Prediction of Acute Kidney Injury in Cardiac Surgery Patients:
Interpretation using Local Interpretable Model-agnostic Explanations
Harry Freitas da Cruz, Frederic Schneider and Matthieu-P. Schapranow
Hasso Plattner Institute, Digital Health Center, Rudolf-Breitscheid-Straße 187, Potsdam, Germany
Keywords:
Clinical Prediction Models, Supervised Learning, Interpretability, Nephrology, Acute Kidney Injury.
Abstract:
Acute kidney injury is a common complication of patients who undergo cardiac surgery and is associated with
additional risk of mortality. Being able to predict its post-surgical onset may help clinicians to better target
interventions and devise appropriate care plans in advance. Existing predictive models either target general
intensive care populations and/or are based on traditional logistic regression approaches. In this paper, we
apply decision trees and gradient-boosted decision trees to a cohort of surgical heart patients of the MIMIC-III
critical care database and utilize the locally interpretable model agnostic approach to provide interpretability
for the otherwise opaque machine learning algorithms employed. We find that while gradient-boosted decision
trees performed better than baseline (logistic regression), the interpretability approach used sheds light on
potential biases that may hinder adoption in practice. We highlight the importance of providing explanations
of the predictions to allow scrutiny of the models by medical experts.
1 INTRODUCTION
Heart patients often have to undergo surgical inter-
ventions during the course of the disease. Particularly
surgeries utilizing a cardiopulmonary bypass place a
significant burden on the patient’s kidneys and may
lead to Acute Kidney Injury (AKI). This condition
occurs in up to 30% of patients following cardio-
surgical treatment and is associated with complica-
tions such as sepsis, an increase of in-hospital and
long-term morbidity, and generally poor patient out-
comes (O’Neal, Jason and others, 2016).
Identifying patients at high risk for developing
AKI before the surgical intervention can assist care
providers in adopting targeted renal-protective strate-
gies, such as increasing renal blood flow and avoid-
ance of nephrotoxins (Rosner and Okusa, 2006).
While there is little consensus on drugs that can ef-
fectively prevent AKI onset in heart patients, early
detection can furthermore be of value for preoperative
patient management and clinical trial recruitment (Ng
et al., 2014). Therefore, a number of studies have
been targeted at developing appropriate risk scores
and Clinical Prediction Models (CPM).
Previous work dealing with the task of predict-
ing heart surgery-associated AKI take into account
biomarkers and/or clinical data before, during and af-
ter the surgical intervention. Particularly concerning
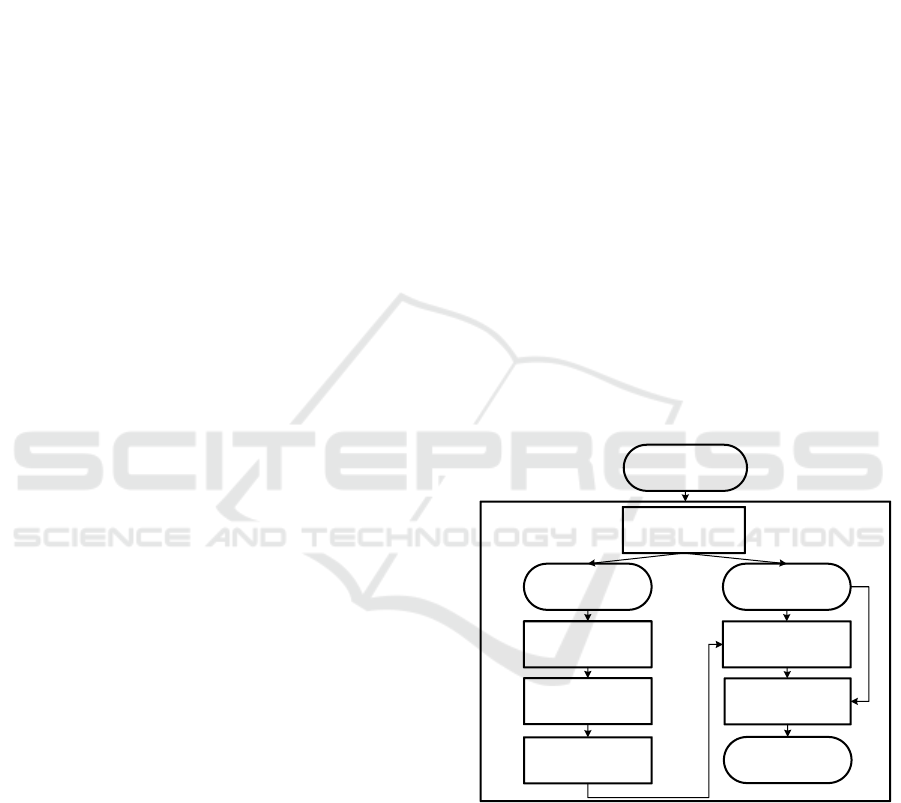
LIME
Preprocessing
& Data split
MIMIC-III
database
Training
Dataset
Validation
Dataset
Hyperparameter
tuning
Cross-validation
Model training
(LR, DT, GBDT
)
Trained model
(LR, DT, GBDT)
Local
Explanations
Feature selection
Submodular Pick
Clinical Prediction
Model
Figure 1: Graphical abstract depicting the set-up of the ex-
periment in this paper as a Fundamental Modeling Concepts
block diagram.
biomarker-based approaches, measurements of inter-
est are usually taken after the surgery (Sawhney et al.,
2015), thus posing barriers for use prior to the inter-
vention. In this paper, we derive a CPM which utilizes
only preoperative variables in order to predict the on-
set of AKI and compare our results to models which
sought to perform the same task. The available mod-
els published to date tend to employ preoperative vari-
ables are often based on linear approaches. While the
380
Freitas da Cruz, H., Schneider, F. and Schapranow, M.
Prediction of Acute Kidney Injury in Cardiac Surgery Patients: Interpretation using Local Interpretable Model-agnostic Explanations.
DOI: 10.5220/0007399203800387
In Proceedings of the 12th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2019), pages 380-387
ISBN: 978-989-758-353-7
Copyright
c
2019 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

results thus achieved are satisfactory, we hypothesize
that is possible to improve them even further using
machine learning approaches.
Indeed, many complex machine learning and pre-
diction modeling techniques, such as neural networks
and ensemble methods, have been shown to out-
perform linear modeling approaches in terms of ac-
curacy and precision, but they typically lack inter-
pretability. This trade-off is a critical hindering fac-
tor for the adoption of such models in high-stakes
domains (Valdes et al., 2016; Letham et al., 2015;
Katuwal and Chen, 2016). Therefore, we employ the
interpretability method Local Model-agnostic Expla-
nations (LIME) to provide intelligible explanations
for the prediction results (Ribeiro et al., 2016).
Our CPM is based on pre-surgery clinical data ob-
tained from the MIMIC-III clinical care database. For
this task, we compare the performance of three dif-
ferent prediction algorithms: a more readily intelligi-
ble Decision Tree (DT) model, a blackbox-type model
Gradient Boosted Decision Tree (GBDT) along with
a Logistic Regression (LR) baseline model. The mod-
els thus trained are internally validated on a held-out
dataset. For interpretation, we select specific classi-
fication instances to be explained by LIME. This pa-
per’s set-up is illustrated by the Fundamental Model-
ing Concepts block diagram in Figure 1.
The remainder of this work is structured as fol-
lows: Section 2 discusses the context of interpretable
classification and prediction models specifically in
medicine as well as the state-of-the-art algorithms for
clinical prediction models. Section 3 details our ap-
proach to data acquisition, preprocessing, modeling,
and use of LIME. In Section 4 we present an overview
of our model’s prediction quality to be able to, in Sec-
tion 5, relate these results to the interpretability of our
two employed models and results.
2 RELATED WORK
2.1 Cardiac Surgery-associated AKI
Research regarding AKI and its occurrence after car-
diac surgery is typically focused on detecting in-
serum and urinary biomarkers, e.g. serum creati-
nine (Latini et al., 2016; Flynn and Dawnay, 2015)
and neutrophil gelatinase-associated lipocalin. Con-
cerning prediction models using electronic health
data, an early approach has been the Cleveland
score (Thakar et al., 2004) derived from a large cohort
of open-heart surgery patients, which was followed by
the publication of the AKICS score (Palomba et al.,
2007) based on a cohort of Brazilian patients. In
a multicentric, multinational study, Mehta et al de-
veloped a score using the National Cardiac Surgery
Database of the Society of Thoracic Surgeons (STS),
achieving satisfactory results. The Simplified Re-
nal Index (SRI) strived to achieve a succinct set of
predictors, but ultimately performed worse than the
Cleveland score on a validation cohort of the Mayo
Clinic (Wijeysundera et al., 2007). Since the publica-
tion of those scores, a literature review recommended
the Cleveland score, as it is the most frequently val-
idated score (Huen and Parikh, 2012). However, the
Cleveland score presented poor discrimination met-
rics when validated in a Chinese cohort, suggesting
limited generalizability for populations not predomi-
nantly Caucasian (Jiang et al., 2017).
The models developed until now have been based
on logistic regression, with the AKICS score present-
ing the best performance upon derivation. Even con-
sidering possible overfitting effects, the Area Under
the Curve (AUC) of most models range from 0.74
to 0.84 (Huen and Parikh, 2012). By using ma-
chine learning algorithms as opposed to simple lo-
gistic regression, we achieved better discriminative
performance for preoperative AKI prediction than ex-
tant models (AUC=0.9). An overview of the results
achieved in comparison with the literature is provided
in Table 2.
2.2 Model Interpretability
Even though blackbox models may promise better
results, specifically in high-stakes domains such as
medical care, the trade-off between model perfor-
mance and intelligibility results in domain experts and
professionals favoring interpretable prediction mod-
els with verifiable outcomes over opaque, machine
learning models (Caruana et al., 2015). As such, in
addition to applying new algorithms on the problem,
we employ the LIME explainer to lend intelligibility
to the algorithms’ predictions.
Model interpretability is a research topic gaining
traction, partially due to ethical concerns and laws
regulating the use of machine learning techniques
on individual-related data (Goodman and Flaxman,
2016). The notion of interpretability, however, is not
yet well-defined and publications often present dif-
ferent characteristics and desiderata for interpretable
models. In this paper, we adopt the notion of post-hoc
interpretability as defined by Lipton, i.e. the avail-
ability of indirect information about a model’s be-
havior or specific results (Lipton, 2016). In effect,
LIME provides exactly such post-hoc interpretabil-
ity on a single-prediction basis using local explana-
tions (Ribeiro et al., 2016).
Prediction of Acute Kidney Injury in Cardiac Surgery Patients: Interpretation using Local Interpretable Model-agnostic Explanations
381
3 METHODS
In the following, we describe the methodological
setup for the development of our CPM, depicted in
Figure 1. We provide implementation details, such
as relevant software libraries, data used for model
training and validation, data preprocessing steps,
employed prediction models and the interpretability
method LIME.
3.1 Experimental Setup
As depicted in Figure 1, we utilized a cohort of in-
tensive care patients from the MIMIC-III critical care
database (Johnson et al., 2016). From this cohort, we
extracted an initial feature set based on expert consul-
tation and analysis of literature. Following a number
of preprocessing steps and data split to obtain training
and validation datasets following the 80:20 ratio, we
proceeded to train tree models, DT, GBDT and LR as
baseline. For each of these algorithms, we applied a
feature selection step using univariate analysis decid-
ing to retain features where p <.001. Subsequently,
we performed hyperparameter optimization with grid-
search using 10-fold cross-validation as score. The
models thus trained were then validated on a held-
out dataset comprised of 20% of the original dataset.
The 10-fold cross-validation discrimination and cal-
ibration metrics for each of the algorithms are com-
pared side-by-side. The LIME explainer takes as in-
put the trained classifier and an instance for explana-
tion. LIME enabled us to analyze the results achieved
not only in terms of raw performance but also in terms
of their medical adequacy.
We implemented all components of the CPM us-
ing the Python programming language at version
3.6.1 (Rossum and Drake, 2010). For data handling,
loading and storing, as well as preprocessing we have
made extensive use of the Python library Pandas at
version 0.23.4 (McKinney, 2010). Furthermore, we
used the DT, GBDT, and LR classifier implementa-
tions of the Python machine learning library scikit-
learn at version 0.19.1 for model development and
evaluation.
3.2 Patient Cohort
The MIMIC-III critical care database contains close
to 59,000 hospital admissions that were recorded over
an eleven year period at the Beth Israel Deaconess
Medical Center in Boston, MA, USA (Johnson et al.,
2016). From this data we selected a cohort of 6,782
admissions of adult patients who underwent cardiac
surgery during their hospital stay for use as labeled
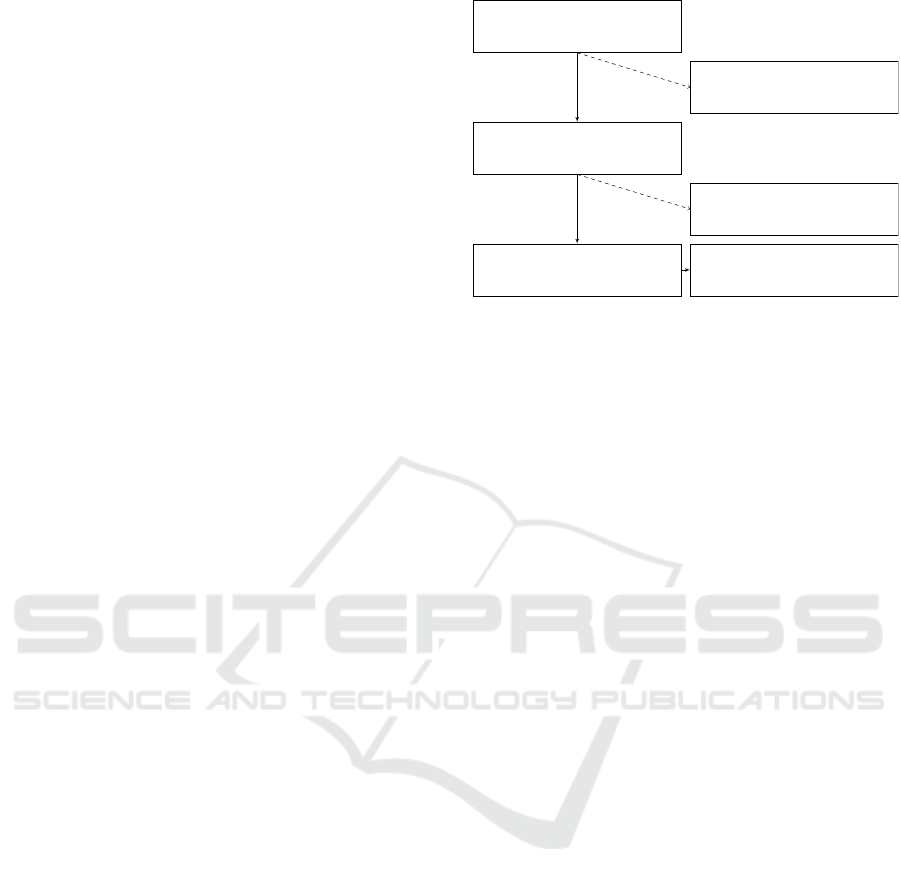
N=58,976
Admissions of
critical care patients
N=7,870
Admissions of non-adult
patients < 18 years
N=51,106
Admissions of adult
critical care patients
N=44,324
Admissions of patients
with no cardiac surgery
N=6,782
Admissions of cardiac
surgery patients
Cardiac surgery admissions
AKI (yes), N=667
AKI (no), N=6,115
Figure 2: Cohort diagram of relevant cases from the
MIMIC-III clinical care database. The data corresponding
to the depicted 6,782 patient admissions is used for model
training and validation of the proposed prediction method.
training and validation data, as depicted in Figure 2.
Surgery cases comprise coronary artery bypass graft
and aortic valve repair and/or replacement.
As concerns target variable for the prediction task,
we defined AKI according to the Acute Kidney In-
jury Network (AKIN) classification occurring after
the surgery. The AKIN classification is used for di-
agnosing AKI and ranks patients’ stages of AKI from
0 to 3, 0 being no injury and 3 being the most severe,
often indicating complete renal failure. The classifi-
cation is based on patients’ measured serum creati-
nine and urine output, and does consider if patients
receive renal replacement therapy (Lopes and Jorge,
2013). In our cohort, the incidence of AKI was ap-
proximately 10%, agreeing with general clinical ob-
servations (O’Neal, Jason and others, 2016).
As per Figure 2, the classes of patients who did
and did not develop AKI following their surgery are
not equally distributed. The data exhibit consider-
able class skew, as the patient cohort comprises ca.
10 times as many negative class cases. Given this
class skew, the robustness of training models towards
skewed training data must be considered.
3.3 Initial Feature Set
The initial feature set from the MIMIC-III database
was derived from consultations with medical experts
and analysis of extant literature as laid out in Sec-
tion 2. The input features include demographical pa-
tient data such as patients’ age and gender, binary
variables indicating the presence of certain comor-
bidities, such as diabetes, and laboratory values. In
addition to the specific comorbidities, we also com-
puted and included the Elixhauser comorbidity score
as a further feature. This score is a scalar value that is
used to assess a patient’s prognosis during their hos-
HEALTHINF 2019 - 12th International Conference on Health Informatics
382

pital stay based on the presence or absence of 30 co-
morbidities. A large value indicates a higher risk to
the patient and therefore, increased urgency for treat-
ment (Elixhauser et al., 1998). The laboratory values
comprised a set of 23 tests, including i.a. serum cre-
atinine, glucose, blood urea nitrogen. For each, we
extracted three values for the last three days leading
up to the time of the patient’s surgery. In total, we
extracted 103 features for modeling, which were then
submitted to preprocessing and feature selection.
3.4 Preprocessing and Imputation
Data preprocessing is comprised of feature scaling
and missing values imputation. The former entails
removing the mean of the individual feature dimen-
sions, i.e. centering the data’s columns, and scaling
to unit variance. The latter is necessary to handle
missing values, which often occur in a clinical con-
text (Bai et al., 2015) and to account for the fact that
the machine learning models employed for this work
do not support missing values. To handle missing val-
ues, we applied the k-Nearest Neighbors (kNN) im-
putation method with k=3, which operates under the
assumption that missing values can be approximated
by samples that are most similar to it. The utilized
the version in the fancyimpute Python library (Rubin-
steyn and Feldman, 2018).
For the examined cohort, the amount of missing
values depends significantly on the considered feature
dimension. For demographical data dimensions such
as gender and age, as well as comorbidity data the
amount of missing data is understandably low varying
from 0% to 1% as the collection of patients’ personal
information is standard procedure and a non-apparent
comorbidity does not result in missing values. Some
laboratory results, however, do exhibit a significantly
large amount of missing values ranging from 50% for
i.a. blood creatinine 1 day before surgery to up to
98% for i.a. blood hematocrit results 3 days before
the date of surgery. It must be noted that the amount
of missing values for laboratory results decreases as
we consider times closer to the time of surgery. That
is, the laboratory results for one day before surgery
typically present the least missing values compared
to values for the same type of test on earlier days.
3.5 Feature Selection
In this processing step, we performed tests using dif-
ferent percentiles of top features, using the full set of
features (103), top 50% and top 25%, reporting the
results for all the algorithms tested. We chose feature
selection based on the mutual information approach,
since it can capture non-linear dependencies among
among variables, unlike an F-test, which can capture
only linear correlations. The list of the top 50% fea-
tures used in the final model after automatic selection
is provided as supplementary material on-line
1
.
3.6 Classification Models
We compared different classification techniques that
were trained and evaluated on the same data using
the same preprocessing pipeline. In the following, we
outline the respective hyperparameters’ configuration
and optimization strategy.
Logistic Regression. Usual parameters to adjust for
LR include regularization strength and the type of
penalty, L1 or L2. Regularization can improve model
performance for unseen data by penalizing large co-
efficients in an effort to reduce overfitting or learning
training data ‘peculiarities’. Higher values for λ can
lead to more sparse models. The library utilized ex-
poses the parameter C defined as the inverse of reg-
ularization strength. The regularization parameters
were set to 10
−6
and the number of iterations before
convergence were set to 300.
Decision Tree. We applied the Gini impurity mea-
sure to calculate optimal splits. Furthermore, we used
class weights of 1:10 (AKI=no; AKI=yes) in order
to compensate for the class skew that is evident in
our training data. Finally, we determined an optimal
maximum tree depth of 6, minimum of 6 samples for
each leaf node, and a minimum of 5 samples for each
split by using a parameter grid-search over a parame-
ter grid of 3 to 10 for tree depth, 3 to 10 for minimum
samples per split, and 1 to 16 for minimum samples
per leaf, and 5-fold cross validation.
Gradient-boosted Decision Trees. This ensemble
classification approach entails using a large set of de-
cision trees or decision tree stumps as weak learn-
ers which are trained iteratively (Gron, 2017). In
our work, the ensemble size was determined using an
early stopping approach after 10 consecutive perfor-
mance decreases at 136 indicating that a set of 126
learners provides the best prediction results. Fur-
thermore, we applied a maximum tree depth for the
stumps of 3 and learning rate of 0.1, which influences
the contribution of each component tree stump.
1
Supplementary material. Top 50% selected model fea-
tures. Available at: https://goo.gl/xnUux2
Prediction of Acute Kidney Injury in Cardiac Surgery Patients: Interpretation using Local Interpretable Model-agnostic Explanations
383

3.7 Local Interpretable Model-agnostic
Explanations
For practical use in high-stakes domains, not only pre-
diction accuracy is relevant but also the level of trust a
model provides. Ensemble models such as GBDT are
typically considered black box models as they lack in-
terpretability due to the fact that the models’ behavior
is determined by a large set of individual classifiers
in a voting process (Valdes et al., 2016; Moon et al.,
2007).
We use the interpretability method LIME to shed
light on the prediction results of the GBDT model.
LIME which uses more intelligible models, such as
linear regression, to approximate the behavior of a
given model in the vicinity of the instance/prediction
being explained. The algorithm generates a number
of perturbed instances close to the instance of interest,
weighing this perturbed input according to a distance
measure. After applying the original model on these
perturbed instances, a linear function is applied to ap-
proximate the thus resulting outputs (Ribeiro et al.,
2016). The coefficients of this linear function repre-
sent the degree of influence of a given feature for the
original prediction we intended to explain. The higher
the number of samples, the higher the fidelity of the
approximate model, but the higher the algorithm run-
time. In this work, we used a sample size of 100.
LIME differs from alternative interpretability
methods, such as mimic learning, in that not the entire
prediction model’s behavior is explained, but rather
one single prediction instance (Che et al., 2016).
Therefore, the explanations provided are faithful lo-
cally but not globally. To make up for this behav-
ior, LIME offers a procedure called submodular pick,
which selects a number representative instances that
can provide some insight into the model’s global be-
havior (Ribeiro et al., 2016).
4 RESULTS
In this section, we present the performance results
achieved using the proposed pipeline along with in-
sights from applying LIME on the best classifier
(GBDT). Besides traditional performance metrics for
classification tasks, such as precision, recall, and
the analysis of Area Under the Receiver Operating
Curve (AUROC), we provide the diagnostic odds ra-
tio (DOR), a performance measure for diagnostic tests
which is prominently used in the medical domain (Be-
wick et al., 2004; Glas et al., 2003).
4.1 Discrimination
Table 1 reports the selected metrics across all feature
selection configurations and models tested, consider-
ing respectively all features, top 50% and top 25%
percentiles. DT performed worse than LR and GBDT
for most metrics, regardless of feature selection, ex-
cept for recall, where it presented a substantial advan-
tage against the other two approaches, e.g. recall of
0.66 as opposed to GBDT’s 0.48 for the top 50% fea-
tures.
The different configurations chosen for feature se-
lection demonstrated that the models achieve a simi-
lar performance even when only half of the available
features are used. Particularly when it comes to the
GBDT, the DOR was substantially improved by re-
moving 50% of the features, from 90.74 to 149.92.
As more features are removed, though, performance
begins to deteriorate perceptibly while not by a large
margin, e.g. a drop of approximately 3% in AUROC
when only 25% of the features are used in the GBDT.
Overall, the GBDT classification method provides
better prediction performance when compared to the
results yielded by conventional decision trees or lo-
gistic regression, most notably when it comes to pre-
cision (40% increase over LR) and AUROC (6% in-
crease over LR), albeit it performs poorly when it
comes to recall. Furthermore, GDBT presents sub-
stantially better results as it refers to DOR with a 7-
fold increase when compared to LR with 50% of fea-
tures.
4.2 Local Interpretability
The LIME method expects a given prediction sample
– or in our case a patient – as along with the trained
model an inputs. Therefore, an expert can inquiry the
model as to ’why’ a given decision was made by the
algorithm. However, to obtain an understanding of the
model as whole, one would have to explain many in-
stances. Since, this task might too consuming, LIME
provide a strategy called submodular pick that pro-
vides the instances that are the most representative of
the overall model’s behavior (Che et al., 2016; Elith
et al., 2008).
For GBDT predictions, LIME provides insight
into which feature dimensions are most relevant to the
results. The exact relevance of dimensions varies de-
pending on the specific data input and outputs, but a
small set of dimensions are found to be relevant for
the model’s decisions quite often across the different
instances; the Elixhauser score, cardiac arrhythmia,
hemoglobin, hematocrit, serum creatinine, and blood
urea nitrogen (BUN) laboratory values before surgery.
HEALTHINF 2019 - 12th International Conference on Health Informatics
384

Table 1: Precision, recall, diagnostic odds ratio (DOR), and area under receiver operating curve (AUROC) for AKI=1 achieved
with the proposed approach employing logistic regression (LR), decision tree classification (DT) and gradient-boosted deci-
sion trees (GBDT) respectively for different feature selection configurations (all features, top 50 and 25%). The results were
obtained by applying the trained models on a hold-out validation dataset made up of 20% of the original dataset.
Metrics
Precision Recall DOR AUCROC
All Top 50% Top 25% All Top 50% Top 25% All Top 50% Top 25% All Top 50% Top 25%
LR 0.63 0.63 0.59 0.28 0.25 0.25 19.55 19.14 16.67 0.84 0.84 0.82
DT 0.33 0.35 0.29 0.67 0.66 0.70 10.86 11.22 10.16 0.80 0.80 0.78
GBDT 0.86 0.90 0.62 0.43 0.48 0.32 90.74 149.92 115.50 0.89 0.90 0.87
The results provided by LIME represent the dis-
cretized coefficients of the regression applied locally
to provide the explanations. We choose to report the
top five features that explain the onset of AKI us-
ing submodular pick of 6 explanations, as displayed
in Figure 3.
5 DISCUSSION
5.1 Discriminative Performance
GBDTs have been found to perform exceptionally
well for classification and prediction tasks in multiple
domains and the results from our work regarding pre-
diction performance illustrate the expected advantage
of the blackbox GBDT model over the simple deci-
sion tree or logistic regression prediction models (Che
et al., 2016; Elith et al., 2008). In the medical domain,
the superior precision and class label discrimination
of the GBDT model effectively means more accu-
rately predicting AKI cases after surgery, which might
empower doctors to adopt targeted kidney-protective
measures.
As exemplified by Table 2, our approach outper-
forms the Cleveland score by a considerable mar-
gin. However, the Cleveland score’s authors utilize
a substantially larger and more diverse cohort. The
same observation applies to the STS score. One could
therefore reasonably argue that those two scores po-
tentially present higher generalizability. As such, our
model must be subject to a validation study in order
to assess its application in different clinical scenarios.
Even though our model performed well across
most metrics, it showed significant drawbacks with
regards to recall. While this issue can possibly be
mitigated by proper model calibration, i.e., by adjust-
ing classification thresholds, it might have critical im-
plications for clinical practice. Since a lower recall
means that patients who will develop AKI might in-
correctly be deemed as not under risk, calibration a
must be conducted, at the expense of possibly harm-
ing patients. This fact speaks for the necessity of a
holistic evaluation of discrimination metrics.
Our model included laboratory values prior to the
surgery. Arguably, these are not always available for
surgery patients, particularly when it comes to emer-
gency surgeries. This fact has led to a high degree of
missing values in our cohort. Even though imputation
has been performed, it is not possible to guarantee that
the model has not been biased in some way or another.
Missing values are widely-discussed topic in clinical
predictive modeling and we intend to compare differ-
ent approaches for imputation side by side.
5.2 Model Interpretation
Since the GBDT performed best in the given task,
we submitted the model to the LIME explainer. In
fact, model interpretability is especially important for
medical practitioners and patients to promote accep-
tance for clinical use and build trust in predictive de-
cision support (Katuwal and Chen, 2016).
Upon examination of the instances chosen by the
submodular pick by LIME, we can observe that a high
Elixhauser score, i.e., over 7, is often implicated with
increasing risk of post-surgical AKI. Note that posi-
tive coefficients are positively correlated with the out-
come and vice-versa. This observation agrees with
the medical significance of this co-mordibities score:
higher values are in general associated with poorer pa-
tient outcomes in general (Austin et al., 2015).
With regards to blood (or serum) creatinine, it is
an important marker of kidney function, being present
in the definition of AKI itself, with higher values in-
dicating deterioration of kidney function (Lopes and
Jorge, 2013). Values between 0.6 and 1.2mg/dL for
creatinine are usually considered normal, and LIME
correctly shows a protective effect of values below
1.4mg/dL. However, the explanations do not include
higher serum creatinine values as a risk factor, possi-
bly casting doubt on the generalizability of the model.
Furthermore, presence of liver disease is gener-
ally implicated in poorer outcomes for kidney pa-
tients (Targher et al., 2008). One would expect the
model explanations to fully reflect medical knowl-
edge. However, the opposite can be verified as per
LIME’s explanations: absence of liver disease often
Prediction of Acute Kidney Injury in Cardiac Surgery Patients: Interpretation using Local Interpretable Model-agnostic Explanations
385
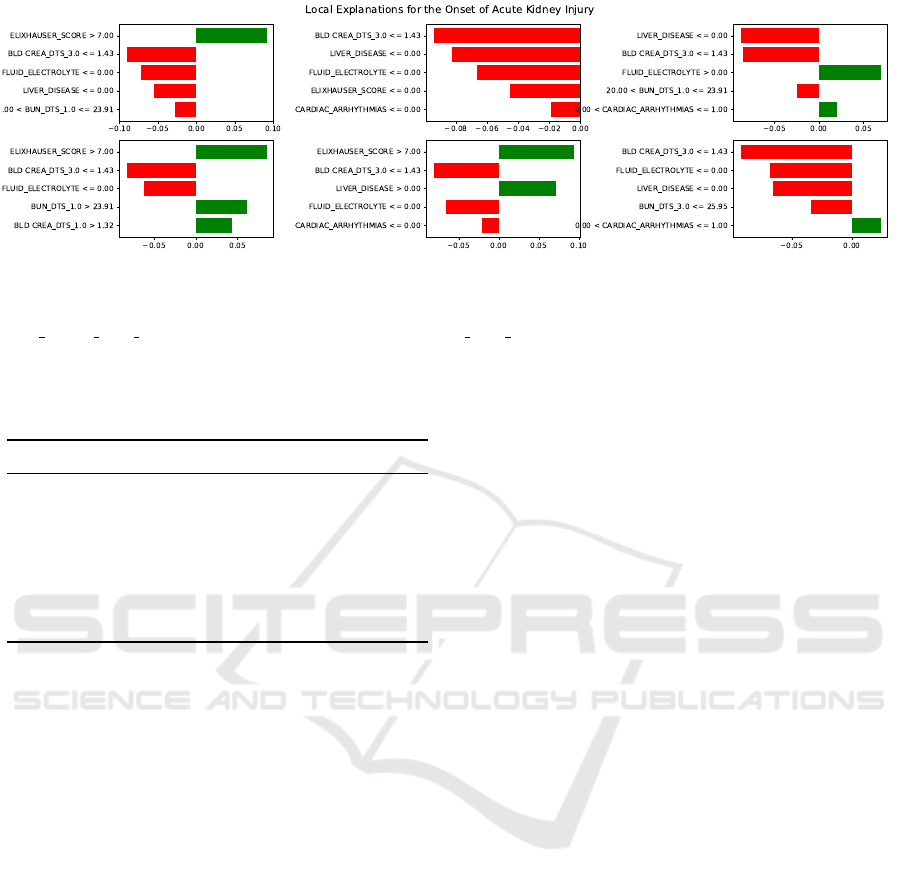
Figure 3: Local explanations provided by LIME. Using submodular pick, LIME chooses the most significant examples for
explanation, i.e, the individual subplots. Each shows the top 5 features chosen by LIME as the most meaningful for the local
predictions. Note that positive coefficients are correlated with increased likelihood of Acute Kidney Injury. Abbreviations:
BLD CREA DTS N=Blood Creatinine N days to surgery; BUN DTS N=Blood Urea Nitrogen N days to surgery.
Table 2: Overview of CPMs for cardiac surgery-associated
AKI. Abbreviations: CPM=Clinical Prediction Model;
N=number of patients; AUC=Area Under the Curve.
CPM N AUC
Cleveland 33,217 0.81
STS score 86,009 0.83
AKICS score 603 0.84
SRI score 2,566 0.78
Ng score 28,422 0.77
Jiang score 7,233 0.74
Our approach 6,782 0.90
appears as a protective factor, with only one expla-
nation displaying the expected behavior. The use the
LIME approach makes it possible to critically analyze
model predictions at the single instance level, i.e., pa-
tient level, revealing potential bias in the models that
might compromise applicability in practice. Correla-
tions that statistically relevant but medically indefen-
sible are not only inconsistent, but potentially danger-
ous for patients. The application of interpretability
approaches can therefore help shed light on the ob-
scure side of black-box models. Finally, it is worth
noting that alternative interpretability methods, such
as mimic learning, have also been applied in previ-
ous research and a thorough comparison of available
interpretability approaches highlighting weaknesses
and strengths constitutes valuable future work in this
field.
6 CONCLUSION
We have devised a clinical prediction model that em-
ploys machine learning methodologies on clinical pa-
tient data in order to assess the risk of AKI in heart
patients before the time of surgery using the MIMIC-
III database. This can allow physicians to make bet-
ter decisions about surgical therapy and plan accord-
ingly for complications in high risk patients, e.g. by
readying renal replacement resources in advance and
avoiding nephrotoxic agents. By comparing the usage
of traditional decision tree models with GBDT pre-
diction models we showed the advantage in predic-
tion quality of a more complex and non-interpretable
black box model over an easily understandable white
box modeling technique such as logistic regression or
decision trees.
Our GBDT model outperformed established clini-
cal scores for post-surgical AKI onset by a significant
margin (AUROC of 0.9 vs. 0.83). While external val-
idation with a bigger and more diverse cohort remains
to be performed for the model to be considered gen-
erally applicable, results suggest GDBT as an appro-
priate algorithm for this specific prediction task. Fu-
ture work shall also consider other algorithms such as
deep learning and random forests, as well as different
strategies for imputation and feature selection.
Despite the promising results, in the light of the
importance of model intelligibility in high-risk do-
mains such as medicine, we also utilized the inter-
pretability method LIME on the GBDT model. Us-
ing this explainer, we regained a significant amount of
meta-information about which features are most rele-
vant for the prediction model’s output. It ultimately
revealed possible incongruencies between model ex-
planations and medical evidence that must be ad-
dressed for such a model to be used in practice.
ACKNOWLEDGMENTS
The authors would like to thank Dr. Alexander Meyer
and his team at the German Heart Center Berlin
for valuable guidance on the medical challenges in-
volved. Parts of the given work were generously sup-
ported by a grant of the German Federal Ministry of
HEALTHINF 2019 - 12th International Conference on Health Informatics
386

Economic Affairs and Energy (01MD15005).
REFERENCES
Austin, S. R. et al. (2015). Why Summary Comorbidity
Measures such as the Charlson Comorbidity Index and
Elixhauser Score Work. Medical care, 53(9):e65.
Bai, B. M., Mangathayaru, N., and Rani, B. P. (2015). An
Approach to Find Missing Values in Medical Datasets.
In Proc. Intl. Conf. on Engineering & MIS, pages
70:1–70:7, New York, NY, USA. ACM.
Bewick, V., Cheek, L., and Ball, J. (2004). Statistics Re-
view 13: Receiver Operating Characteristic Curves.
Critical Care, 8(6):508.
Caruana, R. et al. (2015). Intelligible Models for Health-
Care: Predicting Pneumonia Risk and Hospital 30-day
Readmission. In Intl. Conf. Knowledge Discovery and
Data Mining, pages 1721–1730, New York, NY, USA.
ACM.
Che, Z., Purushotham, S., Khemani, R., and Liu, Y. (2016).
Interpretable Deep Models for ICU Outcome Predic-
tion. AMIA Symposium, 2016:371–380.
Elith, J., Leathwick, J. R., and Hastie, T. (2008). A Working
Guide to Boosted Regression Trees.
Elixhauser, A. et al. (1998). Comorbidity Measures for Use
with Administrative Data. Medical Care, 36(1):8–27.
Flynn, N. and Dawnay, A. (2015). A Simple Electronic
Alert for Acute Kidney Injury. Ann Clin Biochem,
52(2):206–212.
Glas, A. S. et al. (2003). The Diagnostic Odds Ratio: A Sin-
gle Indicator of Test Performance. Journal of Clinical
Epidemiology, 56(11):1129–1135.
Goodman, B. and Flaxman, S. (2016). EU Regulations on
Algorithmic Decision-making and a ”Right to Expla-
nation”. In ICML Workshop on Human Interpretabil-
ity in Machine Learning.
Gron, A. (2017). Hands-On Machine Learning with Scikit-
Learn and TensorFlow. O’Reilly Media, Inc., 1st edi-
tion.
Huen, S. C. and Parikh, C. R. (2012). Predicting Acute
Kidney Injury after Cardiac Surgery: a Systematic Re-
view. Ann Thorac Surg, 93(1):337–47.
Jiang, W. et al. (2016). Dynamic Predictive Scores for Car-
diac Surgery–Associated Acute Kidney Injury. Am
Heart J, 5(8).
Jiang, W. et al. (2017). Validation of Four Prediction
Scores for Cardiac Surgery-Associated Acute Kidney
Injury in Chinese Patients. Braz J Cardiovasc Surg,
32(6):481–486.
Johnson, A. E. et al. (2016). MIMIC-III, a Freely Accessi-
ble Critical Care Database. Scientific Data, 3.
Katuwal, G. J. and Chen, R. (2016). Machine Learning
Model Interpretability for Precision Medicine. ArXiv
e-prints.
Latini, R., Aleksova, A., and Masson, S. (2016). Novel
Biomarkers and Therapies in Cardiorenal Syndrome.
Letham, B. et al. (2015). Interpretable Classifiers us-
ing Rules and Bayesian Analysis: Building a Better
Stroke Prediction Model. Annals of Applied Statistics,
9(3):1350–1371.
Lipton, Z. C. (2016). The Mythos of Model Interpretability.
ArXiv e-prints.
Lopes, J. A. and Jorge, S. (2013). The RIFLE and AKIN
Classifications for Acute Kidney Injury: a Critical and
Comprehensive Review. Clin Kidney J, 6(1):8–14.
McKinney, W. (2010). Data Structures for Statistical Com-
puting in Python. In van der Walt, S. and Millman,
J., editors, Proc. 9th Python in Science Conference,
pages 51 – 56.
Moon, H. et al. (2007). Ensemble Methods for Classifica-
tion of Patients for Personalized Medicine with High-
Dimensional Data. Artif Intell Med, 41(3):197–207.
Ng, S. Y. et al. (2014). Prediction of Acute Kidney Injury
within 30 Days of Cardiac Surgery. J Thorac Cardio-
vasc Surg, 147(6):1875–1883.e1.
O’Neal, Jason and others (2016). Acute Kidney Injury Fol-
lowing Cardiac Surgery: Current Understanding and
Future Directions. Critical Care, 20(1):187.
Palomba, H. et al. (2007). Acute Kidney Injury Prediction
following Elective Cardiac Surgery: AKICS Score.
Kidney International, 72(5):624–631.
Ribeiro, M. T., Singh, S., and Guestrin, C. (2016). ”Why
Should I Trust You?”: Explaining the Predictions of
Any Classifier. In Proc. 22nd Intl. Conf. on Knowl-
edge Discovery and Data Mining, pages 1135–1144,
New York, NY, USA. ACM.
Rosner, M. H. and Okusa, M. D. (2006). Acute Kidney
Injury Associated with Cardiac Surgery. Clin J Am
Soc Nephrol, 1(1):19–32.
Rossum, G. V. and Drake, F. L. (2010). Python Tutorial.
History, 42(4):1–122.
Rubinsteyn, A. and Feldman, S. (2018). fancyim-
pute: A Variety of Matrix Completion and Im-
putation Algorithms Implemented in Python.
https://github.com/iskandr/fancyimpute Accessed:
October, 2018.
Sawhney, S. et al. (2015). Acute Kidney Injury - How Does
Automated Detection Perform? Nephrol. Dial. Trans-
plant., 30(11):1853–61.
Targher, G. et al. (2008). Non-alcoholic fatty liver
disease is independently associated with an in-
creased prevalence of chronic kidney disease and
proliferative/laser-treated retinopathy in type 2 dia-
betic patients. Diabetologia, 51(3):444–450.
Thakar, C. V. et al. (2004). A Clinical Score to Predict
Acute Renal Failure after Cardiac Surgery. J Am Soc
Nephrol, 14(8):2176–7.
Valdes, G. et al. (2016). MediBoost: A Patient Stratification
Tool for Interpretable Decision Making in the Era of
Precision Medicine. Scientific Reports, 6.
Wijeysundera, D. N. et al. (2007). Derivation and Valida-
tion of a Simplified Predictive Index for Renal Re-
placement Therapy After Cardiac Surgery. JAMA,
297(16):1801.
Prediction of Acute Kidney Injury in Cardiac Surgery Patients: Interpretation using Local Interpretable Model-agnostic Explanations
387
