain (Guyon
and Elisseeff, 2003) were investigated, but not further
used as no relevant advantage was detected.
Random Forest. Random forest (RF) (Breiman,
2001) models with 1,000 deep trees were created as
image classifiers. These RF-models were trained us-
ing multi-modal image representation. This is the
concatenation of visual features derived with the BoK
approach and the text features computed using BoW.
To reduce computational time, feature dimension and
noise, Principal Component Analysis (PCA) (Jolliffe,
2011) was applied on the visual representation. Pa-
rameters used to tune BoK and RF are:
• Codebook size: 1,000
• Number of descriptors extracted: 1,000
• Visual representation size: 4,000 (2x2 grid)
• Feature size reduction: 4000 to 100 (Principal
Component Analysis)
• Number of trees (RF): 1,000
• Ensemble method (RF): Bag
Support Vector Machines. Multi-class Support
Vector Machine (SVM) (Burges, 1998) image clas-
sification models were created for comparison. These
SVM-models were trained using the same multi-
modal image representation applied with the Random
Forest models. Parameters used to tune the SVM-
Models are:
• Kernel type: Radial basis function
• Cost parameter: 10
• Gamma: 1 / Number-Of-Features
Classification Schemes. For the ImageCLEF 2009
Medical Annotation Task, 4 different classification
schemes were used for evaluation. These schemes are
derived by using the complete IRMA code, mentioned
in subsection 2.1, as well splitting the code to its’ four
axes.
• (T) Technical-Code: 6 classes
• (D) Directional-Code: 34 classes
• (A) Anatomical-Code: 97 classes
• (B) Biological-System-Code: 11 classes
For the ImageCLEF 2015 Medical Clustering Task, a
classification scheme of four (4) classes was used.
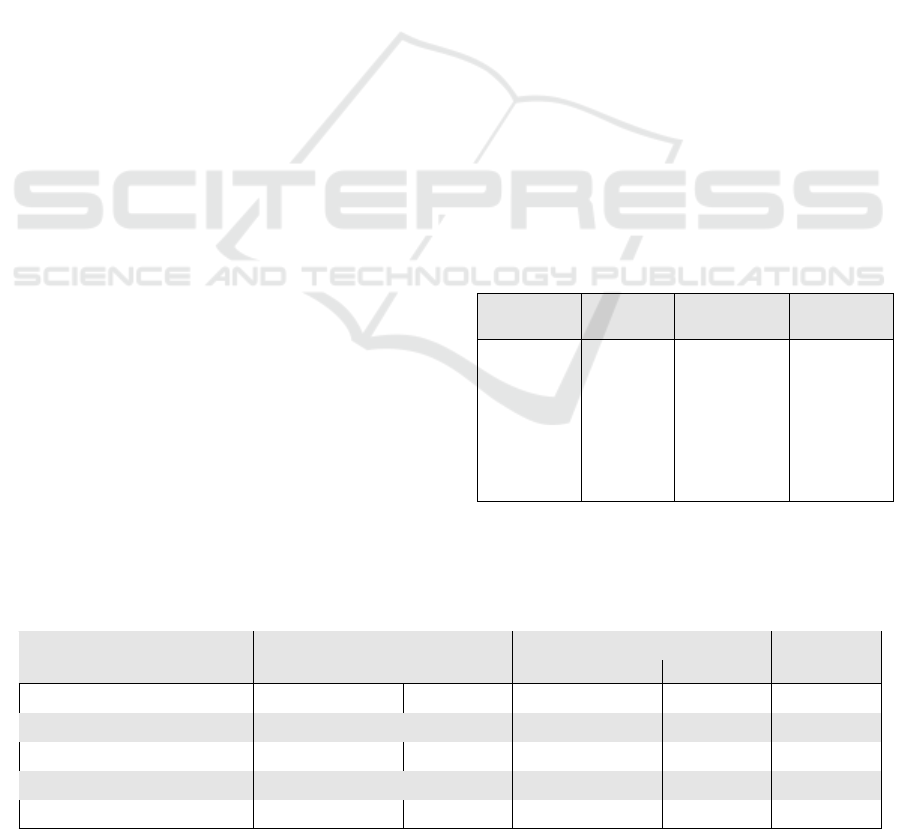
3 RESULTS
Table 2 shows generated keywords grouped to the Im-
ageCLEF 2015 Medical Clustering Task classification
scheme. Figure 9 displays the keywords and corre-
sponding UMLS CUIs, and prediction performance
of the random forest classification model is shown in
table 1.
Table 2: Keywords frequently generated for radiology im-
ages of the ImageCLEF 2015 Medical Clustering Task
Training Set. The keywords are grouped to the classifica-
tion scheme accordingly.
Body Head-
Neck
Lower-
Limb
Upper-
Limb
bodi massiv ray posterior
obstruct right ulna acromion
level neck union ulna
lesion effus humerus shoulder
plain pleural shaft carpus
abdomen disc radius end
distal fractur embrochag midshaft
Table 1: Prediction accuracies obtained using the random forest classification and support vector machine models. Column
’Visual Features’ shows accuracy with visual representation whereas ’Multi-Modal’ shows performance accuracies obtained
with the combination of visual and text representations.
Classification Scheme Visual Features Multi-Modal Test Set
Random Forest SVM Random Forest SVM
Technical-Code 97.00% 86.84% 97.75% 95.35% 1,732
Directional-Code 61.64% 55.31% 62.41% 61.26% 1,732
Anatomical-Code 51.15% 54.16% 54.62% 57.79% 1,732
Biological-Code 90.76% 81.12% 91.74% 82.47% 1,732
Medical Clustering 65.60% 66.40% 70.40% 68.80% 250
KALSIMIS 2018 - Special Session on Knowledge Acquisition and Learning in Semantic Interpretation of Medical Image Structures
184

Figure 9: Generated keywords and the corresponding Uni-
fied Medical Language Systems (UMLS) Concept Unique
Identifiers (CUI). The radiograph used for demonstration
was randomly chosen from the ImageCLEF 2009 Medical
Annotation Task Training Set.
4 DISCUSSION
It can be seen from table 1, that substituting the gen-
erated keywords as text representation improves the
classification prediction accuracy in both datasets and
all classification schemes. This positive increase is
obtained regardless of the classification method, as
both RF-models and SVM-models predicted better
with multi-modal representations.
The prediction performance varies to classifica-
tion scheme and method. The hierarchical approach
of splitting the IRMA-code to its four axes, proved
to be the better way to address this image annotation
task.
As Deep Convolutional Neural Networks have
proven the obtain improved prediction accuracies, an
approach combining generated keywords with fea-
tures extracted from the activation of a deep convo-
lutional network (DeCAF) (Donahue et al., 2014) is
intended. Positive results regarding biomedical image
classification using DeCAF were reported in (Koitka
and Friedrich, 2016).
The presented approach can be utilized for image
structuring and tagging of semantic information. The
generated keywords can be transformed to Unified
Medical Language Systems (UMLS) Concept Unique
Identifiers (CUIs), which is displayed in figure 9. The
conversion was obtained by applying QuickUMLS
(Soldaini and Goharian, 2016). The converted CUIs
are valuable and essential in terms of image retrieval
purposes.
5 CONCLUSION
As multi-modal image representation has proven to
obtain higher prediction results and some image
dataset lack text representation, an approach to gener-
ate keywords utilizing transfer learning was proposed.
To create a keyword generation model, image-caption
pairs of 164,614 biomedical figures distributed at
the ImageCLEFcaption 2017 Caption Prediction Task
was adopted to train Long Short-Term Memory based
Recurrent Neural Network models. The image cap-
tions were preprocessed by removing compound fig-
ure delimiters, single digits, special characters, word
stemming and reducing the captions to nouns and ad-
jectives.
Utilizing the keyword generation model, text rep-
resentation were created for two distinct radiology
datasets: ImageCLEF 2009 Medical Annotation Task
and ImageCLEF 2015 Medical Clustering Task. The
ImageCLEF 2015 Medical Clustering Task training
set contains 500 high resolution radiographs, 250 in
the test set and has a classification scheme with 4
classes. The ImageCLEF 2009 Medical Annotation
Task has 12,671 radiographs in the training set, 1,732
radiographs in the test set, and four classification
schemes with 5, 34, 97, 11 classes, respectively.
The generated keywords were further applied for
image classification purposes. In both image datasets
and all classification schemes, the prediction accura-
cies obtained with multi-modal image representation
outperformed those achieved using just visual fea-
tures. Using these generated keywords, semantic in-
formation in form of Unified Medical Language Sys-
tems (UMLS) Concept Unique Identifiers (CUIs) can
be tagged to the images, which is beneficial and of
assistance to image retrieval solutions. The proposed
work can be further enhanced by extracting image vi-
sual representation using Deep Convolutional Neural
Networks and optimized Bag-of-Words.
REFERENCES
Abadi, M., Agarwal, A., Barham, P., Brevdo, E., Chen, Z.,
Citro, C., Corrado, G. S., Davis, A., Dean, J., Devin,
M., Ghemawat, S., Goodfellow, I., Harp, A., Irving,
G., Isard, M., Jia, Y., Jozefowicz, R., Kaiser, L., Kud-
lur, M., Levenberg, J., Man
´
e, D., Monga, R., Moore,
S., Murray, D., Olah, C., Schuster, M., Shlens, J.,
Steiner, B., Sutskever, I., Talwar, K., Tucker, P., Van-
houcke, V., Vasudevan, V., Vi
´
egas, F., Vinyals, O.,
Warden, P., Wattenberg, M., Wicke, M., Yu, Y., and
Zheng, X. (2015). TensorFlow: Large-Scale Machine
Learning on Heterogeneous Systems. Software avail-
able from tensorflow.org.
Adopting Semantic Information of Grayscale Radiographs for Image Classification and Retrieval
185

Abrao, M. S., Gonc¸alves, M. O. d. C., Dias Jr, J. A.,
Podgaec, S., Chamie, L. P., and Blasbalg, R. (2007).
Comparison between clinical examination, transvagi-
nal sonography and magnetic resonance imaging for
the diagnosis of deep endometriosis. Human Repro-
duction, 22(12):3092–3097.
Amin, M. A. and Mohammed, M. K. (2015). Overview
of the ImageCLEF 2015 Medical Clustering Task. In
Working Notes of CLEF 2015 - Conference and Labs
of the Evaluation forum, Toulouse, France, September
8-11, 2015.
Bengio, Y., Simard, P. Y., and Frasconi, P. (1994). Learn-
ing long-term dependencies with gradient descent is
difficult. IEEE Transactions on Neural Networks,
5(2):157–166.
Bird, S., Klein, E., and Loper, E. (2009). Natural Language
Processing with Python. O’Reilly, first edition.
Bodenreider, O. (2004). The Unified Medical Lan-
guage System (UMLS): integrating biomedical ter-
minology. Nucleic Acids Research, 32(Database-
Issue):267–270.
Breiman, L. (2001). Random Forests. Machine Learning,
45(1):5–32.
Burges, C. J. C. (1998). A tutorial on support vector ma-
chines for pattern recognition. Data Mining Knowl-
edge Discovery, 2(2):121–167.
Codella, N. C. F., Connell, J. H., Pankanti, S., Mer-
ler, M., and Smith, J. R. (2014). Automated med-
ical image modality recognition by fusion of visual
and text information. In Medical Image Computing
and Computer-Assisted Intervention - MICCAI 2014
- 17th International Conference, Boston, MA, USA,
September 14-18, 2014, Proceedings, Part II, pages
487–495.
Csurka, G., Dance, C. R., Fan, L., Willamowski, J., and
Bray, C. (2004). Visual categorization with bags of
keypoints. In Workshop on Statistical Learning in
Computer Vision, European Conference on Computer
Vision, ECCV 2004, Prague, Czech Republic, May 11-
14, 2004, pages 1–22.
Dalal, N. and Triggs, B. (2005). Histograms of oriented
gradients for human detection. In Proceedings of the
2005 IEEE Computer Society Conference on Com-
puter Vision and Pattern Recognition (CVPR’05) -
Volume 1 - Volume 01, CVPR ’05, pages 886–893,
Washington, DC, USA. IEEE Computer Society.
Donahue, J., Jia, Y., Vinyals, O., Hoffman, J., Zhang, N.,
Tzeng, E., and Darrell, T. (2014). Decaf: A deep con-
volutional activation feature for generic visual recog-
nition. In Proceedings of the 31th International Con-
ference on Machine Learning, ICML 2014, Beijing,
China, 21-26 June 2014, pages 647–655.
Eickhoff, C., Schwall, I., de Herrera, A. G. S., and M
¨
uller,
H. (2017). Overview of ImageCLEFcaption 2017
- Image Caption Prediction and Concept Detection
for Biomedical Images. In Working Notes of CLEF
2017 - Conference and Labs of the Evaluation Forum,
Dublin, Ireland, September 11-14, 2017.
Guyon, I. and Elisseeff, A. (2003). An Introduction to
Variable and Feature Selection. Journal of Machine
Learning Research, 3:1157–1182.
Hartigan, J. A. and Wong, M. A. (1979). A k-means cluster-
ing algorithm. JSTOR: Applied Statistics, 28(1):100–
108.
Hinton, G., Deng, L., Yu, D., Dahl, G., Mohamed, A.-
a., Jaitly, N., Senior, A., Vanhoucke, V., Nguyen, P.,
Sainath, T., and Kingsbury, B. (2012). Deep Neural
Networks for Acoustic Modeling in Speech Recog-
nition: The Shared Views of Four Research Groups.
IEEE Signal Processing Magazine, 29(6):82–97.
Hochreiter, S. and Schmidhuber, J. (1997). Long Short-
Term Memory. Neural Computation, 9(8):1735–1780.
Huang, G., Liu, Z., van der Maaten, L., and Weinberger,
K. Q. (2017). Densely Connected Convolutional
Networks. In Proceedings of the IEEE Conference
on Computer Vision and Pattern Recognition, CVPR
2017, Honolulu, USA, July 22-25, 2017.
Indyk, P. and Motwani, R. (1998). Approximate Near-
est Neighbors: Towards Removing the Curse of Di-
mensionality. In Proceedings of the 30th Annual
ACM Symposium on Theory of Computing, STOC ’98,
pages 604–613, New York, NY, USA. ACM.
Jolliffe, I. T. (2011). Principal Component Analysis. In In-
ternational Encyclopedia of Statistical Science, pages
1094–1096. Springer Berlin Heidelberg.
Kalpathy-Cramer, J., de Herrera, A. G. S., Demner-
Fushman, D., Antani, S. K., Bedrick, S., and M
¨
uller,
H. (2015). Evaluating performance of biomedical im-
age retrieval systems - An overview of the medical im-
age retrieval task at ImageCLEF 2004-2013. Comput-
erized Medical Imaging and Graphics, 39:55–61.
Koitka, S. and Friedrich, C. M. (2016). Traditional Fea-
ture Engineering and Deep Learning Approaches at
Medical Classification Task of ImageCLEF 2016. In
Working Notes of CLEF 2016 - Conference and Labs
of the Evaluation forum,
´
Evora, Portugal, 5-8 Septem-
ber, 2016., pages 304–317.
Lazebnik, S., Schmid, C., and Ponce, J. (2006). Beyond
Bags of Features: Spatial Pyramid Matching for Rec-
ognizing Natural Scene Categories. In Proceedings of
the 2006 IEEE Computer Society Conference on Com-
puter Vision and Pattern Recognition, CVPR 2006,
Ney York, USA, June 17-22 2006, pages 2169–2178.
LeCun, Y., Bengio, Y., and Hinton, G. E. (2015). Deep
Learning. Nature, 521(7553):436–444.
Lehmann, T. M., G
¨
uld, M. O., Thies, C., Plodowski, B.,
Keysers, D., Ott, B., and Schubert, H. (2004). IRMA
- Content-Based Image Retrieval in Medical Applica-
tions. In MEDINFO 2004 - Proceedings of the 11th
World Congress on Medical Informatics, San Fran-
cisco, California, USA, September 7-11, 2004, pages
842–846.
Li, F.-F. and Perona, P. (2005). A Bayesian Hierarchi-
cal Model for Learning Natural Scene Categories.
In Proceedings of the 2005 IEEE Computer Society
Conference on Computer Vision and Pattern Recogni-
tion, CVPR 2005, San Diego, USA, June 20-26, 2005,
pages 524–531.
Pan, S. J. and Yang, Q. (2010). A Survey on Transfer Learn-
ing. IEEE Transactions on Knowledge and Data En-
gineering, 22(10):1345–1359.
KALSIMIS 2018 - Special Session on Knowledge Acquisition and Learning in Semantic Interpretation of Medical Image Structures
186

Papineni, K., Roukos, S., Ward, T., and Zhu, W. (2002).
BLEU: a Method for Automatic Evaluation of Ma-
chine Translation. In Proceedings of the 40th An-
nual Meeting of the Association for Computational
Linguistics, July 6-12, 2002, Philadelphia, PA, USA.,
pages 311–318.
Pelka, O. and Friedrich, C. M. (2015). FHDO Biomedi-
cal Computer Science Group at Medical Classification
Task of ImageCLEF 2015. In Working Notes of CLEF
2015 - Conference and Labs of the Evaluation forum,
Toulouse, France, September 8-11, 2015.
Pelka, O. and Friedrich, C. M. (2016). Modality prediction
of biomedical literature images using multimodal fea-
ture representation . GMS Medizinische Informatik,
Biometrie und Epidemiologie, 12(2):1345–1359.
Pelka, O. and Friedrich, C. M. (2017). Keyword Generation
for Biomedical Image Retrieval with Recurrent Neural
Networks. In Working Notes of CLEF 2017 - Confer-
ence and Labs of the Evaluation Forum, Dublin, Ire-
land, September 11-14, 2017.
Porter, M. (1980). An algorithm for suffix stripping.
Program-electronic Library and Information Systems,
14:130–137.
PubMed Central (2017). National institutes of health
NIH, US National Library of Medicine. In
https://www.ncbi.nlm.nih.gov/pmc/ Last accessed:
2017-11-30.
Rahman, M. M., Bhattacharya, P., and Desai, B. C. (2007).
A Framework for Medical Image Retrieval Using Ma-
chine Learning and Statistical Similarity Matching
Techniques With Relevance Feedback. IEEE Trans-
actions on Information Technology in Biomedicine,
11(1):58–69.
Salton, G. and McGill, M. J. (1983). Introduction to Mod-
ern Information Retrieval. McGraw-Hill computer
science series. McGraw-Hill, New York.
Shallue, C. (2017). Im2txt github. In
https://github.com/tensorflow/models/tree/master/
research/im2txt, Last accessed: 2017-11-30.
Soldaini, L. and Goharian, N. (2016). QuickUMLS: a
fast, unsupervised approach for medical concept ex-
traction. In Medical Information Retrieval (MedIR)
Workshop in SIGIR Conference on Research and De-
velopment in Information Retrieval 2016, Pisa, Italy,
July 17-21, 2016.
Szegedy, C., Ioffe, S., Vanhoucke, V., and Alemi, A. A.
(2017). Inception-v4, Inception-ResNet and the Im-
pact of Residual Connections on Learning. In Pro-
ceedings of the Thirty-First AAAI Conference on Arti-
ficial Intelligence, February 4-9, 2017, San Francisco,
California, USA., pages 4278–4284.
Tagare, H. D., Jaffe, C. C., and Duncan, J. S. (1997).
Synthesis of research: Medical image databases: A
content-based retrieval approach. Journal of the
American Medical Informatics Association JAMIA,
4(3):184–198.
Tommasi, T., Caputo, B., Welter, P., G
¨
uld, M. O., and De-
serno, T. M. (2009). Overview of the CLEF 2009
Medical Image Annotation Track. In Multilingual In-
formation Access Evaluation II. Multimedia Experi-
ments - 10th Workshop of the Cross-Language Evalu-
ation Forum, CLEF 2009, Corfu, Greece, September
30 - October 2, 2009, Revised Selected Papers, pages
85–93.
Valavanis, L., Stathopoulos, S., and Kalamboukis, T.
(2016). IPL at CLEF 2016 Medical Task. In Work-
ing Notes of CLEF 2016 - Conference and Labs of
the Evaluation forum,
´
Evora, Portugal, 5-8 Septem-
ber, 2016., pages 413–420.
Vedaldi, A. and Fulkerson, B. (2010). VLFEAT: an open
and portable library of computer vision algorithms. In
Proceedings of the 18th International Conference on
Multimedia 2010, Firenze, Italy, October 25-29, 2010,
pages 1469–1472.
Vinyals, O., Toshev, A., Bengio, S., and Erhan, D. (2015).
Show and Tell: A neural image caption generator.
In IEEE Conference on Computer Vision and Pattern
Recognition, CVPR 2015, Boston, MA, USA, June 7-
12, 2015, pages 3156–3164.
Vinyals, O., Toshev, A., Bengio, S., and Erhan, D. (2017).
Show and Tell: Lessons Learned from the 2015
MSCOCO Image Captioning Challenge. IEEE Trans-
actions on Pattern Analysis and Machine Intelligence,
39(4):652–663.
Xu, Y., Mo, T., Feng, Q., Zhong, P., Lai, M., and Chang,
E. I. (2014). Deep learning of feature representa-
tion with multiple instance learning for medical image
analysis. In IEEE International Conference on Acous-
tics, Speech and Signal Processing, ICASSP 2014,
Florence, Italy, May 4-9, 2014, pages 1626–1630.
Zhang, H., Berg, A. C., Maire, M., and Malik, J. (2006).
SVM-KNN: Discriminative Nearest Neighbor Classi-
fication for Visual Category Recognition. In Proceed-
ings of the 2006 IEEE Computer Society Conference
on Computer Vision and Pattern Recognition, CVPR
2006, Ney York, USA, June 17-22 2006, pages 2126–
2136.
Adopting Semantic Information of Grayscale Radiographs for Image Classification and Retrieval
187
