
Classification of Images of Childhood Pneumonia using Convolutional
Neural Networks
A. A. Saraiva
1 a
, N. M. Fonseca Ferreira
2,3,4 b
, Luciano Lopes de Sousa
5 c
,
Nator Junior C. Costa
5 d
, Jos
´
e Vigno Moura Sousa
5,6 e
, D. B. S. Santos
5 f
, Antonio Valente
2,7
g
and Salviano Soares
7 h
1
UTAD University, Coimbra, Portugal
2
INESC-TEC Technology and Science, Campus da FEUP, Rua Dr. Roberto Frias 378, 4200 - 465 Porto, Portugal
3
Knowledge Engineering and Decision-Support Research Center (GECAD) of the Institute of Engineering,
Polytechnic Institute of Porto, Portugal
4
Department of Electrical Engineering, Institute of Engineering of Coimbra, Polytechnic Institute,
Rua Pedro Nunes, 3031-601 Coimbra, Portugal
5
State University of Piau
´
ı, Piau
´
ı, Brazil
6
University Brazil, S
˜
ao Paulo, Brazil
7
IEETA-UA and School of Science and Technology, University of Tr
´
as-os-Montes and Alto Douro, Vila Real, Portugal
Keywords:
Pneumonia, X-Ray, CNN, K-Fold.
Abstract:
In this paper we describe a comparative classification of Pneumonia using Convolution Neural Network. The
database used was the dataset Labeled Optical Coherence Tomography (OCT) and Chest X-Ray Images for
Classification made available by (Kermany, 2018) with a total of 5863 images, with 2 classes: normal and
pneumonia. To evaluate the generalization capacity of the models, cross-validation of k-fold was used. The
classification models proved to be efficient compared to the work of (Kermany et al., 2018) which obtained
92.8 % and the present work had an average accuracy of 95.30 %.
1 INTRODUCTION
Pneumonia is one of the most common causes of
death in children worldwide, accounting for 15 % of
all deaths of children under 5 years of age (Mathur
et al., 2018). Identifying and treating pneumonia has a
substantial effect on infant mortality. Although meth-
ods utilizing chest X-ray are promising modalities for
radiologic diagnosis, their role in clinical manage-
ment and their impact on outcomes need to be im-
proved (Zar et al., 2017).
a
https://orcid.org/0000-0002-3960-697X
b
https://orcid.org/0000-0002-2204-6339
c
https://orcid.org/0000-0003-0551-4804
d
https://orcid.org/0000-0001-5636-424X
e
https://orcid.org/0000-0002-5164-360X
f
https://orcid.org/0000-0003-4018-242X
g
https://orcid.org/0000-0002-5798-1298
h
https://orcid.org/0000-0001-5862-5706
For an adequate treatment, therefore, an early di-
agnosis of pneumonia is necessary, but it is not always
clear (Saraiva et al., 2018a), (Saraiva et al., 2018e). In
a review of the medical records of patients admitted
with pneumonia, 22 % of the patients presented some
reason for the uncertainty of the diagnosis, which
could result in delays in the delivery of antibiotics.
Furthermore, chest computed tomography is a gold
standard for diagnosis, but it is not always available
and is loaded with a high dose of radiation and high
cost, preventing its use in the routine diagnostic pro-
cess of patients with suspected pneumonia (Cortellaro
et al., 2012) (Saraiva et al., 2018f), (Marques et al.,
2018), (Saraiva et al., 2018c).
Several researchers use imaging for the detection
of pneumonia. Most methods use radiographic imag-
ing as a tool. For example, in (Sharma et al., 2017),
to detect clouds of pneumonia, the Otsu threshold
was used, which will separate the healthy part of the
lung from the nebulous regions infected by pneumo-
112
Saraiva, A., Ferreira, N., Lopes de Sousa, L., Costa, N., Sousa, J., Santos, D., Valente, A. and Soares, S.
Classification of Images of Childhood Pneumonia using Convolutional Neural Networks.
DOI: 10.5220/0007404301120119
In Proceedings of the 12th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2019), pages 112-119
ISBN: 978-989-758-353-7
Copyright
c
2019 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

nia, and a detection method based on neural networks
using tomography models computerized, was used in
(Rajaraman et al., 2018). In addition to these meth-
ods, there are projects that extract information from
the cough sound analysis to diagnose cases of pneu-
monia (Amrulloh et al., 2018).
The doctor, when interpreting the chest x-ray, will
look for white patches in the lungs to detect pneu-
monia. However, such hazy patterns would also be
observed tuberculosis and severe cases of bronchitis.
For the purpose of conclusive diagnosis, the purpose
of this study is to identify pneumonia, through analy-
sis of chest X-ray images, to recognize patterns of the
disease using a neural network.
The classification stage consists of two sub-steps,
where the first one is the training of a Convolutional
Neural Networks (CNN), the second one is the vali-
dation of the model, that is, the test with images not
known by CNN. The method covered ensures a robust
coverage in image recognition (Saraiva et al., 2018d),
(Saraiva et al., 2018g) (Saraiva et al., 2018b), under
certain assumptions that will be clarified throughout
the text. A classification model (CNN) for the pneu-
monia and normal classes is proposed. The results
are compared with the work of (Kermany et al., 2018)
and obtained a slightly higher result, and the present
work presents cross-validation of k-fold. In order to
strengthen the results.
This article is divided into 5 sections, and section
2 is intended for related works. In section 3, it is com-
posed by the methodology, where will be explained
the steps taken for the development of the algorithm.
In section 4, it is assigned to the results obtained and,
following in the next section, in 5, where the conclu-
sion of the work is presented.
2 RELATED WORK
For the detection of pulmonary diseases chest radiog-
raphy is always required to identify pulmonary prob-
lems. Diseases such as tuberculosis, pneumonia and
lung cancer are a major threat to human health. Thus,
in (Khobragade et al., 2016) proposes pulmonary seg-
mentation, extraction of characteristics and its classi-
fication using an artificial neural network for the de-
tection of pulmonary diseases. (Khobragade et al.,
2016) a simple image processing technique with in-
tensity based method was used, and a method based
on the discontinuity to detect pulmonary limits, in this
way, statistical and geometric characteristics were ex-
tracted. Neural networks were used feed forward and
back propagation to detect diseases.
Pneumonia is one of the leading causes of infant
mortality. In developing countries there is little in-
frastructure and doctors in rural areas to provide the
necessary diagnosis. Therefore, in (Barrientos et al.,
2016) proposes a method for automatic diagnosis us-
ing ultrasonography of the lungs. The approach pre-
sented is based on the analysis of patterns in rectangu-
lar segments in the image of the ultrasonography. The
specific characteristics and characteristic vectors were
obtained and classified by a standard neural network.
In (Barrientos et al., 2016) I obtained a sensitivity of
91.5 % and specificity of 100 % but were extracted
from a single patient and only included in the test or
in the training set.
Many researchers have developed several algo-
rithms for the diagnosis of lung diseases through
sound. One of the parameters used for the detection of
pulmonary sound is entropy, so there are differences
in the sound of a normal respiratory system and a sys-
tem with pathologies. In the article (Rizal et al., 2017)
discurses several measures of entropy for a classifica-
tion of pulmonary sounds. The result in (Rizal et al.,
2017) shows that the use of a single entropy could not
achieve high accuracy, so 7 entropies were used and
guaranteed 94.95 % accuracy using multilayer per-
ceptron.
In paper (Rodrigues et al., 2018) suggests a
Structural Co-occurrence Matrix (SCM) approach to
classify malignant nodules or benign nodules and
also their level of malignancy. The structural co-
occurrence matrix technique was applied to extract
characteristics of the nodule images and classify
them. The SCM was applied in gray scale and images
of the Hounsfield unit with four filters, creating eight
different configurations. The classification stage used
classifiers known as the multilayer perceptron, sup-
port vector machine, k-Nearest Neighbors algorithm
and were applied in two tasks: (i) to classify the nod-
ule images as malignant or benign, (ii) to classify the
nodules pulmonary lesions at the level of malignancy
(1 to 5). O (Rodrigues et al., 2018) had a result of
96.7 % for precision and F-score measurements in the
first task and 74.5 % accuracy and 53.2 % F-score in
the second task.
The (Santosh and Antani, 2018) has proposed an
idea that takes into account the alterations of the right
and left lung region in terms of symmetry and auto-
mated the chest X-ray system for the evidence of tu-
berculosis. The proposed method is the observation of
radiological exams leading to bilateral comparisons
in the lung field. In (Santosh and Antani, 2018) an-
alyzed the symmetric lung region using a multiple-
scale shape feature, as well as border texture char-
acteristics. Three different types of classifiers were
used: Bayesian network, multilayer network percep-
Classification of Images of Childhood Pneumonia using Convolutional Neural Networks
113

tron and random forest. The results obtained with an
abnormality detection accuracy of 91 % and area un-
der the ROC curve of 0.96.
Many researchers use various methods for detect-
ing diseases based on lung sound, for example the use
of entropy measurement. Sound of pulmonary snor-
ing is a sound that is discontinuous, of short duration
and appears in the inspiratory, expiratory or in both
cases. Thus, in (Rizal et al., 2016) the Tsallis entropy
was used as the characteristic extraction method to
classify lung sounds. The results were achieved us-
ing at least three Tsallis entropy values with q = 2, 3,
and 4 with MLP as a classifier and three-fold cross
validation at an accuracy of 95.35 %, sensitivity of
90.48 % and 100 % specificity, were achieved.
Lung cancer accounts for 26 % of all cancer
deaths in 2017, accounting for more than 1.5 mil-
lion deaths. Thus, in (d. N
´
obrega et al., 2018) is
proposed to explore the performance of deep transfer
learning to classify pulmonary nodule malignancies.
In this work, we have used a convolutional neural
network such as VGG16, VGG19, MobileNet, Xcep-
tion, InceptionV3, ResNet50, Inception-ResNet-V2,
DenseNet169, DenseNet201, NASNetMobile and
NASNetLarge, where they were used to extract pa-
rameters from an image database of the lung. The
characteristics were classified using Naive Bayes,
Perceptron multilayer, support vector machine, Near
Neighbors and Random Forest. The results obtained
in (d. N
´
obrega et al., 2018) were 88.41 % of ACC and
93.19 % of AUC.
(Paing and Choomchuay, 2017) has the objective
of detecting pulmonary nodules from a series of dig-
itized CT images. The threshold and morphological
operations of Otsu are applied for the segmentation
of nodules. In (Paing and Choomchuay, 2017), after
segmentation, objects that can not be nodes are dis-
carded. In view of this, multilayer Perceptron was
used for the classification and 95 % accuracy was
achieved.
In the work (Kermany et al., 2018) a method was
proposed using Convolutional Neural Networks with
a transfer learning technique. Transfer learning has
proven to be a highly effective technique, particularly
when confronted in domains with limited data. The
main application of the transfer learning algorithm
was in the diagnosis of OCT images of the retina, but
it was also tested in pediatric thoracic radiographs. In
case of retinal OCT, in a multiclass comparison be-
tween choroidal neovascularization, diabetic macular
edema, drusen and normal, obtained a precision of
96.6 %, with a sensitivity of 97.8 %, a specificity of
97, 4 %. In the comparison of chest X-rays with pneu-
monia versus normal, it obtained an accuracy of 92.8
%, with sensitivity of 93.2 % and specificity of 90.1
%.
The convolutional neural networks offer great
help in detecting diseases such as (Lisowska et al.,
2017), (Ponzio et al., 2018) and (Mabaso et al., 2018).
Therefore, a 3D Convolutional Neural Network for
the detection of subtle signs of a Stroke has been pro-
posed in (Lisowska et al., 2017), thus, the architecture
was developed to explore contralateral resources and
anatomical information from the cerebral atlas. In the
work (Ponzio et al., 2018), a deep learning technique
based on Convolutional Neural Networks was pro-
posed to differentiate adenocarcinomas from healthy
tissues and benign lesions. In the article (Mabaso
et al., 2018), it presents an automated way of detect-
ing and counting points in microscopic images based
on a Convolutional Neural Network based on a slid-
ing window for detection of multiple points in micro-
scopic images.
Before these works, in this one, the objective is the
construction of an algorithm that recognizes the pneu-
monia, from x-ray images of the thoracic region. In
the algorithm, a Convolutional Neural Network was
used to recognize patterns in the images of people in-
fected by pneumonia, either by virus or bacteria.
3 METODOLOGY
3.1 Description of the Dataset
In the dataset (Kermany, 2018) used for training and
validation, it contained 5863 X-ray (JPEG) images
and two categories: Normal and Pneumonia. A total
of 5863 patients (Junge and Dettori, 2018)
The radiographic images were from pediatric pa-
tients one to five years old from the Medical Center in
Guangzhou. In this way, radiographs were performed
as part of clinical care.
All images in dataset (Kermany, 2018) underwent
a treatment in order to remove all low-quality scans,
as well as being classified by two specialist physicians
and by a third party specialist, in order to prevent any
misclassification.
3.1.1 Pneumonia (Bacterial and Viral) and
Normal
For the diagnosis of pneumonia, the alveoli become
filled with secretion and appear as a white spot on
the chest radiograph. Pulmonary consolidation means
that the pulmonary alveoli are filled with inflamma-
tory fluid. In radiography, pulmonary consolidation
corresponds to an opacity, that is, the whitish area. As
BIOIMAGING 2019 - 6th International Conference on Bioimaging
114
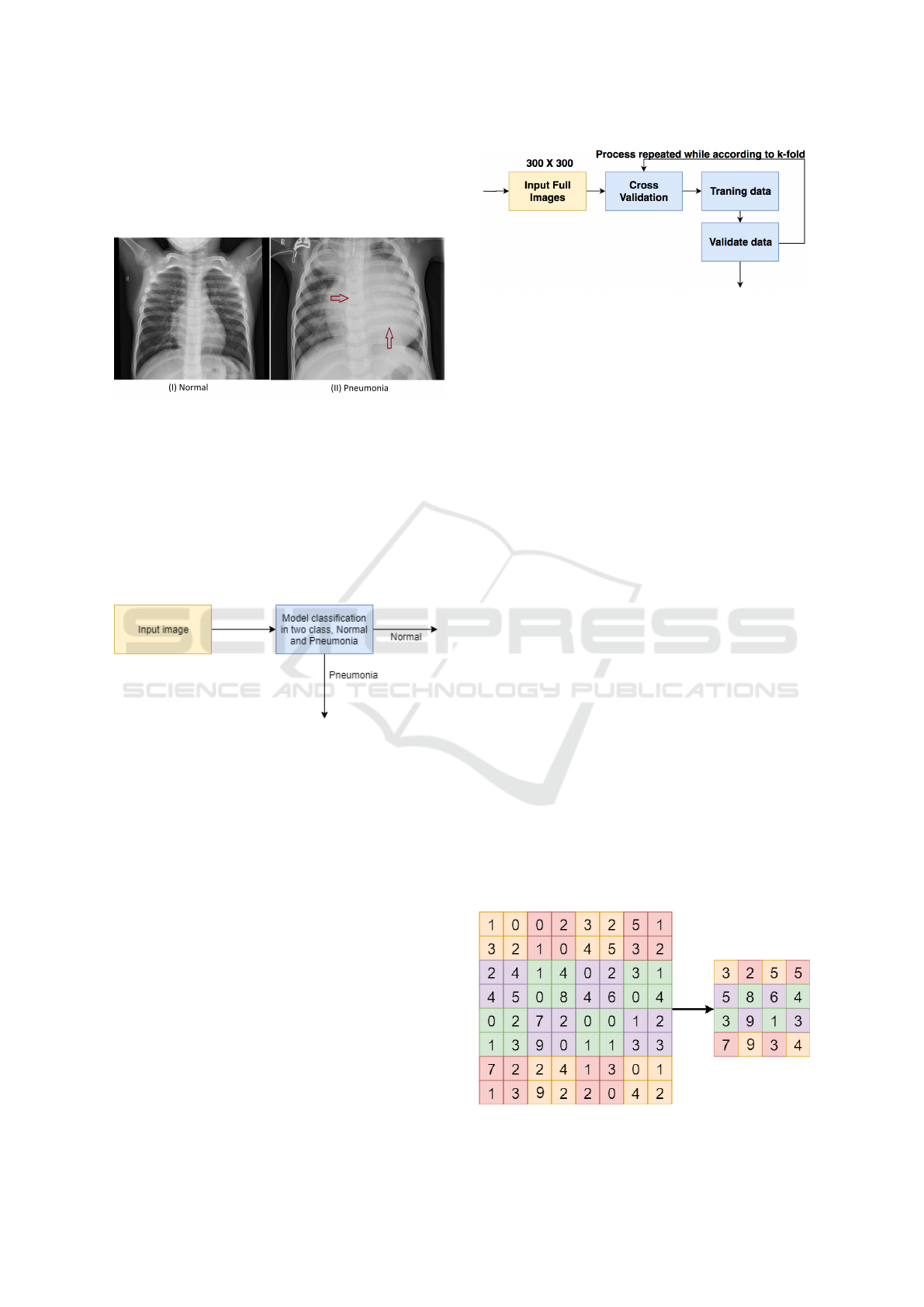
shown in the Figure 1 (II), the X-ray of the thoracic re-
gion of patients presenting with pneumonia and X-ray
of patients under normal conditions, shown in Figure
1 (I).
Figure 1: Exemple normal and pneumonia.
3.2 Input Structure for Learning Model
In Figure 2 the classification structure is represented,
which consists of the image entry in the network, re-
sponsible for classification in pneumonia or normal.
Images are converted to grayscale and normalized so
that they are between 0 and 1, making them a 32-bit
floating point.
Figure 2: Exemple input image.
3.3 Training Structure
The training and validation structure has three steps,
represented in Figure 3, the first one is the division
and normalization of the images. In the second one
the training of the images is realized. Network vali-
dation is performed using the test data.
To validate the model, cross-validation of k-fold
is used, which consists of dividing the images into
two sets: test and training. The process is repeated 5
times, changing the test and training images after the
calculation of k.
3.4 Proposed Model Convolution
Neural Network (CNN)
In the first stage we have the convolutional layer,
which is one of the main layers, where it is the extrac-
tion of characteristics of the image through a series of
Figure 3: Training structure and validation.
filters (kernels). A filter is an array of values, called
weights, trained to detect specific features. The filter
moves over each part of the image to see if the feature
that it should detect is present. When the feature is
present in part of an image, the convolution operation
between the filter and that part of the image results in
a real number with a high value. If the resource is not
present, the resulting value will be low. To provide a
value that represents the confidence that a particular
resource is present, the filter performs a convolution
operation, which is an elementary product and sums
between two arrays.
Given a two-dimensional image, I, and a small ar-
ray, K of size h x w (kernel), the convoked image,
I ∗K, is calculated by superimposing the kernel at the
top of the image all possible forms, and recording the
sum of the elementary products between the image
and the kernel equation 1.
(I ∗ K)
xy
=
h
∑
i=1
w
∑
j=1
K
i j
.I
x+i−1,y+ j−1
(1)
After the convolution layer is the pooling layer,
which is responsible for reducing the spatial size of
the feature map, preserving the resources detected
in a smaller representation. There are several pool-
ing layer options, with maxpooling being the most
popular. Basically, maxpooling operates by locating
the locations in the image that show the strongest
correlation for each resource (the maximum value)
are preserved and those maximum values combine to
Figure 4: Example max-pooling with a 8x8 image.
Classification of Images of Childhood Pneumonia using Convolutional Neural Networks
115

form a smaller space as shown in Figure 4.In general,
they are used after the convolutional layers, both with
the objective of progressively reducing computational
costs in the network, as well as minimizing the prob-
ability of overfitting.
After the pooling layer, we find the fully con-
nected layers (FCN) that are used to make final pre-
dictions. A FCN layer obtains ”resources” in a vector
form from a previous resource extraction layer, mul-
tiplies a weight matrix, and generates a new resource
vector whose computation pattern is a dense matrix-
vector multiplication (Zhang et al., 2018).Some FCNs
are used in a cascade mode that ultimately produce
the CNN classification result that generates a proba-
bility (a number ranging from 0 to 1) for each of the
classification labels that the model is trying to pre-
dict. Sometimes multiple input vectors are processed
simultaneously in a single batch to increase the over-
all throughput as shown in the following expression
when the batch size h is greater than 1. Note that the
FCN layers are also the major components of neu-
ral networks (DNNs) that are widely used in speech
recognition (Zhang et al., 2018).
Out[m][h] =
N
∑
n=0
W [m][n] ∗ In[n][h] (2)
3.4.1 Categorical Cross-entropy
The cross-entropy loss measures the performance of
a classification model, with the output being a proba-
bility value ranging from 0 to 1 (Zhang and Sabuncu,
2018).
H(p, q) = −
∑
x
p(x) log q(x). (3)
3.4.2 CNN Architecture
In Figure5 is the CNN architecture used, it has 10 lay-
ers, the first seven convolutionals and the last three
without convolution with the softmax activation func-
tion (Peng et al., 2017) equation 4, the network in-
put receives a 300x300 pixel image, each convolution
layer has the ReLUs enable function. For the convo-
lution kernel, the 3x3 size was adopted, because this
way it is possible to have a greater precision in the
time to go through the entire image.
After two convolutional layers a Max-pooling
layer is used, this reduces the size of the matrices re-
sulting from the convolution. With this layer it is pos-
sible to reduce the amount of parameters that will be
learned by the network, this way it is done overfitting
control.
In the last layer the softmax activation function is
used (Peng et al., 2017) equation 4, this function is
responsible for making the probabilistic distribution
of the input image belong to each of the classes in
which the network was trained. To reduce the train-
ing time and to avoid overfitting is used dropout in the
layer, ie it is randomly removed at each training inter-
action, a certain percentage of the neurons of a layer,
re-adding them in the following iteration. The loss
function used was equation 3 and the optimization
function used was ADAM (Kingma and Ba, 2014)
σ(z)
j
=
e
z
j
∑
K
k=1
e
z
k
f or j = 1, . . . , K. (4)
4 METRICS OF THE
EVALUATION
4.1 Cross Validation
Cross-validation is one of the fundamental methods
in machine learning for method evaluation and pa-
rameter selection in a machine prediction or learning
task. Thus, K-fold cross validation was used to eval-
uate this model.
In K-fold cross-validation, the sample is randomly
divided into k sets of equal sizes. In each of the k
shares, a single set is separated as the validation data
to test the model, and the remaining sets of k − 1
are used as training data (Fan and Hauser, 2018).
The cross-validation process is then repeated k times,
with each of the k sets used exactly once for valida-
tion (Fan and Hauser, 2018).The mean performance is
then used as the evaluation index of the method stud-
ied. This approach can be computationally expensive,
but it takes full advantage of the entire set of data,
especially when the number of samples is very small
(Men et al., 2018). This approach can also demon-
strate how the trained model is generalizable for un-
seen data in order to avoid deliberate choice of data
with superior test results (Men et al., 2018).
4.2 Confusion Matrix
The confusion matrix is an array that contains correct
and incorrect predictions of the algorithm and the ac-
tual situation. As shown in table 1.
• True Positive: Number of people who actually
have pneumonia according to the algorithm.
• False Negative: Number of people who are ac-
tually with pneumonia but categorized as healthy
according to the algorithm.
BIOIMAGING 2019 - 6th International Conference on Bioimaging
116
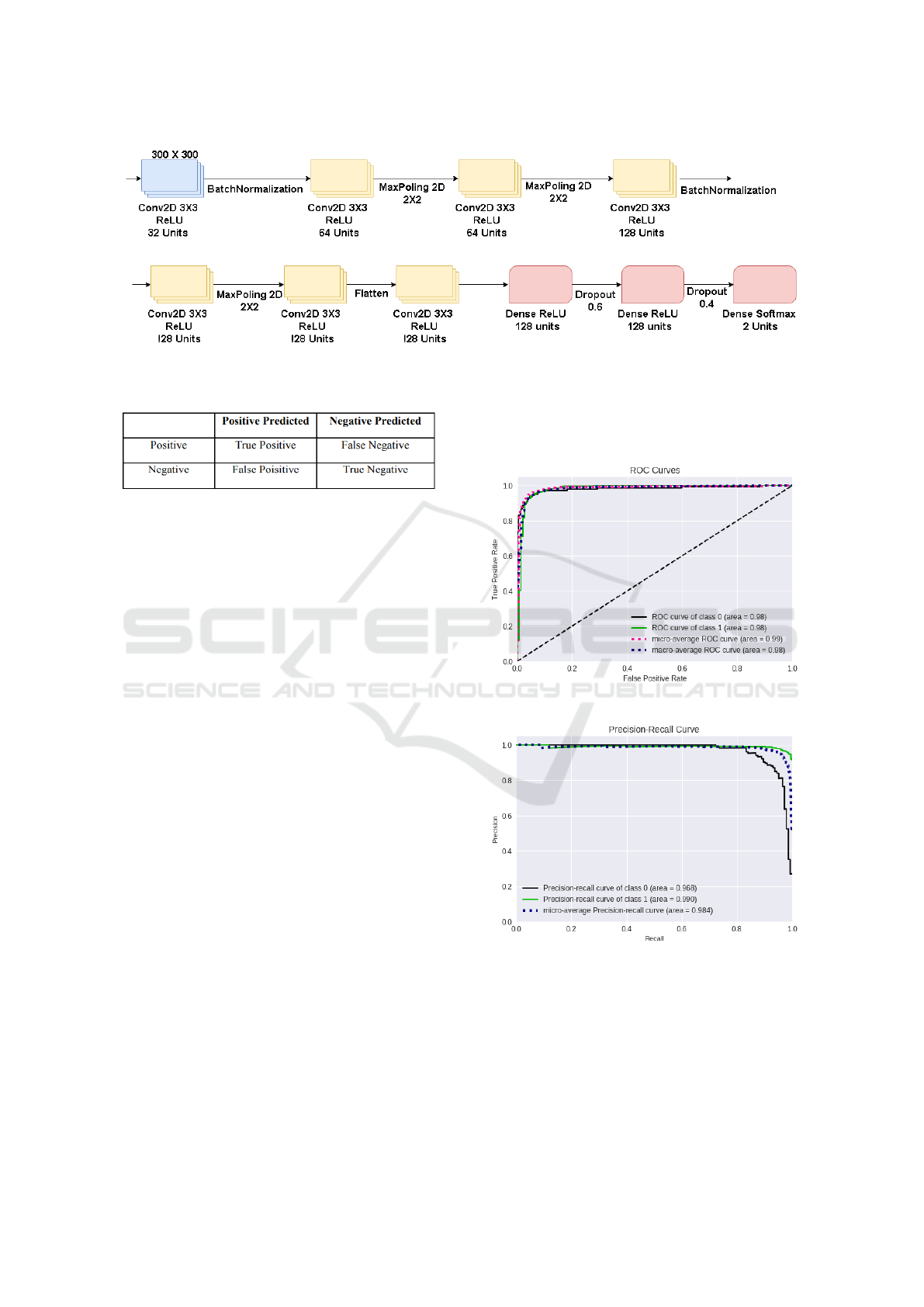
Figure 5: Construction of the CNN training mode.
Table 1: Confusion Matrix.
• False Positive: Number of people who are actu-
ally healthy, but categorized as pneumonia, ac-
cording to the algorithm.
• True Negative: Number of people who are really
healthy and categorized as healthy according to
the algorithm.
4.3 ROC Curve
The ROC curve is a graphical representation that vi-
sualizes the relationship between the true positive rate
and the false positive rate for a classifier under vari-
ous decision thresholds. Thus, a ROC curve is con-
ceptually equivalent to a curve that shows the rela-
tionship between the power of the test and the prob-
ability of error, with the variation of the cutoff value
of a statistical test. Therefore, the ROC curve com-
pares classifier performance across the full range of
class distributions, offering an assessment, covering
a wide range of operating conditions (Brzezinski and
Stefanowski, 2017).
ROC curves are an important tool in assessing the
uncertainty format, and are a valuable method in char-
acterizing the strengths and weaknesses of diagnostic
tests (Junge and Dettori, 2018).
5 RESULTS
In this section will be presented the results obtained
by the classification model described above, The met-
rics used reinforce the results, in the table 2 CNN per-
formance results compared to the work of (Kermany
et al., 2018). In Figure 7 you can view a graph with
the ROC curve. In the figure 8 the confusion matrix
for iteration 1 of the table 2.
Figure 6: ROC curve interaction 1 reference table 2.
Figure 7: Precision Recall curve interaction 1 reference ta-
ble 2.
6 CONCLUSIONS
In this paper we have demonstrated a comparison with
the work of (Kermany et al., 2018) in the detection
and classification of images for the detection of pneu-
monia from the chest X-ray of patients. The Convo-
Classification of Images of Childhood Pneumonia using Convolutional Neural Networks
117
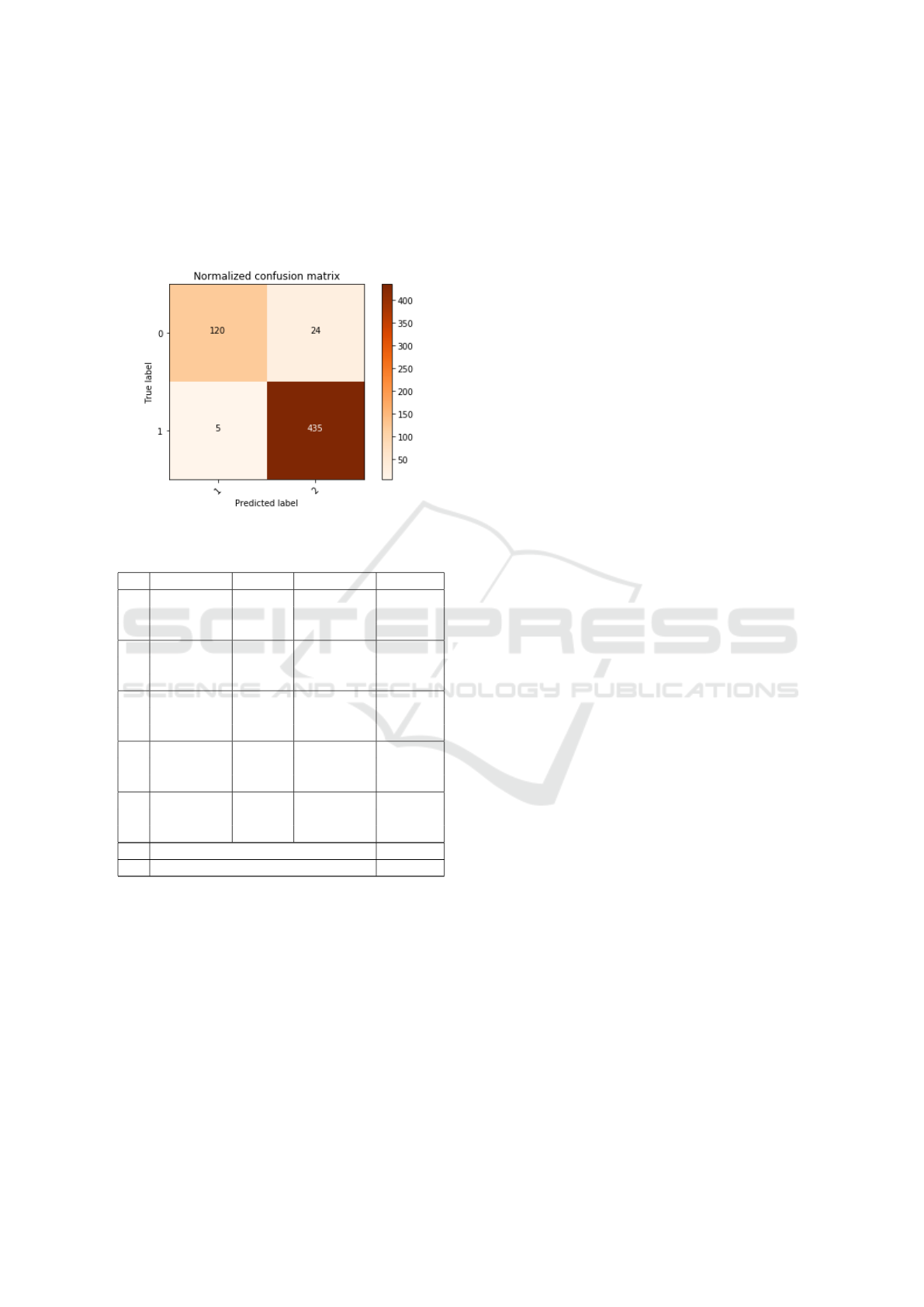
lutional Neural Network was used to train the neural
network and, for the validation of the model, Cross
validation was used. The classification model pre-
sented was efficient in the classification, obtaining an
average accuracy of 95.30 % in the tests against 92.8
% of the work of (Kermany et al., 2018).
Figure 8: Confusion matrix reference interaction 1 table 2.
Table 2: Accuracy of the interactions the model learning.
** Normal Pneumonia ACC
1 Normal 120 24
1 Pneumonia 5 435
1 95.03 %
2 Normal 131 33
2 Pneumonia 2 418
2 94.00 %
3 Normal 117 25
3 Pneumonia 3 439
3 95.20 %
4 Normal 142 17
4 Pneumonia 5 420
4 96.23 %
5 Normal 132 19
5 Pneumonia 2 431
5 96.04 %
** (Kermany et al., 2018) 92.8 %
** Average accuracy this paper 95.30 %
ACKNOWLEDGMENTS
This work is financed by National Funds through the
FCT - Fundac¸
˜
ao para a Ci
ˆ
encia e a Tecnologia (Por-
tuguese Foundation for Science and Technology) as
part of project UID/EEA/00760/2019.
REFERENCES
Amrulloh, Y. A., Abeyratne, U. R., Swarnkar, V., Herath,
D., Triasih, R., and Setyati, A. (2018). Hmm
based cough sound analysis for classifying pneumo-
nia and asthma in pediatric population. In Eskola,
H., V
¨
ais
¨
anen, O., Viik, J., and Hyttinen, J., editors,
EMBEC & NBC 2017, pages 852–855, Singapore.
Springer Singapore.
Barrientos, R., Roman-Gonzalez, A., Barrientos, F., Solis,
L., Correa, M., Pajuelo, M., Anticona, C., Lavarello,
R., Casta
˜
neda, B., Oberhelman, R., Checkley, W.,
Gilman, R. H., and Zimic, M. (2016). Automatic
detection of pneumonia analyzing ultrasound digital
images. In 2016 IEEE 36th Central American and
Panama Convention (CONCAPAN XXXVI), pages 1–
4.
Brzezinski, D. and Stefanowski, J. (2017). Prequential auc:
properties of the area under the roc curve for data
streams with concept drift. Knowledge and Informa-
tion Systems, 52(2):531–562.
Cortellaro, F., Colombo, S., Coen, D., and Duca, P. G.
(2012). Lung ultrasound is an accurate diagnostic tool
for the diagnosis of pneumonia in the emergency de-
partment. Emergency Medicine Journal, 29(1):19–23.
d. N
´
obrega, R. V. M., Peixoto, S. A., d. Silva, S. P. P., and
Filho, P. P. R. (2018). Lung nodule classification via
deep transfer learning in ct lung images. In 2018
IEEE 31st International Symposium on Computer-
Based Medical Systems (CBMS), pages 244–249.
Fan, C. and Hauser, H. (2018). Fast and accurate cnn-based
brushing in scatterplots. In Computer Graphics Fo-
rum, volume 37, pages 111–120. Wiley Online Li-
brary.
Junge, M. R. J. and Dettori, J. R. (2018). Roc solid: Re-
ceiver operator characteristic (roc) curves as a founda-
tion for better diagnostic tests. Global Spine Journal,
8(4):424–429.
Kermany, D. S., Goldbaum, M., Cai, W., Valentim, C. C.,
Liang, H., Baxter, S. L., McKeown, A., Yang, G.,
Wu, X., Yan, F., et al. (2018). Identifying medical
diagnoses and treatable diseases by image-based deep
learning. Cell, 172(5):1122–1131.
Kermany, Daniel; Zhang, K. G. M. (2018). Labeled optical
coherence tomography (oct) and chest x-ray images
for classification. Mendeley Data.
Khobragade, S., Tiwari, A., Patil, C. Y., and Narke, V.
(2016). Automatic detection of major lung diseases
using chest radiographs and classification by feed-
forward artificial neural network. In 2016 IEEE 1st In-
ternational Conference on Power Electronics, Intelli-
gent Control and Energy Systems (ICPEICES), pages
1–5.
Kingma, D. P. and Ba, J. (2014). Adam: A method for
stochastic optimization. CoRR, abs/1412.6980.
Lisowska, A., Beveridge, E., Muir, K., and Poole, I. (2017).
Thrombus detection in ct brain scans using a convolu-
tional neural network. In BIOIMAGING, pages 24–33.
Mabaso, M. A., Withey, D. J., and Twala, B. (2018). Spot
BIOIMAGING 2019 - 6th International Conference on Bioimaging
118

detection in microscopy images using convolutional
neural network with sliding-window approach.
Marques, J. F., das Chagas, Fontenele, A. A., Costa, J.
V. M., De Araujo, N. F., and Valente, A. (2018). Ma-
nipulation of bioinspiration robot with gesture recog-
nition through fractional calculus. IEEE LARS 2018 –
15th Latin American Robotics Symposium.
Mathur, S., Fuchs, A., Bielicki, J., Anker, J. V. D., and
Sharland, M. (2018). Antibiotic use for community-
acquired pneumonia in neonates and children: Who
evidence review. Paediatrics and International Child
Health, 38(sup1):S66–S75. PMID: 29790844.
Men, K., Geng, H., Cheng, C., Zhong, H., Huang, M.,
Fan, Y., Plastaras, J. P., Lin, A., and Xiao, Y. (2018).
More accurate and efficient segmentation of organs-
at-risk in radiotherapy with convolutional neural net-
works cascades. Medical physics.
Paing, M. P. and Choomchuay, S. (2017). A computer aided
diagnosis system for detection of lung nodules from
series of ct slices. In 2017 14th International Con-
ference on Electrical Engineering/Electronics, Com-
puter, Telecommunications and Information Technol-
ogy (ECTI-CON), pages 302–305.
Peng, H., Li, J., Song, Y., and Liu, Y. (2017). Incrementally
learning the hierarchical softmax function for neural
language models. In AAAI, pages 3267–3273.
Ponzio, F., Macii, E., Ficarra, E., and Di Cataldo, S. (2018).
Colorectal cancer classification using deep convolu-
tional networks.
Rajaraman, S., Candemir, S., Kim, I., Thoma, G., and An-
tani, S. (2018). Visualization and interpretation of
convolutional neural network predictions in detecting
pneumonia in pediatric chest radiographs. Applied
Sciences, 8(10).
Rizal, A., Hidayat, R., and Nugroho, H. A. (2016). Pul-
monary crackle feature extraction using tsallis entropy
for automatic lung sound classification. In 2016 1st
International Conference on Biomedical Engineering
(IBIOMED), pages 1–4.
Rizal, A., Hidayat, R., and Nugroho, H. A. (2017). Entropy
measurement as features extraction in automatic lung
sound classification. In 2017 International Confer-
ence on Control, Electronics, Renewable Energy and
Communications (ICCREC), pages 93–97.
Rodrigues, M. B., N
´
obrega, R. V. M. D., Alves, S. S. A.,
Filho, P. P. R., Duarte, J. B. F., Sangaiah, A. K., and
Albuquerque, V. H. C. D. (2018). Health of things
algorithms for malignancy level classification of lung
nodules. IEEE Access, 6:18592–18601.
Santosh, K. C. and Antani, S. (2018). Automated chest
x-ray screening: Can lung region symmetry help de-
tect pulmonary abnormalities? IEEE Transactions on
Medical Imaging, 37(5):1168–1177.
Saraiva, A., Barros, M., Nogueira, A., Fonseca Ferreira, N.,
and Valente, A. (2018a). Virtual interactive environ-
ment for low-cost treatment of mechanical strabismus
and amblyopia. Information, 9(7):175.
Saraiva, A., Miranda de Jesus Castro, F., Ferreira, N.,
and Valente, A. (2018b). Compression of electro-
cardiographs comparative study between the walsh
hadamard transform and discrete cosine transform.
Saraiva, A. A., , N. F. F., Salviano F.S.P. Soares and, M. J.
C. S. R., and Antonio, V. (2018c). Filtering of cardiac
signals with mathematical morphology for qrs detec-
tion. Proceedings of ICAT’18, 7th International Con-
ference on Advanced Technologies.
Saraiva, A. A., Costa, N., Sousa, J. V. M., De Araujo, T. P.,
Ferreira, N. F., and Valente, A. (2018d). Scalable task
cleanup assignment for multi-agents. In Memorias de
Congresos UTP, volume 1, pages 439–446.
Saraiva, A. A., Ferreira, N. M. F., and Valente, A. (2018e).
New bioinspired filter of dicom images. In Proceed-
ings of the 11th International Joint Conference on
Biomedical Engineering Systems and Technologies -
Volume 1: BIODEVICES,, pages 258–265. INSTICC,
SciTePress.
Saraiva, A. A., Nogueira, A. T., Ferreira, N. F., and Valente,
A. (2018f). Application of virtual reality for the treat-
ment of strabismus and amblyopia. In 2018 IEEE 6th
International Conference on Serious Games and Ap-
plications for Health (SeGAH), pages 1–7. IEEE.
Saraiva, A. A., SANTOS, D. S., JUNIOR, F. M., SOUSA,
J. V. M., FERREIRA, N. F., and Valente, A.
(2018g). Navigation of quadruped multirobots by ges-
ture recognition using restricted boltzmann machines.
In Memorias de Congresos UTP, volume 1, pages
431–438.
Sharma, A., Raju, D., and Ranjan, S. (2017). Detection of
pneumonia clouds in chest x-ray using image process-
ing approach. In 2017 Nirma University International
Conference on Engineering (NUiCONE), pages 1–4.
Zar, H. J., Andronikou, S., and Nicol, M. P. (2017). Ad-
vances in the diagnosis of pneumonia in children.
BMJ, 358.
Zhang, C., Sun, G., Fang, Z., Zhou, P., Pan, P., and Cong,
J. (2018). Caffeine: Towards uniformed representa-
tion and acceleration for deep convolutional neural
networks. IEEE Transactions on Computer-Aided De-
sign of Integrated Circuits and Systems, pages 1–1.
Zhang, Z. and Sabuncu, M. R. (2018). Generalized cross
entropy loss for training deep neural networks with
noisy labels. CoRR, abs/1805.07836.
Classification of Images of Childhood Pneumonia using Convolutional Neural Networks
119
