
Feature Space Reduction for Multimodal Human Activity Recognition
Yale Hartmann, Hui Liu and Tanja Schultz
Cognitive Systems Lab, University of Bremen, Germany
Keywords:
Human Activity Recognition, Biosensors, Multi-channel Signal Processing, Feature Space Reduction,
Stacking.
Abstract:
This work describes the implementation, optimization, and evaluation of a Human Activity Recognition (HAR)
system using 21-channel biosignals. These biosignals capture multiple modalities, such as motion and muscle
activity based on two 3D-inertial sensors, one 2D-goniometer, and four electromyographic sensors. We start
with an early fusion, HMM-based recognition system which discriminates 18 human activities at 91% recog-
nition accuracy. We then optimize preprocessing with a feature space reduction and feature vector stacking.
For this purpose, a Linear Discriminant Analysis (LDA) was performed based on HMM state alignments. Our
experimental results show that LDA feature space reduction improves recognition accuracy by four percentage
points while stacking feature vectors currently does not show any positive effects. To the best of our knowl-
edge, this is the first work on feature space reduction in a HAR system using various biosensors integrated into
a knee bandage recognizing a diverse set of activities.
1 INTRODUCTION
Arthrosis is the most common joint disease world-
wide and causes a noticeable reduction in quality of
life. The research and development efforts to sup-
porting arthrosis patients have increased significantly
over the last years. Efforts range from active mo-
tion support systems via exoskeletons (Fleischer and
Reinicke, 2005)(Liu et al., 2017), novel markers and
methods for arthrosis diagnostics (Mezghani et al.,
2017) to automatic offline and online recognition of
human activities associated with causing strain on the
knee (Rebelo et al., 2013)(Liu and Schultz, 2018)(Liu
and Schultz, 2019). Additionally, HAR systems have
been developed to detect and take action against func-
tional decline based on the Stair Climb Power Test
(Hellmers et al., 2017).
The mentioned works typically achieve recogni-
tion accuracies in the high ninety percent. (93%
(Hellmers et al., 2017), 97% (Liu and Schultz, 2018)
and 98% (Rebelo et al., 2013)). While these are
great results, the scope of these studies is very spe-
cific, usually recognizing a couple of different activi-
ties and using similar sensor types. Sensors typically
found in HAR Systems based around the knee and
also HAR Systems, in general, include Accelerome-
ters, Gyroscopes, (Bi-polar) Electromyography sen-
sors and Electrogoniometers. Less typical setups have
included Magnetometers and Barometers (Hellmers
et al., 2017) or Piezoelectric and Airborne micro-
phones (Teague et al., 2016)(Lukowicz et al., 2004).
Our goal is to technically assist the early treatment
of arthrosis using a HAR system to measure and re-
flect on knee straining behavior. Furthermore, we aim
to widen the scope by introducing a set of different
and diverse activities and by contributing a base sys-
tem to evaluate the benefit of sensors and features to
the discrimination of these activities. In this paper, we
will focus on that base system.
To achieve this goal we are following up on our
work using biosensors integrated into a knee bandage
(Liu and Schultz, 2018) and are using the same frame-
work for data acquisition and annotation, but with a
larger set of activities, sensors and newly evaluated
parameters. We continue to model activities using
Hidden Markov Models (HMMs), which is a widely
adopted approach. Examples include the recognition
of assembly and maintenance tasks (Lukowicz et al.,
2004) or 3D handwriting recognition (Amma et al.,
2010). However, differently, to most HAR systems we
need to model very short activities using small win-
dows, resulting in parameters similar to those found
in HMM-based Automatic Speech Recognition (ASR)
systems.
Hartmann, Y., Liu, H. and Schultz, T.
Feature Space Reduction for Multimodal Human Activity Recognition.
DOI: 10.5220/0008851401350140
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 4: BIOSIGNALS, pages 135-140
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
135

2 EQUIPMENT AND SETUP
We chose the biosignalsplux Research Kits
1
as a
recording device. One PLUX hub can process signals
from 8 channels (each up to 16 bits) simultaneously.
Therefore, we used three hubs connected via a cable
to ensure synchronization during the entire session.
Similar to (Mathie et al., 2003) and (Liu and
Schultz, 2018), we used two tri-axial accelerome-
ters, four bipolar EMG sensors and both channels of
one bi-axial electrogoniometer, as they were proven
to be effective and efficient. Additionally, combined
with the accelerometer into two Inertial Measurement
Units (IMU), two tri-axial gyroscopes were used. We
used both channels of an electrogoniometer to mea-
sure both the frontal and sagittal plane since we in-
tend to recognize rotational movements of the knee
joint in activities like ”curve-left” and ”curve-right”.
Moreover, we used a piezo and airborne microphone
like (Teague et al., 2016) and (Lukowicz et al., 2004)
and included an additional force sensor.
EMG signals, as well as the microphones, require
a high sampling rate, whereas the other biosignals
are slow in nature. Therefore, they were recorded
with different sampling rates. The slower signals used
100Hz and were up-sampled to match the 1000Hz
used for the faster signals.
2.1 Sensor Placement
We use the Bauerfeind GenuTrain knee bandage
2
shown in Figure 1 as the wearable carrier of the
biosensors. Table 1 lists all measured muscles and
sensor positions. The sensor positioning was decided
in collaboration with kinesiologists of the Institute
of Sport and Sports Science at Karlsruhe Institute of
Technology and designed to capture human everyday
and sports activities relevant to gonarthrosis.
Table 1: Sensor placement and captured muscles.
Sensor Position / Muscle
IMU1 Thigh, proximal ventral
IMU2 Shank, distal ventral
EMG1 Musculus vastus medialis
EMG2 Musculus tibialis anterior
EMG3 Musculus biceps femoris
EMG4 Musculus gastrocnemius
Goniometer Right Knee, lateral
Microphones Bandage inside/outside, medial
Force Sensor Between patella and bandage
1
biosignalsplux.com/researcher
2
www.bauerfeind.de/en/products/supports-
orthoses/knee-hip-thigh/genutrain.html
Figure 1: The knee bandage used as carrier.
3 DATASET
We recorded a dataset of eighteen activities from
seven male subjects in a controlled lab environment
using the previously introduced ASK framework (Liu
and Schultz, 2018). We had to drop three of the seven
subjects’ recordings due to technical issues, resulting
in a total of 40 minutes of usable semi-automatically
annotated data. While this is a limited amount, further
recordings are underway.
Table 2 gives occurrences, minimum and maxi-
mum length of the eighteen activities.
Most activities in Table 2 are self-explanatory, the
others are defined thusly:
Curve-X-Spin is a fast 90
◦
body turn in one step.
Curve-X-Step is a 90
◦
turn using several walking
steps.
V-Cut-X is a direction change with an acute angle at
jogging speed.
Lateral-shuffle-X are repeated lateral steps starting
with the right/left foot, the other following.
Jump-one-leg means jumping 1m forward using the
bandaged leg.
Jump-two-legs means jumping in place using both
legs.
Run means several steps passed at constant jogging
speed.
An imbalance of occurrences can be observed for
the activities ”Run” and ”Walk” and is explained by
BIOSIGNALS 2020 - 13th International Conference on Bio-inspired Systems and Signal Processing
136

their repeated use in different lists, which we used
similarly to (Liu and Schultz, 2018). However, this
imbalance is welcomed as it reflects expectations in
uncontrolled settings as well as allowing for a more
detailed model better discriminating similar activities.
Table 2: Number of occurrences, minimum and maximum
length of each activity.
Activity Occ. Min Max.
curve-left-spin 54 0.725s 3.249s
curve-left-step 44 1.759s 4.269s
curve-right-spin 47 0.666s 2.279s
curve-right-step 45 1.649s 3.789s
jump-one-leg 47 0.749s 2.159s
jump-two-leg 47 0.999s 1.969s
lateral-shuffle-left 47 1.129s 4.089s
lateral-shuffle-right 47 0.969s 4.379s
run 86 1.179s 3.139s
sit 38 1.329s 5.089s
sit-to-stand 42 0.939s 3.589s
stair-down 45 1.769s 5.259s
stair-up 43 1.989s 5.159s
stand 37 1.759s 5.129s
stand-to-sit 42 1.029s 3.449s
v-cut-left 43 0.709s 2.209s
v-cut-right 37 0.679s 1.699s
walk 198 1.579s 5.179s
4 BASELINE HUMAN ACTIVITY
RECOGNIZER
We developed a baseline recognizer using our in-
house HMM Decoder BioKIT with simple features
and tuned the parameters to achieve a good baseline.
Our recognizer uses a forward topology commonly
found in HAR and ASR, where each state allows for
a transition to itself or the next(Rebelo et al., 2013).
The emission probability for each state is modeled us-
ing Gaussian Mixture Models (GMMs). For our base
system, we model each activity with the same num-
ber of states and each state with the same number of
Gaussians per mixture.
4.1 Windowing and Feature Extraction
Feature Extraction is straightforward. For each chan-
nel, we use a rectangular window function with some
overlap, then calculate the Root Mean Square (RMS)
and Average (avg) on each window and z-normalize
the whole activity. Due to the lack of spectral features,
no smoothing window function is required.
Denoting the sample sequence of a window as
(x
1
, ...x
n
) and N = n the number of samples in that
window, the average is defined as:
avg =
1
N
N
∑
n=1
x
n
, (1)
The Root Mean Square is defined as:
RMS =
s
1
N
N
∑
k=1
x
2
n
(2)
While our framework supports both early and late
fusion of biosignals, we opted to use an early fu-
sion in order to allow feature combinations of dif-
ferent channels. The resulting multi-channel feature
vector has 42 dimensions since there are 21 channels
and per channel two features are calculated. This
approach differs from our previous work (Liu and
Schultz, 2018) where RMS was only used for the
EMG channels and Average for all others.
4.2 Parameter Tuning
The research on gait analysis commonly distinguishes
two phases into eight events, as described in (Whit-
tle, 2014) and further discussed in (Whittle, 1996)
and (Mezghani et al., 2013). Therefore, we expected
a HMM topology with eight states to perform best.
However, in evaluation it was outperformed by a six-
state topology, which is what we then continued with.
This unexpected result might be due to the GMMS be-
ing better fitted in the six-state topology or due to not
all activities corresponding to a gait cycle. Further
discussion is left to future work.
The overall shortest activity is an instance of
”curve-right-spin” (See Table 2) at 666ms. Therefore,
requiring a maximum window length of 111ms to
be recognizable with a six-state HMM. Assuming an
equal distribution of samples across the HMM when
aligned, we opted to use a window length of 10ms
with an overlap of 2ms, resulting in an absolute mini-
mum of twelve samples per state. Using these param-
eters we evaluated several numbers of Gaussians per
mixture, finding that seven Gaussians perform reason-
ably well and having enough data to fit the Gaussians
properly. In the future, we want to use a merge and
split estimation here, adaptively adjusting the amount
of Gaussians to use the present data optimally.
To summarize, our base system used a multi-
channel feature signal rectangularly framed with a
window length of 10ms and an overlap of 2ms. Cal-
culating the Average and RMS on each window and
z-normalizing the result. HMMs are modeled using
Feature Space Reduction for Multimodal Human Activity Recognition
137

six states with a forward topology and each state us-
ing a GMM with seven Gaussians. Evaluating this
setup with a randomized ten-fold cross-validation the
mean accuracy is 91%.
5 FEATURE SPACE REDUCTION
Reducing the feature space dimension has several
benefits. Firstly, GMMs can be fitted more effec-
tively. Secondly, more (sensor specific) features can
be added easily, since the LDA is used to transform
them into a smaller maximum discriminating fea-
ture space. Effectively reducing redundancy between
channels and producing a consistent feature space di-
mension independent from sensor and feature setup.
Reducing the original multi-channel feature vec-
tor directly with a LDA could not preserve non-linear
relations and would interfere with the sequential mod-
eling of our HMMs. Therefore, we align the sam-
ples to states using the Viterbi algorithm and using
each activities’ state as target for the aligned feature
vector. This approach allows for a linear supervised
reducing function, which generally outperform unsu-
pervised options like a Principal Component Analysis
(PCA). Similar experiments using non-linear reduc-
tion methods like Neural Networks (NN) combined
with a PCA have shown to improve performance(Hu
and Zahorian, 2010).
Figure 2: Evaluation: Feature Dimension. Setup: 10ms
window length; 2ms overlap; 6 States per HMM; 7 Gaus-
sians per state.
Applying this technique to our base system, the
system has a performance peak when reducing to a
13-dimensional feature space as shown in Figure 2.
Increasing the accuracy by four percentage points to
94.9% compared to the approach without dimension
reduction. Similarly clear is the decrease in perfor-
mance when reducing to too few dimensions as too
few meaningful features are used. More surprising
is the steady decline of performance after 13 dimen-
sions. There are several possible reasons for this,
including too high a dimension to fit the Gaussians
properly as well as sensors that might provide contra-
dictory information. A deeper analysis of sensors and
features is planned for the future.
The recognizer using 13-dimensional features
tends to predict walking and running over curve based
activities as seen in Figure 3. Several Steps and Spins
are recognized as ”Walk”. The ”V-Cuts” are more
than once predicted to be ”Run”. Additionally, the
recognizer confuses left and right ”V-Cuts”. This is
consistent behavior to what we have seen in the base-
line recognizer.
Figure 3: Confusion matrix of recognition results. Setup:
10ms window length; 2ms overlap; 6 States per HMM; 7
Gaussians per state; 13-dimensional feature space.
6 FEATURE VECTOR STACKING
Another possible approach to improve performance is
to add context to each feature vector in the preprocess-
ing step by prepending the n previous and appending
the n following feature vectors. This process is called
stacking.
If evaluated naively without a feature space reduc-
tion, the performance decreases with increasing con-
text. This behavior is expected due to the significantly
increasing feature vector dimensions as shown in Ta-
ble 3 compared to only few data samples.
Running the same experiment with a fixed feature
space dimension of 13 does not increase the perfor-
mance (Figure 4). On the contrary, 0-stacking fea-
tures enhance performance in our case. Incidentally,
0-stacking here uses the same configuration as the
recognizer shown in Figure 3. Additionally, the per-
BIOSIGNALS 2020 - 13th International Conference on Bio-inspired Systems and Signal Processing
138
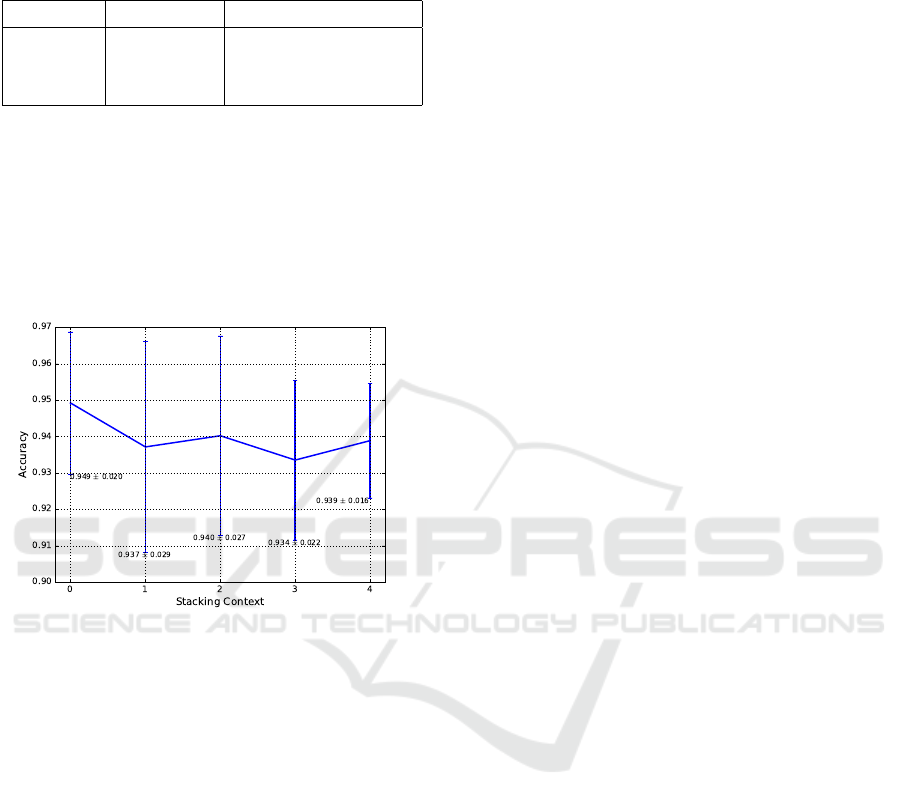
Table 3: Results of naive stacking. Window length: 10ms;
overlap: 2ms; 6 States per HMM; 7 Gaussians per state;
without feature space reduction.
Context Accuracy Vector Dimension
0 0.92 42
1 0.80 126
2 0.74 210
formance between different context sizes is not sig-
nificant as a statistical analysis via T-Test indicates.
These results are obtained on a local optimum
with a 13-dimensional feature space using solely sta-
tistical features. Therefore, we will investigate this
behavior for temporal features and further dimensions
in the future.
Figure 4: Evaluation: stacking. Setup: 10ms window
length; 2ms overlap; 6 States per HMM; 7 Gaussians per
state; 13-dimensional feature space.
7 CONCLUSION
In this paper, we successfully implemented, evaluated
and improved an offline, early fusion HAR system us-
ing a 21-dimensional biosignal comprised of different
sensors placed onto a knee bandage. The base system
performed very well with a 91% accuracy using only
simple features. We showed, that the performance
could be improved by four percentage points to 94.9%
using a LDA trained with HMM state aligned labeled
data and reducing the feature space dimension. Fur-
thermore, we found that in our case stacking feature
vectors to improve context did not increase perfor-
mance but instead slightly decreased it with respect
to not stacking at all, which is one topic for further
investigation.
In the future, we will evaluate additional more so-
phisticated features targeted to the specific sensors
and their influence on the overall performance as well
as the feature space reduction specific performance.
Furthermore, we will create and evaluate different
topologies for different activities and investigate the
performance of our system using a person indepen-
dent evaluation on a larger dataset. To the best of our
knowledge, this is the first work on feature space re-
duction in a HAR system using various biosensors in-
tegrated into a knee bandage recognizing a diverse set
of activities.
REFERENCES
Amma, C., Gehrig, D., and Schultz, T. (2010). Airwrit-
ing recognition using wearable motion sensors. In
First Augmented Human International Conference,
page 10. ACM.
Fleischer, C. and Reinicke, C. (2005). Predicting the in-
tended motion with emg signals for an exoskeleton
orthosis controller. In 2005 IEEE/RSJ International
Conference on Intelligent Robots and Systems (IROS
2005), pages 2029–2034.
Hellmers, S., Kromke, T., Dasenbrock, L., Heinks, A.,
Bauer, J. r. M., Hein, A., and Fudickar, S. (2017). Stair
Climb Power Measurements via Inertial Measurement
Units. pages 1–9.
Hu, H. and Zahorian, S. A. (2010). Dimensionality reduc-
tion methods for HMM phonetic recognition. In 2010
IEEE International Conference on Acoustics, Speech
and Signal Processing, pages 4854–4857. IEEE.
Liu, H. and Schultz, T. (2018). Ask: A framework for data
acquisition and activity recognition. In 11th Interna-
tional Conference on Bio-inspired Systems and Signal
Processing, Madeira, Portugal, pages 262–268.
Liu, H. and Schultz, T. (2019). A Wearable Real-time Hu-
man Activity Recognition System using Biosensors
Integrated into a Knee Bandage. In Proceedings of
the 12th International Joint Conference on Biomedi-
cal Engineering Systems and Technologies, pages 47–
55. SCITEPRESS - Science and Technology Publica-
tions.
Liu, X., Zhou, Z., Mai, J., and Wang, Q. (2017). Multi-
class SVM Based Real-Time Recognition of Sit-to-
Stand and Stand-to-Sit Transitions for a Bionic Knee
Exoskeleton in Transparent Mode. In The Semantic
Web - ISWC 2015, pages 262–272. Springer Interna-
tional Publishing, Cham.
Lukowicz, P., Ward, J. A., Junker, H., St
¨
ager, M., Tr
¨
oster,
G., Atrash, A., and Starner, T. (2004). Recognizing
Workshop Activity Using Body Worn Microphones
and Accelerometers. In Pervasive Computing, pages
18–32. Springer, Berlin, Heidelberg, Berlin, Heidel-
berg.
Mathie, M., Coster, A., Lovell, N., and Celler, B. (2003).
Detection of daily physical activities using a triaxial
accelerometer. In Medical and Biological Engineer-
ing and Computing. 41(3):296—301.
Mezghani, N., Fuentes, A., Gaudreault, N., Mitiche, A.,
Aissaoui, R., Hagmeister, N., and De Guise, J. A.
Feature Space Reduction for Multimodal Human Activity Recognition
139

(2013). Identification of knee frontal plane kinematic
patterns in normal gait by principal component anal-
ysis. Journal of Mechanics in Medicine and Biology,
13(3).
Mezghani, N., Ouakrim, Y., Fuentes, A., Mitiche, A., Hage-
meister, N., Vendittoli, P.-A., and De Guise, J. A.
(2017). Mechanical biomarkers of medial compart-
ment knee osteoarthritis diagnosis and severity grad-
ing: Discovery phase. Journal of Biomechanics,
52:106–112.
Rebelo, D., Amma, C., Gamboa, H., and Schultz, T. (2013).
Activity recognition for an intelligent knee orthosis.
In 6th International Conference on Bio-inspired Sys-
tems and Signal Processing, pages 368–371. BIOSIG-
NALS 2013.
Teague, C. N., Hersek, S., Toreyin, H., Millard-Stafford,
M. L., Jones, M. L., Kogler, G. F., Sawka, M. N.,
and Inan, O. T. (2016). Novel Methods for Sens-
ing Acoustical Emissions From the Knee for Wear-
able Joint Health Assessment. IEEE Transactions on
Biomedical Engineering, 63(8):1581–1590.
Whittle, M. W. (1996). Clinical gait analysis: A review.
Human Movement Science, 15(3):369–387.
Whittle, M. W. (2014). Gait Analysis. Normal Gait.
Butterworth-Heinemann.
BIOSIGNALS 2020 - 13th International Conference on Bio-inspired Systems and Signal Processing
140
