
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
Gonc¸alo Santos
1,∗
, Cl
´
audia Alves
1
, Ana Carolina P
´
adua
1
, Susana Palma
1
,
Hugo Gamboa
2
and Ana Cec
´
ılia Roque
1
1
UCIBIO, Departamento de Qu
´
ımica, Faculdade de Ci
ˆ
encias e Tecnologia da Universidade NOVA de Lisboa,
2829-516 Caparica, Portugal
2
Laborat
´
orio de Instrumentac¸
˜
ao Engenharia Biom
´
edica e F
´
ısica da Radiac¸
˜
ao (LIBPhys-UNL), Departamento de F
´
ısica,
Faculdade de Ciencias e Tecnologia da Universidade NOVA de Lisboa, Monte da Caparica, 2829-516 Caparica, Portugal
Keywords:
Electronic Nose, Volatile Organic Compounds, Machine Learning, Biomaterials.
Abstract:
Electronic noses (E-noses), are usually composed by an array of sensors with different selectivities towards
classes of VOCs (Volatile Organic Compounds). These devices have been applied to a variety of fields, includ-
ing environmental protection, public safety, food and beverage industries, cosmetics, and clinical diagnostics.
This work demonstrates that it is possible to classify eleven VOCs from different chemical classes using a
single gas sensing biomaterial that changes its optical properties in the presence of VOCs. To accomplish this,
an in-house built E-nose, tailor-made for the novel class of gas sensing biomaterials, was improved and com-
bined with powerful machine learning techniques. The device comprises a delivery system, a detection system
and a data acquisition and control system. It was designed to be stable, miniaturized and easy-to-handle. The
data collected was pre-processed and features and curve fitting parameters were extracted from the original
response. A recursive feature selection method was applied to select the best features, and then a Support
Vector Machine classifier was implemented to distinguish the eleven distinct VOCs. The results show that the
followed methodology allowed the classification of all the VOCs tested with 94.6% (± 0.9%) accuracy.
1 INTRODUCTION
The human olfactory system is sophisticated and is
able to detect spoiled food, to identify dangerous en-
vironments or pleasant experiences. Highly trained
humans can even develop this sense beyond the usual
limits. Hippocrates (430 B.C.), considered the father
of medicine, could diagnose diseases by smelling pa-
tient’s biological samples (R
¨
ock et al., 2008). Odours
consist of sets of volatile organic compounds (VOCs).
When VOCs flow through the human’s nasal cavity,
they reach the olfactory epithelium, where olfactory
sensory neurons define action potentials up their own
axons, until the olfactory bulb, which then transmits
the signal to the brain, where recognition occurs (Bar-
bosa et al., 2018).
The intriguing olfactory system has inspired re-
searchers to develop technologies mimicking natural
olfaction (Wasilewski et al., 2017; He et al., 2012;
P
´
adua et al., 2018b).
The best example of artificial olfaction is the con-
cept of electronic nose (E-nose). E-noses comprise
of an array of semi-selective sensors (e.g. metal ox-
ide semiconductor or MOS-based sensors, conduct-
ing polymers, piezoelectric quartz crystals) which
change properties in the presence of VOCs (Arshak
et al., 2004). Physico-chemical changes that occur in
the sensors during VOCs exposure are typically con-
verted to electrical signals by the transduction system.
The acquired data is further analysed and processed
andd VOC recognition is performed.
A set of features is firstly extracted and selected
from the signals obtained. (Yan et al., 2015) reviewed
many feature extraction methods, including features
extraction from the original response curves, curve
fitting parameters and transform domains. As con-
clusion, the authors stated that the best method de-
pends on the situation. Nevertheless, some advice
was given: for steady-state features, normalization
models are the best option; transient features, such as
derivatives and integrals of original response curves,
have more information than steady-state features (Yan
et al., 2015).
After features selection, advanced computational
techniques can be applied. Examples of linear clas-
sification methods are principal component analysis
(PCA), linear discriminant analysis (LDA), functional
discriminant analysis (FDA), partial least square dis-
36
Santos, G., Alves, C., Pádua, A., Palma, S., Gamboa, H. and Roque, A.
An Optimized E-nose for Efficient Volatile Sensing and Discrimination.
DOI: 10.5220/0007390700360046
In Proceedings of the 12th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2019), pages 36-46
ISBN: 978-989-758-353-7
Copyright
c
2019 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

criminant analysis (PLS-DA) and cluster analysis
(CA); and examples of non-linear models include
fuzzy logic and artificial neural networks (ANN)
(Scott et al., 2007). Some articles reported the use
of Support Vector Machine (SVM) for data acquired
by E-noses, namely in classification problems e.g.
to evaluate the quality of olive oil (Ordukaya and
Karlik, 2017), to recognize fruit odors (Uc¸ar and
¨
Ozalp, 2017), and to discriminate alcoholic and non-
alcoholic beers (Ghasemi-Varnamkhasti et al., 2015).
SVM was also applied to address regression prob-
lems, such as the prediction of the concentration of
formaldehyde (Xu et al., 2016).
Our research group has developed a new class
of biomaterials for gas sensing. The sensing gels
are composed of liquid crystal (LC) droplets self-
assembled in the presence of ionic liquid, dispersed
inside a biopolymeric matrix. These materials pos-
sess enormous versatility and have unique stimuli-
responsive properties, changing their optical and elec-
trical properties, when interacting with VOCs (Hus-
sain et al., 2017). In order to explore the applica-
tion of these materials in gas sensing, different E-
nose versions were designed and assembled by our
research group (P
´
adua et al., 2018b). After process-
ing and analysing signals obtained from the interac-
tion of VOCs with the sensing materials VOCs detec-
tion and identification can be made. The final goal is
to achieve an accurate, miniaturized and user-friendly
device that could run sample analysis in an automated
way.
In the past, it was shown that 3 different composi-
tions of biomaterials could accurately distinguish 11
VOCs, as seen by PCA (Hussain et al., 2017). In this
work an optimized E-nose device combined with so-
phisticated signal processing methods, aimed to show
that a single biomaterial sensor could automatically
classify 11 different VOCs.
2 THE OPTIMIZED E-NOSE
DEVICE
The presented E-nose explores the optical properties
of the sensing films. Therefore, a light source and a
light detector are required, separated by two crossed
polarizers and a sensing film. Without sensing film,
light does not reach the light detectors due the pres-
ence of crossed polarizers. When the sensing film is
present, its optical properties rotate polarized light al-
lowing it to reach the phototectors. This is the de-
tection principle of the E-nose V3. Previous E-nose
versions used handmade solder boards with through-
hole components, which would usually led to align-
ment problems between the light emitters and the
light detectors. Those versions used LDRs (Light
Dependent Resistors) as light detectors. Present ver-
sion uses PCBs (Printed Circuit Boards) with SMD
(Surface Mount Devices) components combined with
tailor-made 3D printed structures to guarantee proper
alignment. Also, the LDRs were replaced by photo-
diodes which are reported to be more sensitive. These
changes allowed to assemble an improved version
with increased viability.
The E-nose and its operation method are shown in
Figure 1(a) and 1(b), respectively. The apparatus is
composed of a delivery system, a detection system,
and an acquisition and control system. All presented
CAD images were designed using Fusion 360 by Au-
todesk and printed with PLA (polylactid acid) using
an Ultimaker 3.
2.1 Delivery System
The delivery system is composed by two air pumps,
the sample chamber and the relays (see Figure 1(a)).
The sample chamber is inline with one of the
pumps, being called the exposure pump, before con-
necting to the detection chamber. This allows the air
containing VOCs in the sample chamber to be carried
into the detection chamber. The other pump, the re-
covery pump, connects directly to the detection cham-
ber as its purpose is to restore the initial conditions in
this last. Both pumps use ambient air.
In a hierarchical order, the control system informs
the relays when and which pump should they power
on, then the pump either carry the VOCs to the detec-
tion chamber or renew the air inside it, depending on
which pump was powered. By switching the activa-
tion of the pumps, the VOC exposure/recovery cycles
are generated.
2.2 Detection System
The detection chamber is composed of an external
chamber with a lid, and an internal structure. Inside
this last, an optical detection system serves as a trans-
ducer to the sensors that change their optical proper-
ties on Polarised lightin contact with VOCs.
2.2.1 External Chamber
The 3D drawings of the external chamber with a lid
are shown in Figure 2(a).
The main goal of the external detection chamber
(see Figure 2(b)) is to apply pressure on the glass
chamber (part of the internal chamber), compress-
ing it between the EPDM (ethylene propylene diene
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
37

Figure 1: (a) Schematic of E-nose V3, composed by (i) Delivery system, (ii) Detection system, and (iii) Acquisition and
control system. (b) Flow of instructions given by the embedded system.
Figure 2: (a) 3D drawing of the external detection cham-
ber. (b) Printed external detection chamber, enclosing the
printed internal structure and respective connections. Lid is
open for illustration purpose.
monomer) rubber strip of the lid and the films support.
This compression is enough to close the chamber her-
metically, for the pressures being applied in the sys-
tem. Besides, it also increases the sensor robustness
and isolates the light sensors from ambient light.
2.2.2 Internal Structure
The internal structure (see 3D drawings in Figure
3) serve as support to the PCBs, the polarizing fil-
ters and the sensing films support. It has two slots
to insert two PCBs: one that has an emission cir-
cuit with LEDs, and other that has a detection circuit
based on photodiodes. These PCBs are symmetrically
aligned in front of each other. As such, each LED
is placed exactly in front of its corresponding photo-
diode. Crossed polarizers are placed in between the
PCBs, filtrating the light that is not polarized by the
sensing probes. Moreover, the light guiders prevent
interference of the light from other neighbor LEDs on
the results of each individual LED/photodiode pair.
Figure 3(e) is a picture of the fully assembled internal
structure.
Emission Circuit
The emission circuit is composed by six LEDs,
wired in parallel with the voltage source. It is a basic
LED circuit where the current is, theoretically, evenly
distributed through all the LEDs (i.e. all LEDs emit
light with equal intensity). Also, a transistor in the
common ground line acts as a switch to control LEDs.
The LEDs circuit schematic can be found in Fig-
ure 4.
Detection Circuit
Photodiodes are one of the most popular sensor
types for light-based measurements. In the present
case, the analyte detection relies on photodiodes for
precise light intensity measurement. The photodiodes
circuit schematics can be found in Figure 5.
Photodiodes generate a current proportional to the
photons that strike their active area. This current
needs to be converted into voltage, since the micro-
controller (Arduino Due) analog input pins read volt-
age variations, and not current variations. Therefore,
a circuit using a current-to-voltage converter (in this
case a transimpedance amplifier) was implemented.
Here, there is a need to point out the importance of
V
DIV
(Figure 5) voltage. The value of V
DIV
is estab-
lished as a compromise between the output voltage
window and the reverse polarization of the photodi-
odes. The maximum output voltage is defined di-
rectly by V
DIV
through the opamp (Operational Am-
plifier) positive input, and the minimum is 0V, being
BIODEVICES 2019 - 12th International Conference on Biomedical Electronics and Devices
38

Figure 3: Main internal structure (light grey) and sensing films support (dark grey). The sensing films support is removable to
ease the sensing films placement. (a) 3D drawing of the symmetrical internal structure. (b) Cross-section revealing one of the
PCBs slot and respective light-guiders for the six LED/photodiode pairs. (c) Middle cross-section revealing the sensing films.
(d) Middle cross-section revealing how the glass chamber fits in the sensing films support. (e) Assembled internal structure
with respective components and connections.
Figure 4: Schematic of the LED´s circuit designed using
Eagle software. Only one LED is represented for illustra-
tion purposes.
the lowest value read by the arduino analog inputs
(3.3 - 0V range). On the other hand, photodiode´s
reverse polarization voltage is the result of V
DD
minus
the opamp negative input voltage (or V
DIV
, since both
inputs share the same voltage). Increasing V
DIV
will
increase the output window but negatively impact the
performance of the photodiodes, as they benefit from
higher polarization voltages (become more sensitive
to light). Opposite behaviour occurs if V
DIV
is de-
creased.
Figure 5: Schematic of photodiode´s circuit designed using
the software Eagle. (a) Generic voltage divider to gener-
ate V
DIV
. (b) Schematic of photodiode´s reverse polarizing
circuit. One replica per photodiode.
A viable way to also control the system sensitiv-
ity to light is by resizing the resistor in the negative
feedback loop, according to Equation 1 (Ohm Law).
V
DROP
is the voltage drop at the output, I
PD
the cur-
rent generated by the photodiode (PD) and R the re-
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
39

sistor value. The resizing of this resistor can be seen
as an adjusting scale for the output signal, avoiding
signal saturation.
V
DROP
= R.I
PD
(1)
To confirm that the detection circuit is properly
working without signal loss, either through signal sat-
uration or insufficient sensitivity of the photodiodes
to the lowest amounts of photons, one more test was
performed.
A new setup (Figure 6) was assembled, including
polarizing filters, and where a single LED/photodiode
pair had the ability to rotate over the other. The ex-
periment aimed to confirm that the photodiodes were
sensitive enough to detect small light variations, spe-
cially close to the darkest point (i.e. when polariz-
ers are crossed at 90°) corresponding to the maximum
output voltage.
2.3 Acquisition and Control System
The instructions to control the device are given by an
embedded system (Raspberry Pi 3 Model B) and exe-
cuted by a microcontroller (Arduino Due). When the
experiment begins, the LEDs are turned on, and the
pumps work alternately while the photodiodes receive
the signal and the microcontroler acquires them at a
90Hz sampling rate. At the end of the experiment, the
LEDs are turned off and data is stored in a text file for
later analysis (see Figure 1(b)). In terms of software,
the latest version of the open access language Python
(Python 3.6) was used with Pymata library, that al-
lows the communication between the embedded sys-
tem and the microcontroller.
3 CLASSIFICATION TOOLS
Together with the optimization of the device, anal-
ysis tools based on Python libraries (SciPy, sklearn
and novainstrumentation) were also developed. A
machine learning algorithm was trained with a set
of selected features in order to distinguish 11 VOCs.
In these tests, a single sensing film composition was
used.
Before extracting the features of the signal, the
signal was filtered using the median filter from SciPy
library, kernel size equal to 11, to remove inductive
noise caused mainly by the activation of the pumps.
Then, the signal was filtered with smooth function
from the novainstrumentation library. The E-nose
records information that indicates when the recovery
or the exposure pumps are working. This information
was used to split the signal and obtain the individual
cycles (a cycle corresponds to exposure to gas sam-
ple followed by a recovery period, in which air re-
stores the radial configuration of the LC-IL droplets,
see Figure 7. Full phenomenon is described by (Hus-
sain et al., 2017).). Every time there is an indication
that the exposure pump is on, a cycle begins and a cut
in the signal is made (Figure 8(a)).
After splitting the signal in individual cycles,
those were normalised by Equation 2.
signal
normalized
=
∑
n
value
n
− min(signal)
max(signal) − min(signal)
(2)
A time domain analysis was performed, combin-
ing two types of features: morphological features and
parameters of curve fitting models.
A typical output signal, regarding an individual
cycle, starts to rise when the sensors are being ex-
posed to VOCs, until a maximum is reached which
corresponds to the darkest state. On the other hand,
the output signal decreases as the VOCs leave the
chamber, restoring the radial configuration of the
droplets and corresponding to the brightest state. This
leads to a typical shape in the derivatives. Figure 8(b)
represents a single cycle and its respective derivatives.
The first derivative has two prominent peaks, with
their position corresponding to the inflection points
in the rise (positive peak) and the decrease (nega-
tive peak) of the signal. The second derivative has
four prominent peaks; two positives when the signal
is concave up and two negatives where it is concave
down. The relative position of these peaks in relation
to the beginning of the cycle, as well at its amplitudes,
correspond to the first thirteen extracted features.
A suggestion made by R. Dutta and col-
leagues (Dutta and Dutta, 2006) was followed. Skew-
ness and kurtosis which are measures of the asymme-
try of the probability distribution of a random vari-
able around its mean were extracted. Although the
signal is not a probability density function, extracting
these features helped increase the accuracy of their
electronic nose classifier. Hence, they were included
to see if could also be fitted for the current problem.
For the fitting model, the function considered was
proposed by Holmberg, M. et al. (Equation 3) where
θ
1
corresponds to signal amplitude, θ
2
to the rising
slope, θ
3
to the position where the signal reaches half
the rise, θ
4
to the descent slope and θ
5
to position
where the signal reaches half of the descend (Holm-
berg et al., 1998). This model is based on the logis-
tic function (Equation 4). The logistic function de-
scribes something that initially grows and that even-
tually starts to saturate and converge to a maximum
value (in this case, total darkness, that occur when the
droplets’ configuration becomes isotropic). In Equa-
BIODEVICES 2019 - 12th International Conference on Biomedical Electronics and Devices
40

Figure 6: 3D printed structures designed to verify the detection circuit. (a) Separated pieces revealing the interior of the round
chamber. (b) When together they create an isolated dark chamber, where the center point is the light-guider of the respective
LED/photodiode pair. (c) Cross-section revealing the PCB slot. (d) Printed and assembled structure as used in the test.
Figure 7: (a) Polarized Optical Microscopy image of a
droplet when it interacts with a VOC. (b) Illustration of con-
figuration changes when the VOC interacts with a droplet.
Adapted from (Hussain et al., 2017).
tion 4, parameter a corresponds to that maximum
value, b is the steepness of the curve and c indicates
when in time the curve reaches its mid point.
f (x) = θ
1
.
1
1 + e
θ
2
.(θ
3
−x)
.
1 −
1
1 + e
θ
4
.(θ
5
−x)
(3)
f (x) =
a
1 + e
b.(c−x)
(4)
The wrapper method Recursive Feature Elimina-
tion with Cross Validation (RFECV) was used for
feature selection. This algorithm recursively elimi-
nates features and in each iteration trains a classifier
to ascertain the effect of the removed feature. It stops
when a good classification score is obtained. It then
ranks feature performance as 1 if the remaining subset
of features gave a good classification result. A. Jovic
et al. stated that linear Support Vector Machines were
feasible to use with wrapper methods (Jovic et al.,
2015). Therefore, it was the learning model used.
Stratified 10-fold cross-validation was used to obtain
a classification score. In which the dataset is ran-
domly divided in ten subsets, avoiding unbalanced set
by mantaining class percentage, and using one tenth
of the set as validation set and the rest as training set.
Then the process is repeated ten times and the result
is the average of the accuracies obtained.
Accuracy, given by Equation 5, was chosen as the
scoring method, since it provides a measure of global
performance.
Accuracy =
T P + T N
T P + T N + FP + FN
(5)
It gives the number of correctly classified exam-
ples: the sum of True Positives (TP) and True Nega-
tives (TN) divided by the total number of examples
(TP, TN, False Positives (FP) and False Negatives
(FN)).
Finally, a Support Vector Machine classifier was
chosen as the classification model. The standard
SVM, illustrated in Figure 9, is a binary classifier. It
finds a hyperplane that best separates the two classes
and creates a decision boundary. The examples of the
classes that are closer to the hyperplane are called the
support vectors. The hyperplane can be described by
the set of points that satisfy w
T
x = 0, w being the
vector perpendicular to the hyperplane. The points
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
41
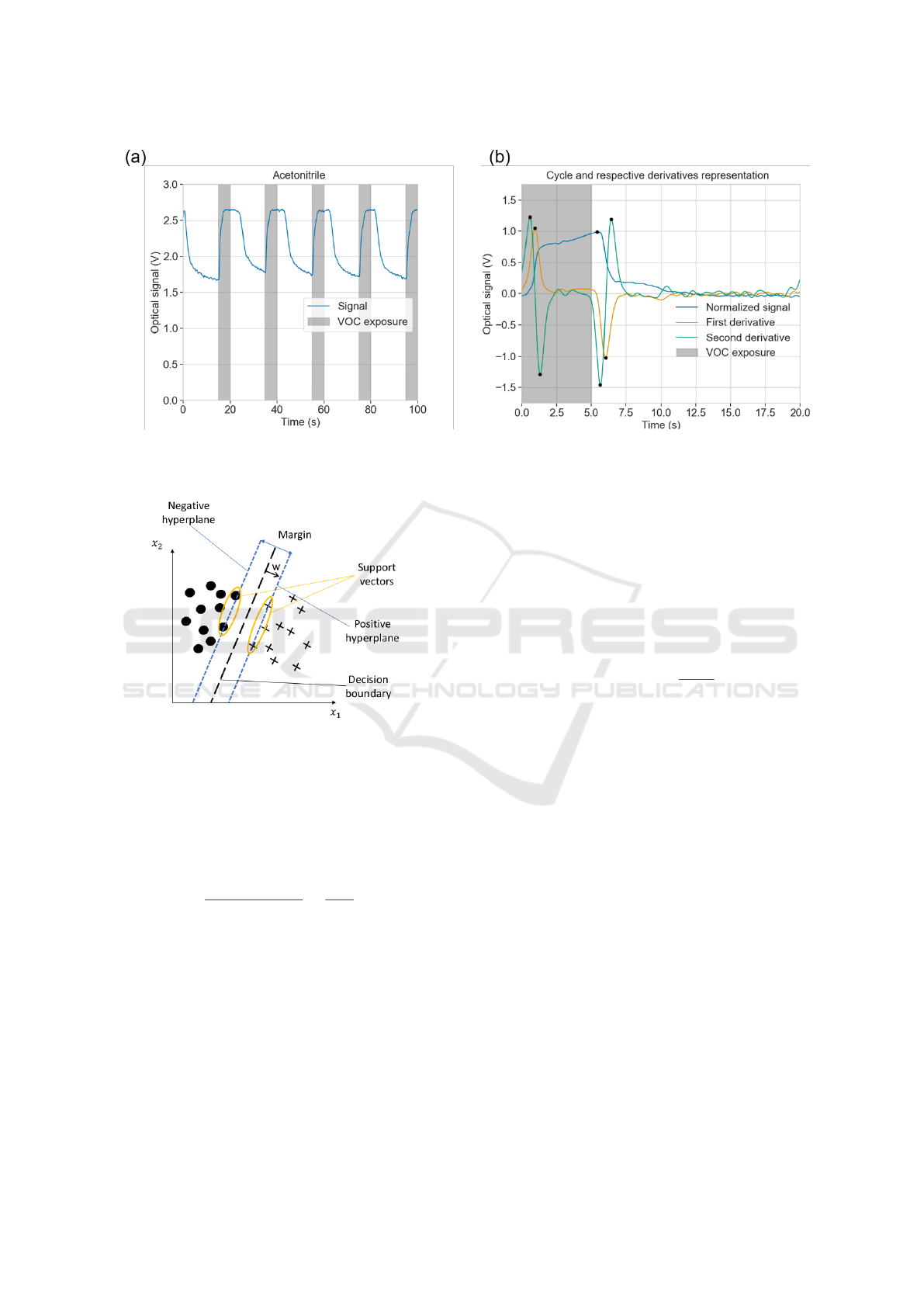
Figure 8: (a) Representation of some cycles with their exposure and recovery times. (b) Example of an individual cycle and
its first and second derivatives. Black dots mark the peaks of interest and the darkened part of the plot correspond to the
exposure time.
Figure 9: Schematic representation of Support Vector Ma-
chines. Adapted from (Sebastian, 2015).
that satisfy w
o
+ w
T
x
pos
= 1 form the positive hyper-
plane, and the ones that satisfy w
o
+ w
T
x
neg
= −1
form the negative hyperplane. By subtracting those
expressions and normalizing by the length of the vec-
tor w, Equation 6 is obtained.
w
T
(x
pos
− x
neg
)
||w||
=
2
||w||
(6)
The left term of Equation 6 is the distance between
hyperplanes, the objective is to maximize it.
The performance of the SVM classifier depends
on the kernel, the kernel parameter γ (if kernel is non-
linear) and the parameter C.
The variable C influences the number of support
vectors used by the model, a larger C means a higher
cost for misclassification (Sebastian, 2015).
The kernel is used to transform the original fea-
tures and project them onto a higher dimensional
space via a mapping function (Sebastian, 2015). The
kernels available in the scikit-learn library were:
polynomial kernel, radial basis function kernel, and
sigmoid kernel.
The radial basis function kernel, the one used in
this work, is defined by Equation 7. The parameter γ
influences the reach of the training examples, a higher
γ makes the boundaries closer to the examples. By
default, its value is 1/nr
samples
.
K(x, x
0
) = e
−
||x−x
0
||
2
2σ
2
= e
(
−γ||x−x
0
||
2
)
(7)
Parameters C and γ should be tuned together. For
that, is possible to use GridSearchCV from scikit-
learn library that performs a grid search with cross
validation to choose the best parameters. This func-
tion was used with: values 0.1, 1, 10 and 100 for C;
values 0.01, 0.8, 1 and 10 for γ; and the kernels avail-
able, already mentioned. Other parameters from the
SVC scikit-learn library used the default values given.
4 RESULTS
4.1 Features of E-nose V3
E-nose V3 is an optimized version of E-nose V0, the
first prototype. The system was improved, having a
faster and cheaper data acquisition system composed
by an Arduino microcontroller and a Raspberry Pi
(under 100 euros) as opposed to the previous system
composed by a computer and a NI myDAQ (above
600 euros). Now it possess a single interface making
BIODEVICES 2019 - 12th International Conference on Biomedical Electronics and Devices
42
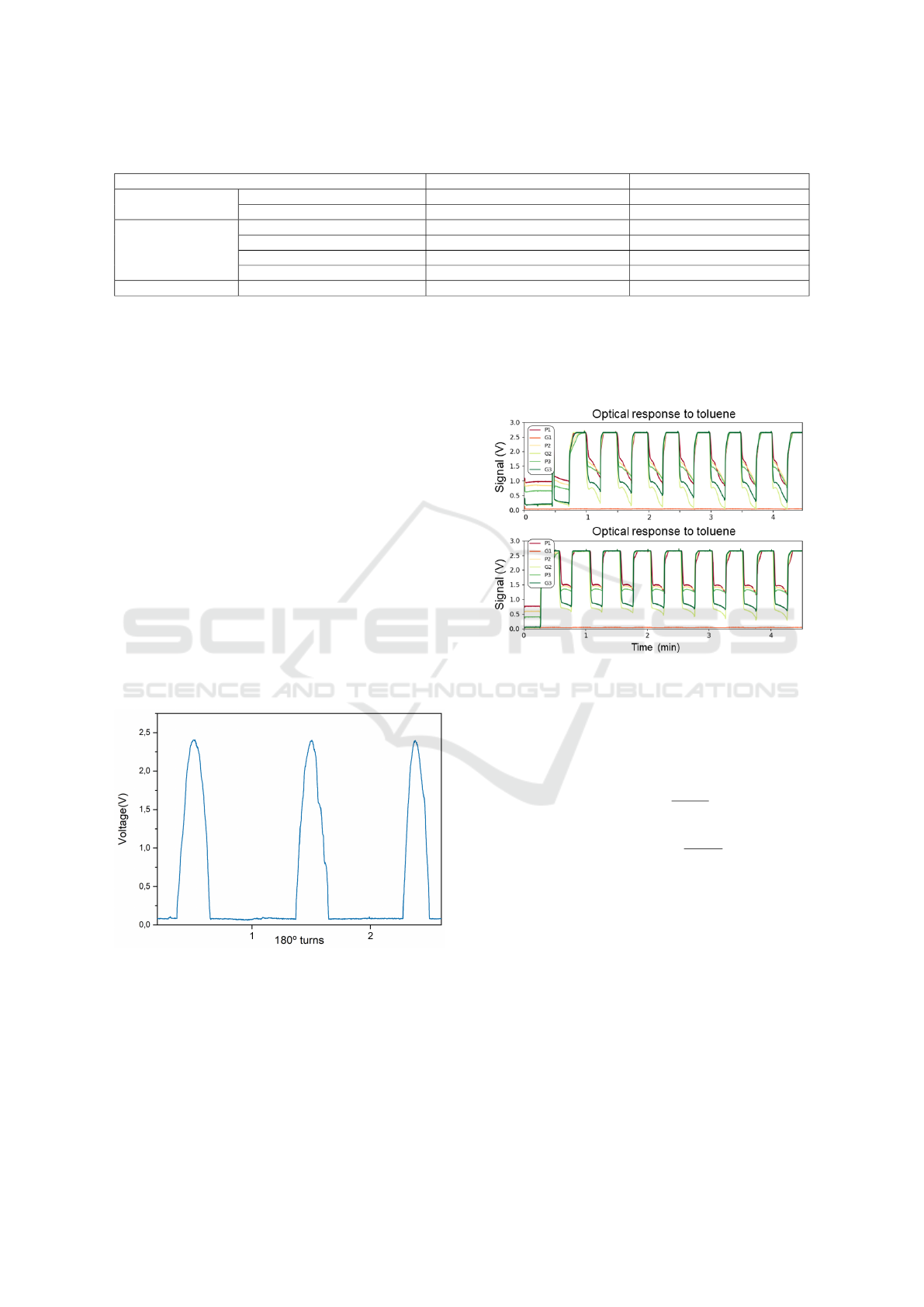
Table 1: Comparative table of different e-nose versions.
E-nose Version First version (P
´
adua et al., 2018b) Latest version (present work)
Transduction system
Sampling rate (Hz) 10 90
Data acquisition elements NI myDAQ + Computer Arduino Due + Raspberry Pi3
Detection chamber
Detection sensors Photoresistors Photodiodes
Array capacity (nr sensors)
4 6
Stability and scaling (scale 1-5)* 1 4
Chamber volume 2L 28mL
Delivery system Analysis frequency 0.37 cycles/min 3 cycles/min
* Stability and scaling scale:
1: Open and close the chamber was not practical. Chamber with flow losses.
2: Robust and more easy-to-use chamber. Difficult to change sensing films between experiments. Flow losses verified.
3: Easy to change sensing films. Hermetic. PCBs with through-hole technology. components.
4: Easy to change sensing films. Hermetic.. PCBs with surface-mount technology components. Has slots to insert the PCBs and support for sensing
films. Has light guiders for LEDs.
it more user-friendly (P
´
adua et al., 2018a). Regarding
the detection chamber, the number of sensors was in-
creased and the chamber became more robust, stable
and hermetic. Moreover, improvements in the deliv-
ery system allowed a 10 times faster analysis, com-
paring to the initial prototype.
Regarding the delivery system, pumps flow rate
is 3 L/min, the flow rate at chamber entrance is 1.8
L/min for the exposure circuit, and 2.5 L/min for the
recovery circuit. Table 1 summarizes the main differ-
ences between E-nose versions.
Considering the implementation of a new detector
system, the results for the validation of the photodi-
odes are represented in Figure 10. It demonstrates
that the photodiodes are suitable for the application
and that no signal is lost at any given light intensity.
Figure 10: Proof of concept of the photodiodes sensitivity.
The experiment consisted in the signal acquisition during
three 180 degree turns. The absence of a saturation line at
the maximum values approves the detection system.
Signals were acquired (Figure 11) to test the new
version of the E-nose. A preliminary experiment was
made to measure signal to noise ratio. Five sens-
ing films were sequentially exposed to two different
VOCs (acetone and toluene), the remaining position
Figure 11: Signals acquired to test the new E-nose version.
Position G1 corresponds to the control measurement.
of the array was used as a control measurement.
The signal to noise ratio (SNR) was calculated by
Equation 8. The result was approximately 50 dB.
SNR
dB
= 10log
P
signal
P
noise
= 20log
A
signal
A
noise
2
!
(8)
4.2 Classification Results
Three sensing gels, with the exact same composition,
were exposed to a sequence of 11 VOCs (hexane, hep-
tane, toluene, chloroform, dichloromethane, acetoni-
trile, acetone, diethyl ether, ethyl acetate, ethanol and
methanol) in cycles of 5 seconds of exposure and 15
seconds of recovery. 15 mL of VOC were placed in
the sample chamber and heated at 37°C durign 15
minutes before beggining the experiment to ensure
headspace saturation. Each sample interacted with
the sensors in a total of 15 minutes. The remaining
positions of the electronic nose were used as control
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
43

measurements. After the sequence was completed,
sensing gels were stored. Two months later, the same
procedure was repeated with the same sensing gels.
The first cycles of each volatile (the first 5 for the first
time the gels were exposed and the first 6 for the sec-
ond time) were not considered due to the presence of
noise, caused by the opening of the valve that allows
VOC intake. This resulted in a total of approximately
2600 cycles for analysis (236 cycles per VOC).
The features were extracted, however, the curve
fitting algorithm was not able to converge into a solu-
tion for the parameters of the approach of the article
(Equation 3). In order to fit a model to the signal,
it was necessary to simplify the approach. The solu-
tion was to divide the signal in two parts (from cycle
beginning until its maximum and from the maximum
until its end) and apply the logistic function (Equation
4) to each part of the signal (Figure 12).
Figure 12: Estimated curves for (a), the first part of the cy-
cle and for (b), the second part. The red point marks the
coordinates where the signal bisects with the value of half
the rise/descend.
After obtaining a set of features, RFECV algo-
rithm used a quarter of the data and the rest remained
untouched for validation purposes. The rank of the
features is shown in Table 2. Features ranked as 1 are
the ones with best performance, and therefore were
the ones selected.
When tuning the classifier parameters, the best
performance was obtained with the radial basis func-
tion kernel and parameters C: 100 and γ : 0.1.
Finally, to validate the classifier with unseen data,
the validation set was used. The classifier managed
to obtain a global accuracy of 94.6% with a standard
deviation of 0.9%. Figure 13 shows the correspondent
normalized confusion matrix.
Figure 13: Confusion matrix obtained for the classification
of the 11 VOCs. Accuracy of 94.6% (± 0.9%).
5 CONCLUSION AND FUTURE
WORK
E-nose V3 is a tailor-made device using as gas sen-
sors an innovative class of biomaterials which opti-
cal properties vary in the presence of VOCs (Hus-
sain et al., 2017). This new optimized device, to-
gether with the appropriate use of signal processing
tools, allowed the accurate classification of 11 VOCs
from different chemical classes using sensing films
with the exact same composition. The results show
that our sensor possess advantages over other types
of sensors such as MOS-sensors or conducting poly-
mer sensors, as these normally have limited selectiv-
ity towards VOC classes. In addition, our materials
and sensing device operate at ambient conditions as
opposed to MOS (300°C)(Wilson, 2012). In addition,
used sensing materials are biodegradable and reusable
with the potential to recycle its individual compo-
nents, having a lower carbon footprint.
The detection chamber is hand-size and hermetic
for the pressures being applied. Its low volume allows
the use of short cycle periods, and consequently a fast
analysis. The advantages of using photodiodes is that
they are more sensible and have a quicker response
time than photoresistors.
The transduction system based on Arduino and
Raspberry Pi constitute a low-cost and independent
BIODEVICES 2019 - 12th International Conference on Biomedical Electronics and Devices
44
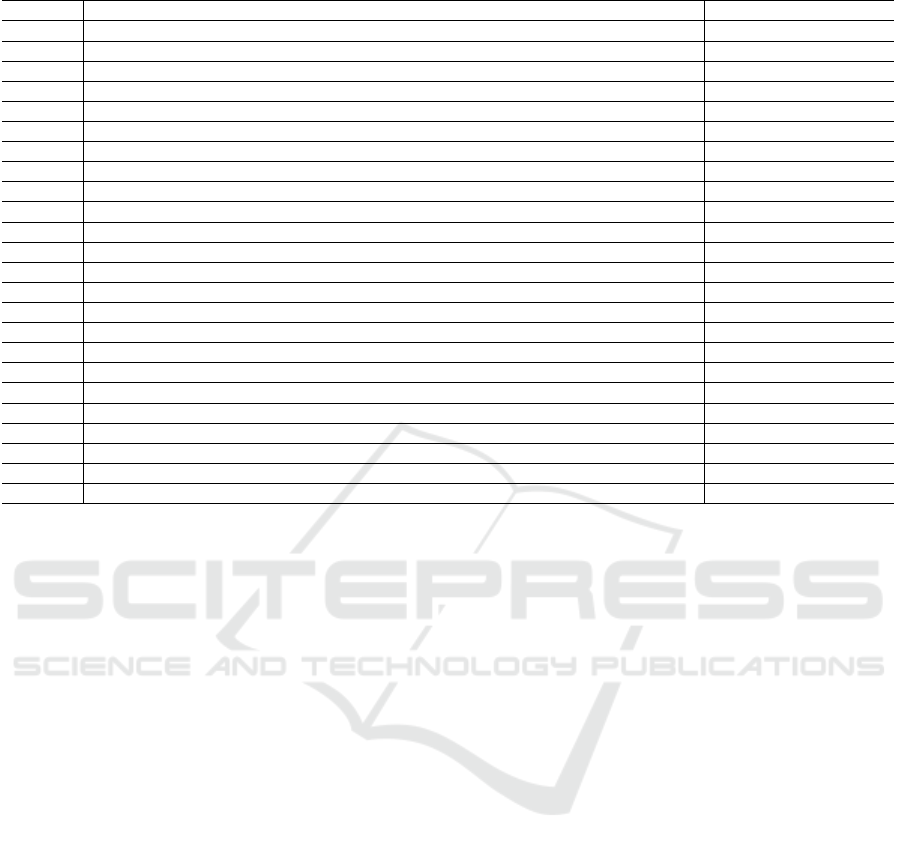
Table 2: Ranking of the selected features by RFECV with 10-fold cross validation.
Number Description 10-fold validation score
1 Time to reach the maximum value of the cycle 7
2 Time to reach the maximum of the first derivative of the signal 4
3 Time to reach the minimum of the first derivative of the signal 1
4 Maximum value of the first derivative of the signal 3
5 Minimum value of the first derivative of the signal 2
6 Time to reach the maximum of the first half of the second derivative of the signal 1
7 Time to reach the minimum of the first half of the second derivative of the signal 10
8 Maximum value of the first half of the second derivative of the signal 1
9 Minimum value of the first half of the second derivative of the signal 1
10 Time to reach the maximum of the second half of the second derivative of the signal 11
11 Time to reach the minimum of the second half of the second derivative of the signal 9
12 Maximum value of the second half of the second derivative of the signal 1
13 Minimum value of the second half of the second derivative of the signal 1
14 Area under the signal 1
15 Skewness 1
16 Kurtosis 1
17 Parameter a from the first half of the signal 13
18 Parameter b from the first half of the signal 1
19 Parameter c from the first half of the signal 1
20 Error associated with the fit from the first half of the signal 5
21 Parameter a from the second half of the signal 12
22 Parameter b from the second half of the signal 1
23 Parameter c from the second half of the signal 6
24 Error associated with the fit from the second half of the signal 8
unit. The sampling rate of 90 Hz was enough to guar-
antee informative signal acquisition.
After data collection, two different feature extrac-
tion methods were used: features extraction from the
original response curves and curve fitting parameters.
The most informative features extracted from the sig-
nals were identified and reported in bold in Table 2.
Applying those features, the accuracy rate for SVM
was 94.6% ± 0.9%. Future work should include com-
parison of these results with other machine learning
classification techniques.
The assembled system was able to distinguish the
11 tested VOCs. Although the identification of the
VOCs was not made in real time, a step towards mak-
ing this E-nose a device that is able to make automatic
classification was taken.
The E-nose V3 device is robust and user-friendly.
We are aware that there is still room for improvement.
For example, in the future the detection system could
benefit from the use of variable resistors, or multiple
resistors per photodiode, enabling auto scaling or sim-
ply by easing the actual scaling where multiple PCBs
assembled with different resistor values, are chosen to
best suit the sensing probes in use based on the out-
put voltage. Also, a longer array of LED/photodiodes
pairs would allow for a wider combination of different
sensing probes, or a larger number of replicas, which
in both cases have a positive impact in the VOCs pre-
diction algorithm.
Using the optimized e-nose version, the necessary
studies to evaluate more complex scenarios are pos-
sible, such as testing concentration limits, the effects
of room conditions in the material and classification
of complex samples where many different VOCs are
mixed together.
ACKNOWLEDGEMENTS
This project has received funding from the European
Research Council (ERC) under the EU Horizon 2020
research and innovation programme (grant agree-
ment No. SCENT-ERC-2014-STG-639123). This
work was supported by the Applied Molecular Bio-
sciences Unit-UCIBIO which is financed by national
funds from FCT/MCTES (UID/Multi/04378/2013)
and co-financed by ERDF under the PT2020 Partner-
ship Agreement (POCI-01-0145-FEDER-007728).
The authors thank FCT/MEC for the research fellow-
ship PD/BD/105752/2014 for A.P.
REFERENCES
Arshak, K., Moore, E., Lyons, G., Harris, J., and Clifford,
S. (2004). A review of gas sensors employed in elec-
tronic nose applications. Sensor Review, 24(2):181–
198.
Barbosa, A. J., Oliveira, A. R., and Roque, A. C. A. (2018).
An Optimized E-nose for Efficient Volatile Sensing and Discrimination
45

Protein- and peptide-based biosensors in artificial ol-
faction. Trends in Biotechnology, 36:1244–1258.
Dutta, R. and Dutta, R. (2006). Intelligent Bayes Classi-
fier (IBC) for ENT infection classification in hospital
environment. BioMedical Engineering Online, 5.
Ghasemi-Varnamkhasti, M., Mohtasebi, S. S., Siadat, M.,
Ahmadi, H., and Razavi, S. H. (2015). Research pa-
per: From simple classification methods to machine
learning for the binary discrimination of beers using
electronic nose data. Engineering in Agriculture, En-
vironment and Food, 8:44 – 51.
He, Q., Yan, J., Shen, Y., Bi, Y., Ye, G., Tian, F., and
Wang, Z. (2012). Classification of Electronic Nose
Data in Wound Infection Detection Based on PSO-
SVM Combined with Wavelet Transform. Intelligent
Automation and Soft Computing, 18(7):967–979.
Holmberg, M., Gustafsson, F., H
¨
ornsten, E. G., Winquist,
F., Nilsson, L. E., Ljung, L., and Lundstr
¨
om, I.
(1998). Bacteria classification based on feature ex-
traction from sensor data. Biotechnology Techniques,
12(4):319–324.
Hussain, A., Semeano, A. T. S., Palma, S. I. C. J., Pina,
A. S., Almeida, J., Medrado, B. F., P
´
adua, A. C.
C. S., Carvalho, A. L., Dion
´
ısio, M., Li, R. W. C.,
Gamboa, H., Ulijn, R. V., Gruber, J., and Roque,
A. C. A. (2017). Tunable gas sensing gels by co-
operative assembly. Advanced Functional Materials,
1700803:1–9. ¡p¿n/a¡/p¿.
Jovic, A., Brkic, K., and Bogunovic, N. (2015). A re-
view of feature selection methods with applications.
In 2015 38th International Convention on Information
and Communication Technology, Electronics and Mi-
croelectronics (MIPRO), pages 1200–1205.
Ordukaya, E. and Karlik, B. (2017). Quality control of olive
oils using machine learning and electronic nose. Jour-
nal of Food Quality, Vol 2017 (2017).
P
´
adua, A. C., Os
´
orio, D., Rodrigues, J., Santos, G., Porteira,
A., Palma, S., Roque, A., and Gamboa, H. (2018a).
Scalable and easy-to-use system architecture for elec-
tronic noses. In Proceedings of the 11th International
Joint Conference on Biomedical Engineering Systems
and Technologies, pages 179–186.
P
´
adua, A. C., Palma, S., Gruber, J., Gamboa, H., and
Roque, A. C. A. (2018b). Design and evolution of
an opto-electronic device for vocs detection. Pro-
ceedings of the 11th International Joint Conference on
Biomedical Engineering Systems and Technologies,
pages 48–55.
R
¨
ock, F., Barsan, N., and Weimar, U. (2008). Electronic
nose: Current status and future trends. Chemical Re-
views, 108(2):705 – 725.
Scott, S. M., James, D., and Ali, Z. (2007). Data analy-
sis for electronic nose systems. Microchimica Acta,
156(3/4):183 – 207.
Sebastian, R. (2015). Python Machine Learning. Packt Pub-
lishing Ltd.
Uc¸ar, A. and
¨
Ozalp, R. (2017). Efficient android elec-
tronic nose design for recognition and perception of
fruit odors using kernel extreme learning machines.
Chemometrics and Intelligent Laboratory Systems,
166:69 – 80.
Wasilewski, T., Ge¸bicki, J., and Kamysz, W. (2017).
Bioelectronic nose: Current status and perspectives.
Biosensors and Bioelectronics, 87:480–494.
Wilson, A. D. (2012). Review of electronic-nose tech-
nologies and algorithms to detect hazardous chemi-
cals in the environment. Procedia Technology, 1(First
World Conference on Innovation and Computer Sci-
ences (INSODE 2011)):453 – 463.
Xu, L., He, J., Duan, S., Wu, X., and Wang, Q. (2016).
Comparison of machine learning algorithms for con-
centration detection and prediction of formaldehyde
based on electronic nose. Sensor Review, 36(2):207.
Yan, J., Xiuzhen, G., Shukai, D., Pengfei, J., Lidan, W.,
Chao, P., and Songlin, Z. (2015). Electronic nose fea-
ture extraction methods: A review. Sensors, Vol 15,
Iss 11, Pp 27804-27831 (2015), (11):27804.
BIODEVICES 2019 - 12th International Conference on Biomedical Electronics and Devices
46
