
Antioxidant Activity from Lygodium Microphyllum Aerial Parts
Hadi Kuncoro, Laode Rijai
Laboratorium Penelitian dan Pengembangan Kefarmasian Farmaka Tropis, Fakultas Farmasi Universitas Mulawarman,
Samarinda, Indonesia, 75117, Phone : +62541739491
Keywords: L. microphyllum, Antioxidant, DPPH, IC
50
Abstract: Lygodium microphyllum is an invasive plant known as another name “Old World Climbing Fern” or
considered a weed plant. Research on L. microphyllum related pharmacological activity is limited. This
study aims to evaluate antioxidant activity from Methanol extract and Fractionation using n-hexane, Ethyl
acetate and water solvent with a different polarity from L. microphyllum plant. Methods: Aerial part of L.
microphyllum was macerated with methanol solvent, the extract obtained in fractionation using solvents
with different polarity levels and was tested in invitro free radical using 1, 1-diphenyl-2-picrylhydrazyl
(DPPH) free radical. The IC50 result was 49.328 ± 0.242 ppm for methanol extract, n-hexane extract was
32.041 ± 0.770 ppm, and ethyl acetate extract 8,732 ± 0.204 ppm and water extract 93.330 ± 1.931 ppm.
The conclusion from this study reveals the results of extraction using ethyl acetate solvent showed the
highest value antioxidant activity against DPPH.
1 INTRODUCTION
Lygodium microphyllum is an invasive plant known
as “Old World Climbing Fern” or is considered a
weed plant. L. microphyllum plants develop and
growing fast in large colonies that dominate an area
and cause disruption of the ecosystem the original
flora and degrades critical ecosystem services and
habitats of rare and endangered species (Ferriter
2001; Langeland & Hutchinson, 2001; Pemberton,
1998).
L. microphyllum spread to the nearby plant and
live in an open place to get sunshine (Bower, 2010).
Some studies showed L. microphyllum have
medicinal properties including antibacterial (Wang
et al 2014), Hepatoprotector (Gnanaraj et al, 2017),
Cytotoxic activity against P-388 Murine Leukemia
cells (Kuncoro, et al 2017), and antiplasmodial
against Plasmodium falciparum (Ratri et al, 2018).
The chemical composition of L. microphyllum has
been reported including steroid and glycoside
flavonoids ( Kuncoro, et al 2017).
Reports of the antioxidant activity from other
species Lygodium in one genus have been reported.
Methanol extract from L. flexuosum has antioxidant
activity (Jeetendra & Manish, 2011).
The aim of this studies to evaluated Antioxidant
activities aerial part from L. microphyllum against
DPPH.
2 MATERIAL AND METHOD
.
2.1 Material
L. microphyllum aerial part was obtained from
Lempake Village, Samarinda, Kalimantan Timur.
Specimens were identified and deposit by staff at the
Dendrology laboratory, Foresty Faculty,
Mulawarman University.
2.2 Extraction Process
Aerial parts of L. microphyllum are cleaned and
dried and powdered to obtain 3.52 Kg. The dry
powder was macerated with methanol solvent at
room temperature. The methanol extract was
obtained and concentrated 526 g with a Rotary
Evaporator (Buchi
®
). The methanol extract obtained
was dissolved in water (1:1) and partitioned
successively with n-hexane solvent, ethyl acetate
solvent, and Aqua solvent. The resulting extract with
solvent was evaporated and obtained n-hexane
Kuncoro, H. and Rijai, L.
Antioxidant Activity from Lygodium microphyllum Aerial Parts.
DOI: 10.5220/0009845400002406
In Proceedings of BROMO Conference (BROMO 2018) - Symposium on Natural Product and Biodiversity, page 1
ISBN: 978-989-758-347-6
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
1

extract (59 g), ethyl acetate extract (72 g), and aqua
extract (296,5 g).
2.3 Antioxidant Assay
2.3.1 Preparation
The DPPH crystals were weighed 4 mg to be
dissolved in 100 mL of methanol in the measuring
flask for obtaining a DPPH solution with a
concentration of 0.004% or 40 ppm (part per
million) used in the test. The solution is stored in a
tightly sealed place and protected from light.
2.3.2 Concentration Series Determination
The concentration series used was 0.2,4,6,8,10 ppm..
These concentration variations were used in
antioxidant activity testing by DPPH method.
Preparation of stock solution with a concentration of
1000 ppm (10 mg dry extract diluted with methanol
to 10 mL). Furthermore, dilution to obtain the
concentration and Duplo repetition or two
repetitions and a negative control that DPPH
solution and methanol (without the addition of
extract).
2.3.3. Determination of the Maximum
Absorption Wavelength DPPH
2 mL of 0.004% DPPH solution was added with 2
mL of methanol. After being left for 30 minutes in
the dark, Absorption of the solution was measured
by UV-Vis spectrophotometer at 515 -520 nm
wavelength to obtain the maximum wavelength.
2.3.4 Antioxidant assay
The antioxidant test was carried out through a series
of sample solutions from the methanol extract and
the three fractions with 2 repetitions using a
methanol solvent. Each solution plus 2 mL of DPPH
solution, in order to obtain a solution of a
predetermined concentration, was allowed to stand
for 30 minutes (calculated after addition of DPPH
solution), measured its absorbance at the maximum
wavelength. The absorbance data obtained is used to
determine % inhibition (damping). Through sample
concentration curve versus % inhibition, IC
50
extract
value can be obtained with statistical analysis using
linear regression. There is also a measure of
absorbance of blanks (methanol). The antioxidant
test indicator is the color change of DPPH. The data
in this study is the percentage of DPPH radical
reduction obtained using the formula:
% AA=
𝐴𝑏𝑠. 𝐶𝑜𝑛𝑡𝑟𝑜𝑙 − 𝐴𝑏𝑠. 𝑆
𝐴𝑏𝑠. 𝐶𝑜𝑛𝑡𝑟𝑜𝑙
× 100%
Data analysis using linear regression equation
using the formula:
y = b (x) + a
Explanation:
y: % AA
x: log concentration
a: intercept
b: slope
3 RESULT AND DISCUSSION
In this study conducted testing of antioxidant
activity of aerial parts of L. microphyllum. Dry
powder aerial parts of L. microphyllum macerated
using methanol to obtain methanol extract. Methanol
extract in fractionation based on polarity level using
n-hexane, ethyl acetate, methanol and water. Each
extract obtained was tested for its antioxidant
activity using DPPH method. This method was
chosen because is simple, easy, fast and sensitive
and requires only a little extract. This method is
often used to detect the anti-radical ability of a
compound because the result proves to be accurate,
reliable, relatively fast and practical. The principle
of this method is the measurement of synthetic free
radical capture in polar organic solvents such as
ethanol or methanol at room temperature by a
compound having antioxidant activity. This process
of radical capture through the mechanism of taking
hydrogen atoms from antioxidant compounds by
free radicals so that free radicals capture an electron
from antioxidants. With the arrest of these radicals,
the diazo double bond in DPPH decreases, resulting
in decreased absorbance. The free radical used is
DPPH (2,2-diphenyl-1-picrylhydrazyl).
DPPH reacts with antioxidant compounds by
taking hydrogen atoms from antioxidant compounds
to obtain electron pairs. The compounds that can
potentially as antioxidants are from the class of
flavonoids, alkaloids, phenols, and tannins. Before
the testing of antioxidant activity, the first
determination of the maximum wavelength of DPPH
used with the wavelength range 515-520 nm. The
BROMO 2018 - Bromo Conference, Symposium on Natural Products and Biodiversity
2
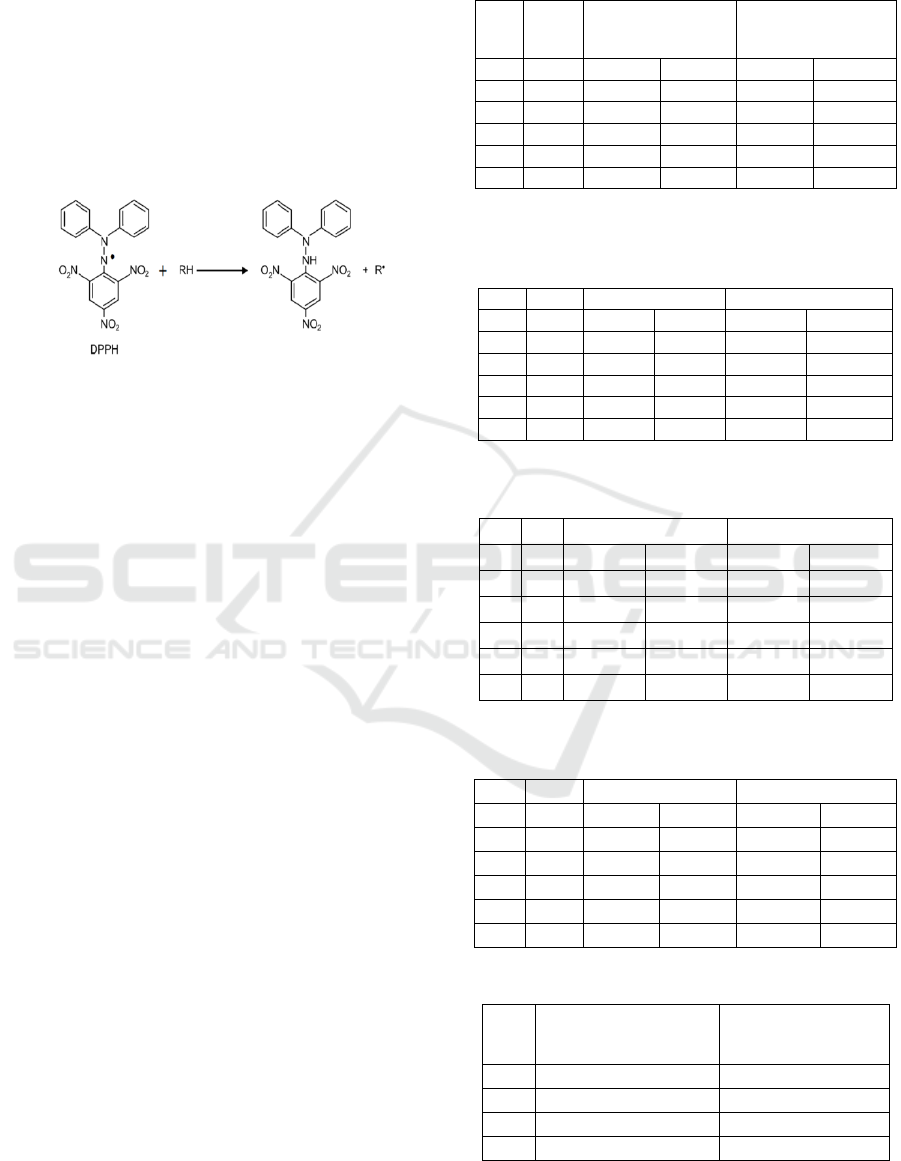
maximum wavelength is obtained from the
maximum absorbance of 515 nm. This wavelength
will be used in subsequent antioxidant testing. The
compound that reacts as a radical catcher will reduce
DPPH which can be observed by the color change of
DPPH to yellow from purple when the odd electron
from the DPPH radical has paired with hydrogen.
From the free radical captured compound which will
form the reduced DPPH-H (Molyneux, 2004) seen
in Figure 1.
Figure 1: DPPH Radical Reaction with Antioxidant
Compound (Molyneux, 2004)
All electrons in DPPH free radicals become in
pairs, then the color of the solution changes to bright
yellow from dark purple and absorbance at 515 nm
wavelength will be lost. These changes can be
measured stoichiometrically according to the
number of electrons or hydrogen atoms captured by
DPPH molecules due to the presence of antioxidants
(Gurav, 2007). The calculation used in the
determination of radical catch activity is the value of
IC
50
(Inhibition Concentration 50%). This value
describes the concentration of test compounds that
can capture radicals by 50%. IC
50
values in this
study were obtained by using a linear regression
equation which expresses the relationship between
sample concentrations (extract test) with the symbol
(x) with radical capture activity (y) from series
replication measurement. The smaller the value of
IC
50
obtained, the test sample has more effectiveness
as a better radical catcher. Molyneux (2004) states
that a substance has antioxidant properties if the IC
50
value is less than 200 ppm. If the value of IC
50
obtained ranges from 200-1000 ppm, then the
material is less active but still potentially as an
antioxidant substance.
From the test result on methanol extract, n-
hexane, ethyl acetate, and water obtained absorbance
value and% inhibition.
Table 1: Absorbance and % inhibition of antioxidant
activity (DPPH) of methanol extract.
No.
ppm
Absorbance
% inhibition
1.
0
0,7745
0,7745
0,0000
0,0000
2.
2
0,7559
0,7684
2,4015
0,7876
3.
4
0,7404
0,7605
4,4028
1,8076
4.
6
0,7160
0,7255
7,5533
6,3267
5.
8
0,7116
0,7116
8,1214
8,1214
6.
10
0,6973
0,6958
9,9677
10,1614
Table 2: Absorbance and % inhibition of antioxidant
activity (DPPH) of n-hexane extract.
No.
ppm
Absorbance
% inhibition
1.
0
0,8478
0,8478
0,0000
0,0000
2.
2
0,8068
0,8241
4,8360
2,7955
3.
4
0,7678
0,7776
6,0156
8,2803
4.
6
0,7573
0,7549
10,6747
10,9578
5.
8
0,7478
0,7478
11,7952
11,7952
6.
10
0,7114
0,7114
16,0887
16,0887
Table 3: Absorbance and % inhibition of antioxidant
activity (DPPH) of ethyl acetate extract.
No
ppm
Absorbance
% inhibition
1.
0
0,8820
0,8820
0,0000
0,0000
2.
2
0,7994
0,8033
9,3651
8,9229
3.
4
0,6788
0,7070
23,0386
19,7732
4.
6
0,5718
0,5749
35,1701
34,8186
5.
8
0,4704
0,4828
46,6667
45,2608
6.
10
0,3689
0,3849
58,1746
56,3605
Table 4: Absorbance and % inhibition of antioxidant
activity (DPPH) of water extract..
No.
ppm
Absorbance
% inhibition
1.
0
0,9879
0,9879
0,0000
0,0000
2.
2
0,9849
0,9849
0,3037
0,3037
3.
4
0,9624
0,9624
2,5812
2,5812
4.
6
0,9607
0,9489
2,7533
3,9478
5.
8
0,9439
0,9454
4,4539
4,3021
6.
10
0,9396
0,9388
4,8892
4,9701
Table 5: Antioxidant activity (IC
50
)
No.
Extract
IC
50
(ppm)
1
Methanol
49.328 ± 0.242
2
n-hexane
32.041 ± 0.770
3
Ethyl Acetate
8,732 ± 0.204
4
Water
93.330 ± 1.931
Antioxidant Activity from Lygodium microphyllum Aerial Parts
3

From data assay against DPPH obtained that the
extract of ethyl acetate has the highest IC
50
value
8,732 ± 0204 ppm. Based on this, the aerial part
extract of L. microphyllum has an activity of an
antioxidant with an IC
50
value of less than 200 ppm.
4 CONCLUSIONS
Ethyl acetate fraction of aerial parts L. microphyllum
has the highest antioxidant activity against DPPH
with an IC
50
value of 8,732 ± 0,204 ppm.
ACKNOWLEDGMENTS
Thanks to Fakultas Farmasi Universitas
Mulawarman Samarinda and Central Laboratory
Padjadjaran University for Antioxidant assay against
DPPH.
REFERENCES
Bower, F. O. 2010. The Fern (Filicales). Cambridge
University Press. Cambridge
Ferriter, A. 2001. Lygodium Management Plant for
Florida. Florida: Florida Exotic Pest Plant Council
Lygodium Task Force.
Gnanaraj, C., Shah, M.D., Song, T.T. and Iqbal, M., 2017.
Hepatoprotective mechanism of Lygodium
microphyllum (Cav.) R. Br. through ultrastructural
signaling prevention against carbon tetrachloride
(CCl4)-mediated oxidative stress. Biomedicine &
Pharmacotherapy, 92, pp.1010-1022.
Gurav, S., Deshkar, N., Gulkari, V., Duragkar, N., & Patil,
A. 2007. Free radical scavenging activity of Polygala
chinensis Linn. Pharmacology Line, 2, 245-253.
Hutchinson, J.T. & Langeland, K.A., 2013. Susceptibility
Of Old World Climbing Fern (Lygodium
microphyllum) Gametophytes To Metsulfuron Methyl.
Invasive Plant Science and Management 6:304-309.
Jeetendra, N. E. H. E. T. E., & Manish, B. H. A. T. I. A.
(2011). Correlation of antioxidant activity with
phenolic content and isolation of antioxidant
compound from Lygodium flexuosum (l.) Sw.
Extracts. Int J Pharm Pharm Sci, 3(2), 48-52.
Kuncoro, H., Farabi, K. and Rijai, L., 2017. Steroids and
isoquercetin from Lygodium microphyllum. Journal of
Applied Pharmaceutical Science Vol, 7(11), pp.136-
141.
Kuncoro, H., Rijai, L., Julaeha, E., & Supratman, U.,
2017, Cytotoxic Activity Against P-388 Murine
Leukemia Cell From Lygodium microphyllum HERB,
Jurnal Farmasi Galenika Volume 3 No. 1; p 13-16
Langeland, K.A. & Hutchinson, J., 2001. Natural Area
Weeds: Old World Climbing Fern (Lygodium
microphyllum). University of Florida Cooperative
Extension Service, Institute of Food and Agricultural
Sciences, EDIS.
Molyneux, P., 2004. The use of the stable free radical
diphenylpicrylhydrazyl (DPPH) for estimating
antioxidant activity. Songklanakarin J. Sci. Technol,
26(2), pp.211-219.
Pemberton, R. W. 1998. The Potential Of Biological
Control To Manage Old World Climbing Fern
(Lygodium microphyllum), An Invasive Weed In
Florida. American Fern J. 88: 176-182.
Pemberton, R. W., and Ferriter, A. P. 1998. Old World
Climbing Fern (Lygodium microphyllum), A
Dangerous Invasive Weed In Florida. American Fern
J. 88: 165-175.
Ratri, T. R., Harlia, & Widiyantoro, A., 2018,
Characterization of Antimalarial Compounds from
Lygodium microphyllum Ethyl Acetate Fraction
against Plasmodium falciparum, Jurnal Kimia
Khatulistiwa, 7(2): 89-95
Wang, R.L., Zheng, Z.H., Lu, J., Shao, H., Zhang, H., Su,
Y.J., & Cai, Y.F. 2014. Allelopathic potential of
invasive climbing fern Lygodium microphyllum
against native plants and antibacterial activity of
essential oils. Allelopathy Journal. 33(1), 97–106.
BROMO 2018 - Bromo Conference, Symposium on Natural Products and Biodiversity
4
